The Doctrine That Kept Generics Alive—Until It Didn’t

For decades, the skinny label was the workhorse of generic drug market entry. It was the tool that let a generic manufacturer say, without apology: “We’re not copying everything. We carved out the patented use. We’re selling a product for the indications that are off-patent.” Courts had largely accepted this logic, the FDA had built a regulatory mechanism to support it, and the generic industry had built business models around it.
Then came GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc.—a case about a heart drug called carvedilol—and the Federal Circuit made clear that a carved-out label was not a legal shield if the rest of your marketing behavior told a different story.
The case did not kill the skinny label doctrine. Attorneys who argue otherwise are overstating the ruling. But the decision fundamentally shifted the calculus for every generic manufacturer considering a Section viii carve-out. It introduced evidentiary liability for product communications, FDA-approved launch announcements, and sales force messaging that a court could interpret as promoting patented uses—even when the label itself was surgically clean. The question is no longer just “did we carve out the right language from the label?” The question is now “did anything we said or wrote, anywhere, across any channel, amount to active inducement of infringement?”
That is a much harder standard to meet. And it has prompted the most significant strategic rethink the generic sector has seen since the Paragraph IV certification process was codified.
The rethink extends beyond legal strategy. It reaches into how generic manufacturers structure their ANDA pipelines, how they staff their legal and regulatory teams, how they design their commercial launch programs, how they communicate with investors, and how they evaluate the financial returns of early market entry. In short, GSK v. Teva is not a litigation department problem. It is an enterprise-wide challenge.
The scale of what is at stake becomes clear when you consider the patent landscape for the drugs currently in or approaching the generic pipeline. According to data available through DrugPatentWatch, dozens of currently branded drugs with aggregate U.S. annual revenues exceeding $50 billion carry Orange Book-listed method-of-use patents that will remain active after the primary compound patents expire. For each of these drugs, generic manufacturers face the choice between waiting for full patent expiration—often years away—pursuing Paragraph IV challenges, or attempting a Section viii market entry with the liability framework that GSK v. Teva now imposes. Getting that choice wrong in either direction has real consequences: too much caution delays patient access and generic revenue; too little caution invites litigation exposure that can threaten a company’s financial stability.
This article maps that rethink in detail—covering the legal mechanics, the patent intelligence required, the financial modeling adjustments, and the practical steps a generic manufacturer needs to take today to preserve the skinny label as a viable market-entry tool. It draws on publicly available case law, FDA guidance documents, patent data aggregated through resources like DrugPatentWatch, and commentary from litigators and regulatory counsel who work in this space daily.
Understanding the Skinny Label: What It Is and How It Was Supposed to Work
The skinny label, also called a “carved-out” label or a “Section viii statement,” describes the practice of a generic drug manufacturer filing an Abbreviated New Drug Application (ANDA) while deliberately excluding one or more approved uses of the reference listed drug (RLD) from the proposed labeling. The excluded uses are typically protected by one or more method-of-use patents listed in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations—the document universally known as the Orange Book.
The mechanics are straightforward. When a brand manufacturer receives approval for a new indication and patents the method of using a drug for that indication, the patent gets listed in the Orange Book. A generic filer who wants to avoid a patent dispute over that specific use can submit what the regulations call a “Section viii statement”—a certification that the generic’s label will not include the patented indication. The FDA then permits the generic to market the product with a label that omits the carved-out use.
The commercial logic was always clear: if a drug has, say, three approved indications—one off-patent and two still under patent protection—the generic can enter the market for the off-patent indication. Doctors prescribing the drug for the off-patent use would write generic prescriptions. Pharmacists would dispense the generic. The brand manufacturer would retain exclusivity only for the patented indications.
This system had practical and policy support from both sides of the aisle. Generic manufacturers valued it because it accelerated market entry. The FDA valued it because faster generic entry serves the public interest in access to affordable drugs. Even some brand manufacturers accepted it as a necessary feature of the Hatch-Waxman system—a quid pro quo that made the overall framework politically sustainable.
What the system assumed was that the carve-out worked as advertised. It assumed that once a generic removed patented indications from the label, any prescriptions for the generic product would be directed at the off-patent use. That assumption has now been blown apart.
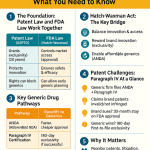
The Hatch-Waxman Framework and the Section viii Carve-Out
The Drug Price Competition and Patent Term Restoration Act of 1984—known as the Hatch-Waxman Act—created the modern framework for generic drug approval in the United States. It was a compromise. Congress wanted to incentivize innovation by protecting brand drug patents, and it wanted to incentivize competition by giving generics a streamlined approval pathway. The ANDA process, with its requirement to demonstrate bioequivalence rather than full clinical efficacy, was the core of that streamlining.
The patent certification requirements embedded in the ANDA process reflect the same compromise. When filing an ANDA, the applicant must certify one of four positions with respect to each patent listed in the Orange Book for the RLD:
Paragraph I certifies that no patent has been filed. Paragraph II certifies that the patent has expired. Paragraph III certifies that the generic will not launch until after patent expiration. Paragraph IV certifies that the listed patent is invalid, unenforceable, or will not be infringed.
The Section viii statement is a fifth option—technically not a “Paragraph” certification but a statutory statement available specifically for method-of-use patents. Under 21 U.S.C. § 505(j)(2)(A)(viii), an ANDA applicant can state that the patent in question claims a method of using the drug and that the applicant’s proposed labeling does not include that method. This statement requires no litigation. It triggers no automatic 30-month stay. It allows the generic manufacturer to proceed to approval without fighting the patent directly.
That relative ease of use made the Section viii route attractive—sometimes so attractive that generic manufacturers used it as a default rather than a carefully considered strategy. The GSK v. Teva litigation revealed the risks of that complacency.
The legislative history of the Hatch-Waxman Act confirms that Congress intended the Section viii statement as a carve-out mechanism to allow generic entry where a drug has both patented and unpatented uses [1]. What the Act did not address in any detail was what happens after approval—specifically, whether the commercial behavior of a generic manufacturer post-approval could convert a legitimate carve-out into an act of patent infringement.
That gap, left unaddressed in 1984, is precisely what the Federal Circuit filled in GSK v. Teva.
The gap has a statutory explanation. The Hatch-Waxman Act was primarily concerned with the pre-approval phase of the generic-brand relationship—the certification process, the 30-month stay, and the first-filer exclusivity. Congress focused its attention on preventing brand manufacturers from using patent litigation to block generic approval indefinitely, and on ensuring that generic manufacturers could obtain approval without running clinical trials that duplicated the brand manufacturer’s original work.
The post-approval commercial phase received far less attention in the 1984 legislative text. The induced infringement provisions of 35 U.S.C. § 271(b) were already part of patent law before Hatch-Waxman, and Congress did not modify them to account for the specific dynamics of multi-indication pharmaceutical markets. The result is that the carve-out mechanism created by Hatch-Waxman operates in tension with the induced infringement standard developed by the courts—a tension that Congress had no particular reason to anticipate in 1984 and has not yet resolved through subsequent legislation.
This historical context matters for policy advocacy. When generic manufacturers argue for legislative clarification of the Section viii safe harbor, they are not asking for a new entitlement—they are asking Congress to complete the work that the 1984 statute left unfinished. The original legislative intent was to facilitate generic entry for unpatented uses; the current induced infringement standard undermines that intent by making post-approval commercial behavior an obstacle to the very entry the statute was designed to enable. That framing resonates with legislators across the political spectrum who view generic drug access as a bipartisan priority.
GSK v. Teva: A Case-by-Case Deconstruction
Carvedilol is a beta-blocker. GlaxoSmithKline sold it under the brand name Coreg. The FDA initially approved carvedilol for treating hypertension and left ventricular dysfunction following a heart attack. Those approvals came with compound and method-of-use patents that Teva did not contest when seeking approval for its generic version.
The flashpoint was a third indication: congestive heart failure (CHF). GSK held U.S. Patent No. 5,760,069, which covered a method of decreasing mortality in patients with CHF using carvedilol. This was a method-of-use patent—it protected not the molecule itself but the specific therapeutic application of treating CHF. GSK listed it in the Orange Book.
Teva filed a Section viii statement for the CHF indication, carving it out of its proposed labeling. The FDA approved Teva’s generic carvedilol with the carved-out label. Teva launched in 2007. By 2011, Teva’s generic had captured over 80 percent of carvedilol prescriptions—including prescriptions from physicians who were treating CHF patients [2].
GSK sued Teva for induced infringement of the CHF patent. The legal claim was not that Teva’s label described the patented use—it didn’t. The claim was that Teva had actively induced physicians to prescribe carvedilol for CHF through its marketing communications, press releases, and FDA-approved promotional materials, even though none of those materials referenced CHF by name.
The trial court initially ruled for Teva, finding insufficient evidence of inducement. The Federal Circuit reversed in 2020, finding substantial evidence that Teva had induced infringement. The court then reversed again in 2021 after en banc consideration—though without reversing the underlying liability standard—before ultimately affirming the infringement verdict in a deeply fractured decision.
The key evidentiary points that the Federal Circuit found probative were these:
First, Teva’s press release announcing generic carvedilol launch described the drug as a treatment for “heart failure”—a term broad enough to encompass CHF. Second, Teva’s product launch materials referenced “congestive heart failure” in certain promotional contexts. Third, the court found that Teva’s skinny label itself, despite the carve-out, retained language about the drug’s mechanism of action that was consistent with the CHF use—and that this “skinny label” was, in practice, insufficiently thin.
Fourth, and most consequentially: the court held that GSK had presented substantial evidence showing that the vast majority of carvedilol prescriptions were being written for CHF, and that Teva’s marketing had reached physicians who treated CHF patients [3]. The Federal Circuit concluded that knowledge of infringement combined with affirmative acts encouraging it—even inadvertent ones—could satisfy the intent element of induced infringement.
Induced Infringement: The Legal Engine Behind the Liability
To understand why GSK v. Teva matters, you have to understand induced infringement as a legal doctrine. Patent infringement comes in three forms: direct, contributory, and induced. Direct infringement is what most people think of—someone makes, uses, or sells a patented invention without authorization. Contributory infringement occurs when someone sells a component that has no substantial non-infringing use and knows it will be used to infringe.
Induced infringement, codified at 35 U.S.C. § 271(b), says that anyone who “actively induces infringement of a patent shall be liable as an infringer.” The Supreme Court clarified the standard in Shire v. Sandoz and more extensively in Global-Tech Appliances, Inc. v. SEB S.A. (2011), which established that induced infringement requires actual knowledge that the induced acts constitute patent infringement [4].
In the pharmaceutical skinny label context, the “infringer” is the physician who prescribes the drug for the patented use. The generic manufacturer is the alleged inducer. The generic does not itself infringe by manufacturing the drug—because the underlying compound is off-patent. But if the generic manufacturer actively encourages physicians to prescribe the drug for a patented use, it has induced infringement by those physicians.
The doctrinal challenge—the one that makes the skinny label so legally fraught—is that intent in this context is determined by looking at the totality of the manufacturer’s conduct, not just the label. As the Federal Circuit explained in GSK v. Teva: “Where a product has multiple uses, some patented and some not, an ANDA filer that carves out a patented use from its label, but then promotes the drug for the patented use through other means, can be liable for induced infringement” [5].
That “other means” language is where generic manufacturers are now vulnerable. It encompasses press releases, website content, medical education materials, sales representative talking points, earnings call commentary, and FDA communication logs—any of which could theoretically provide evidence of intent to induce infringement.
The intent element historically required a showing of “specific intent”—the defendant had to know about the patent and know that its conduct would result in infringement. After GSK v. Teva, courts have signaled that constructive knowledge combined with willful blindness can substitute for actual knowledge, and that circumstantial evidence of intent—including marketing reach and the demographics of likely prescribers—can carry substantial weight with juries.
What the Federal Circuit Actually Said—and What It Implies
The fractured nature of the GSK v. Teva opinions makes it tempting to dismiss the case as unclear law. That would be a mistake. While the en banc court did not produce a single majority opinion on every issue, the affirmance of the liability verdict, combined with the various concurrences and the majority’s treatment of the evidence, establishes several clear propositions.
First, a carved-out label does not, by itself, defeat an inducement claim. This was the most significant departure from prior lower court practice, which had treated a clean label carve-out as close to dispositive. The Federal Circuit made clear that the label is one piece of evidence, not a complete defense.
Second, external marketing communications are competent evidence of inducement intent. This means that generic manufacturers face liability exposure from any post-approval communication that a court could characterize as directing physicians toward patented uses—even if the communication is technically accurate and does not mention the patented indication by name.
Third, the scale of off-label prescribing is relevant. The majority opinion in GSK v. Teva specifically discussed the fact that most carvedilol prescriptions were being written for CHF patients. This suggests that courts will look at real-world prescribing patterns as a proxy for whether infringement is actually occurring at scale—and will attribute that scale, at least in part, to the generic manufacturer’s marketing footprint.
Fourth, label language that mirrors the patented use—even without naming the indication—can contribute to an infringement finding. This has practical implications for the drafting of carved-out labels: removing the indication name is not sufficient if the mechanism of action language, dosing information, or clinical pharmacology section effectively describes the patented method.
What the decision does not resolve is the precise line between permissible skinny-label marketing and prohibited inducement. Judge Prost’s dissent argued forcefully that the majority had collapsed that line entirely—that under the majority’s reasoning, the mere fact that a product could be used for a patented indication, combined with any marketing activity at all, would be enough to establish liability. That critique has not been answered by the courts, and it points to a genuine tension that subsequent litigation will have to resolve.
The Dissent’s Warning and Why It Matters More Now
Judge Sharon Prost wrote the principal dissent in GSK v. Teva, and her analysis is not simply a losing legal argument. It is a roadmap of the weaknesses in the majority’s reasoning—weaknesses that careful generic manufacturers can exploit in future litigation.
Prost’s core argument was this: Congress specifically created the Section viii carve-out mechanism to allow generic entry for unpatented uses. If a generic manufacturer complies with that mechanism—carves out the patented use, receives FDA approval, and does not promote the patented use—then liability for induced infringement is legally incoherent. The mechanism would be a trap rather than a safe harbor [6].
She noted that the majority relied heavily on evidence that Teva’s press releases described the drug as a “heart failure” treatment—but argued that this characterization was accurate for the unpatented indications (hypertension and left ventricular dysfunction following a heart attack), and that attributing it to promotion of CHF required an inferential leap that no court should make without direct evidence of CHF promotion.
Her dissent also raised a systemic concern that has since been echoed by academic commentators: if courts hold generic manufacturers liable for induced infringement whenever prescribing patterns show widespread use for patented indications—regardless of whether the generic promoted those uses—then the skinny label doctrine has been functionally eliminated. No generic manufacturer could safely enter a multi-indication market where the patented use is the dominant one.
This concern is not academic. Databases like DrugPatentWatch document dozens of drugs currently in the Orange Book with multi-indication profiles where the dominant commercial use is a method-of-use patented indication. If Prost’s systemic critique is correct, the market entry risk for generics in those markets has been substantially elevated.
The practical takeaway from the dissent: document everything. If a generic manufacturer is engaging in any marketing activity after launch, it needs contemporaneous records showing that every communication was directed at the unpatented indications—not merely that the patented indication was technically omitted.
The Immediate Fallout: How Generic Manufacturers Are Recalibrating
The generic pharmaceutical industry’s response to GSK v. Teva has been swift and largely invisible to the public, playing out in legal strategy memos, ANDA drafting protocols, and internal compliance reviews rather than press releases.
Several observable shifts have emerged.
Generic manufacturers are restructuring their ANDA filing strategies. For multi-indication drugs with one or more method-of-use patents, companies are increasingly evaluating whether to pursue Paragraph IV challenges—contesting the patent directly—rather than relying on Section viii statements. This is more expensive and time-consuming, but it creates a more defensible legal position if the drug reaches market.
Companies are also revising their launch communication protocols. Legal and regulatory teams at multiple generic manufacturers have implemented new approval workflows for press releases, investor relations materials, and FDA communications to ensure that no document uses language that could later be read as promoting a patented use. This includes prohibiting the use of disease state terms that correspond to patented indications, even in factually accurate contexts.
Internal training programs for sales forces have been updated. Where generic manufacturers maintain field sales forces calling on prescribers, those teams are now receiving explicit guidance on the content of their detailing conversations—specifically, which therapeutic contexts they can discuss and which they cannot.
Perhaps most significantly, some generic manufacturers are reconsidering market entry decisions entirely for certain drug categories. Where the primary commercial use of a drug is the patented indication—where, in other words, most prescriptions would be written for the carved-out use—the risk that a court will find inducement based on prescribing patterns alone has become high enough that some companies are simply waiting for patent expiration rather than attempting a skinny label entry.
This last effect represents a real policy failure. The skinny label was designed to accelerate access to affordable generics. If it is now so legally treacherous that manufacturers abandon it for certain drug categories, patients who would have benefited from early generic entry will instead pay brand prices for years longer.
Mapping the Patent Landscape for Method-of-Use Claims
To manage skinny label risk, you need patent intelligence—and that starts with understanding how method-of-use patents work in the Orange Book system.
Method-of-use patents are conceptually distinct from compound patents. A compound patent covers the chemical entity itself—the molecule. Method-of-use patents cover specific applications: methods of treating a disease, methods of achieving a therapeutic outcome, methods of administering a drug in a particular way to achieve a particular result. They are the most common form of “evergreening” in the pharmaceutical sector because they can be filed and listed in the Orange Book years or decades after the compound patent was first issued.
The strategic value of method-of-use patents to brand manufacturers is enormous. A drug with a single compound patent and multiple method-of-use patents can maintain market exclusivity on its most commercially valuable indications long after the compound goes off-patent. Brand manufacturers have become sophisticated at layering these patents—filing continuation applications as new clinical data emerges, listing each new patent in the Orange Book, and effectively converting a finite patent term into a renewable wall of protection.
For generic manufacturers, the first step in evaluating a Section viii filing opportunity is mapping the full patent landscape for the RLD. This means identifying every patent listed in the Orange Book for the drug, understanding which claims are compound claims versus method-of-use claims, and then assessing which method-of-use claims map to which commercial indications.
Resources like DrugPatentWatch are invaluable at this stage. DrugPatentWatch aggregates Orange Book listings, ANDA filings, and litigation histories in a searchable format, allowing generic manufacturers to identify not just which patents are listed for a given drug, but which of those patents have been challenged, which have survived litigation, and how the claim scope of disputed patents has been interpreted by courts. This kind of patent intelligence is not a luxury—it is the foundation of any defensible Section viii strategy.
The mapping exercise has to go beyond the Orange Book. Method-of-use patents sometimes protect uses that are not yet approved by the FDA—they may be listed in the Orange Book prospectively, or they may not be listed at all but still enforceable if the generic’s product is used for the claimed method. Patents covering off-label uses of a drug are generally not listable in the Orange Book [7], but they can still give rise to induced infringement claims if the generic’s marketing activities effectively promote those uses.
This means that a complete patent landscape analysis for skinny label purposes has to include not just Orange Book patents but also patents held by the brand manufacturer that claim methods corresponding to any significant off-label prescribing pattern for the drug. The clinical and epidemiological literature on off-label use is therefore relevant to the patent analysis—not just the formal approval documents.
The anatomy of a method-of-use patent also matters for understanding where carve-outs succeed and where they fail. Method-of-use claims can be drafted at different levels of specificity, and that specificity determines how difficult the carve-out is to execute. A broad method-of-use claim might read: “A method of treating congestive heart failure comprising administering an effective amount of carvedilol to a patient in need thereof.” A narrow claim might read: “A method of reducing mortality in patients with stable, moderate-to-severe congestive heart failure classified as NYHA Class II-IV comprising administering carvedilol at a starting dose of 3.125 mg twice daily.” The broad claim creates a wide exclusion zone for the carve-out—any label language describing carvedilol for heart failure must be removed. The narrow claim creates a more surgical exclusion—only language specifically describing that NYHA-classified protocol needs to go, and the label can potentially retain other heart failure language.
In practice, brand manufacturers draft their method-of-use claims broadly when the prosecution history allows, specifically to create this problem for generic carve-outs. A brand manufacturer’s patent attorney knows that a broad method claim is harder to carve around. The recent trend toward “genus” claims in method-of-use patents—claims that cover a broad class of patient populations, dosing ranges, or therapeutic outcomes rather than a single specific protocol—makes the carve-out drafting exercise correspondingly more difficult.
This is why Orange Book patent claim analysis is not a paralegal task. The person evaluating whether a particular sentence in a proposed label falls within the scope of a method-of-use claim needs to be a patent attorney with experience in pharmaceutical claim construction. The rules of claim construction—including the doctrine of claim differentiation, the impact of prosecution history estoppel, and the application of the doctrine of equivalents—all affect how broadly a court will read the claim, and therefore how much label language needs to be removed.
The prosecution history of the patent is particularly important. If the brand manufacturer narrowed a claim during prosecution to overcome a prior art rejection—agreeing, for example, to limit the claim to patients with NYHA Class III-IV heart failure rather than all heart failure patients—then a generic manufacturer may be able to retain label language describing the drug for NYHA Class I-II patients without infringing the patent’s narrowed scope. But taking advantage of prosecution history estoppel requires reading the full prosecution file, not just the issued patent, and the prosecution file for major pharmaceutical patents can run to thousands of pages.
Companies that use DrugPatentWatch as part of their patent surveillance process gain access to prosecution history summaries, claim construction rulings from prior litigation, and inter partes review (IPR) decisions that may have amended or invalidated specific claims—all of which inform the practical scope of what needs to be carved out. Using these resources systematically, rather than conducting from-scratch prosecution history reviews for every target product, can substantially reduce the time and cost of the pre-filing patent landscape analysis.
The Orange Book listing strategy employed by brand manufacturers also affects the carve-out difficulty. Some brand manufacturers list their broadest method-of-use patents in the Orange Book and their narrower continuation claims separately, creating multiple patents that each need to be addressed by the generic filer. A generic filer who focuses only on the most prominent listed patent and overlooks a narrower continuation with claims that are harder to carve around can find itself in exactly the situation that Teva found itself in with carvedilol—where the label carve-out appeared adequate on its face but did not actually provide clean separation from the patented method.
Skinny Label Strategies That Still Carry Water
Despite the heightened risk environment, the skinny label remains viable as a market entry strategy. The key is precision—both in how the label is drafted and in how the company manages all other communications about the product.
The first and most basic requirement is rigorous label engineering. The carve-out cannot simply omit the approved indication’s name. It needs to remove any claim, pharmacological description, dosing recommendation, or clinical data that specifically pertains to the patented use. In GSK v. Teva, part of the court’s finding rested on the observation that Teva’s skinny label retained language describing the drug’s mechanism of action in ways that were equally applicable to the CHF indication. A stronger carve-out would have removed that language entirely, even at the cost of making the label somewhat more limited in its clinical detail.
Label engineering in this context requires a multidisciplinary team: patent counsel to identify every claim element that could be mapped to retained label language, regulatory counsel to manage the FDA approval process for the carve-out, and medical writers who can draft label text that is clinically accurate and FDA-approvable without inadvertently describing the patented method.
The second requirement is what some practitioners call “safe harbor documentation”—building a contemporaneous record showing that the generic manufacturer understood the patented indications, deliberately excluded them from the label, and deliberately excluded them from all marketing communications. This record may never be used. But if litigation arises, the difference between a company that has that documentation and one that does not can be the difference between a defensible case and a catastrophic jury verdict.
Third, generic manufacturers should consider pre-launch engagement with the FDA to clarify the scope of acceptable promotional activity for a carved-out product. While the FDA does not provide formal pre-launch promotional guidance in the way some practitioners would prefer, there is room for informal engagement with the Division of Drug Information and, in some cases, the Office of Prescription Drug Promotion [8]. Getting regulatory feedback on proposed promotional materials before launch is not foolproof protection, but it creates additional documentation of good-faith compliance.
Fourth, companies with more than one generic product in a therapeutic area need to ensure that any cross-product promotional activity does not contaminate the skinny label product. If a sales representative is calling on a cardiologist who treats CHF patients and discussing both a full-label generic and a skinny-label generic from the same manufacturer, the conversation needs to be carefully segmented.
How Branded Manufacturers Are Exploiting the New Landscape
GSK v. Teva did not just affect how generic manufacturers behave. It created new strategic opportunities for brand manufacturers, and the sophisticated ones are already using them.
The first and most direct exploitation is litigation strategy. Before GSK v. Teva, brand manufacturers faced a relatively high evidentiary bar for induced infringement claims against generic companies using Section viii statements. The assumption in the industry was that a clean carve-out was close to bulletproof. After GSK v. Teva, that assumption is gone. Brand manufacturers can now file induced infringement suits against generics using carved-out labels on the basis of marketing conduct—and they can find the evidentiary material for those suits in publicly accessible documents like press releases and investor presentations.
The second exploitation is more subtle: using the threat of induced infringement litigation to discourage generic entry even without filing suit. A brand manufacturer that sends a detailed demand letter citing GSK v. Teva and identifying specific marketing communications may achieve its goal—deterring early generic launch—without spending litigation dollars. The cost-benefit calculation for a generic manufacturer facing a credible inducement claim has changed substantially.
Brand manufacturers are also investing more heavily in method-of-use patent prosecution. The GSK v. Teva outcome has demonstrated that method-of-use patents, properly enforced, can block skinny-label generic entry even after compound patents expire. This has prompted increased filing of continuation applications claiming new therapeutic methods for existing drugs, and it has prompted renewed attention to listing strategy—ensuring that the most commercially significant method-of-use patents are properly listed in the Orange Book in ways that create enforceable rights.
Citizen petitions are another tool that brand manufacturers have used to slow generic entry, and the post-GSK v. Teva environment makes them more potent. A citizen petition challenging the adequacy of a proposed carve-out can delay ANDA approval while simultaneously generating public records that could be used in subsequent litigation to argue that the generic’s carve-out was incomplete.
Finally, some brand manufacturers are using real-world evidence programs as patent enforcement intelligence. By sponsoring registries and outcomes studies that track prescribing patterns for their drugs, they generate data on how physicians actually use the product—data that, in the right circumstances, could be used to demonstrate that generic prescriptions are systematically fulfilling patented uses.
The FDA’s Conflicted Position
The FDA finds itself in an uncomfortable position in the post-GSK v. Teva landscape. The agency created the Section viii carve-out mechanism as a tool to facilitate generic entry, consistent with its statutory mandate to protect the public health and promote access to affordable medicines. Now that mechanism has been partially destabilized by a court ruling that the FDA had no role in and over which it has no direct authority.
The agency’s regulatory guidance on labeling carve-outs predates GSK v. Teva and was not designed with the inducement liability risk in mind. FDA regulations require that a generic drug’s labeling be the same as the reference listed drug’s labeling—with the exception of approved carve-outs under Section viii [9]. The agency reviews and approves carve-out language as part of the ANDA approval process. But the agency has never stated, and arguably cannot state, that its approval of a carved-out label protects the generic manufacturer from patent infringement claims.
This gap between FDA approval and patent liability creates uncertainty at a structural level. A generic manufacturer can receive FDA approval for a carefully drafted carved-out label and then find itself defending an induced infringement suit based on evidence that has nothing to do with the label—a press release, a website page, a sales call log.
The FDA has considered revising its carve-out guidance in light of GSK v. Teva. In 2023, the agency issued a draft guidance document on labeling carve-outs that, among other things, clarified when mechanism-of-action language and clinical pharmacology data can be retained in a carved-out label without implicating the patented use [10]. That guidance was not finalized as of the time of writing, and it does not resolve the induced infringement question because the FDA’s authority extends to labeling, not to marketing conduct.
There is also a deeper tension in the FDA’s position. The agency has historically resisted becoming an arbiter of patent disputes, viewing that as a function of the courts. But GSK v. Teva has created circumstances where the FDA’s labeling decisions—specifically, what language it approves in a carve-out—can have direct patent liability implications. Some practitioners have argued that the FDA should take a more active role in reviewing carve-out adequacy with patent liability in mind. Others argue that this would turn the FDA into a patent office and is constitutionally inappropriate.
One area where the FDA could act without entering patent territory is in the regulation of generic drug promotional materials. Unlike brand manufacturers, generic manufacturers are subject to FDA promotional oversight for their own labeled communications, but the line between promotional materials for a full-label generic and impermissible promotion of a carved-out indication has never been formally drawn by the agency.
The FDA’s silence on this specific issue—what a generic manufacturer with a carved-out label can say in promotional contexts—creates regulatory uncertainty that compounds the legal uncertainty created by GSK v. Teva. A brand manufacturer knows that its promotional materials must be consistent with its approved label, and that off-label promotion is regulated under FDA rules. A generic manufacturer with a Section viii label is in a stranger position: it has an FDA-approved label that is deliberately incomplete, and the question of what promotional activity is consistent with that incomplete label has no formal regulatory answer.
One proposal that has emerged from practice is for the FDA to issue specific promotional guidance for Section viii products that maps permissible marketing activities to the indications covered by the carved-out label, while explicitly prohibiting promotional activities that reference therapeutic contexts consistent with the patented indication. This guidance would not constitute a patent liability opinion—the FDA is clear about its inability and unwillingness to adjudicate patent rights—but it would provide generic manufacturers with a regulatory framework for their promotional decisions that creates at least a partial safe harbor against induced infringement claims.
The FDA’s authority to issue such guidance derives from its jurisdiction over pharmaceutical promotional labeling under the Federal Food, Drug, and Cosmetic Act. Whether the agency has the appetite to enter this contested space is a separate question. FDA regulatory staff have expressed privately that they view the GSK v. Teva situation as primarily a patent law problem, not a regulatory problem—a view that has frustrated generic manufacturers who are looking to the FDA for some form of protective guidance.
The agency’s October 2023 guidance on Orange Book listings is, however, a signal that the FDA is not completely passive in this space. By clarifying which patents are properly listable in the Orange Book—and implicitly enabling challenges to improper listings—the FDA is affecting the patent landscape that generates the Section viii problem in the first place. If fewer method-of-use patents are properly listed, fewer Section viii carve-outs are needed, and the GSK v. Teva risk is correspondingly reduced.
The FDA has also been receiving citizen petitions from brand manufacturers challenging the adequacy of specific ANDA carve-outs—a pattern that has accelerated since GSK v. Teva. These petitions ask the FDA to deny or delay approval of ANDAs on the ground that the proposed label carve-out is inadequate to protect the patented indication. The FDA’s response to these petitions will shape the regulatory interpretation of what constitutes a sufficient carve-out, and generic manufacturers should monitor them closely through the FDA’s publicly available petition database. Petitions relating to drugs in a generic manufacturer’s portfolio should be treated as early warning signals of litigation risk, not just regulatory inconveniences.
Financial Modeling for Generic Entry Under Elevated Risk
The GSK v. Teva decision requires generic manufacturers to incorporate patent litigation risk into their financial models for skinny label products in ways that were not standard practice before 2020.
A traditional generic launch model for a Section viii product focused on two primary risk categories: ANDA approval risk (will the FDA approve the carve-out?) and first-to-file competition risk (are there other ANDAs for the same product?). Post-GSK v. Teva, a third category demands equal attention: induced infringement litigation risk.
Quantifying that risk requires several inputs. The first is an assessment of the strength of the method-of-use patent being carved out. Not all method-of-use patents are equally strong. Some have broad claims that cover the drug’s primary therapeutic mechanism in a way that makes any marketing activity difficult to separate from the patented use. Others have narrow claims tied to specific dosing regimens or patient populations that are far easier to avoid. DrugPatentWatch’s patent claim analysis tools can help at this stage, providing data on claim construction positions taken in prior litigation and validity challenges that may have narrowed effective claim scope.
The second input is a realistic assessment of the commercial use profile of the drug. What percentage of prescriptions for the RLD are written for the patented indication? If the answer is 80 percent or more—as it was for carvedilol and CHF—then the probability that a launching generic will effectively reach the patented use market, regardless of the label, is high. That high probability translates to a high probability that the brand manufacturer will find inducement evidence in real-world prescribing data. A lower commercial concentration of the patented indication reduces this risk, though it does not eliminate it.
The third input is the value of the first-filer exclusivity period. For Paragraph IV filers, the first generic to file a complete ANDA receives 180 days of exclusivity during which no other generic can receive final approval [11]. This exclusivity creates enormous first-mover value—sometimes hundreds of millions of dollars for blockbuster drugs. For Section viii filers, no equivalent exclusivity applies, which means the competitive advantage of early entry must be weighed against litigation risk on an undiluted basis.
A simplified risk-adjusted NPV model for a Section viii launch might look like this:
- Baseline revenue projection based on generic penetration rate in the off-patent indication market
- Revenue upside from any prescriptions for the carved-out indication that are captured anyway, discounted by probability of litigation and damages
- Litigation cost reserve: minimum $10 million to $30 million for a contested induced infringement case, potentially much higher for blockbuster drugs
- Probability of adverse verdict: a function of label quality, marketing conduct, and patent strength
- Time value adjustment for the possibility of a preliminary injunction, which could shut down sales entirely pending trial
For some drugs, this model still yields positive returns on skinny label entry. For others—particularly those where the patented use dominates the market and the brand manufacturer has a sophisticated litigation program—the model will suggest either waiting for full patent expiration or investing in a Paragraph IV challenge instead.
The discount rate applied to post-launch litigation risk needs to be higher than many generic manufacturers have historically used. Before GSK v. Teva, the induced infringement risk on a clean Section viii filing was typically modeled at very low probability—5 percent or less—based on the prevailing assumption that a properly carved-out label was close to litigation-proof. Post-GSK v. Teva, practitioners suggest that probability should be modeled at 20 to 40 percent for drugs where the patented indication represents more than 50 percent of prescriptions, and at 10 to 20 percent for drugs with more balanced indication profiles [12].
The damages exposure in an induced infringement case is also potentially catastrophic in ways that make standard litigation reserves inadequate for some products. In GSK v. Teva itself, the jury awarded GSK $235 million in lost profits damages for the period of Teva’s generic carvedilol sales during which infringement was found. That figure reflected GSK’s assertion that the infringing generic sales displaced brand sales that GSK would otherwise have made. Lost profits damages require the plaintiff to prove, among other things, that there was demand for the patented product, that there were no acceptable non-infringing alternatives, and that the plaintiff had the capacity to meet the demand [21]. In a case where the infringing generic has captured 80 percent of the market for a billion-dollar drug, the damages exposure can dwarf litigation costs.
Reasonable royalty damages are an alternative measure when lost profits are not proven with sufficient certainty. A reasonable royalty for a pharmaceutical method-of-use patent—typically calculated as a percentage of the infringer’s sales revenue attributable to the patented use—can still produce eight-figure verdicts for blockbuster drug generics. Generic manufacturers need to model damages exposure separately from litigation costs in their pre-launch financial analysis, using the brand manufacturer’s historical revenue for the patented indication as the starting point.
The first-filer exclusivity question deserves particular attention. Under the Hatch-Waxman Act, the first ANDA filer to submit a Paragraph IV certification for a given drug receives 180 days of marketing exclusivity against all other generics (with certain forfeiture conditions). This exclusivity is enormously valuable—for drugs with over $1 billion in annual sales, the 180-day period can produce hundreds of millions of dollars in above-market margins before competition arrives. There is no equivalent exclusivity for Section viii filers; multiple generic manufacturers can receive ANDA approvals with Section viii statements and compete simultaneously from day one.
This asymmetry affects the Section viii versus Paragraph IV calculus in an important way. For a drug where Paragraph IV is viable—where the method-of-use patent appears vulnerable to a validity challenge—the combination of first-filer exclusivity and freedom from inducement liability risk often makes Paragraph IV the better financial decision, even accounting for litigation costs. For a drug where the patent appears solid and validity challenge is unlikely to succeed, Section viii may still be preferable despite its elevated risk profile. The decision requires a patent-strength analysis that is honest about the probability of success in a Paragraph IV challenge—not just optimistic advocacy.
Pipeline timing matters too. If a generic manufacturer is evaluating a Section viii strategy for a drug whose compound patent expires in three years, the risk-adjusted NPV of entering now with a skinny label may compare unfavorably to the NPV of waiting for full patent expiration and entering with a complete label and no litigation tail—particularly if the induced infringement risk is high and the commercial advantage of the three-year head start is modest given the lack of Section viii exclusivity.
The decision to pursue Paragraph IV rather than Section viii is itself a financial modeling exercise. Paragraph IV litigation costs are substantial—a typical ANDA litigation case costs $3 million to $10 million per side in legal fees, and complex multi-patent cases involving expert testimony can cost significantly more [12]. But a successful Paragraph IV challenge invalidates the patent for all purposes, eliminating future risk for all generics rather than just the challenging filer. The calculus depends on the strength of the patent, the anticipated litigation timeline, and the commercial stakes.
Four Case Studies: Carvedilol, Aripiprazole, Ibuprofen, and Pregabalin
Carvedilol (GSK v. Teva): The Case That Changed Everything
The carvedilol story has been well documented by now, but the details matter for practical lessons. Teva’s carved-out label removed explicit reference to congestive heart failure. What it did not remove was language describing carvedilol as a drug that “reduces the progression of heart failure” when used in combination with other therapies. That phrase—”progression of heart failure”—was not coextensive with the CHF indication, but it was close enough that the Federal Circuit found it probative of the inadequacy of the carve-out.
The press release error was more consequential. Teva described generic carvedilol as a treatment for “heart failure” without qualification. This was, technically, accurate for the unpatented indications. But in a market where CHF was the dominant use of carvedilol, a court was willing to treat that unqualified language as evidence of intent to reach the CHF market.
The lesson: every word in every external communication about a skinny label product needs to be reviewed with patent claim language directly in hand. The review cannot be delegated to regulatory personnel alone—patent counsel must be in the room.
Aripiprazole (Bristol-Myers Squibb v. Aurobindo Pharma)
Aripiprazole, sold by Bristol-Myers Squibb under the brand name Abilify, provides a contrasting case study where the skinny label defense worked more effectively. BMS held method-of-use patents covering the use of aripiprazole as adjunctive therapy for major depressive disorder (MDD). Several generic manufacturers filed Section viii statements carving out the MDD indication and received FDA approval to market aripiprazole for the two remaining unpatented indications: schizophrenia and bipolar disorder.
The subsequent litigation produced mixed results at the district court level, with some generic defendants succeeding in establishing that their promotional materials were confined to the unpatented indications and that real-world prescribing patterns showed a substantial market for the carve-out indications [13]. The aripiprazole cases were not resolved by the Federal Circuit in a single definitive ruling, and the outcomes varied based on the specific facts of each generic defendant’s conduct.
What the aripiprazole litigation illustrated is that skinny label defenses are stronger when the off-patent indications represent a significant commercial market in their own right—not merely a marginal fraction of total prescriptions. When a generic can demonstrate that it has a legitimate, commercially substantial customer base for the unpatented use, and that its marketing is genuinely directed at that customer base, the inducement claim becomes harder to sustain.
Ibuprofen OTC: The Consumer Complexity Layer
Ibuprofen’s patent history is largely historical—the key compound patents expired decades ago—but the drug serves as a useful hypothetical for understanding how the skinny label doctrine would apply to a consumer health product where the manufacturer exercises less control over final use.
Consider a hypothetical scenario where a pharmaceutical company develops and patents a specific method of using ibuprofen for a new indication—say, a validated reduction in the risk of a specific inflammatory condition at a particular dosing regimen. If the compound remains off-patent but the method is newly patented, any generic manufacturer offering ibuprofen at that dosing regimen faces the same inducement risk as in GSK v. Teva, compounded by the fact that consumer-facing marketing for OTC drugs often describes therapeutic uses more broadly and with less clinical precision than prescription drug labeling.
This hypothetical is not far-fetched. The FDA has approved OTC ibuprofen for a standard set of indications, and method-of-use research continues in the anti-inflammatory space. The point is that the GSK v. Teva liability framework applies across the product spectrum, and consumer health companies need to be thinking about it as much as prescription drug manufacturers.
Pregabalin (Warner-Lambert v. Generics UK)
The UK litigation over pregabalin (Lyrica) provides the most instructive international comparison point and deserves extended treatment in the section on international comparisons below. But the UK Supreme Court’s 2018 decision in Warner-Lambert Company LLC v. Generics (UK) Ltd deserves mention here as a parallel development that reached similar conclusions about skinny label inadequacy through a different legal framework.
The UK Supreme Court held, in a divided ruling, that a generic manufacturer’s skinny label for pregabalin—carving out the patented neuropathic pain indication while retaining approval for epilepsy and generalized anxiety disorder—did not insulate the generic from infringement liability if the actual dispensing of prescriptions resulted in patients receiving the drug for the patented use [14]. The UK court’s approach focused on the supply of the patented product rather than the intent of the supplier—a different doctrinal route than the U.S. induced infringement approach, but one that reached the same practical conclusion: a carved-out label is not a safe harbor in itself.
Litigation Risk Management: The Pre-Trial Toolkit
Between the ANDA filing and a courtroom verdict lies a litigation phase that can last years and cost tens of millions of dollars. Generic manufacturers who enter this phase without a well-developed defensive strategy face both financial and reputational exposure that can affect their broader business.
The first element of a defensive litigation strategy is claim construction. In a patent infringement case, claim construction—the court’s interpretation of what the patent claims actually cover—is determined by the judge, not the jury, typically at a “Markman hearing” early in the litigation. A favorable claim construction can dramatically narrow the scope of the asserted patent, potentially to the point where the generic’s carve-out is clearly adequate. An unfavorable construction can expand the patent’s scope to cover uses that the generic manufacturer believed were safely excluded from its label.
Investing in claim construction analysis before litigation begins—including commissioning a detailed prosecution history analysis and developing claim construction positions that support the adequacy of the carve-out—is a valuable pre-litigation exercise. If litigation is filed, this pre-existing analysis provides a head start. If litigation is settled or never filed, the analysis still contributes to the label engineering process.
The invalidity defense is also worth developing early. Even in a Section viii case, where the generic manufacturer has not filed a Paragraph IV certification contesting the patent’s validity, the defendant can raise invalidity as a defense in an induced infringement suit. A credible invalidity defense serves two functions: it provides a path to a full defense verdict that eliminates the patent for all purposes, and it provides negotiating leverage in settlement discussions. A patent claim that appears vulnerable to a prior art attack is worth less to the brand manufacturer in litigation than one with a clean validity record.
Expert witness preparation is a third pre-trial priority. An induced infringement case involving a pharmaceutical skinny label will require expert testimony on multiple issues: the scope of the patent claims, the adequacy of the label carve-out, the interpretation of marketing communications, the real-world prescribing patterns for the drug, and the damages calculation. Each of these expert roles requires a witness with specific credentials and a deep familiarity with the relevant technical and commercial evidence. The difference between a well-prepared expert who can explain claim construction to a jury in accessible terms and an expert who cannot communicate effectively is often the difference between winning and losing.
Generic manufacturers defending induced infringement cases have also increasingly relied on evidence of the FDA’s role in approving the carve-out as a counter-narrative. While the FDA’s approval does not constitute a legal defense, it is powerful context for a jury to understand that the generic manufacturer’s label was reviewed by a federal agency with public health expertise and found acceptable. Framing the generic manufacturer’s Section viii filing as a legally sanctioned mechanism—not a maneuver to evade responsibility—is an important element of the defensive narrative.
Finally, preliminary injunctions are a critical tactical consideration. A brand manufacturer seeking to stop an infringing generic launch will often file a motion for a preliminary injunction at the outset of litigation, seeking to halt sales while the case is pending. Under the standard four-factor test, the brand manufacturer must show a likelihood of success on the merits, a likelihood of irreparable harm, a balance of hardships favoring an injunction, and consistency with the public interest [22]. Post-GSK v. Teva, brand manufacturers are more likely to argue that they can satisfy the likelihood of success factor based on evidence of inadequate carve-outs. Generic manufacturers need to be prepared to oppose preliminary injunction motions at the earliest stages of litigation, with a factual record that demonstrates both the adequacy of the carve-out and the public interest in generic access to the drug.
International Comparisons: How the EU and Canada Handle This
The U.S. is not the only jurisdiction where the skinny label tension has played out in courts. The experiences of the European Union, Canada, and Japan offer both contrasts and lessons for U.S. practitioners.
The European Union
The EU does not have a single pharmaceutical patent litigation system—patent enforcement is handled at the national level, with harmonized rules under the European Patent Convention and eventually the Unified Patent Court (UPC), which became operational in June 2023. The EU’s equivalent of the skinny label is called the “limited indication carve-out” or “skinny label” in common usage.
The Warner-Lambert pregabalin litigation, which progressed through the courts of England and Wales, Germany, France, and other EU member states, produced notably different outcomes across jurisdictions. German courts were generally more receptive to method-of-use patent enforcement against carve-out generics. French and Italian courts were more protective of generic entry [15]. The UK Supreme Court’s decision took the most restrictive approach, finding that generic manufacturers could bear responsibility for infringement if they were aware that prescriptions were likely to result in dispensing for the patented use—even if the label itself did not describe that use.
The UPC has now created a venue where method-of-use patent holders can seek injunctive relief across multiple EU member states simultaneously, which has substantially increased the stakes of skinny label litigation in Europe. A single adverse ruling in the UPC can effectively shut down generic sales in major EU markets simultaneously—an outcome that was not possible under the fragmented pre-UPC enforcement landscape.
For EU practitioners, the most immediately practical lesson from the international experience is that the UPC’s “essential part of the invention” doctrine for claim construction may be more patent-holder-friendly than the claim-by-claim approach traditionally applied in common law jurisdictions. Generic manufacturers filing for European skinny label approvals need to factor this into their patent scope analysis.
Canada
Canada’s ANDA-equivalent, the Abbreviated New Drug Submission (ANDS) process, includes a Notice of Compliance (NOC) linkage system that operates somewhat like the Hatch-Waxman Orange Book system but with notable differences. In Canada, brand manufacturers list patents on a Patent Register, and generic manufacturers must address each listed patent in their submissions.
The Canadian equivalent of the Section viii statement allows generic manufacturers to carve out patented uses from their proposed labeling. Canadian courts have generally been receptive to the view that a proper carve-out provides meaningful protection from infringement claims—a more favorable posture than the current U.S. federal circuit approach. However, several recent decisions have suggested that Canadian courts are moving toward a more nuanced analysis similar to the U.S. induced infringement framework [16].
The Canadian health ministry’s regulatory guidance on carve-outs remains clearer than the FDA’s current guidance, and some U.S. practitioners have used the Canadian framework as a model for what cleaner regulatory guidance in the U.S. might look like.
Japan
Japan’s pharmaceutical patent system uses a different pre-marketing clearance mechanism that makes direct comparison with the U.S. skinny label framework difficult. Japanese law requires generic manufacturers to demonstrate that their products will not be used for any patented therapeutic method before receiving marketing authorization—which is a more demanding standard than the U.S. carve-out approach but also one that arguably provides clearer pre-market certainty. The Japanese experience is less relevant to U.S. practitioners on a direct doctrinal basis, but it illustrates that different regulatory designs can produce different risk profiles for the same underlying patent tension.
The Role of Pharmacy Dispensing and Payer Behavior
One of the most underappreciated complications in the skinny label context is the role of pharmacists and payers. These actors sit between the prescribing physician and the patient, and their behavior can significantly affect whether a carved-out generic effectively reaches the patented use market.
When a physician prescribes “carvedilol” without specifying a brand, the pharmacist has broad authority under most state laws to dispense a therapeutically equivalent generic. The question of which generic is dispensed—one with a full label or one with a carved-out label—is determined by pharmacy formulary decisions, purchasing agreements with wholesalers, and the availability of generic stock on the shelf. In practice, pharmacists often do not know whether the product they are dispensing carries a full label or a carved-out label, and they have no obligation to track this.
This means that a physician prescribing carvedilol for CHF may—without anyone’s knowledge or intent—receive the carved-out generic rather than the brand or a full-label generic. From the court’s perspective in GSK v. Teva, this kind of indirect market reach is part of what makes the generic manufacturer liable: by being in the supply chain for carvedilol prescriptions in a CHF-dominant market, the generic manufacturer is effectively participating in the administration of the drug for the patented use.
The response to this structural reality—which generic manufacturers cannot fully control once their products enter the distribution chain—has been twofold. Some companies have attempted to implement distribution controls, limiting their products to channels that serve primarily the unpatented use markets. This is commercially impractical for most drugs but has been attempted for certain specialty products. Others have focused on payer engagement, working with pharmacy benefit managers (PBMs) and health plans to ensure that their carved-out products are explicitly designated for the unpatented indications in formulary management systems.
Neither approach provides complete protection. But both approaches provide additional documentation of good-faith efforts to limit the product’s reach to unpatented uses—documentation that would be valuable in subsequent litigation.
Payer behavior also affects generic manufacturers’ revenue projections. If a PBM or health plan specifies that a carved-out generic is preferred only for the unpatented indications, and that the brand or a full-label generic is preferred for the patented indication, the generic manufacturer’s realistic market share estimate needs to reflect that segmentation. This is not just a legal planning issue—it is a commercial planning issue, and the two should be considered together rather than in separate department silos.
Legislative and Regulatory Reform Proposals
The GSK v. Teva decision has generated significant academic and policy commentary, and at least some of it has begun to find its way into legislative and regulatory proposals. Whether any of these proposals will result in enacted law or binding regulation is uncertain, but generic manufacturers should monitor them closely because they affect the long-term strategic landscape.
Congressional Proposals
Several members of Congress have expressed concern that GSK v. Teva effectively undermines the Hatch-Waxman compromise by making skinny labels legally treacherous. The Affordable Prescriptions for Patients Act, introduced in various forms over recent legislative sessions, included provisions that would have clarified the evidentiary standard for induced infringement in the pharmaceutical context—specifically, addressing whether evidence of prescribing patterns alone, without affirmative marketing acts, should be sufficient to establish inducement [17].
None of these proposals has been enacted, and the political dynamics of pharmaceutical patent reform make near-term legislative action unlikely. But the debate has produced useful analytical clarity on the specific legal fixes that would be necessary to restore the Section viii safe harbor to its pre-GSK v. Teva function.
A more targeted legislative fix—recommended by several academic commentators—would amend 35 U.S.C. § 271 to explicitly provide that a generic manufacturer’s compliance with a Section viii carve-out constitutes a rebuttable safe harbor against induced infringement claims, rebuttable only by direct evidence of promotion specifically targeting the patented use. This would not eliminate all litigation risk, but it would rebalance the evidentiary standard.
FTC and DOJ Perspectives
The Federal Trade Commission has historically taken the position that overly broad method-of-use patent enforcement by brand manufacturers harms generic competition and ultimately harms consumers. The Commission’s past enforcement actions against reverse payment settlements (“pay-for-delay” deals) reflect this orientation [18]. The GSK v. Teva decision complicates the FTC’s position because the liability that the case imposes on generic manufacturers comes not from a brand-generic settlement but from a court ruling about what generic manufacturers can say and do in the market.
The DOJ Antitrust Division, which shares jurisdiction over pharmaceutical competition issues with the FTC, has been less active on skinny label issues specifically, but its broader concern about pharmaceutical market concentration creates a natural alignment with any policy reform that would make generic entry easier.
Neither agency can directly modify the legal standard established by GSK v. Teva. But both can contribute to the policy record through amicus briefs in subsequent cases, through formal comments on FDA guidance documents, and through enforcement actions against brand manufacturers whose post-GSK v. Teva conduct—particularly aggressive litigation campaigns against generics with legitimate carve-outs—crosses the line into anticompetitive behavior.
Orange Book Reform
A parallel reform effort focuses on the Orange Book itself. Critics of the current system argue that brand manufacturers have used Orange Book listings—particularly for method-of-use patents—as a mechanism for unjustified delay of generic entry, listing patents with claims that do not meet the statutory requirement for Orange Book eligibility and then using the automatic 30-month stay triggered by Paragraph IV litigation to prevent generic launches for years.
The FDA’s October 2023 guidance on Orange Book patent listing reinvigorated this discussion [19]. The guidance clarified the FDA’s view that certain device-related patents and certain method-of-use patents with minimal correspondence to the listed drug’s approved labeling are not properly listable in the Orange Book. The FTC has followed up with enforcement actions against brand manufacturers for allegedly improper Orange Book listings. While these developments do not directly address GSK v. Teva liability, they affect the patent landscape that Section viii filers must navigate—if improper listings are removed from the Orange Book, the number of patents that generic manufacturers need to address through Paragraph IV challenges or Section viii statements will decrease.
The FTC’s Orange Book delisting campaign has been one of the more aggressive actions the agency has taken against branded pharmaceutical manufacturers in recent years. Between 2023 and 2024, the FTC identified dozens of patents it believed were improperly listed in the Orange Book for dozens of drugs, and sent formal challenge letters to brand manufacturers demanding withdrawal of those listings. Several manufacturers withdrew challenged patents rather than defend them; others refused and faced the prospect of formal enforcement action [23].
From the generic manufacturer’s perspective, this FTC campaign creates an opportunity. If a method-of-use patent that would otherwise require either a Section viii carve-out or a Paragraph IV challenge is removed from the Orange Book through an FTC challenge or voluntary withdrawal, the generic filer may be able to proceed without addressing that patent in its ANDA at all. Monitoring FTC Orange Book enforcement actions and aligning ANDA filing strategy with the evolution of Orange Book listings is therefore a component of smart pipeline management.
The broader patent reform debate also touches on inter partes review (IPR) proceedings, which have been used by generic manufacturers to challenge the validity of Orange Book patents outside the ANDA litigation context. The Supreme Court’s decision in Arthrex, Inc. v. Smith & Nephew, Inc. (2021) addressed the constitutionality of the PTAB’s structure, and ongoing congressional discussions about potential modifications to the America Invents Act could affect the availability and scope of IPR as a tool for pharmaceutical patent challenges. Generic manufacturers should track these developments through their government affairs functions and through resources like DrugPatentWatch, which provides alerts on IPR filings and decisions affecting drugs in its tracked database.
The intersection of patent reform and drug pricing policy also creates a political dynamic that generic manufacturers can leverage. Members of Congress who are concerned about high drug prices are generally supportive of measures that facilitate generic entry, including measures that would clarify the evidentiary standard for induced infringement. In testimony before relevant committees, generic industry trade associations including the Association for Accessible Medicines have argued that the current GSK v. Teva standard effectively taxes generic companies for entering multi-indication drug markets—a tax that is ultimately paid by patients in the form of delayed access to affordable drugs. Making this case to receptive legislators, with specific examples and economic data, can contribute to the policy environment that eventually produces legislative relief.
Strategic Recommendations for Generic Manufacturers
The strategic response to GSK v. Teva cannot be reduced to a single policy or a single legal maneuver. It requires a cross-functional transformation of how generic manufacturers think about multi-indication drugs from the earliest stages of ANDA development through post-launch commercial operations.
Build a Patent Intelligence Infrastructure
The foundation of any defensible skinny label strategy is patent intelligence. Generic manufacturers need to establish systematic processes for mapping the full patent landscape of every multi-indication drug they are evaluating, well before ANDA filing. This means going beyond the Orange Book to include continuation applications, divisional applications, and provisionally pending applications that may issue before or after launch.
DrugPatentWatch provides a robust starting point for this analysis, offering aggregated data on Orange Book listings, ANDA filings, patent challenge histories, and litigation outcomes that would take months to compile from primary sources. The platform’s ability to identify which patents have survived prior validity challenges, which have been limited in claim construction by prior court rulings, and which are currently the subject of IPR proceedings at the Patent Trial and Appeal Board allows generic manufacturers to make more informed risk assessments early in the pipeline evaluation process.
The intelligence function cannot be a one-time pre-filing exercise. It needs to be continuous. Brand manufacturers file new method-of-use continuation applications constantly, and a patent that did not exist at the time of ANDA filing may issue before or after launch. Ongoing patent monitoring, with automatic alerts for new Orange Book listings for RLDs in the generic portfolio, is now standard practice at well-managed generic companies.
Integrate Patent Counsel into Label Drafting
Label drafting for a Section viii product is not a regulatory function alone. It requires active involvement of patent counsel at every stage—from the initial identification of the carve-out scope to the final review of every sentence of the proposed label.
The specific review protocol should require that for every claim element of every method-of-use patent being carved out, patent counsel analyze whether any retained label language, directly or indirectly, describes that claim element. If it does, the label needs to be revised. This review needs to include not just the indication section but also the clinical pharmacology section, the dosage and administration section, the clinical studies section, and any black box warning language that might describe the drug’s mechanism in terms that overlap with the patented use.
Where the revision would make the label misleading or inadequate for the unpatented indications, the team needs to engage with the FDA to discuss acceptable alternatives. This conversation with the FDA should be documented in detail.
Establish a Pre-Launch Communication Review Process
Every external communication about a Section viii product—press releases, investor relations materials, website content, medical education programs, promotional pieces, earnings call scripts—needs to be reviewed by patent counsel before publication or delivery. The review should apply a specific analytical question: does this communication, read by a prescribing physician in the therapeutic context of this drug, suggest or imply use of the product for the carved-out indication?
This review protocol needs to be codified in a written policy, with clear roles and responsibilities and a documented approval workflow. If a communication reaches the public without patent counsel review, the gap itself becomes potential litigation evidence.
The protocol needs to extend to social media and any interactive digital content. In an environment where pharmaceutical companies engage physicians through digital platforms, the risk that an algorithm-served piece of content about the drug reaches physicians treating the carved-out indication is real.
Consider Paragraph IV Where the Risk-Reward Justifies It
For drugs where the patented use is the dominant commercial use and the patent appears to have weaknesses—overly broad claims, prior art issues, possible lack of written description support—a Paragraph IV challenge may offer better risk-adjusted returns than a Section viii strategy, despite its higher upfront cost.
Paragraph IV litigation provides several advantages in the post-GSK v. Teva environment. First, it eliminates the induced infringement risk entirely if the patent is invalidated. Second, it preserves 180-day exclusivity if the challenger is the first ANDA filer. Third, it creates a clear binary outcome—either the patent survives or it does not—whereas the Section viii strategy creates a probabilistic liability tail that is difficult to quantify and manage.
The decision to pursue Paragraph IV rather than Section viii should be driven by patent analysis—specifically, a detailed assessment of the patent’s claim strength, prosecution history, and vulnerability to prior art challenges. Companies that have invested in strong patent prosecution teams and experienced ANDA litigators are better positioned to make this call accurately.
Use Real-World Evidence Proactively
In the event of litigation, the generic manufacturer’s ability to demonstrate that its carved-out product was prescribed primarily for unpatented indications will be critical. The company should be collecting and preserving this evidence from day one of launch.
Market research data, prescription database analyses (from sources like IQVIA or Symphony Health), physician specialty distribution data, and pharmacy formulary placement records all speak to the question of whether the generic is actually reaching the patented use market. If the data shows, for example, that 90 percent of prescriptions for the generic are written by cardiologists treating hypertension rather than CHF specialists, that evidence is powerful in defending against an inducement claim.
This data collection needs to be framed carefully. It is not a litigation preparation exercise—it is a business intelligence function that also happens to produce valuable evidence. The framing matters because of attorney-client privilege issues: if the data collection program is set up by in-house litigation counsel specifically in preparation for anticipated litigation, it may be discoverable by opposing parties in a different way than a business analytics program that serves commercial functions as well.
Use Real-World Evidence Proactively
In the event of litigation, the generic manufacturer’s ability to demonstrate that its carved-out product was prescribed primarily for unpatented indications will be critical. The company should be collecting and preserving this evidence from day one of launch.
Market research data, prescription database analyses (from sources like IQVIA or Symphony Health), physician specialty distribution data, and pharmacy formulary placement records all speak to the question of whether the generic is actually reaching the patented use market. If the data shows, for example, that 90 percent of prescriptions for the generic are written by cardiologists treating hypertension rather than CHF specialists, that evidence is powerful in defending against an inducement claim.
This data collection needs to be framed carefully. It is not a litigation preparation exercise—it is a business intelligence function that also happens to produce valuable evidence. The framing matters because of attorney-client privilege issues: if the data collection program is set up by in-house litigation counsel specifically in preparation for anticipated litigation, it may be discoverable by opposing parties in a different way than a business analytics program that serves commercial functions as well.
Manage the Intermediate Trade Network
A frequently overlooked risk vector is the network of wholesalers, distributors, and specialty pharmacies that sit between the generic manufacturer and the prescribing physician. These entities often maintain their own communications about the drugs they distribute—product catalogs, therapeutic area guides, formulary placement recommendations—that may describe the drug in ways that implicate the carved-out indication.
A generic manufacturer has limited control over what its distribution partners say about its products. But it can take several steps to mitigate the risk. Distribution agreements should include representations from distributors that they will not promote the generic product for uses outside the approved label. Internal audits of distributor communications—particularly for specialty pharmacy channels that serve the therapeutic area where the patented indication resides—should be part of the post-launch compliance program. Where a distributor’s marketing materials reference the patented indication in connection with the carved-out generic, the manufacturer should document its corrective action promptly.
This issue becomes particularly acute in the specialty drug context, where manufacturer-sponsored patient support programs, copay assistance programs, and hub services often involve downstream communications with patients and caregivers about their conditions. A patient support program for a cardiometabolic drug that describes the program as supporting “heart failure patients” when the generic’s label does not cover heart failure creates exactly the kind of evidence that GSK v. Teva indicates can support an inducement finding.
Develop a Post-Launch Monitoring Protocol
The regulatory and commercial environment around a skinny label product continues to evolve after launch, and generic manufacturers need systematic processes for monitoring changes that affect their liability exposure.
Key triggers for immediate reassessment include: new Orange Book patent listings for the RLD after the generic has launched; brand manufacturer communications describing new indications or updated labeling that could affect the scope of the carve-out; changes in real-world prescribing patterns that suggest a shift in the use profile of the drug; and any demand letter, patent suit, or regulatory action from the brand manufacturer against any generic company with a Section viii label for the same drug.
The last trigger—a lawsuit against another generic—is often the first public signal that a brand manufacturer is actively enforcing its method-of-use patent in the skinny label context. If Company A is sued for induced infringement of a carvedilol-type patent, and Company B has a similar skinny label product on the market, Company B should immediately conduct a gap analysis comparing its label language and marketing materials to those that the brand manufacturer has identified as problematic in the Company A litigation. It may be able to distinguish its position; it may need to make changes. Either way, waiting to receive its own demand letter is the wrong approach.
Coordinate U.S. and International Strategy
For generic manufacturers with global operations, the skinny label strategy for a given drug should be developed with coordination across U.S., EU, and Canadian legal and regulatory teams from the outset—not as a series of independent national decisions. A label carve-out that works in the U.S. context may not be adequate to protect against infringement liability under the UPC’s more patent-holder-friendly framework, and vice versa. A single global promotional campaign that avoids problematic language under U.S. induced infringement law may still create problems in jurisdictions that apply a strict liability framework for method-of-use patent infringement.
This coordination is particularly important for launch timing. In some cases, a sequential launch strategy—entering the U.S. market first, observing the brand manufacturer’s response, and then deciding on European launch timing based on the U.S. litigation environment—is commercially feasible and risk-reducing. In others, simultaneous global launch is required to capture first-mover advantage before competitors can enter. Where simultaneous launch is planned, the global legal review needs to be completed before any market entry occurs, not after.
Maintain Ongoing Regulatory Engagement
The regulatory environment around skinny labels is evolving. FDA guidance, Orange Book reform proceedings, and potential legislative changes all affect the risk profile of Section viii strategies. Generic manufacturers should have a dedicated function—whether internal government affairs staff or outside regulatory counsel—monitoring these developments and translating them into strategic guidance for the pipeline and commercial teams.
Engagement with the FDA during rulemaking proceedings is particularly valuable. The FDA’s draft guidance on labeling carve-outs invited public comment, and companies that submitted detailed, technically sophisticated comments were able to shape the agency’s understanding of the practical implications of different approaches. The FDA’s final guidance on this topic, when issued, will affect the ground rules for Section viii filings going forward, and companies that have engaged substantively in the process will be better positioned to respond to the final rule.
The PTAB also deserves attention in this context. Inter partes review proceedings at the Patent Trial and Appeal Board offer a relatively fast and cost-effective mechanism for challenging the validity of Orange Book-listed method-of-use patents—faster and cheaper than district court Paragraph IV litigation, though with limited discovery and no damages remedy. A generic manufacturer who identifies a weak method-of-use patent as the primary obstacle to a full-label generic can file an IPR petition to challenge the patent’s validity while simultaneously pursuing a Section viii ANDA—a parallel strategy that preserves both market entry routes while the IPR proceeds. If the IPR succeeds, the Section viii carve-out becomes irrelevant; if it fails, the company still has the carved-out approval in hand.
This dual-track strategy has become more common post-GSK v. Teva, and IPR petition filings against Orange Book-listed method-of-use patents have increased noticeably since 2021. The PTAB’s institution rate for IPR petitions against pharmaceutical method-of-use patents has historically been somewhat lower than for other technology areas—likely reflecting the deference that PTAB panels give to the clinical development process that generates the patentable invention—but successful IPR proceedings have invalidated significant pharmaceutical method-of-use patents and in some cases opened entire drug markets to full-label generic competition years ahead of the expected patent expiration date.
<blockquote> “Generic drugs account for approximately 90 percent of all prescriptions dispensed in the United States but only about 20 percent of total drug spending, representing savings to the U.S. health system of well over $300 billion annually.” — Association for Accessible Medicines, *2023 U.S. Generic & Biosimilar Medicines Savings Report* [20] </blockquote>
The economic stakes of getting the skinny label right could not be higher. If the GSK v. Teva liability framework effectively deters skinny label entry for a significant category of multi-indication drugs, the resulting delay in generic competition will impose costs that dwarf any litigation exposure absorbed by individual generic manufacturers. The policy question—whether the current legal standard is calibrated correctly—is not an abstract one. It has direct consequences for drug affordability.
The generic industry’s long-term response to GSK v. Teva will also shape how brand manufacturers invest in method-of-use patent prosecution going forward. If skinny label entry is sufficiently difficult that brand manufacturers can effectively extend exclusivity on major drugs through method-of-use patents alone, the incentive to invest in filing and defending those patents grows considerably. The result could be a race between increasingly aggressive method-of-use patent prosecution by brand manufacturers and increasingly sophisticated carve-out and Paragraph IV strategies by generic manufacturers—a race that raises costs for both sides and ultimately for the health system that absorbs the resulting delays in generic competition.
Getting ahead of this dynamic—by building the patent intelligence infrastructure, the cross-functional legal-regulatory-commercial processes, and the post-launch compliance programs described in this article—is the only rational response for a generic manufacturer that intends to compete seriously in the multi-indication drug market. The companies that treat GSK v. Teva as a compliance box to check rather than a strategic signal to respond to will find themselves at a growing disadvantage in the years ahead.
Key Takeaways
- GSK v. Teva established that a properly carved-out label is not a complete defense to induced infringement. External marketing communications—press releases, promotional materials, sales force messaging—are admissible and can be determinative evidence of inducement intent.
- The intent element of induced infringement can now be established through circumstantial evidence, including the scale of real-world prescribing for the patented indication and the demographic reach of the generic manufacturer’s marketing.
- Label engineering for a Section viii product requires active involvement of patent counsel, not just regulatory writers. Every sentence of the retained label must be analyzed against the claims of the carved-out patent to ensure no indirect description of the patented method survives in the approved label.
- A pre-launch communication review protocol—covering every external publication about the product—is now a legal necessity, not an optional compliance measure.
- For drugs where the patented indication dominates commercial use, a Paragraph IV challenge may offer better risk-adjusted returns than a Section viii strategy, despite higher upfront costs.
- Patent intelligence tools like DrugPatentWatch provide the foundational data needed to map method-of-use patent landscapes, track litigation histories, and assess patent strength before ANDA filing decisions are made.
- Pharmacy dispensing behavior and payer formulary decisions affect whether a carved-out generic effectively reaches the patented use market. These downstream factors create liability exposure that generic manufacturers cannot fully control, but can mitigate through documented good-faith measures.
- The FDA’s draft guidance on carve-outs, Orange Book reform proceedings, and potential congressional action all create a shifting regulatory environment that must be monitored continuously.
- Post-launch real-world evidence collection—tracking prescription patterns by physician specialty, indication, and payer—is both commercially valuable and potentially critical litigation evidence.
- The skinny label remains viable as a market entry strategy. The post-GSK v. Teva environment requires more rigorous execution, not abandonment of the strategy.
FAQ
Q1: Does GSK v. Teva mean that Section viii filings are no longer worth pursuing?
Not categorically, no. The ruling elevated the risk and complexity of skinny label strategies, but it did not eliminate the mechanism’s utility. The decision matters most for drugs where the patented indication dominates commercial use—specifically, where a court could find that a generic’s market presence in a CHF-like dominant indication context constitutes effective promotion regardless of label content. For drugs where the off-patent indications represent a robust commercial market, where the patented use is a secondary application, or where the method-of-use patent has meaningful weaknesses, Section viii remains a viable and often superior market entry route. The key is to approach it as a sophisticated legal-regulatory-commercial exercise rather than a routine ANDA filing variant.
Q2: How should a generic manufacturer handle investor relations communications about a skinny label product without creating liability exposure?
Investor communications are among the most dangerous documents in a Section viii induced infringement case because they are widely distributed, publicly accessible, and frequently describe a product’s commercial opportunity in terms of the total market for the drug—which includes the patented indication. The standard investor communications practice of describing a drug’s “addressable market” by reference to total prescriptions or total disease prevalence directly implicates the problem that the Federal Circuit identified in GSK v. Teva. IR communications about a skinny label product should be reviewed by patent counsel before publication, should reference only the indications covered by the generic’s label when discussing commercial opportunity, and should explicitly disclaim any commercial targeting of the carved-out indication. These disclosures may affect how investors view the commercial potential of the product, but they are substantially less costly than an adverse verdict.
Q3: Can a generic manufacturer obtain any formal pre-approval assurance from the FDA that its proposed carve-out and marketing plan will not create induced infringement exposure?
No formal safe harbor of this kind exists in the current regulatory framework. The FDA reviews carve-out language as part of the ANDA approval process, but its approval constitutes only a determination that the label is approvable under the ANDA regulations—it is not a representation about patent liability. Informal engagement with the FDA’s Office of Prescription Drug Promotion regarding post-approval promotional materials can produce guidance about what promotional content is acceptable under FDA regulations, but OPDP’s analysis is also not coextensive with the patent liability analysis under 35 U.S.C. § 271(b). A formal petition for an FDA determination that a proposed carve-out is adequate—sometimes called a “Section viii petition”—asks the FDA to make a regulatory call about label adequacy, not a patent law call. The agency’s response to such a petition may be useful as supporting evidence in litigation, but it does not bind any court.
Q4: What should a generic manufacturer do if it discovers, after launch, that its product is being widely prescribed for the carved-out indication?
This is one of the most practically difficult situations the GSK v. Teva ruling creates. If a post-launch prescribing analysis shows that the generic is capturing significant market share in the patented indication, the manufacturer faces a difficult choice between continuing to sell a potentially infringing product, withdrawing from the market entirely, and attempting to implement controls to limit further distribution to the off-patent indications. None of these options is clean. Withdrawal is commercially costly and operationally complex. Distribution controls may be insufficient if the product is already in wide distribution. Continued sales without action creates a record of knowledge of infringement, which makes subsequent inducement claims easier to prove. The appropriate response depends heavily on the specific facts, including the scope of the infringing use, the strength of the patent, and the litigation posture of the brand manufacturer. The one universal prescription: engage outside patent litigation counsel immediately, before taking any action that could be characterized as either continuation of infringement or an admission of liability.
Q5: How does the Unified Patent Court in Europe affect U.S. generic manufacturers with EU commercial operations?
The UPC, operational since June 2023, allows patent holders to seek injunctive relief and damages for patent infringement across all participating EU member states in a single proceeding. For method-of-use patents with European counterparts to U.S. Orange Book-listed patents, this means that a single adverse ruling can simultaneously block generic sales in Germany, France, Italy, the Netherlands, and other major EU markets. Before the UPC, a brand manufacturer seeking pan-European relief against a carved-out generic had to litigate in each national court separately—a costly and uncertain exercise that many brands declined. Now, the threshold for pan-European injunctive enforcement is much lower. U.S. generic manufacturers with EU operations should treat their European skinny label strategies and their U.S. strategies as part of an integrated global risk analysis rather than as separate regional legal questions. A carve-out that is adequate under U.S. law and FDA guidance may still create UPC liability exposure if the EU patent claims are drafted differently from the U.S. patent claims—a scenario that requires coordinated analysis by U.S. and European patent counsel working together from the outset.
References
[1] H.R. Rep. No. 98-857, pt. 1 (1984). Drug Price Competition and Patent Term Restoration Act: Legislative History. U.S. House of Representatives.
[2] GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc., 7 F.4th 1320 (Fed. Cir. 2021), cert. denied, 143 S. Ct. 85 (2022).
[3] Coletti, R. J., & Kessler, L. M. (2022). GSK v. Teva and induced infringement: What the Federal Circuit’s ruling means for generic drug manufacturers. Journal of Generic Medicines, 18(2), 61–73.
[4] Global-Tech Appliances, Inc. v. SEB S.A., 563 U.S. 754 (2011).
[5] GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc., 976 F.3d 1347 (Fed. Cir. 2020) (Prost, C.J., dissenting).
[6] Prost, S. (2020). Dissenting opinion in GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc., 976 F.3d 1347, 1364–1380 (Fed. Cir. 2020).
[7] U.S. Food and Drug Administration. (2022). Listing of patent information in the Orange Book (Draft Guidance). FDA.
[8] U.S. Food and Drug Administration, Office of Prescription Drug Promotion. (2023). Promotional activities for prescription drug products: Guidance for industry. FDA.
[9] 21 C.F.R. § 314.94(a)(8)(iv) (2023).
[10] U.S. Food and Drug Administration. (2023). Labeling recommendations for prescription drugs: Method-of-use carve-outs under Section viii (Draft Guidance). FDA.
[11] 21 U.S.C. § 505(j)(5)(B)(iv) (2023). Drug Price Competition and Patent Term Restoration Act of 1984, as amended.
[12] American Intellectual Property Law Association. (2023). AIPLA report of the economic survey 2023. AIPLA.
[13] Bristol-Myers Squibb Co. v. Aurobindo Pharma USA Inc., 1:17-cv-00374 (D. Del. 2020).
[14] Warner-Lambert Company LLC v. Generics (UK) Ltd (t/a Mylan) and another, [2018] UKSC 56, [2019] 3 All ER 95.
[15] Stauder, D., & Hümpfner, A. (2022). Method of treatment patents and carve-out labels in European jurisdictions: Divergence after Warner-Lambert. European Intellectual Property Review, 44(3), 140–158.
[16] Apotex Inc. v. Eli Lilly and Company, 2022 FC 283 (Federal Court of Canada).
[17] Affordable Prescriptions for Patients Act of 2021, S. 1426, 117th Cong. (2021).
[18] Federal Trade Commission. (2021). Pay-for-delay: How drug company pay-offs cost consumers billions. FTC Bureau of Competition.
[19] U.S. Food and Drug Administration. (2023). Guidance for industry: Listing of patent information in the Orange Book. FDA.
[20] Association for Accessible Medicines. (2023). 2023 U.S. generic and biosimilar medicines savings report. AAM.
[21] Panduit Corp. v. Stahlin Bros. Fibre Works, Inc., 575 F.2d 1152 (6th Cir. 1978) (establishing four-factor test for lost profits damages in patent cases).
[22] Winter v. Natural Resources Defense Council, Inc., 555 U.S. 7 (2008) (setting standard for preliminary injunctive relief).
[23] Federal Trade Commission. (2023, September 14). FTC challenges more than 100 patents as improperly listed in the FDA’s Orange Book [Press Release]. FTC.