How AI, blockchain, multi-sourcing, and smarter policy can cut costs, eliminate fragility, and keep patients supplied
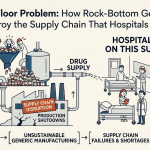
Generic drugs fill 89% of all U.S. prescriptions but account for only 26% of total drug spending [1]. That ratio is either an extraordinary public health achievement or a trap, depending on which side of the supply chain you sit on. For patients, it is the former. For the manufacturers, wholesalers, and logistics operators who keep those drugs moving, it increasingly looks like the latter.
The same price competition that makes generic drugs affordable creates margins so thin that companies cannot sustain the infrastructure investments needed to keep supply reliable. The result is a system that works magnificently until it doesn’t, at which point drug shortages cascade, patients are harmed, and regulators issue warnings that manufacturers struggle to act on quickly. The FDA’s shortage list has rarely been short.
This piece examines how the generic drug supply chain actually works, why it breaks down, and what the organizations succeeding in this space are doing differently. It covers operational, technological, and policy dimensions, and it pulls in hard data rather than consulting-firm generalizations. Readers include supply chain VPs, business development leads at generic manufacturers, pharmacy network executives, and institutional investors who need to price the operational risk embedded in generic drug portfolios.
Tools like DrugPatentWatch are built specifically to give pharmaceutical professionals competitive intelligence on patent expirations, ANDA filings, and market entry timing, and understanding that competitive context is inseparable from understanding how the supply chain gets stressed.
Part I: The Machine and Its Mechanics
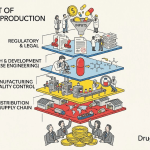
1. What the Generic Drug Supply Chain Actually Is
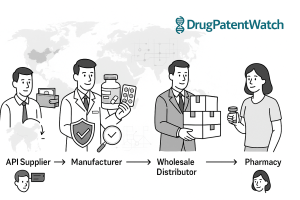
A supply chain, at its most direct, is every person, process, and organization required to get a product from raw material to end consumer [2]. For generic drugs, that chain has four primary nodes: manufacturers, wholesale distributors, pharmacies, and Pharmacy Benefit Managers (PBMs). Each plays a distinct economic role, and understanding those roles precisely matters because the financial incentives at each node shape behavior in ways that can either stabilize or destabilize the overall system.
Manufacturers produce the drug. Their responsibilities begin earlier than most people outside the industry appreciate, starting with patent monitoring and bioequivalence development and running through commercial-scale production and post-market surveillance. They typically distribute first to wholesale distributors and, for large retail chains or specialty networks, sometimes directly [2].
Wholesale distributors sit between manufacturers and pharmacies. They buy in bulk, manage logistics, handle inventory, and provide financial services including payment processing. Their compensation is often calculated as a percentage of Wholesale Acquisition Cost (WAC), with additional discounts for prompt payment or high-volume purchasing [2]. Critically, distributors also carry significant responsibility for compliance across the chain, including product authentication and proper storage.
Pharmacies — retail, specialty, and hospital — are the dispensing endpoint. Inventory management at this level requires precise tracking of expiration dates, temperature controls, and product authentication. Smaller pharmacies often operate with lean inventories, which makes them acutely vulnerable when upstream supply falters [3].
PBMs are the most complex and least understood node. They began as simple claims processors. Today they manage formularies, negotiate manufacturer rebates, determine generic and therapeutic substitutions, and operate mail-order pharmacy services [2]. One fact that is frequently lost in broad coverage of drug pricing: generic manufacturers almost never negotiate rebates with PBMs. That practice is largely confined to brand-name drugs [1]. The financial architecture of the generic market is therefore fundamentally different from the brand market, and conflating the two produces flawed analysis.
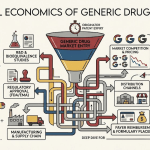
2. The Economics of Generic Drug Competition
Generic drugs compete primarily on price. The multi-competitor model is not a bug; it is the design. When a brand-name drug loses patent exclusivity, multiple generic manufacturers file Abbreviated New Drug Applications (ANDAs) with the FDA. As more entrants receive approval, competition intensifies and prices decline. This is the mechanism that generates the 26% spending figure against 89% prescription volume [1].
The problem is that price erosion does not stop at a level that allows manufacturers to fund robust supply chain infrastructure. It continues until some manufacturers exit the market or until only a small number of suppliers remain, often concentrated in low-cost manufacturing geographies. The FDA’s data on drug shortages consistently shows that low-priced generics are disproportionately affected [4]. That is not a coincidence. It is the market’s cost-optimization function operating at the expense of supply resilience.
Reimbursement structures amplify this dynamic. Public payers like Medicare and Medicaid use Maximum Allowable Cost (MAC) limits to cap what they will pay for a generic drug, regardless of manufacturer [5]. When input costs rise, whether from API price increases, regulatory compliance requirements, or tariffs, manufacturers cannot pass those costs through. They absorb them, reduce investment, or exit. All three outcomes weaken the supply chain.
The generic market also captures a smaller share of supply chain revenue than the brand market. Generic manufacturers capture 64% of total revenue from the supply chain compared to 76% for brand manufacturers [1]. That 12-point gap represents money that is not available for quality systems, redundant sourcing, or domestic manufacturing investment.
3. How a Generic Drug Gets Made: From ANDA to Aisle
The development of a generic drug is considerably more demanding than the phrase ‘copy of a branded drug’ implies. Regulatory requirements are stringent, the science is genuinely difficult, and the timelines are long.
Patent and market analysis is where everything begins. Generic developers monitor patent expirations, exclusivity periods, and competitive ANDA filings to identify commercially viable opportunities. Platforms like DrugPatentWatch are used extensively at this stage to map the IP landscape and assess first-to-file advantages that can translate into 180-day market exclusivity under the Hatch-Waxman framework.
Formulation development requires scientists to reverse-engineer the Reference Listed Drug (RLD) — the FDA-approved branded product against which the generic is measured. This process, called de-formulation, is complex because the generic manufacturer does not have access to the brand’s proprietary formulation or manufacturing specifications [4]. They must infer critical quality attributes from public data and their own testing.
Bioequivalence studies are the defining scientific hurdle. The generic must demonstrate that it delivers the active ingredient into the bloodstream at essentially the same rate and in the same amount as the branded product. Regulators permit differences in inactive ingredients, manufacturing processes, and sometimes even the salt or ester form of the active ingredient, so long as bioequivalence is established [4]. For complex formulations — modified-release products, narrow therapeutic index drugs, inhaled or topical products — this is a substantial scientific challenge that creates meaningful barriers to entry.
Scale-up and tech transfer take the formulation from laboratory batch to commercial production. This phase is where many programs encounter delays. Maintaining product performance at production scale requires careful process control, and the transfer of that control from R&D to manufacturing is often imperfect on the first attempt.
ANDA submission and FDA review can take years. The FDA has worked to reduce review times, but a significant backlog built up during and after the COVID-19 pandemic. Approval timelines remain a meaningful source of market uncertainty for generic developers.
Post-market surveillance does not end at launch. Manufacturers must monitor adverse events, track product performance, and respond to FDA requests for additional data. This ongoing obligation carries costs that are rarely visible in the unit economics of a generic product but are real.
Part II: Where It Breaks Down
4. The Affordability Paradox and Why Cheap Drugs Create Fragile Chains
There is a structural contradiction at the center of the generic drug market. The very mechanism that makes generics affordable — intense price competition driven by multi-supplier markets — systematically undermines the supply chain’s ability to sustain itself.
When prices fall to the point where margins are too thin for quality investment, three things happen. First, manufacturers rationalize their supplier relationships, concentrating purchases with the lowest-cost API suppliers, which are overwhelmingly located in China and India [6]. Second, they defer investment in manufacturing quality systems, backup production lines, and domestic sourcing redundancy. Third, the least financially viable manufacturers exit the market, reducing competition and, paradoxically, reducing supply availability for the drugs whose shortage risk was highest.
The drugs most likely to go into shortage are the cheapest ones [4]. A 2023 ASPE analysis covering 2018 to 2023 found persistent shortages concentrated in generic injectables and other essential medicines with low price points [7]. These are often the drugs hospitals and oncologists most urgently need. <blockquote> ‘The generic drug market’s reliance on a limited number of manufacturers for specific products, combined with narrow profit margins, creates an environment where disruptions — even minor ones — can cascade into significant shortages affecting patient care across entire health systems.’ — ASPE Analysis of Drug Shortages, 2018–2023 [7] </blockquote>
This is not a market failure in the technical sense. The market is doing exactly what market competition does: driving prices toward marginal cost. The failure is in the policy architecture that treats the lowest price as the only objective, without weighting supply reliability or quality resilience. Procurement decisions by PBMs, hospitals, and pharmacy chains that select the lowest-cost generic supplier without considering that supplier’s quality history, geographic concentration, or manufacturing redundancy are importing risk directly into the supply chain.
5. Geographic Concentration: The China-India Problem
The concentration of Active Pharmaceutical Ingredient manufacturing in China and India is well-documented and widely discussed. What is less often examined is precisely how this concentration translates into supply chain risk.
By 2020, 74% of establishments manufacturing active pharmaceutical ingredients for the U.S. market were located overseas [8]. A significant share of those are in China and India. This matters for several reasons. First, both countries are subject to geopolitical risks that could interrupt exports with little warning. Trade tensions, regulatory actions, or diplomatic disputes can restrict the flow of APIs at the border. Second, natural disasters and disease outbreaks disproportionately affect concentrated production regions. The COVID-19 pandemic demonstrated how a single country’s lockdown policies could simultaneously reduce API supply and surge demand for certain drugs.
Third, and perhaps most important for day-to-day supply chain management, regulatory oversight of these overseas facilities is structurally weaker than for domestic sites. The FDA must apply for visas to conduct inspections, is often required to give advance notice, and in crisis conditions has to triage its inspection capacity to ‘mission-critical’ sites [8]. The practical result is that quality problems at overseas facilities can persist undetected longer than they would at domestic sites.
The 2008 heparin contamination crisis, which resulted in 149 deaths, originated at a Chinese API supplier [8]. The N-nitrosamine contamination in angiotensin II receptor blockers (ARBs) that triggered widespread recalls beginning in 2018 also involved overseas manufacturing. Neither crisis was inevitable, but both were made more likely by the structural conditions of concentrated overseas sourcing and limited regulatory reach.
6. Regulatory Complexity and the Compliance Cost Spiral
Generic drug manufacturers operate under a regulatory regime that is among the most demanding in any manufacturing industry. Good Manufacturing Practice (GMP) requirements, ANDA submission standards, Drug Supply Chain Security Act (DSCSA) serialization requirements, and post-market reporting obligations all impose real costs.
Those costs fall disproportionately on smaller manufacturers and on manufacturers competing in low-margin categories. A large generic company with scale efficiencies and a diversified portfolio can absorb compliance costs across many products. A smaller company with a narrow product line in a commodity generic category has fewer options. The result is consolidation — fewer, larger manufacturers dominating the generic landscape — which reduces supply redundancy even as it improves average quality management capability.
The DSCSA requirements deserve particular attention. Phased in over a decade, the DSCSA requires serialization of individual drug packages, electronic data interoperability using Electronic Product Code Information Services (EPCIS), and verification mechanisms throughout the supply chain [9]. Full compliance requires technology investment across all trading partners, and smaller pharmacies and distributors have faced significant implementation challenges. The FDA has extended compliance deadlines multiple times, reflecting genuine industry difficulty with the requirements rather than lax enforcement.
International regulatory divergence adds another layer of complexity. A generic manufacturer selling in both the U.S. and EU markets must meet FDA requirements for ANDA approval and EMA requirements for European market authorization, which differ in meaningful ways including bioequivalence study design standards. Harmonization efforts, including the ICH M13A guideline, aim to reduce duplication but progress is slow [10].
7. The Invisible Equivalence Problem
Generic drugs are bioequivalent to their branded counterparts by definition — that is what regulatory approval requires. But bioequivalence is established in clinical pharmacokinetic studies measuring drug exposure in the bloodstream. It does not directly measure everything that matters clinically.
Patients and prescribers sometimes report differences in tolerability, side effect profiles, or perceived effectiveness when switched between generic manufacturers’ products. Some of these reports reflect pharmacological reality — particularly for narrow therapeutic index drugs like warfarin, levothyroxine, or certain anticonvulsants — and some reflect nocebo effects or other perceptual factors. The scientific debate about when inter-manufacturer substitution is genuinely problematic is ongoing and unresolved.
What is clear is that the current market structure makes quality differences ‘invisible’ to purchasers. Once multiple manufacturers have FDA approval, PBMs, pharmacy chains, and hospital systems select primarily on price. The specific manufacturer supplying a product can change from one dispensing cycle to the next without any notification to the prescribing physician or patient. Quality differences that are real but sub-clinical may never be detected at the individual patient level, even if they manifest statistically across large populations [8].
This is a legitimate concern for drug categories where formulation precision matters. The 2018 ARB recalls, for instance, revealed that manufacturing process variations at multiple overseas plants had introduced carcinogenic N-nitrosamine impurities that were not part of original regulatory submissions and were not being monitored. The contamination persisted for years before testing identified it [8].
Part III: The Operational Layer — Where Efficiency Gets Lost
8. Demand Forecasting: The Spreadsheet Isn’t Enough
Accurate demand forecasting is foundational to everything else in the supply chain. Too little inventory means patients go without medication. Too much means capital tied up in product that may expire before it can be dispensed.
Generic drug demand is particularly difficult to forecast. It is influenced by brand-to-generic conversion rates, competitor market entry, seasonal disease patterns, prescriber behavior, formulary changes by PBMs, and population health dynamics. Traditional forecasting methods that rely on historical sales data alone consistently underperform in this environment, particularly when the input data is itself shaped by past supply constraints [11].
The consequences of forecast error are not symmetric. Stockouts directly harm patients. Excess inventory incurs carrying costs and expiration risk. For temperature-sensitive products, excess inventory that expires represents a double loss: the product cost and the cold chain cost. For injectable generics produced in sterile manufacturing environments — already among the most expensive products to make — the cost of a forecasting error is substantial.
A 2024 AI-driven pharmacy chain implementation achieved 94% forecast accuracy while reducing stockouts by 15% during a peak cold and flu season [12]. A hospital network deploying AI inventory optimization cut holding costs by 23% while simultaneously reducing emergency orders by 35% [12]. These are not marginal improvements; they represent meaningful P&L impact for organizations operating at thin margins.
Advanced Planning and Scheduling (APS) systems have demonstrated 30% lead time reductions and 25% decreases in inventory carrying costs in pharmaceutical applications [13]. The technology is available. The barrier is organizational — generic drug manufacturers, many of them mid-sized companies with legacy ERP systems, frequently lack the data infrastructure to feed these systems effectively.
9. Temperature Control Failures: $35 Billion Per Year in Smoke
Cold chain failures cost the pharmaceutical industry an estimated $35 billion annually, with approximately one-third of compromised shipments destroyed outright rather than salvaged [14]. The environmental cost compounds the financial one: producing and shipping replacement product can double the greenhouse gas emissions associated with a single shipment.
The problem is not that the technology to monitor temperature-sensitive shipments doesn’t exist — IoT-enabled temperature sensors are widely available and relatively inexpensive. The problem is that real-time monitoring data is frequently siloed within individual logistics providers, not integrated across the full chain of custody. A shipment may be perfectly monitored by the manufacturer’s 3PL partner but then transferred to a last-mile carrier without continuity of monitoring. Temperature excursions that occur in the final stretch of delivery — the point at which the supply chain is most fragmented and least visible — go undetected until the product reaches the pharmacy.
Predictive analytics in cold chain management has reduced temperature excursions by up to 30% in pilot programs [13]. Optimal safety stock models built around cold chain reliability data are expected to reduce stockouts by 85% while cutting overall inventory costs by 30% [13]. The ROI on cold chain technology investment is straightforward in the sectors where product values are high; the challenge is making the case for full-chain monitoring in lower-value generic categories where per-unit margins are minimal but aggregate impact is large.
10. Inventory Shrinkage, Expiration, and Recall Readiness
Pharmaceutical products are theft targets. They are high-value, portable, and in significant demand. Manual inventory processes and outdated tracking systems create the conditions for shrinkage that is not discovered until reconciliation periods, by which point the loss is difficult to trace and the regulatory exposure has already materialized.
Expiration management is equally demanding. Every drug dispensed beyond its expiration date is both a patient safety issue and a regulatory liability. For pharmacies managing thousands of SKUs across multiple manufacturers, manual expiration tracking is unreliable. Automated systems that flag approaching expiration dates and trigger reorder or return processes are necessary but not universally deployed, particularly among independent pharmacies.
Recall readiness is where many organizations are most unprepared. A recall requires the ability to identify, within hours, exactly which batches of a product are affected, where they are physically located across the supply chain, and which patients received the product. Without lot-level traceability integrated across all trading partners, that process takes days or weeks — time during which patients may continue receiving potentially unsafe medication.
The DSCSA was designed to address this gap through serialization and interoperable data systems, but the timeline to full implementation has been extended repeatedly. Until lot-level traceability is universal and connected, recall speed will remain a significant operational risk for the industry.
Part IV: The Playbook — What the Best Operators Actually Do
11. Multi-Sourcing: The Most Direct Fix for Concentration Risk
The most immediate way to reduce geographic concentration risk is to source from multiple suppliers in multiple geographies. This is obvious in principle and difficult in practice, primarily because of cost and qualification time.
Qualifying a new API supplier requires analytical testing, audit visits, regulatory submissions, and often stability data that takes 12 to 24 months to generate. For a generic drug with thin margins, the cost of maintaining a second or third qualified supplier can appear to exceed the expected risk reduction. That calculation looks different after a shortage.
Multi-sourcing models work best when they are built proactively, before a disruption creates urgency. Organizations that have already developed multi-tier supply networks with pre-screened supplier profiles can switch sources significantly faster than those responding to a crisis without an existing alternative [6]. The upfront investment in qualification is essentially an insurance premium — paid regularly and invisibly when nothing goes wrong, essential and retrospectively obvious when it does.
Geographic diversification complements multi-sourcing. Reducing concentration in China and India means actively developing qualified suppliers in other regions, including domestic U.S. suppliers, European manufacturers, and emerging markets that are building pharmaceutical manufacturing capacity [6]. This is expensive. The unit cost of API from a domestic U.S. manufacturer is typically higher than from an Asian supplier. But the risk-adjusted cost, accounting for supply disruption probability and impact, is often competitive — particularly for products where a shortage would be clinically severe.
The U.S. government has begun to recognize this. Policy discussions around reshoring pharmaceutical manufacturing have accelerated since COVID-19, and several initiatives aim to incentivize domestic API production through tax benefits, grant programs, and long-term government purchasing commitments [15]. The FDA’s Essential Medicines List and related policy work reflect an acknowledgment that market forces alone will not produce the supply redundancy the public health system needs.
12. Strategic Buffer Stock: How Much Is Enough?
Buffer stock — inventory held above immediate demand to absorb supply disruption — is a well-understood concept whose application in the pharmaceutical supply chain is inconsistent and often inadequate.
For many generic drugs, the entire supply chain — from raw material to dispensed product — operates with 60 to 90 days of inventory at best [16]. That seems substantial until you realize that an API quality problem, a manufacturing facility shutdown, or a logistics disruption can take six months or more to resolve. A three-month buffer against a six-month disruption is a three-month shortage.
Mathematical optimization models for safety stock in pharmaceutical supply chains have to account for demand variability, lead time variability, and the cost of a stockout — which in drug supply is not just lost sales but patient harm, including medication errors, adverse events, and treatment delays [16]. When those patient harm costs are quantified and included in the model, the optimal safety stock level is almost always higher than what most commercial decisions produce.
Optimal safety stock models, when properly calibrated, are projected to reduce stockouts by 85% while decreasing overall inventory costs by 30% simultaneously [13]. The paradox is that holding more of the right inventory can reduce total inventory costs if it prevents the expensive emergency procurement and expedited shipping that stockouts trigger.
Hospital systems and large integrated delivery networks (IDNs) are better positioned than independent pharmacies to maintain buffer stock, because they have the storage capacity, capital, and scale to absorb carrying costs. Policy proposals to provide financial incentives for hospitals to maintain strategic reserves for essential drugs are worth taking seriously precisely because they would address the structural mismatch between who can hold buffer stock and who currently does.
13. Lean Manufacturing in the Generic Drug Plant
Lean manufacturing principles — eliminating waste, implementing pull-based production, reducing work-in-process inventory, and shortening cycle times — have a long track record in automotive and other discrete manufacturing industries. Their application in pharmaceutical manufacturing is more recent and more complex, largely because of regulatory constraints that limit rapid process changes.
But where lean has been applied thoughtfully in generic drug manufacturing, the results are significant. An Indian generic startup applying lean principles increased production output by 50% without adding floor space or major capital equipment [17]. Case studies in conventional tablet manufacturing show reductions of 30-40% in cycle times and meaningful improvements in equipment utilization through structured waste reduction programs [18].
The barriers to lean adoption in pharmaceutical manufacturing are partly cultural. Pharmaceutical plants historically valued stability over efficiency, and process change — even clearly beneficial change — required regulatory notification and sometimes prior approval. The FDA has worked to create pathways for post-approval manufacturing changes that are less burdensome, but regulatory caution about process variation remains a powerful conservative force in the industry.
The most productive applications of lean in generic manufacturing tend to focus on changeover time reduction (critical in facilities making many different products), batch scheduling optimization, and quality systems improvements that reduce rework and batch failure rates. These areas offer improvement without requiring regulatory submissions for process changes.
14. AI and Machine Learning: Where the Real Gains Are
Artificial intelligence in pharmaceutical supply chains is generating a great deal of coverage, much of it vague. Let us be specific about where the evidence actually supports meaningful gains.
Demand forecasting is the most mature application. ML algorithms that incorporate epidemiological data, prescription trends, formulary changes, and competitive ANDA filing data — the kind of data tracked by DrugPatentWatch — consistently outperform statistical time-series methods. A 94% forecast accuracy rate achieved by a major pharmacy chain using AI exceeded previous performance by a substantial margin [12]. The mechanism is straightforward: ML can process far more input variables than traditional forecasting models and can identify non-linear relationships that humans and conventional statistics miss.
Predictive maintenance is the second well-validated application. Manufacturing equipment failure is a significant source of production disruption in pharmaceutical plants, where scheduled downtime for calibration and cleaning is already substantial. IoT sensors monitoring equipment vibration, temperature, pressure, and other parameters feed ML models that predict imminent failures before they cause unplanned downtime. Merck reported a 20% improvement in production uptime after implementing IoT-based predictive maintenance across its manufacturing network [19].
Quality control vision systems are producing impressive results in solid dose manufacturing. High-resolution cameras with deep learning algorithms can inspect hundreds of tablets per minute, detecting physical defects — cracks, chips, color variations, coating defects — with an accuracy of 99.2% in documented implementations [12]. That is far superior to manual inspection and substantially reduces batch rejection rates, which in some facilities have dropped by 35% [12].
Route optimization for distribution is well-established in other industries and is being applied with good effect in pharmaceutical last-mile logistics. AI routing that incorporates real-time traffic data, temperature constraints, delivery windows, and regulatory requirements consistently outperforms static routing plans, particularly for specialty drug distribution where delivery windows are clinically important [12].
What AI cannot yet do well in pharmaceutical supply chains is make strategic decisions about supplier relationships, respond to novel disruptions for which there is no historical training data, or substitute for regulatory judgment. Organizations that understand these limitations and apply AI to well-defined operational problems where good training data exists will capture real value. Organizations that treat AI as a general solution to supply chain complexity will be disappointed.
15. Blockchain for Traceability: Past the Hype, Into Specifics
Blockchain in pharmaceutical supply chains has been discussed for nearly a decade. Early implementations often struggled with scalability, integration complexity, and the fundamental challenge that blockchain cannot make off-chain data reliable — the ‘garbage in, garbage out’ problem applies to distributed ledgers as much as to centralized databases.
The most compelling current applications are narrower and more specific than early visions of a fully blockchain-enabled pharmaceutical supply chain. Serialization data management — recording the creation, transfer, and dispensing of serialized drug packages in a manner that is tamper-evident and auditable — is a natural fit for blockchain architecture. The immutability of the ledger means that once a serialization event is recorded, it cannot be altered without detection, which is precisely what anti-counterfeiting and recall readiness require [20].
A major pharmaceutical company implementing a blockchain-based drug authentication system reduced counterfeit drug incidents by 47% across its distribution network [12]. Blockchain implementations in European markets have reduced compliance delays by 25% by automating the verification of trading partner credentials and product authentication [17].
The DSCSA interoperability requirements create a powerful regulatory driver for blockchain adoption. The Act requires that serialization data flow electronically between all trading partners, and blockchain provides one architecture for that interoperability that avoids requiring all parties to use the same central database [9]. Whether blockchain proves to be the dominant architecture for DSCSA compliance or whether other interoperability approaches prevail, the underlying requirement for electronic lot-level traceability across the supply chain is not going away.
16. Real-World Data: The Most Underused Intelligence Source
Real-World Data — patient-level data from claims databases, electronic health records, and pharmacy dispensing records — has well-established applications in pharmacovigilance and outcomes research. Its applications in supply chain management are less developed but potentially as valuable.
RWD can map geographic demand patterns for specific drugs in ways that prescription-level sales data cannot, because it reflects actual patient utilization rather than what distributors ordered. During a shortage, that difference matters. Distributor ordering patterns can reflect hoarding behavior, just-in-case purchasing, and allocation gaming that distorts the signal. Patient-level dispensing data tells you where patients are actually receiving — or failing to receive — specific medications [21].
RWD can also identify supply disruption signals earlier than conventional inventory reports. A statistically significant increase in therapeutic substitutions — patients being switched from their usual medication to an alternative — is a leading indicator of shortage conditions, sometimes visible weeks before distributor reports acknowledge a supply problem [21].
For generic manufacturers considering market entry or exit decisions, RWD provides a ground-truth picture of actual patient utilization that complements the patent expiration and competitive ANDA data available through platforms like DrugPatentWatch. Combining IP intelligence with utilization intelligence gives a more complete picture of commercial opportunity and risk.
Part V: Logistics, Distribution, and the Last Mile
17. The 3PL Relationship: Beyond Transactional Contracting
Third-party logistics providers manage the physical distribution of most generic drugs in the U.S. market. The relationship between generic manufacturers and 3PLs is often treated as a commodity procurement decision — price, coverage, and basic service levels — rather than as a strategic partnership.
That approach costs money and introduces risk. Lupin Pharmaceuticals changed this calculus when it partnered with UPS Healthcare to restructure its entire U.S. generic drug supply chain. UPS completed the transition in seven weeks — against a typical timeline of up to five months — moving 5,000 pallets with minimal disruption [22]. The partnership gave Lupin access to UPS’s specialized pharmaceutical warehousing, temperature monitoring, and distribution network. By converting 6% of its air freight volume to ground transport at equivalent delivery speed, Lupin reduced transportation costs by $0.50 per pound [22]. Speed-to-market for new product launches improved. Back-orders declined.
The Lupin-UPS example illustrates what a genuinely strategic 3PL relationship can deliver: not just physical movement of product but active inventory management, demand signal integration, and operational agility that a manufacturer building those capabilities internally would require years and significant capital to develop.
The supply chain question for a generic manufacturer is not ‘what is the cheapest 3PL?’ but ‘which logistics partner creates the most defensible operational position in markets where speed and reliability are differentiators?’ That is a strategy question, not a procurement question.
18. Good Distribution Practice: The Baseline That Isn’t Always Met
Good Distribution Practices (GDP) define the minimum requirements for the proper distribution of medicinal products: storage conditions, chain of custody documentation, temperature maintenance, product authentication, and handling of returns and recalls. In Europe, GDP is a formal regulatory standard. In the U.S., equivalent requirements flow through GMP regulations and the DSCSA.
Meeting GDP is the baseline, not the goal. Organizations that treat GDP compliance as their distribution quality objective are managing to the floor. The operational question is what above-baseline practices are worth investing in, given that generic drug margins constrain what is affordable.
The answer depends heavily on the product category. Temperature-sensitive generics — and the category is growing as more complex drug products reach generic approval — require real-time monitoring, qualified containers, and validated shipping lanes. Failure to maintain these controls produces product losses and potential patient harm that dwarf the cost of the controls themselves. For room-temperature solid dose generics with high production volumes and low per-unit values, the economic calculus is different, and the monitoring infrastructure can be proportionally simpler.
What GDP compliance and genuine quality distribution practice share is the need for documentation that is complete, accurate, and retrievable on demand. In a shortage, a recall, or a regulatory inspection, the ability to reconstruct the chain of custody for every unit is not optional.
19. Innovating at the Last Mile
Last-mile delivery in pharmaceutical distribution has historically been the least technologically advanced segment of the chain. The product travels through sophisticated global supply networks and then gets delivered by a pharmacy technician counting tablets into a bottle by hand.
Direct-to-consumer models and online pharmacy platforms are changing the last-mile calculus. Mail-order pharmacy, long established for maintenance medications, is growing. Specialty pharmacy models with home delivery are expanding into categories previously limited to hospital or clinic dispensing. These channels have different operational requirements than traditional retail pharmacy — higher individual delivery precision, tighter temperature control in transit, and more sophisticated patient adherence support — but they also offer data capture opportunities that traditional retail cannot match [23].
AI-powered route optimization for pharmaceutical delivery is reducing delivery times and operational costs for organizations that have implemented it [12]. For specialty drugs and biosimilars, where delivery windows and temperature controls are critical, optimized routing is not an efficiency nice-to-have but a product integrity requirement.
The supply chain visibility opportunity in last-mile pharmaceutical delivery is significant and largely untapped. Real-time tracking at the individual shipment level, integrated with pharmacy dispensing systems, would enable the kind of end-to-end visibility that the DSCSA envisions but that current systems struggle to deliver in practice.
Part VI: Policy, Collaboration, and the Systemic Fixes
20. What the Policy Debate Is Actually About
The policy debate around pharmaceutical supply chain resilience is frequently framed as a choice between domestic manufacturing mandates and free-market efficiency. That framing is unhelpful because it treats resilience and cost as inherently opposed, which they are not over any time horizon that accounts for shortage costs.
A more productive framing asks: what specific market failures cause the generic supply chain to under-invest in resilience, and what policy interventions would correct those failures at acceptable cost?
The answers point to three specific interventions. First, reimbursement reform: MAC pricing and similar mechanisms that cap generic drug prices below a level sustainable for quality investment need to be modified for drugs with concentrated supply or shortage histories [4]. This does not mean allowing manufacturers to price generics freely; it means recognizing that the lowest possible price is not the only relevant variable when public health is downstream of the supply decision.
Second, regulatory cost reduction targeted at resilience investments: the FDA’s Generic Drug User Fee program (GDUFA) charges application fees that are significant for smaller manufacturers. Reducing or restructuring these fees for manufacturers who demonstrate supply chain resilience — through domestic production, multi-source qualification, or buffer stock commitments — creates a direct financial incentive for the behavior the public health system needs [4].
Third, increased inspection capacity: the FDA’s overseas inspection program remains understaffed relative to the volume of overseas manufacturing it is responsible for oversight. Increased funding for overseas inspections, combined with unannounced audit programs like those already in use domestically, would reduce the inspection advantage that overseas facilities currently have in preparing for regulatory visits [8].
These are not politically easy recommendations. Reimbursement reform requires negotiating with PBMs and public payers. Regulatory cost reduction requires FDA budget decisions. Increased inspection requires congressional appropriations. But they address actual market failures, not symptoms.
21. Public-Private Partnerships: What Works and What Doesn’t
Government-industry partnerships to support domestic pharmaceutical manufacturing have a mixed track record. Some have succeeded in building manufacturing capacity that would not otherwise exist. Others have subsidized capacity that was not actually needed, at prices that were not actually sustainable.
The partnerships most likely to succeed share several characteristics. They are structured around long-term purchasing commitments rather than one-time grants, because predictable revenue is what makes capital investment viable. They include quality and performance requirements that prevent recipients from claiming incentives without delivering reliable supply. They are targeted at specific drugs or drug categories where supply concentration creates genuine public health risk, rather than broadly subsidizing domestic manufacturing regardless of strategic need.
The Biomedical Advanced Research and Development Authority (BARDA) has developed public-private partnership structures for pandemic preparedness that offer useful templates. BARDA commitments to purchase specific quantities of a product at specified prices, contingent on the partner maintaining FDA compliance and production readiness, create the kind of revenue certainty that pharmaceutical manufacturers need to justify domestic facility investment.
Similar structures could be applied to essential generic medicines where supply concentration creates unacceptable shortage risk. A government commitment to purchase, say, six months of national consumption of a specific sterile injectable generic at a price sufficient to support domestic production would create a viable business model for domestic manufacturing that the unsubsidized generic market currently cannot support.
22. The Collaboration Imperative: Sharing Data Without Sharing Competitively Sensitive Information
The generic drug supply chain is a network of organizations that are simultaneously interdependent and competitive. Manufacturers compete against each other for the same market. Distributors compete for manufacturer relationships. Pharmacies compete for patient business. This competition is healthy and creates the efficiency benefits that generics deliver.
But it also creates hoarding behavior, information asymmetry, and coordination failures that worsen supply disruptions. During the early stages of the COVID-19 pandemic, multiple distributors simultaneously over-ordered key drugs in anticipation of shortage. The result was that available supply was absorbed into pipeline inventory rather than reaching patients, creating shortage conditions before any actual production shortfall had occurred [16].
Better information sharing — specifically, aggregate demand signals and pipeline inventory data — would allow the system to coordinate more effectively without requiring manufacturers or distributors to reveal individually competitively sensitive information. The FDA’s drug shortage early warning system moves in this direction but is incomplete. Voluntary information sharing programs exist but participation is inconsistent.
Technology platforms that aggregate and anonymize supply chain data from multiple participants, providing market-level visibility without revealing individual company positions, are a promising approach. The precedent exists in other industries where pre-competitive data sharing has improved market function without compromising commercial competition.
23. Sustainability, ESG, and the Supply Chain
The pharmaceutical industry’s carbon footprint is substantial. Approximately 4.4% of the industry’s emissions come from supply chain activities [24]. Cold chain operations are a significant contributor: refrigerated transport is energy-intensive, and temperature excursions that lead to product destruction add the carbon cost of replacement production.
Sustainability investments and supply chain resilience investments often target the same operational failures. Reducing cold chain failures reduces both waste and emissions. Optimizing transport routes reduces both cost and fuel consumption. Reducing packaging weight and converting to recyclable materials reduces both shipping costs and environmental impact — one analysis showed packaging optimization can reduce shipping costs by up to 10% [24].
This alignment means that ESG commitments and supply chain efficiency goals, sometimes framed as competing priorities, are often mutually reinforcing in pharmaceutical logistics. A manufacturer that invests in real-time cold chain monitoring to prevent temperature excursions simultaneously improves product quality, reduces waste, and reduces emissions. A distributor that optimizes routing simultaneously cuts costs and reduces fuel use.
The ESG dimension also affects procurement decisions increasingly. Large hospital systems and integrated delivery networks are beginning to incorporate supplier sustainability metrics into their contracting processes, alongside traditional quality, compliance, and cost criteria. Generic manufacturers that can credibly demonstrate supply chain sustainability performance will have an advantage in those procurement processes that goes beyond the current price-dominant framework.
Part VII: The Competitive Intelligence Dimension
24. Patent Intelligence as a Supply Chain Tool
Patent expiration is the event that creates the generic drug market opportunity. Understanding exactly when and how a patent expires — which claims are most commercially significant, whether there are secondary patents that could extend market exclusivity beyond the primary patent date, and how many other manufacturers are positioned to file ANDAs — is the foundation of every viable generic drug development program.
DrugPatentWatch provides detailed patent expiration data, FDA Orange Book information, ANDA filing history, and market exclusivity tracking that pharmaceutical companies use to make these strategic entry decisions. But patent intelligence is not only a pre-launch planning tool. It is also a supply chain management input.
When a new generic competitor gains ANDA approval, the competitive landscape changes rapidly. Additional entrants drive prices down further, which compresses margins, which forces manufacturers to make cost reduction decisions that often affect supply chain investment. Patent surveillance data lets manufacturers anticipate competitive entry and plan supply chain adjustments proactively rather than reactively.
The relationship runs in the other direction too. Supply chain data can inform patent strategy. A manufacturer monitoring a competitor’s supply chain problems — production site issues, API supplier changes, FDA warning letters — has intelligence that informs both competitive positioning and supply chain risk planning. If a major supplier of a generic API is facing regulatory action, manufacturers who source from that supplier need to know before their supply is interrupted, and DrugPatentWatch’s tracking of regulatory actions and market dynamics provides relevant context.
25. Market Entry Timing and Supply Chain Readiness
The economics of generic drug market entry are heavily front-loaded. The first manufacturer to achieve ANDA approval and commercial supply may capture a 180-day exclusivity period that generates most of the commercial value of a generic product’s lifetime. Being second or third to market is significantly less valuable. Being late to market because of supply chain failures is economically catastrophic.
This creates an interesting interaction between supply chain capability and competitive strategy. Manufacturers who can reliably bring commercial supply to market within weeks of ANDA approval capture far more value than those who need months to establish distribution relationships, qualify API suppliers, and build up inventory. Supply chain readiness is a competitive differentiator, not just an operational function.
The organizations that consistently capture first-to-market advantages in generic drugs have typically made the supply chain investments — pre-qualified suppliers, established distribution relationships, built-up inventory of API — before regulatory approval, betting that approval will come and that the supply chain cost of early preparation is worth the revenue upside of fast commercial launch. That is a calculated risk, but it is calculable, and the upside of getting it right is substantial.
Part VIII: The Numbers
26. Quantifying the Stakes
The financial scale of the generic drug supply chain makes the investment case for improvement straightforward, even if individual ROI calculations require company-specific data.
Start with the waste: cold chain failures alone cost the industry $35 billion per year [14]. If real-time monitoring and predictive analytics can reduce temperature excursions by 30%, that is more than $10 billion in annual waste that becomes recoverable product and averted replacement cost.
Add inventory carrying costs: a hospital network that reduced inventory holding costs by 23% using AI-driven optimization was capturing real working capital [12]. At the scale of the U.S. hospital system — which spends roughly $40 billion per year on pharmaceuticals — a 23% reduction in inventory costs across even a subset of products represents billions of dollars.
Consider recall costs: product recalls in the pharmaceutical industry routinely cost manufacturers $500 million to $1 billion per event, including direct product write-offs, remediation costs, and revenue loss during supply interruption [25]. A 70% reduction in product recalls, attributable to real-time visibility platforms, is not a minor efficiency gain [26].
Factor in shortages: drug shortages cost the U.S. healthcare system an estimated $230 million annually in direct costs — expedited procurement, therapeutic substitution, additional clinical management — and the indirect costs, including patient harm and associated liability, are substantially higher [7].
The aggregate financial opportunity from supply chain optimization in generic drugs is measured in tens of billions of dollars annually. The investment required to capture a meaningful share of it — in data infrastructure, supplier qualification, monitoring technology, and analytics capability — is significant but dwarfed by the potential return.
27. Case Studies That Actually Tell Us Something
Three case studies deserve closer examination because they illustrate different dimensions of supply chain improvement and are specific enough to draw conclusions from.
Lupin-UPS Healthcare is the most instructive example of what a transformed logistics partnership looks like operationally. Lupin, importing thousands of pallets of generic pharmaceuticals annually, faced the operational complexity common to mid-sized generic manufacturers: fluctuating demand, multi-market distribution, tight launch timelines. The seven-week transition of its entire U.S. supply chain to UPS Healthcare — against a five-month industry standard — required intensive coordination but produced measurable results: eliminated back-orders, faster new product launches, and a $0.50 per pound reduction in transportation cost through mode shift [22]. The case demonstrates that supply chain transformation does not require years; it requires the right partner and the organizational will to move quickly.
Merck’s IoT-powered factories illustrate what predictive maintenance actually delivers in a real manufacturing environment. IoT sensors monitoring equipment parameters feed analytics models that flag impending failures before they cause unplanned downtime. The result was a 20% improvement in production uptime [19]. In a capital-intensive pharmaceutical manufacturing environment where a single product line going down can trigger shortage conditions, 20% uptime improvement is not just a financial win — it is a supply reliability win.
Novartis’s digital twin adoption shows how manufacturing process innovation can accelerate tech transfer without requiring physical trials. Digital twins simulate production processes computationally, allowing engineers to test process changes, troubleshoot quality problems, and validate scale-up decisions before committing to physical production runs [19]. The time and cost savings from avoiding failed scale-up attempts are significant. The supply chain implication is shorter lead times from formulation change to commercial supply.
These are not edge cases or aspirational examples. They are documented outcomes from organizations that made specific technology and partnership investments. The pattern across all three is the same: targeted investment in a specific operational problem, with measurable returns that justify the investment within a defined timeframe.
Part IX: Looking Forward
28. Where the Industry Is Heading
The generic drug supply chain in 2030 will look substantially different from the one that exists today. Three structural changes are already in motion.
First, domestic manufacturing capacity in the U.S. will grow, driven by a combination of policy incentives, geopolitical risk awareness, and technological changes that make domestic production more economically viable. Advanced manufacturing technologies — continuous manufacturing, novel synthesis routes, AI-driven process optimization — are narrowing the cost gap between high-cost domestic production and low-cost overseas production [15]. Not all API categories will shift, but strategically critical ones will, and the supply chain implications are significant.
Second, data infrastructure will become a competitive requirement rather than a competitive advantage. The DSCSA interoperability requirements, full implementation of serialization, and the growing use of AI-driven supply chain management tools all require data systems that are integrated, real-time, and interoperable. Organizations that delay this investment will face mounting compliance risk and operational disadvantage. The window to build these capabilities proactively, rather than reactively under regulatory pressure, is narrowing.
Third, ESG considerations will be embedded in procurement and contracting across the supply chain. Large health system purchasers are already beginning to incorporate environmental and social governance performance into supplier evaluation. Generic manufacturers that have invested in sustainable supply chain practices — cold chain efficiency, packaging optimization, supplier labor standards — will have an advantage in these procurement processes that compounds over time.
29. The Regulatory Evolution
The FDA’s Center for Drug Evaluation and Research (CDER) has signaled through multiple policy initiatives that it intends to continue raising the bar on manufacturing quality and supply chain resilience for generic drugs. The CDER’s quality management maturity initiative, which encourages manufacturers to adopt sophisticated quality management systems and report quality performance data to the FDA, is an early step toward a tiered quality recognition system that could eventually influence market access or reimbursement.
International regulatory harmonization will continue, slowly. The FDA-EMA collaborative review program for generic drugs and the ICH M13A bioequivalence guideline represent genuine progress toward reducing the duplication that generic developers face when seeking approval in multiple markets. The practical effect will be faster market access for well-managed generic programs, which benefits manufacturers who have invested in robust regulatory affairs capabilities [10].
The DSCSA interoperability deadline has been extended multiple times, but it will ultimately be enforced. The industry should treat each extension as a window to implement rather than as evidence that the deadline will move again indefinitely. The manufacturers and distributors that achieve full DSCSA compliance early will have a material operational advantage in the market for several years before laggards catch up.
30. The Essential Conversation About Drug Pricing
Any serious discussion of generic drug supply chain resilience eventually arrives at drug pricing, and that conversation is uncomfortable for an industry that has spent decades defending the principle that competitive pricing is the mechanism that makes generics work.
The discomfort is understandable. The same pricing pressure that creates affordability also, as documented above, creates fragility. Acknowledging that connection does not require endorsing price controls or accepting that generic drugs should be expensive. It does require acknowledging that a system optimized solely for the lowest possible price will produce an increasing frequency of shortages and quality failures, and that the public health cost of those outcomes is real.
The policy debate needs to move from ‘how low can we push generic drug prices?’ to ‘what is the price floor that sustains reliable supply of essential medicines?’ Those are different questions with different answers, and the second question has not received adequate attention in policy circles that are focused primarily on the first.
Generic manufacturers, PBMs, hospital systems, and pharmacy chains all have legitimate roles in that conversation. So does the public, which ultimately pays for both the drugs and the shortage costs. The conversation will be easier to have productively if it is grounded in supply chain economics rather than political positioning — which is precisely why detailed, factual analysis of the kind that DrugPatentWatch and similar intelligence platforms enable is valuable to all parties at the table.
Conclusion: The Integrated Imperative
The generic drug supply chain does not have a single broken component. It has a structural misalignment between the incentives that drive commercial behavior and the outcomes the public health system requires. Fixing that misalignment demands changes at every level: individual company operations, industry collaboration, and policy architecture.
At the operational level, the path forward is clear. Multi-sourcing for APIs and finished product. AI-driven demand forecasting and inventory optimization. Predictive maintenance and AI quality control in manufacturing. Real-time cold chain monitoring. Blockchain-based lot-level traceability. None of these is speculative; all have documented performance data. The barrier to adoption is not uncertainty about whether they work. It is organizational capability, capital, and data infrastructure.
At the industry level, pre-competitive information sharing — aggregate demand signals, pipeline inventory visibility, early shortage warning — would reduce the coordination failures that worsen supply disruptions without requiring individual companies to expose competitively sensitive information.
At the policy level, reimbursement reform that reflects the true cost of reliable supply, regulatory incentives for domestic manufacturing and supply chain resilience, and increased FDA oversight capacity for overseas facilities are the interventions most likely to address the structural causes of generic drug shortages.
This is not a call to make generic drugs expensive. It is a call to stop pretending that maximizing supply chain fragility is an acceptable price to pay for minimizing unit cost. The two objectives — affordable generics and reliable generic supply — are not inherently opposed. Achieving both requires smarter policy, better technology, and supply chain investment that goes beyond what the current market structure rewards.
The organizations that will win in the generic drug market over the next decade are those building that capability now.
Key Takeaways
- Generic drugs fill 89% of U.S. prescriptions at 26% of total drug spending, but the pricing mechanism that produces affordability systematically under-funds supply chain resilience.
- Geographic concentration of API manufacturing — 74% of establishments overseas by 2020 — creates regulatory oversight gaps and geopolitical supply risk that is directly traceable to documented drug shortages.
- AI-driven demand forecasting achieves 94% accuracy in documented implementations, reduces stockouts by 15%, and cuts inventory holding costs by 23%. These are available, proven tools, not aspirational technologies.
- Cold chain failures cost the industry $35 billion annually. Real-time IoT monitoring and predictive analytics can reduce temperature excursions by 30% and make a significant portion of that cost recoverable.
- Multi-sourcing for APIs and finished product is the single most direct structural fix for concentration risk. The upfront qualification cost is an insurance premium, not a sunk cost.
- Blockchain-based serialization reduces counterfeit incidents by documented margins of 47% in corporate implementations and is becoming a regulatory necessity under DSCSA requirements.
- Policy changes — specifically, reimbursement reform that sets a price floor for essential medicines above the level that triggers supply exit, and regulatory fee restructuring tied to resilience investment — would address root causes rather than symptoms.
- ESG investments and supply chain resilience investments often target the same operational failures. They are complementary, not competing, objectives.
FAQ
Q1: Why do generic drug shortages keep recurring despite regulatory attention and industry awareness?
The recurrence reflects a structural problem, not a failure of attention. The generic market’s price competition drives margins to levels that don’t support the infrastructure investment needed for resilient supply. Until reimbursement structures create a floor price that reflects the true cost of reliable supply for essential medicines, the economic incentive to under-invest in supply chain redundancy remains intact. Regulatory attention and public concern are insufficient to overcome a market structure that systematically punishes the investments needed to prevent shortages.
Q2: How should a generic manufacturer prioritize between multi-sourcing, AI adoption, and blockchain when capital is limited?
Start with multi-sourcing for your highest-volume and lowest-margin products, because those are the categories most at risk of shortage and the most vulnerable to API disruption. AI-driven demand forecasting and inventory optimization typically generates positive ROI within 12 to 18 months and requires less capital than physical supply chain restructuring. Blockchain investment should be timed around DSCSA compliance requirements — there is no advantage in paying for implementation twice, so align blockchain investment with your DSCSA compliance timeline.
Q3: What does the DSCSA actually require from manufacturers versus distributors versus pharmacies, and when does full compliance kick in?
Manufacturers must serialize individual drug packages using a National Drug Code, lot number, expiration date, and serial number in a 2D barcode. They must transmit Transaction Information (TI), Transaction History (TH), and Transaction Statement (TS) data electronically when transferring product to trading partners. Distributors must verify this data when receiving product, maintain it, and pass it forward. Pharmacies must verify product at point of dispensing. Full electronic interoperability — meaning systems must communicate directly rather than passing paper-based or email-based documentation — is the most demanding requirement and the one with the most extended implementation history. The FDA has indicated it will enforce interoperability requirements, but manufacturers should verify current enforcement timelines directly with FDA guidance documents, as these have been revised multiple times.
Q4: Can patent intelligence from platforms like DrugPatentWatch actually improve supply chain decisions, or is it primarily useful for market entry strategy?
Both. The obvious application is market entry: tracking patent expirations, ANDA filing counts, and exclusivity periods to identify commercial opportunities. The less obvious application is supply chain risk management. When DrugPatentWatch data shows multiple ANDA approvals pending for a drug you manufacture, that is a signal that price compression is coming and that supply chain cost structures need to be reviewed. When regulatory action data suggests a major API supplier is under compliance pressure, that is an early warning to qualify alternative suppliers. Patent and regulatory intelligence is supply chain intelligence, just presented in a different frame.
Q5: How should hospital systems and integrated delivery networks think about their role in generic drug supply chain resilience?
Hospital IDNs are the purchasers whose contracting decisions most directly shape the financial viability of generic drug supply chains. Purchasing decisions that select the absolute lowest-cost supplier regardless of quality history, geographic concentration, or supply chain resilience are contributing to the fragility they later complain about during shortages. IDNs can positively influence supply chain resilience by incorporating supplier quality and resilience metrics into contracting, maintaining strategic buffer stocks for essential medicines in exchange for preferred pricing, participating in voluntary shortage early warning programs, and advocating with payers for reimbursement models that reflect the true cost of reliable supply. None of this requires IDNs to pay more than market price. It requires them to treat supply reliability as a purchasing criterion alongside price.
References
[1] Association for Accessible Medicines. (2024). The generic drug supply chain. https://accessiblemeds.org/resources/blog/generic-drug-supply-chain/
[2] Datex Corporation. (2024). How does the pharmaceutical supply chain work? https://www.datexcorp.com/pharmaceutical-supply-chain-2/
[3] DrugPatentWatch. (2025, August 20). Streamlining the generic drug supply chain: Best practices. https://www.drugpatentwatch.com/blog/streamlining-the-generic-drug-supply-chain-best-practices/
[4] DrugPatentWatch. (2024). Optimizing the generic drug supply chain: Strategies for success. https://www.drugpatentwatch.com/blog/optimizing-the-generic-drug-supply-chain-strategies-for-success/
[5] University of Wisconsin School of Pharmacy. (2025, June 11). The future of generics. https://pharmacy.wisc.edu/2025/06/11/the-future-of-generics/
[6] DrugPatentWatch. (2024). Sourcing key starting materials (KSMs) for pharmaceutical active pharmaceutical ingredients (APIs): A strategic imperative for resilience. https://www.drugpatentwatch.com/blog/sourcing-the-key-starting-materials-ksms-for-pharmaceutical-active-pharmaceutical-ingredients-apis/
[7] U.S. Department of Health and Human Services, ASPE. (2023). Analysis of drug shortages, 2018–2023. https://aspe.hhs.gov/reports/drug-shortages-2018-2023
[8] Chua, K. P., Conti, R. M., & Shrank, W. H. (2021). Price and quality in the generic pharmaceutical market. Circulation, 144(1), 1–3. https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.121.057727
[9] Frontier Scientific Solutions. (2024). Understanding the Drug Supply Chain Security Act (DSCSA). https://www.fs2.com/understanding-the-drug-supply-chain-security-act-dscsa/
[10] RAPS. (2025, February). Expanding global access to complex generics: The case for regulatory convergence. https://www.raps.org/news-and-articles/news-articles/2025/2/expanding-global-access-to-complex-generics-the-ca
[11] PLAIO. (2024). What is demand forecasting? Guide for pharma companies. https://www.plaio.com/articles/what-is-demand-forcasting/
[12] Svitla Systems. (2024). 7 outstanding examples of digital transformation in the pharmaceutical industry. https://svitla.com/blog/digital-transformation-in-the-pharma-industry/
[13] Number Analytics. (2024). Future of supply chain resilience for healthcare and pharma. https://www.numberanalytics.com/blog/future-supply-chain-resilience-healthcare-pharma
[14] Tive. (2024). Pharma sustainability: The top strategies and tactics in pharma supply chains. https://www.tive.com/blog/pharma-sustainability-the-top-strategies-tactics-in-pharma-supply-chains
[15] Pharma Almanac. (2024). Building resilient pharma supply chains in an uncertain world. https://www.pharmasalmanac.com/articles/building-resilient-pharma-supply-chains-in-an-uncertain-world
[16] National Academies of Sciences, Engineering, and Medicine. (2022). Causes and consequences of medical product supply chain disruptions. https://www.ncbi.nlm.nih.gov/books/NBK583734/
[17] Kaizen Institute. (2024). Generic drug production improvement project. https://kaizen.com/insights/pharmaceutical-industry-generic-production/
[18] ResearchGate. (2014). Improvement of manufacturing operations through a lean management approach: A case study in the pharmaceutical industry. https://www.researchgate.net/publication/268377274
[19] Coherent Solutions. (2025). AI in pharma and biotech: Market trends 2025 and beyond. https://www.coherentsolutions.com/insights/artificial-intelligence-in-pharmaceuticals-and-biotechnology-current-trends-and-innovations
[20] Tandfonline. (2022). Blockchain implementation in pharmaceutical supply chains: A review and conceptual framework. https://www.tandfonline.com/doi/full/10.1080/00207543.2022.2125595
[21] PurpleLab. (2024). Decoding the generic supply chain: Why pharma data analytics matters. https://purplelab.com/pharma-data-analytics-generic-drugs/
[22] UPS Healthcare. (2024). Lupin Pharmaceuticals case study. https://es-us.ups.com/us/en/healthcare/learning-center/case-studies/lupin-pharmaceuticals
[23] Express It Delivery. (2024). How to optimize pharmaceutical distribution and delivery. https://expressitdelivery.com/blog/ways-to-optimize-pharmaceutical-distribution/
[24] PCI CDMO. (2024). Sustainable pharmaceutical supply chains. https://pci.com/resources/sustainable-pharmaceutical-supply-chain/
[25] DrugPatentWatch. (2024). The role of quality assurance in generic drugs. https://www.drugpatentwatch.com/blog/the-role-of-quality-assurance-in-generic-drugs/
[26] ScikIQ. (2024). Real-time visibility in supply chain management is an edge. https://scikiq.com/blog/how-emerging-technologies-are-driving-real-time-visibility-in-the-supply-chain/