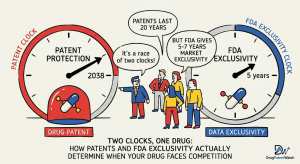
Every pharmaceutical executive knows the word “patent cliff.” Fewer can accurately define the legal machinery underneath it. Fewer still understand that a patent expiration and an FDA exclusivity expiration are not the same event, do not occur on the same timeline, and do not produce the same competitive consequences.
This confusion is not academic. When the wrong clock runs out, it can cost a brand manufacturer hundreds of millions of dollars in premature generic competition. When investors misread which protection layer a drug actually relies on, they misprice both the branded stock and the generic challenger.
This article separates the two mechanisms completely, explains how they interact in the real world, and shows you how to read both clocks correctly. It draws on patent data from DrugPatentWatch, FDA Orange Book and Purple Book records, and litigation histories to give you a framework that works in practice, not just in theory.
The Two Systems Are Built on Different Laws and Answer Different Questions
A patent is a private property right granted by the U.S. Patent and Trademark Office (USPTO) under 35 U.S.C. It gives the patent holder the right to exclude others from making, using, selling, or importing the patented invention for a fixed period. A patent does not require FDA approval to exist, and the FDA does not grant it. The FDA does not even know a drug has a patent unless the sponsor tells the agency.
FDA regulatory exclusivity is a government-conferred market protection created under the Federal Food, Drug, and Cosmetic Act (FD&C Act). It operates by preventing the FDA from approving, or in some cases even accepting, a competitor’s application for a defined period after a drug’s approval. It has nothing to do with the USPTO. It cannot be challenged in a patent lawsuit. It is, in legal terms, a separate beast entirely.
The two systems answer different questions. A patent answers: Does someone else have the right to make or sell this molecule or this method? FDA exclusivity answers: Can the FDA legally approve a competitor’s application right now? Both questions matter. Only one of them is often correctly understood.
The Practical Consequence of Conflating the Two
When analysts say a drug “loses patent protection” in a given year, they often mean only one layer of protection expires. The other layer may still be running. Conversely, a drug’s FDA exclusivity may expire years before its core composition-of-matter patent, which means the FDA can approve a generic application, but the generic manufacturer cannot launch without facing immediate patent infringement litigation.
The result is a landscape where a drug can simultaneously be generic-approvable and effectively launch-blocked. Or, in the opposite scenario, it can be patent-expired but exclusivity-protected, preventing the FDA from even accepting a generic application. Knowing which scenario you are looking at changes every downstream decision.
How Pharmaceutical Patents Actually Work
A pharmaceutical company can file patents on a drug at almost any stage of development. The most common types protect four distinct things.
The Four Core Patent Types in Pharma
- Composition-of-matter patents cover the active molecule itself. These are the most valuable and the hardest to design around. A composition-of-matter patent on a new chemical entity (NCE) means that no generic manufacturer can produce that molecule legally, full stop.
- Formulation patents cover how the drug is delivered: the tablet matrix, the coating system, the nanoparticle structure, the extended-release mechanism. These are easier to challenge because a generic manufacturer may achieve bioequivalence through a different delivery mechanism.
- Method-of-use patents protect the specific therapeutic application. They cover what the drug does, not what it is. A generic can often carve out a skinny label that avoids the patented indication and still get approved.
- Process patents cover manufacturing methods. They rarely block generic entry directly because a generic manufacturer can often use a different synthesis process, but they matter considerably in biosimilar disputes where manufacturing is integral to the product itself.
The 20-Year Clock and Its Complications
A U.S. patent runs for 20 years from its filing date, not from its grant date. In pharmaceutical development, where the average drug takes 10 to 15 years to go from filing to approval, this means a brand manufacturer often has only 5 to 10 years of patent-protected market life remaining by the time the FDA approves the drug. That economic reality is what drove Congress to create the Hatch-Waxman Act.
Patent Term Extension Under Hatch-Waxman
The Drug Price Competition and Patent Term Restoration Act of 1984, known universally as Hatch-Waxman, created a mechanism for brand manufacturers to recover some of the patent term consumed during FDA review. Under 35 U.S.C. §156, a sponsor can apply to the USPTO for a patent term extension (PTE) of up to five years. The total remaining patent life after the extension cannot exceed 14 years from the drug’s approval date [1].
The calculation uses a formula that accounts for time spent in clinical trials and in FDA review. Only one patent per drug can receive the extension, and only a patent that was in force at the time of approval qualifies. The sponsor must apply within 60 days of FDA approval. Miss that window and the extension is gone.
DrugPatentWatch tracks patent term extensions across approved drugs and provides expiration dates adjusted for these extensions, which matters because the raw patent filing date understates the remaining protection life for most brand products.
FDA Regulatory Exclusivity: Five Types, Five Different Purposes
FDA exclusivity is not a single thing. There are five distinct types, each created for a different policy purpose, each running on a different clock, and each blocking competition in a slightly different way. Conflating them produces analytical errors.
| Exclusivity Type | Duration | Trigger | Competition Effect |
|---|---|---|---|
| New Chemical Entity (NCE) | 5 years | First approval of new active ingredient | Blocks ANDA submission (4 yrs for Para IV) |
| New Clinical Investigation | 3 years | New indication, formulation, condition of use | Blocks final ANDA approval for that change |
| Orphan Drug | 7 years | Rare disease designation (<200,000 patients) | Blocks same drug for same orphan indication |
| Pediatric Exclusivity | +6 months added | Completion of FDA-requested pediatric studies | Extends all existing patent/exclusivity dates |
| Biologic (BLA) – BPCIA | 12 years (+ 4-yr data exclusivity) | First licensure of reference biologic | Blocks biosimilar approval |
FDA Exclusivity Types: Duration, Trigger, and Competitive Effect
New Chemical Entity Exclusivity: The Five-Year Shield
When the FDA approves an NCE, a drug with an active ingredient that has never been approved before, the sponsor receives five years of NCE exclusivity. During this period, the FDA will not accept an ANDA or a 505(b)(2) application referencing that drug. The exception is a Paragraph IV certification challenging the drug’s patents, which can be submitted after four years [2].
NCE exclusivity is independent of patents. A sponsor can have robust patent coverage and still receive NCE exclusivity. The exclusivity provides a floor of protection that patents alone cannot guarantee, because patents can be challenged and invalidated. FDA exclusivity cannot be invalidated through litigation.
Three-Year Clinical Investigation Exclusivity
When a sponsor conducts new clinical investigations essential to approval of a supplement, such as a new indication, new dosage form, or new condition of use, the FDA grants three years of exclusivity on that specific change. This exclusivity blocks final approval of a competitor’s ANDA for that particular change, not for the entire drug. It is narrower in scope than NCE exclusivity and expires faster.
Brand manufacturers use three-year exclusivity as a lifecycle management tool. Each new supplement backed by clinical data resets the exclusivity clock for that modification. It is not a loophole but an intended incentive for continued development investment.
Orphan Drug Exclusivity: Seven Years of Targeted Protection
Under the Orphan Drug Act of 1983, a drug approved for a rare disease affecting fewer than 200,000 people in the United States receives seven years of orphan drug exclusivity (ODE) [3]. During this period, the FDA cannot approve another application for the same drug for the same orphan indication, with narrow exceptions for clinical superiority claims.
ODE can be extraordinarily valuable for high-priced specialty drugs. Drugs like Spinraza, approved for spinal muscular atrophy, and Kalydeco, approved for specific CFTR mutations in cystic fibrosis, relied on ODE as a critical layer of protection in markets where the patient population is too small to attract immediate generic competition anyway.
The interaction between orphan exclusivity and patents gets complicated when a drug receives orphan status for multiple indications. Each designated indication can carry its own ODE, stacking protection across different disease states.
Pediatric Exclusivity: The Six-Month Bonus
Pediatric exclusivity is not a standalone protection. It is an addition to existing patent or exclusivity protection. When a sponsor completes FDA-requested pediatric studies under the Best Pharmaceuticals for Children Act (BPCA), the FDA grants six additional months of protection, tacked onto whatever patent or exclusivity protection already exists [4].
Six months on a blockbuster drug translates into real money. When AstraZeneca received pediatric exclusivity on Nexium (esomeprazole), the six-month extension was worth an estimated $1.2 billion in additional revenue given the drug’s sales volume at the time [5].
Biologics Exclusivity: Twelve Years Under the BPCIA
For biologics, the Biologics Price Competition and Innovation Act of 2010 (BPCIA) created a separate exclusivity framework. Reference product sponsors receive 12 years of exclusivity from the date of first licensure, with a four-year data exclusivity period during which biosimilar applicants cannot even submit an application [6].
The BPCIA framework is fundamentally different from Hatch-Waxman. The patent dispute process under BPCIA, sometimes called the “patent dance,” is optional, not mandatory. The litigation timelines and procedural rules differ substantially. And the 12-year exclusivity applies regardless of patent status.
The Real-World Timeline: How Patents and Exclusivity Overlap
The most important concept to master is that patents and FDA exclusivity are independent variables that happen to apply to the same drug. They do not start at the same time, they do not end at the same time, and the one that expires last is the one that controls the competitive outcome.
The Four Scenarios That Determine Launch Timing
In any given drug, one of four situations applies.
- Patents expire before exclusivity. The FDA may be able to accept a competitor’s ANDA after the exclusivity period ends, but the patent situation is clean. Generic manufacturers can launch freely once exclusivity expires. This is common with drugs whose patents were filed early and whose development took many years.
- Exclusivity expires before patents. The FDA can accept and approve a competitor’s application, but any generic launch would face immediate patent infringement litigation. This creates the situation where generics are technically approved but commercially blocked. The rational strategy for generic manufacturers is to challenge the remaining patents via Paragraph IV.
- Both expire at approximately the same time. This is the clearest patent cliff scenario. The brand faces simultaneous loss of both protection layers, and generic competition typically arrives within weeks of expiration.
- Patents are challenged and invalidated while exclusivity remains intact. The FDA can approve a generic, but it cannot do so before the exclusivity expires. Paragraph IV litigation outcomes that invalidate patents do not accelerate FDA exclusivity expiration.
Case Study: Lipitor (Atorvastatin)
Pfizer’s Lipitor was the best-selling drug in pharmaceutical history. Its patent and exclusivity story is a textbook example of how layered protection works and how it unravels.
The primary composition-of-matter patent on atorvastatin, U.S. Patent No. 4,681,893, was filed in 1986 and had a basic expiration in 2006. Pfizer obtained a patent term extension that pushed the effective expiration to March 2010. Separately, Pfizer held several formulation and method-of-use patents with later expiration dates.
Generic manufacturers, led by Ranbaxy Laboratories, filed Paragraph IV certifications challenging Pfizer’s patents years before expiration. After protracted litigation, Ranbaxy prevailed on some claims. Pfizer settled with Ranbaxy, granting Ranbaxy a license to launch a generic in November 2011, with an exclusivity-from-settlement period that kept other generics out for 180 days.
By the time Lipitor went fully generic in May 2012, Pfizer had already begun the revenue transition. The patent cliff in this case was real but not sudden: it was managed across multiple years through litigation timelines and settlement strategy.
Case Study: Humira (Adalimumab) and the Biosimilar Delay
AbbVie’s Humira is the highest-revenue drug in history, earning over $20 billion annually at peak sales. It held its market position through a combination of biologic BPCIA exclusivity and an aggressive patent portfolio that industry analysts, using data tracked by sources including DrugPatentWatch, described as a patent thicket comprising more than 160 patents.
Despite FDA approval of multiple biosimilar adalimumab products years earlier, no biosimilar launched in the United States until January 2023. The reason was not BPCIA exclusivity, which expired in 2023, but litigation settlements that AbbVie reached with each biosimilar manufacturer, licensing each of them to launch no earlier than specific negotiated dates.
The Humira case separated the two protection mechanisms clearly. The FDA exclusivity timeline was manageable. The patent thicket required individual negotiation. AbbVie’s strategy was to use patents where FDA exclusivity could not reach.
The Hatch-Waxman Act: The Legal Architecture of Generic Entry
No discussion of pharmaceutical IP is complete without Hatch-Waxman. The 1984 law did two things simultaneously that seem contradictory: it made it easier for generic drugs to get approved, and it created new protections for brand manufacturers to compensate for time lost during clinical development.
The ANDA Pathway and the Orange Book
Before Hatch-Waxman, generic manufacturers had to conduct their own full clinical trials to prove safety and efficacy. The law created the Abbreviated New Drug Application (ANDA), which allows generics to rely on the brand manufacturer’s safety and efficacy data by demonstrating bioequivalence. This dramatically lowered the cost and time to bring a generic to market.
As a counterbalance, the law required brand manufacturers to list their relevant patents in the FDA’s Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations). When a generic files an ANDA, it must certify its position relative to each listed patent in one of four ways, called Paragraph I through Paragraph IV certifications.
DrugPatentWatch aggregates Orange Book patent listings and provides expiration dates, patent types, and Paragraph IV challenge histories for approved drugs, making it a critical tool for tracking when listed patents actually expire versus when generic challenges have been filed.
The Paragraph IV Challenge Mechanism
A Paragraph IV certification is the mechanism by which a generic manufacturer challenges a listed patent. By certifying that the brand’s patent is invalid, unenforceable, or not infringed by the generic’s formulation, the ANDA filer invites litigation. The brand manufacturer then has 45 days to file a patent infringement lawsuit [7].
If the brand sues within that 45-day window, it triggers an automatic 30-month stay on FDA final approval of the generic. Even if the generic manufacturer would win on the merits, the FDA cannot approve the application for 30 months from the date of the original ANDA filing. The stay can be shortened by a court order or extended in limited circumstances.
The 180-Day Generic Exclusivity Incentive
Hatch-Waxman created an incentive for generic manufacturers to challenge patents: the first filer of a Paragraph IV certification earns 180 days of generic exclusivity upon launch. During those 180 days, no other generic manufacturer can receive final FDA approval.
This 180-day exclusivity has real economic value because a generic launching without competition can price higher than it would in a fully competitive generic market. The first generic typically captures 70 to 80 percent market share in volume terms within its exclusivity window. After the window closes and multiple generics enter, prices drop rapidly, often by 80 to 90 percent compared to the brand price.
The 180-day exclusivity is forfeited if the first filer fails to launch within specific timeframes or if a court rules the challenged patent valid and infringed. The forfeiture rules are complex and have been litigated extensively.
Patent Cliff vs. Exclusivity Cliff: Two Different Events
Financial analysts frequently use “patent cliff” to describe any point at which a major drug faces generic competition. The term flattens a distinction that matters enormously for valuation.
When the Real Cliff Happens
A true competitive cliff occurs when both patent protection and FDA exclusivity have expired or been circumvented. Before that point, one or both layers of protection remain operative. A drug can be patent-expired but exclusivity-protected. It can be challenged on patents via Paragraph IV while exclusivity blocks ANDA acceptance. It can have exclusivity expired but face no patent challenges.
The practical question is not when the first protection expires but when the last meaningful barrier falls. For most brand drugs, the answer requires reading the entire Orange Book patent listing, the NCE or other exclusivity expiration date, any pediatric exclusivity additions, and the litigation history of any Paragraph IV challenges, then determining which of those events controls actual generic entry.
The Strategic Implication for Valuation
“Generic drugs account for 90% of U.S. prescriptions but only 18% of total drug spending, with branded drugs commanding the premium through the period of exclusivity protection.”
— Association for Accessible Medicines, U.S. Generic and Biosimilar Medicines Savings Report, 2022 [8]
That price premium evaporates quickly after generic entry. A drug generating $3 billion annually can see revenues drop to $300 million or less within two years of full generic competition. A one-year error in estimating the effective protection expiration translates directly into a $2-3 billion valuation error on the brand side and an equally large timing error for the generic entrant.
The correct valuation approach for any drug nearing its protection expiration is to identify the controlling protection layer, account for the probability and expected timing of any Paragraph IV challenges, and model the impact of 180-day generic exclusivity on the shape of the revenue decline curve. Ignoring any one of these variables produces a wrong answer.
Biologics: A Different Battlefield Entirely
Biosimilars are not generics. The regulatory science is different, the legal framework is different, and the competitive dynamics are different. Treating biosimilar competition as equivalent to small-molecule generic competition is one of the most common analytical errors in pharmaceutical investing.
Why the BPCIA and Hatch-Waxman Are Not Analogous
The ANDA pathway under Hatch-Waxman was designed for small molecules that can be fully characterized and shown to be bioequivalent through pharmacokinetic studies. Biologics are large, complex molecules made in living cells. Their structure, post-translational modifications, and higher-order properties cannot be fully characterized by current analytical methods. The FDA approves biosimilars as “highly similar” to the reference product with no clinically meaningful differences, not as identical.
This scientific difference has legal consequences. The BPCIA patent dance process is optional, not mandatory. There is no mandatory 30-month stay equivalent triggered by biosimilar litigation. The interchangeability designation, which allows substitution at the pharmacy level without prescriber intervention, requires additional clinical data and has been granted to fewer biosimilars than the basic biosimilar designation.
The Twelve-Year Exclusivity and Its Real-World Effect
The 12-year exclusivity under the BPCIA has held firm despite multiple legislative attempts to shorten it to seven years. For reference product sponsors, this means that even if every patent in a biologic’s portfolio were invalidated tomorrow, no biosimilar could be approved until the 12-year clock runs out.
Biologic developers have had less incentive to build dense patent thickets because the exclusivity itself provides a long baseline. AbbVie’s strategy with Humira was unusual in its aggressiveness. Most biologic sponsors rely more heavily on the BPCIA exclusivity as the primary protection and on manufacturing complexity as a secondary barrier.
The Purple Book: The Biologic Equivalent of the Orange Book
The FDA’s Purple Book (the Lists of Licensed Biological Products) functions similarly to the Orange Book for biologics. Unlike the Orange Book, the Purple Book did not originally list patents or exclusivity dates in a machine-readable format, which made competitive intelligence significantly harder. The BPCIA Amendments of 2019 improved transparency requirements, but the data remains less structured than the Orange Book patent listings [9].
DrugPatentWatch covers both small-molecule Orange Book data and biologic patent portfolios, enabling competitive analysis across both drug types in a single platform. For biosimilar manufacturers assessing reference product protection, this cross-coverage is essential because the Purple Book alone does not show the full patent picture.
Brand Manufacturer Strategies: How to Extend Market Exclusivity Legally
Brand pharmaceutical companies deploy several strategies to extend their period of protected revenue. Some are legitimate incentive mechanisms built into the law. Others have attracted FTC scrutiny and litigation.
Lifecycle Management and the Evergreening Debate
Lifecycle management refers to a brand manufacturer’s strategy of developing new formulations, delivery mechanisms, or combinations of an existing drug to obtain new patents and new periods of FDA exclusivity as the original drug’s protection expires. Critics call this “evergreening.” Supporters point out that the new formulations often provide genuine patient benefit through improved dosing convenience, better tolerability, or extended release.
The legal framework explicitly incentivizes this activity. The three-year clinical investigation exclusivity exists precisely to reward sponsors who invest in new clinical data. Patent law explicitly allows patents on improvements to existing inventions. Whether the improvement is meaningful enough to justify the pricing advantage it creates is a policy debate, not a legal one.
Authorized Generics: Competing With Yourself
An authorized generic is a copy of a brand drug, manufactured by or for the brand manufacturer, and sold at generic prices through a subsidiary or partner. Brand manufacturers launch authorized generics at the beginning of the first generic’s 180-day exclusivity window, which cuts into the first generic’s revenue advantage without technically violating the exclusivity (because the authorized generic is the brand).
The FTC has examined authorized generics extensively. Its 2011 report found that authorized generics reduce the first filer’s revenues during the 180-day window by an average of 50 percent. They are legal, they complicate the economics of Paragraph IV challenges, and they are widely used [10].
Pay-for-Delay Settlements and Their Legal Status
When a brand manufacturer settles a Paragraph IV litigation by paying the generic challenger to delay launch, the arrangement is called a “pay-for-delay” or “reverse payment” settlement. The Supreme Court’s 2013 decision in FTC v. Actavis held that such settlements are subject to antitrust scrutiny under the rule of reason, rejecting the position that any settlement within the scope of the patent is automatically lawful [11].
Since Actavis, pay-for-delay settlements have become structurally more complex. Direct cash payments are less common. Value transfers take the form of supply agreements, marketing arrangements, or co-promotion deals. The legal analysis turns on whether the value transferred exceeds what can be explained by avoiding litigation costs.
Generic and Biosimilar Manufacturer Strategies: Reading the Landscape
For generic manufacturers, the core strategic task is identifying which drugs have protection expiring on a near-term horizon, which protection layer actually controls the entry date, and whether the first-filer 180-day exclusivity is worth the cost and risk of patent litigation.
Identifying Targets: What to Look For
The ideal generic target has a large commercial market, a composition-of-matter patent that is either expired or potentially weak, NCE exclusivity that has expired or is expiring within the next two years, and no Paragraph IV challenge already on file.
Services like DrugPatentWatch allow generic manufacturers to screen the entire Orange Book by exclusivity expiration date, patent type, and challenge history. The ability to identify drugs where NCE exclusivity expires before core patents do, and where formulation or method-of-use patents are the remaining barriers, helps focus patent counsel’s attention on the challenges most likely to succeed.
The Biosimilar Assessment Problem
Assessing biosimilar entry timelines requires evaluating four factors that do not apply to small-molecule generics: the BPCIA exclusivity expiration date, the manufacturing complexity of the reference product, the patent estate (often not in the Purple Book), and the probability of achieving interchangeability status. Each variable shifts the timeline.
The track record shows that biosimilar entry has lagged small-molecule generic entry by several years on average after the regulatory pathway opened. First-mover biosimilars capture smaller market shares than first-mover generics, and pricing erosion is slower. The investment required to develop and manufacture a biosimilar is far higher than for a small-molecule generic.
What Investors and Analysts Need to Read Correctly
When a pharmaceutical company discloses patent or exclusivity expiration dates in an SEC filing, it rarely explains which protection layer controls entry or what the probability distribution of early generic entry looks like. The number given is typically the latest possible protection date, not the expected competitive entry date.
The Gap Between Disclosed Expiration and Expected Entry
A drug with a patent listed as expiring in 2030 may face generic entry in 2026 if a Paragraph IV challenge succeeds and the 30-month stay runs without a favorable court ruling. A drug whose core patent expired in 2022 may face no generic competition until 2025 if NCE exclusivity protects it through that date.
The correct analytical framework has two steps. First, identify the last-expiring protection that actually blocks generic entry (not just the latest patent date in the portfolio). Second, assess the probability and timing of Paragraph IV challenges, using the history of challenges to similar compounds as a base rate.
The Information Sources That Matter
The FDA Orange Book provides the official list of patents and exclusivity dates by drug. The USPTO Patent Center provides the actual patent text and prosecution history. SEC filings disclose pending Paragraph IV litigation. DrugPatentWatch aggregates all of these sources and adds alert tracking, making it possible to monitor entire portfolios for patent challenges, newly listed patents, and exclusivity changes. For investors managing position risk around a specific drug’s exclusivity cliff, that alert infrastructure is the operational tool.
Historical Base Rates for Paragraph IV Success
Paragraph IV challengers succeed (i.e., the challenged patent is found invalid or not infringed) in roughly half of litigated cases, though success rates vary by patent type. Formulation patents are challenged more successfully than composition-of-matter patents. Method-of-use patents have mixed records [12].
The probability of a Paragraph IV challenge being filed at all correlates with market size. Drugs with annual revenues above $250 million face Paragraph IV challenges with much higher frequency than drugs below that threshold. For large-market drugs, assume a challenge will be filed; the question is whether it will succeed.
The Global Picture: Why the U.S. Framework Does Not Transfer Directly
Patent and regulatory exclusivity law varies significantly across major pharmaceutical markets. A drug with strong protection in the United States may face generic competition years earlier in Europe, or vice versa.
Data Exclusivity in Europe vs. the United States
The European Medicines Agency (EMA) grants eight years of data exclusivity from initial marketing authorization, plus two additional years of market exclusivity blocking generic approval. There is no equivalent to the U.S. five-year NCE exclusivity for all new active substances, but the eight-plus-two framework produces a broadly similar period of protection for most drugs [13].
European patent term extensions, called Supplementary Protection Certificates (SPCs), function similarly to U.S. patent term extensions under 35 U.S.C. §156 but are administered through national patent offices in each EU member state, not a central authority. An SPC can extend a European patent for up to five years past its expiration.
Regulatory Exclusivity in Emerging Markets
Most major emerging markets, including Brazil, India, China, and South Korea, have data exclusivity provisions of varying scope and enforceability. India’s data exclusivity framework remains contested, and pharmaceutical companies have challenged the country’s narrow patentability standard under Section 3(d) of the Indian Patents Act, which prohibits patents on new forms of known substances without demonstrating enhanced efficacy [14].
For global pharmaceutical companies, the effective competitive lifetime of a drug is market-specific. A drug may be fully generic in India while patent-protected in the United States and SPC-protected in Germany. Global revenue forecasting that applies a single protection expiration date across all markets will be wrong.
REMS Programs: The Third Barrier That Is Often Overlooked
Risk Evaluation and Mitigation Strategies (REMS) are FDA-required safety programs for drugs with serious risks that require specific management. They include restricted distribution systems, mandatory training for prescribers, and enrollment requirements for patients.
REMS programs can function as a competitive barrier because a generic manufacturer must have access to the brand drug’s REMS infrastructure, including samples for bioequivalence testing, to complete its ANDA. Some brand manufacturers have used REMS programs as a reason to deny sample access to generic challengers, arguing that sharing samples would compromise the safety program’s requirements.
The FTC filed a lawsuit against Shire ViroPharma in 2017 alleging use of a REMS program as a mechanism to delay generic entry. Congress addressed this partially through the CREATES Act of 2019, which created a legal pathway for generic manufacturers to compel access to drug samples when brand manufacturers deny access without a legitimate REMS reason [15].
REMS programs are not patents and are not FDA exclusivity. They are a third layer that can interact with both protection mechanisms and that investors frequently overlook in assessing competitive entry timing.
Patents vs. FDA Exclusivity: Side-by-Side
| Attribute | Patent | FDA Exclusivity |
|---|---|---|
| Legal Basis | U.S. Patent Act (35 U.S.C.) | FD&C Act |
| Granted By | USPTO | FDA |
| Duration | 20 years from filing | 3, 5, 7, or 12 years from approval |
| What It Blocks | Making, using, or selling the invention | Submission or approval of competitor applications |
| Can Be Challenged? | Yes (IPR, litigation, Paragraph IV) | No direct challenge mechanism |
| Tied to Approval? | No | Yes |
| Can Coexist? | Yes | Yes |
Core Differences Between U.S. Patent Protection and FDA Regulatory Exclusivity
Key Takeaways
| Key Takeaways: Patents vs. FDA Exclusivity |
| 1. Patents (granted by USPTO) and FDA exclusivity (granted by FDA) are separate legal mechanisms operating on independent timelines. Expiration of one does not mean expiration of the other. |
| 2. The effective protection period for any drug is controlled by whichever mechanism expires last — which requires reading both the Orange Book patent listings and the FDA exclusivity database simultaneously. |
| 3. The five types of FDA exclusivity (NCE, 3-year clinical, orphan, pediatric, and biologic) each have different durations, different triggers, and different competitive effects. |
| 4. Paragraph IV patent challenges can invalidate patents without accelerating FDA exclusivity expiration. A generic can be FDA-approved and still be commercially blocked. |
| 5. The 180-day first-generic exclusivity shapes the competitive entry timeline for most major branded drugs and explains why brand manufacturers use authorized generics and settlement agreements to manage revenue decline. |
| 6. Biologics operate under the BPCIA, not Hatch-Waxman. The 12-year exclusivity and slower biosimilar market penetration make biologic IP analysis categorically different from small-molecule analysis. |
| 7. Authorized generics, pay-for-delay settlements, and REMS programs are additional variables that affect the actual competitive entry date beyond what the formal patent and exclusivity dates suggest. |
| 8. Global pharmaceutical IP protection is market-specific. Applying a U.S. expiration date to global revenue projections produces systematic forecasting errors. |
Frequently Asked Questions
1. Can a generic drug be FDA-approved before the brand’s patent expires?
Yes. The FDA can approve an ANDA based on the applicant’s Paragraph IV certification that the brand’s listed patent is invalid or not infringed. FDA approval of the ANDA is a separate event from the resolution of any patent litigation. The generic manufacturer cannot launch commercially without risking an infringement lawsuit, but FDA approval itself is independent of patent status. This is why “FDA-approved generic” does not automatically mean “available in the market.”
2. If a brand manufacturer gets a new patent during a drug’s lifecycle, does it extend FDA exclusivity?
No. A new patent listed in the Orange Book extends potential patent protection if valid and infringed, but it does not restart the FDA exclusivity clock. FDA exclusivity is tied to specific regulatory events: initial NCE approval, new clinical investigations supporting a supplement, orphan designation, or pediatric study completion. Filing a new patent on an existing drug does not trigger any of these events.
3. How does the 30-month litigation stay interact with FDA exclusivity expiration?
The two run on parallel tracks. If a brand’s NCE exclusivity expires during an ongoing 30-month stay, the generic ANDA can receive final FDA approval as soon as the exclusivity expires (assuming the stay has not been lifted by court order). The stay does not extend exclusivity. Whichever blocks approval for longer is the operative constraint at any given point.
4. Why do biosimilars take so much longer to reach the market than small-molecule generics after protection expiration?
Four factors combine to slow biosimilar entry. Biosimilar development costs are 10 to 100 times higher than generic small-molecule development, which limits the number of manufacturers who can enter. Physician and payer acceptance of biosimilars has historically lagged, reducing commercial incentives. Interchangeability designation requires additional clinical data beyond the basic biosimilar package. Finally, the patent landscapes for biologics tend to be denser and more litigated through settlement agreements, as AbbVie demonstrated with Humira.
5. Does DrugPatentWatch show FDA exclusivity expiration dates alongside patent data?
Yes. DrugPatentWatch integrates Orange Book patent listings with FDA exclusivity data and provides expiration dates for both in a searchable format. For a given drug, you can see the full set of listed patents, their expiration dates (adjusted for patent term extensions where applicable), the exclusivity expiration date, and any Paragraph IV challenge history. This integrated view makes it possible to identify the controlling protection layer without manually cross-referencing multiple FDA and USPTO databases.
Citations
- [1] Drug Price Competition and Patent Term Restoration Act of 1984, Pub. L. No. 98-417, 98 Stat. 1585. 35 U.S.C. §156.
- [2] U.S. Food and Drug Administration. (2015). Paragraph IV certifications. FDA Orange Book Guidance Documents. https://www.fda.gov/drugs/abbreviated-new-drug-applications-andas/paragraph-iv-certifications
- [3] Orphan Drug Act of 1983, Pub. L. No. 97-414, 96 Stat. 2049. 21 U.S.C. §360bb.
- [4] Best Pharmaceuticals for Children Act of 2002, Pub. L. No. 107-109, 115 Stat. 1408.
- [5] Grabowski, H., Long, G., & Mortimer, R. (2014). Recent trends in brand-name and generic drug competition. Journal of Medical Economics, 17(3), 207–214. https://doi.org/10.3111/13696998.2014.877451
- [6] Biologics Price Competition and Innovation Act of 2009, Pub. L. No. 111-148, 124 Stat. 804. 42 U.S.C. §262.
- [7] U.S. Food and Drug Administration. (2022). Guidance for industry: Abbreviated new drug applications — patent and exclusivity questions and answers. https://www.fda.gov/media/72251/download
- [8] Association for Accessible Medicines. (2022). U.S. generic and biosimilar medicines savings report. https://accessiblemeds.org/resources/reports/2022-aam-savings-report
- [9] Biologics Price Competition and Innovation Act Amendments, Further Consolidated Appropriations Act of 2020, Pub. L. No. 116-94.
- [10] Federal Trade Commission. (2011). Authorized generic drugs: Short-term effects and long-term impact. https://www.ftc.gov/reports/authorized-generic-drugs-short-term-effects-long-term-impact-report-federal-trade-commission
- [11] FTC v. Actavis, Inc., 570 U.S. 136 (2013).
- [12] Hemphill, C. S., & Sampat, B. N. (2012). Drug patents at the supreme court. Science, 339(6126), 1386–1387. https://doi.org/10.1126/science.1228319
- [13] European Medicines Agency. (2023). Data exclusivity, market protection and paediatric rewards. https://www.ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/data-exclusivity-market-protection-paediatric-rewards
- [14] Novartis AG v. Union of India, (2013) 6 SCC 1 (Supreme Court of India). Addressing Section 3(d) of the Indian Patents Act, 1970.
- [15] Creating and Restoring Equal Access to Equivalent Samples Act of 2019 (CREATES Act), Pub. L. No. 116-94, Div. N, Title I, Subtitle F.