What Canada’s Patent Linkage System Actually Does
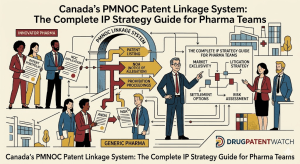
Canada ties the regulatory approval of generic and biosimilar drugs directly to the resolution of patent disputes. That single sentence describes the entire architecture of the Patented Medicines (Notice of Compliance) Regulations — the PM(NOC) Regulations — enacted under Section 55.2(4) of the Patent Act. A generic manufacturer that files a regulatory submission referencing an innovator’s drug cannot receive a Notice of Compliance (NOC) from Health Canada until it satisfies the patent obligations tied to every patent listed on the Patent Register against that reference product.
The system does not attempt to pre-adjudicate the validity or infringement of every listed patent before a generic enters the market. It creates a procedural gate: if an innovator files a court action within 45 days of receiving a Notice of Allegation (NOA), a 24-month statutory stay on generic approval activates automatically. The stay runs whether or not the innovator’s patents survive scrutiny on the merits. That asymmetry — a stay granted on the act of filing, not the strength of claims — is the central policy lever that pharma IP teams need to understand when valuing a Canadian patent portfolio or modeling generic entry timelines.
Key Takeaways: Section Overview
- The PM(NOC) Regulations are the sole statutory mechanism linking patent protection to Health Canada regulatory approval.
- The 24-month automatic stay is procedural, not merits-based.
- Patent listing on the Health Canada Patent Register is a prerequisite for any stay right.
- Biosimilars are nominally within the system’s scope but lack several of the structural protections small-molecule generics face.
Legislative Architecture: PMNOC Regulations Under the Patent Act
Historical Origin and the Hatch-Waxman Divergence
Canada’s patent linkage framework emerged in 1993, modeled broadly on the U.S. Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman). The architects copied the linkage concept — tie regulatory approval to patent clearance — but rejected the Orange Book’s broader patent eligibility rules and the 30-month stay’s longer duration. The result was a system that looked superficially similar to Hatch-Waxman but produced materially different litigation incentives from the outset.
The original PMNOC Regulations used summary proceedings before the Federal Court, modeled on judicial review rather than full patent infringement actions. Summary proceedings were fast in theory but generated perverse outcomes in practice. Evidentiary records were thin, expert witnesses rarely testified live, and the dual-track system meant innovators could lose a PMNOC summary proceeding and then relitigate the same patents in a separate infringement action under the Patent Act. Generic manufacturers faced years of cumulative uncertainty even after winning in the first forum. That structural dysfunction — profitable for Bay Street litigators, costly for generic entry timelines and, by extension, public drug budgets — is what drove the 2017 overhaul.
The September 2017 Amendments: What Changed
The amendments that took effect September 21, 2017, were Canada’s implementation of pharmaceutical IP obligations under CETA’s Article 20.28, which required Canada to provide an ‘effective and non-discriminatory’ mechanism for patent dispute resolution during the marketing authorization process, with equivalent rights of appeal. Health Canada and Justice Canada translated that treaty obligation into a full structural redesign.
Summary proceedings became full patent infringement actions under the Federal Courts Act. Parties gained rights to documentary discovery, examinations for discovery, live expert and fact witnesses, and binding decisions on both infringement and invalidity. The dual-track system was formally eliminated: a finding of infringement or invalidity in a PMNOC action is binding on any subsequent proceeding between the same parties on the same patents and the same drug. The 45-day window for innovators to commence a section 6(1) action remained, as did the 24-month stay — but the proceeding behind that stay became substantively equivalent to conventional patent litigation.
The 2017 amendments also introduced section 8.1, which allows a biosimilar or generic manufacturer with a reasonable belief that its drug might be alleged to infringe a listed patent to commence a declaratory action for invalidity or non-infringement before it even files a regulatory submission. Samsung Bioepis used section 8.1 against Janssen’s ustekinumab patents before filing the PYZCHIVA submission — a pre-emptive strike that is now standard practice for well-resourced biosimilar sponsors.
The April 2021 Clarifications
Amendments published in the Canada Gazette in April 2021 clarified the Minister of Health’s role in maintaining the Patent Register. The changes addressed a persistent gray area: when a patent is submitted for listing after a generic or biosimilar manufacturer has already filed its regulatory submission, the second person is not required to serve an NOA addressing that patent. Two Federal Court of Appeal decisions — one involving Janssen’s ustekinumab, one involving EMD Serono’s cladribine (MAVENCLAD) — confirmed that this timing rule applies even where the Minister’s listing decision itself arrives after the second person’s submission date. The patent listing date that matters is the date the Minister exercises the listing discretion, not the date of submission to OPML.
Key Takeaways: Section Overview
- Canada’s 1993 system copied linkage from Hatch-Waxman but diverged on stay duration, patent eligibility, and litigation format.
- The 2017 CETA-driven amendments replaced summary proceedings with full infringement actions, eliminating dual-track litigation.
- Section 8.1 pre-filing invalidity actions are now a standard tool for generic and biosimilar sponsors.
- The 2021 amendments resolved timing ambiguity: a patent added to the Register after a second person files is not addressable by that second person.
The Patent Register: Eligibility, Listing Mechanics, and IP Valuation
What Can and Cannot Be Listed
Not every patent covering a marketed drug qualifies for the Patent Register. To qualify, a patent must contain at least one eligible claim — specifically, a claim to the medicinal ingredient itself, a claim to the formulation containing the medicinal ingredient, a claim to the dosage form, or a claim to a use of the medicinal ingredient for a specific approved indication. Method-of-manufacture patents are explicitly excluded. Process patents that do not claim the composition or use of the drug are excluded. Patents covering packaging, device components, or manufacturing equipment are excluded.
This eligibility filter is more restrictive than the U.S. Orange Book, which historically permitted listing of method-of-manufacturing patents and resulted in Orange Book ‘stuffing’ — a practice that Canadian courts and regulators intentionally foreclosed. The Federal Court has applied the eligibility criteria strictly. In Amgen’s motion against Bayer’s aflibercept (EYLEA), the Court found that a listed patent failed the ‘product specificity’ requirement because it was asserted against a supplemental new drug submission for a formulation that differed from the specific DIN-level product the patent was listed against. Bayer discontinued its appeal in August 2025, effectively confirming the Court’s narrower reading.
The Form IV Submission and Timing Rules
Innovators submit a Form IV patent list either with their original New Drug Submission (NDS) or within 30 days of the patent’s date of grant. Late additions permitted within the 30-day window give innovators a structured mechanism to capture patents issued after market approval. In practice, this window is the primary vehicle for adding second-generation patents — improved formulations, new dosage forms, new approved indications — to the Register for an already-marketed drug.
The 2021 Federal Court of Appeal decisions on listing timing establish a critical operational constraint: once a biosimilar or generic sponsor files its regulatory submission, any patent added to the Register on or after that filing date creates no obligation for that sponsor. IP teams managing Canadian portfolios should treat the window between a product’s NDS approval and the first anticipated generic or biosimilar regulatory submission as the high-value period for Form IV filings. A patent listed one day before a competitor files carries full stay rights; the same patent listed one day after carries none against that specific competitor.
IP Valuation: Patent Register Position as a Core Asset
For portfolio managers and deal teams valuing a Canadian pharmaceutical asset, Patent Register position is not an administrative footnote — it is a direct determinant of revenue duration. A patent listed on the Register against a high-revenue drug, combined with the ability to trigger a 24-month stay on generic approval, has a calculable NPV contribution. The rough model: annual Canadian revenue of the reference product, multiplied by the probability that the stay period produces a valid final judgment of infringement, multiplied by 24 months of uncontested market presence.
Where the asset carries a Certificate of Supplementary Protection (CSP) in addition to listed patents, the valuation extends further. The CSP provides up to 24 months of additional exclusivity beyond the underlying patent’s expiry and is itself eligible for listing, which means the CSP’s expiry date sets the outer boundary for generic entry that any litigation strategy must defend to.
For assets without CSP coverage but with multiple listed patents of differing expiry dates — the classic ‘patent thicket’ — the valuation model must account for the serial nature of NOA obligations. Each listed patent can generate its own NOA-and-stay cycle, so a well-structured patent portfolio can produce sequential 24-month stays. Sandoz, Apotex, and Auro — the three most active challengers by number of drugs in the 2024-2025 period — understand this arithmetic well, which is why their initial NOAs typically challenge the weakest patents in a portfolio first, attempting to exhaust stays before the commercially critical composition or formulation patents come into play.
Key Takeaways: Section Overview
- Only composition, formulation, dosage form, and use claims are Register-eligible; method-of-manufacture patents are excluded.
- The 30-day Form IV window after patent grant is the primary lifecycle management tool for adding second-generation IP to the Register.
- Patents added after a second person files create no obligation for that specific filer — timing of Form IV submissions is a high-stakes operational decision.
- Patent Register position directly maps to quantifiable NPV contribution; CSP listing extends that valuation window by up to 24 months beyond base patent expiry.
Certificates of Supplementary Protection (CSPs): The Real Numbers
Mechanics and Eligibility
CSPs, introduced under the Certificate of Supplementary Protection Regulations as part of CETA compliance, compensate innovators for the time lost between patent filing and the first Canadian NOC for a new medicinal ingredient or combination. The maximum CSP term is 2 years. The minimum is zero — if the time between patent filing and NOC grant is less than the regulatory processing period used as a floor, no CSP issues. Eligible patents must contain a claim to the medicinal ingredient or combination, and the CSP attaches to that specific medicinal ingredient as marketed in the first approved Canadian product.
The scope restriction matters strategically. A CSP cannot be obtained on a formulation-only patent or a use patent. It requires a patent with a medicinal ingredient claim — the same type of composition patent that forms the core of a small-molecule franchise’s IP estate. Reformulated products, new dosage forms, and second-indication approvals do not anchor new CSPs unless they involve a genuinely new medicinal ingredient or combination. This means CSP coverage is concentrated in first-in-class and new chemical entity (NCE) launches, not in lifecycle extension products.
Canada vs. EU Supplementary Protection Certificates: Key Differences
Canada’s CSP system is structurally similar to the EU’s Supplementary Protection Certificates (SPCs) but narrower in two respects. First, the maximum Canadian CSP term is 24 months, while EU SPCs can extend for up to 5 years plus a 6-month pediatric extension. Second, EU SPCs can be obtained on active ingredient combinations and new therapeutic applications under Regulation (EC) 469/2009, giving European innovators a broader basis for exclusivity extension. Canada’s stricter scope means that the CSP tool is less useful for biologic franchises with complex combination products, and essentially unavailable for devices and software-enabled drug delivery systems that lack a medicinal ingredient claim in their core patents.
For Canadian pharma asset valuation, a CSP that runs to its 24-month maximum on a drug with CAD 400-600 million in annual Canadian revenue adds a material line item to any acquisition or licensing model. The Patented Medicine Prices Review Board’s CompassRx data shows that specialty biologics now drive the majority of public payer drug plan expenditures, which makes CSP status on a biologic franchise especially significant in deal analysis.
CSP Listing on the Patent Register
A CSP, once granted, is eligible for listing on the Patent Register, and its expiry date becomes the effective horizon for generic or biosimilar market entry planning. The strategic implication is that a second person filing a regulatory submission after a CSP is listed must serve an NOA addressing the CSP as a distinct instrument. A CSP is not automatically subsumed by the underlying patent NOA — it requires its own allegation of invalidity or non-infringement, which triggers its own 45-day window and 24-month stay. In practice, generic and biosimilar manufacturers challenge CSPs on validity grounds, arguing that the medicinial ingredient requirement is not met or that the CSP was granted on a patent that does not disclose the marketed product. These challenges have produced limited litigation to date, but as CSPs approach expiry on major small-molecule franchises approved in 2018-2022, dedicated CSP validity litigation will increase.
Key Takeaways: Section Overview
- CSP maximum term is 24 months, capped at compensation for regulatory delay between patent filing and first Canadian NOC.
- CSP eligibility requires a medicinal ingredient claim — formulation-only and use-only patents do not qualify.
- A listed CSP generates its own NOA obligation and 24-month stay, separate from any underlying patent.
- EU SPCs cover a broader scope and longer term than Canadian CSPs; deal teams should not conflate the two when modeling cross-border IP protection.
The NOA-to-Trial Pipeline: Every Step, Every Clock
Step 1: Second Person Files a Regulatory Submission
When a generic manufacturer files an Abbreviated New Drug Submission (ANDS) — or a biosimilar sponsor files an NDS referencing a Canadian Reference Product (CRP) — Health Canada checks the Patent Register for listed patents. If listed patents exist, the submission triggers second-person status under the PM(NOC) Regulations, and the second person must serve an NOA on each patentee identified in the Register.
The NOA must contain, for each patent, a detailed statement of the legal and factual basis for the allegation. The allegation will be one of four types: the patent is invalid; the patent will not be infringed by the second person’s drug; the patent has expired; or the patent is not eligible for listing. The quality of the NOA — specifically the specificity of the invalidity or non-infringement theory — sets the factual record for the section 6(1) action. A poorly particularized NOA constrains the generic’s ability to rely on certain invalidity arguments at trial.
Step 2: The 45-Day Window
On receipt of a complete NOA, the first person (patentee/innovator) has 45 days to commence a section 6(1) action in the Federal Court. Filing within 45 days automatically stays Health Canada’s approval of the second person’s drug for 24 months from the date of service of the NOA. The stay is mechanical: it applies to every patent in respect of which a section 6(1) action was filed within 45 days, regardless of how strong the patentee’s infringement position is.
Failing to file within 45 days forfeits the stay. The patent remains listed and the second person must still address it, but the second person may proceed to approval without waiting for trial resolution on that patent. This creates an important asymmetry in multi-patent portfolios: patentees that file on all listed patents preserve maximum stay coverage, while selective filing — choosing only the strongest patents for litigation — risks leaving undefended patents that clear the NOA hurdle without a stay.
Step 3: The 24-Month Stay
The 24-month stay runs from the date of NOA service, not from the date the section 6(1) action is commenced. If the patentee files on day 44 of the 45-day window, the effective stay period is approximately 23 months and 17 days. The stay cannot be extended. If the Federal Court has not rendered a trial decision within 24 months, the stay lapses and Health Canada may issue the NOC. The second person can then enter the market, subject to whatever risk it is willing to assume regarding a pending infringement finding.
Post-2017 data shows that the Federal Court resolves the minority of cases that reach trial within the 24-month window. The majority of section 6(1) actions — 264 of approximately 326 actions commenced since September 2017 through year eight — were resolved without a decision on the merits. That resolution rate reflects settlement, discontinuance, or consent order, and in many cases reflects patent settlements that carry their own regulatory and competition law implications under section 79 of the Competition Act.
Step 4: Trial, Evidence Standards, and Claim Construction
Post-2017 PMNOC trials follow Federal Court of Canada Rules for full patent actions. Parties exchange affidavits of documents, conduct examinations for discovery, and file expert reports on both validity and infringement. Unlike U.S. district courts, Canadian Federal Court judges do not conduct Markman-style claim construction hearings as a pre-trial event. Claim construction occurs as part of the trial, with claim charts and written argument on construction submitted in opening briefs. This procedural difference has substantive consequences: there is no early claim construction ruling that might precipitate settlement, and parties carry the uncertainty of construction through the entire evidentiary record.
The burden of proof on infringement rests with the patentee on a balance of probabilities. The burden on invalidity rests with the party asserting invalidity — typically the generic or biosimilar sponsor — also on a balance of probabilities. This contrasts with the U.S. standard, where a patent is presumed valid and invalidity requires clear and convincing evidence. The Canadian standard is lower, which means that invalidity arguments are more likely to succeed in Canadian proceedings than equivalent arguments under Hatch-Waxman in U.S. district courts.
Step 5: Section 8 Damages for Wrongful Stay
If the patentee commences a section 6(1) action and loses — either on invalidity or non-infringement — the second person may claim section 8 damages for losses suffered during the stay period. Section 8 damages are the Canadian equivalent of the Hatch-Waxman section 271(e)(4) damages framework: the second person is compensated for the market share it would have captured had the stay not been triggered. Quantifying section 8 damages requires modeling a but-for scenario — the counterfactual market absent the stay — which involves contested assumptions about generic price erosion rates, market penetration curves, and provincial formulary listing timelines.
Three section 8 actions were pending as of late 2024. Pharmascience’s abiraterone action was scheduled for trial in May 2025. As of December 2025, Pharmascience’s fampridine section 8 claim was discontinued. No section 8 action has produced a final merits decision since the 2017 amendments. The absence of a post-2017 section 8 precedent means the damages quantum for a wrongful 24-month stay under the current full-action framework remains untested — a material uncertainty for patentees modeling the financial risk of filing on weak patents.
Key Takeaways: Section Overview
- The 45-day window is absolute; missing it forfeits stay rights on that patent regardless of portfolio strength.
- 264 of 326 section 6(1) actions since 2017 resolved without a merits decision, indicating that the stay itself — not trial victory — is often the strategic objective.
- Canadian validity challenges succeed on a balance of probabilities, not clear and convincing evidence — a lower bar than in U.S. ANDA litigation.
- No post-2017 section 8 damages award on the merits exists, leaving wrongful stay damages quantum as an open litigation risk variable.
Litigation Outcomes: Eight Years of Post-2017 Data
Case Volume and Trends
Health Canada’s statistical report for fiscal year ending March 31, 2025, recorded 33 section 6(1) actions filed in 2024-2025, down from 39 in 2023-2024 and 53 in 2022-2023. The decline represents the second consecutive year of reduced filings — not evidence of a less active patent landscape, but of a maturing post-2017 system in which parties have better calibrated which patents are worth litigating and which are more efficiently resolved through agreement.
From September 2017 through year eight (September 2025), approximately 326 total section 6(1) actions were commenced. Of those, 33 reached a decision on the merits of infringement and/or invalidity. Innovators prevailed in 17 of those 33 merits decisions. The 51% innovator success rate on the merits is higher than early observers expected and reflects the post-2017 system’s tendency to filter: cases that reach trial have typically survived early invalidity challenges during discovery, leaving a more favorable factual record for the patentee. The cases that settle or discontinue skew toward weaker patents.
Active Challengers in 2024-2025
Sandoz, Apotex, and Auro were tied as the most active challengers by drug count in the year ending September 2025, each challenging patents for three separate drugs. This is consistent with the competitive dynamics of Canada’s generic industry: Apotex, with 712 products listed on the Ontario formulary as of November 2024, operates at a scale that justifies systematic patent challenge strategies. Pharmascience and Teva also maintain active challenge portfolios. The concentration of challenge activity in a handful of large generics means that innovators with commercially significant Canadian portfolios face repeat litigation from the same adversaries — an environment that rewards institutional litigation knowledge on both sides.
The Only 2025 Merits Decision
In April 2025, the Federal Court granted Alexion (now part of AstraZeneca) a declaration of infringement and a permanent injunction against Amgen’s eculizumab biosimilar BEKEMV — a biosimilar of Alexion’s SOLIRIS. Amgen discontinued its appeal. This outcome has particular significance for the biologics IP landscape: it confirms that the PMNOC framework, as applied to biosimilars, can produce permanent injunctions on the merits, not merely on interim or interlocutory grounds. Eculizumab’s IP estate — which includes formulation and dosage regimen patents — now stands as a post-2017 test case for how the Federal Court treats complex biologics IP in a full trial context.
The Supreme Court of Canada: Pharmascience’s Methods-of-Treatment Appeal
On October 9, 2025, the Supreme Court of Canada heard Pharmascience’s appeal on the patentability of methods of medical treatment. The case directly challenges whether a patent claim directed to a dosing regimen or therapeutic protocol constitutes patentable subject matter under the Patent Act — a question with massive downstream consequences for use claims listed on the Patent Register. If the SCC narrows the scope of patentable methods of treatment, a significant category of currently-listed patents could become ineligible, collapsing the stay rights attached to them and accelerating generic entry on affected products. The decision, expected in 2026, is the single most consequential pending judicial development for Canadian pharma IP strategy.
Key Takeaways: Section Overview
- Section 6(1) filings declined for two consecutive years through 2024-2025; 33 actions filed in 2024-2025 vs. 53 in 2022-2023.
- Innovators won 17 of 33 merits decisions since 2017 — roughly 51% — suggesting post-2017 trials represent a pre-screened, higher-quality patent set.
- The 2025 Alexion/Amgen eculizumab (SOLIRIS/BEKEMV) decision is the only post-2017 merits ruling on a biologic and demonstrates that permanent injunctions are available in this framework.
- The SCC’s Pharmascience methods-of-treatment case, heard October 2025, may invalidate a category of use-claim patents currently listed on the Register.
Comparative Analysis: Canada vs. Hatch-Waxman, BPCIA, and the EU SPC Regime
Canada vs. Hatch-Waxman: Six Material Differences
Stay duration. Canada’s 24-month stay is 6 months shorter than the 30-month stay under Hatch-Waxman. For a drug generating CAD 100 million per year in Canada, the 6-month difference represents approximately CAD 50 million in foregone stay-period revenue — not a trivial calculation for deal teams valuing Canadian vs. U.S. patent portfolios.
Patent eligibility. Canada excludes method-of-manufacture patents and imposes a product-specificity requirement at the DIN level. The U.S. Orange Book lists use codes and patent numbers without a DIN-equivalent granularity requirement, permitting a broader set of patents to generate stay rights. The Bayer/aflibercept (EYLEA) patent listing dispute turned entirely on this DIN-specificity requirement.
Invalidity standard. Canadian courts apply balance of probabilities; U.S. courts apply clear and convincing evidence. This 20+ percentage point difference in invalidity success probability materially affects the settlement calculus in both jurisdictions. A generic with a strong obviousness case is more likely to achieve summary resolution in Canada than in Delaware.
First-filer exclusivity. The U.S. grants 180 days of market exclusivity to the first ANDA filer that makes a Paragraph IV certification and successfully defends the challenge. Canada has no equivalent. This is analyzed in detail in Section 11.
Binding effect. Post-2017, a Canadian PMNOC merits decision binds subsequent litigation between the same parties on the same patents and drug. U.S. ANDA litigation does not produce binding estoppel in the same way — the dual-track issue that plagued the pre-2017 Canadian system mirrored U.S. persistent dual-track exposure before the Orange Book reform discussions.
Methods of treatment. The U.S. permits patenting of methods of treatment with minimal restriction. Canada’s position is the subject of the pending SCC appeal. A decision narrowing Canadian method-of-treatment patentability would create the most significant Canada-U.S. patent scope divergence since the 1993 reforms.
Canada vs. BPCIA: Biologic IP and the ’12-Dance’ Problem
The U.S. Biologics Price Competition and Innovation Act (BPCIA) governs biosimilar patent disputes and provides a 12-year data exclusivity period, a complex pre-litigation information exchange (the ‘patent dance’), and a 30-month stay on biosimilar approval for patents identified in that exchange. Canada provides no statutory data exclusivity for biologics beyond the PMNOC stay framework, and the absence of a structured equivalent to the BPCIA ‘dance’ means biosimilar sponsors in Canada are not required to disclose their manufacturing process details pre-litigation. For innovators seeking to identify process-related infringement theories before trial, this gap complicates litigation strategy.
The 12-year U.S. data exclusivity alone makes the U.S. far more protective of biologic franchises than Canada’s framework. A biologic drug approved in the U.S. and Canada simultaneously can face biosimilar competition in Canada up to 8 years earlier than in the U.S., purely as a function of data exclusivity rules.
Canada vs. EU SPC Regime
EU SPCs are available for medicinal products and plant protection products under Regulation (EC) 469/2009. They provide up to 5 years of additional exclusivity, extendable by 6 months for pediatric indications. The EU does not have a direct equivalent to the PMNOC stay framework; instead, each EU member state’s patent enforcement system handles linkage through national courts, with varying efficiency. Canada’s unified Federal Court jurisdiction for both PMNOC and general patent actions produces more consistent outcomes than the EU’s fragmented multi-jurisdictional landscape.
For global deal teams, the relevant comparison is total exclusivity duration: a drug in both Canada and the EU with a strong patent estate plus protection instruments can expect 22-27 years of exclusivity in the EU (20-year patent term plus 5-year SPC plus 6-month pediatric extension) versus approximately 22 years in Canada (20-year term plus 24-month CSP). The gap narrows when Canada’s data protection provisions — 8 years for small molecules under the Food and Drug Regulations data protection scheme — are factored alongside patent and CSP coverage.
Key Takeaways: Section Overview
- The U.S.-Canada stay duration gap (30 months vs. 24 months) translates to a quantifiable revenue difference on high-volume branded products.
- Canada’s lower invalidity standard (balance of probabilities) increases generic success rates in validity challenges relative to U.S. Hatch-Waxman litigation.
- The absence of U.S.-style 12-year biologic data exclusivity leaves Canadian biologic franchises significantly more exposed to early biosimilar competition.
- EU SPCs provide up to 5 years of additional protection vs. Canada’s 24-month CSP maximum — a material difference in exclusivity duration modeling.
Biologics and Biosimilars: The System’s Biggest Structural Gap
Why Biosimilars Sit Awkwardly in the PMNOC Framework
The PM(NOC) Regulations nominally cover biosimilars. A biosimilar sponsor that files an NDS referencing a Canadian Reference Product (CRP) is a ‘second person’ under section 5 of the Regulations, obligated to serve NOAs on patentees with Register listings against the CRP. This much is clear.
The structural gap lies in what happens before and after the NOA. Biosimilar sponsors face no statutory stay on approval pending patent challenge unless the patentee files a section 6(1) action within 45 days. Unlike small molecules, where the generic entry model is well-tested and the competitive landscape is predictable, biologic patent portfolios are denser, the manufacturing comparability arguments are more complex, and the interplay between formulation patents, dosing regimen patents, and device patents is harder to resolve in a 24-month window. The Federal Court’s 2025 eculizumab decision demonstrated that a full merits trial on a biologic can complete within the stay period — but eculizumab is not representative; the aflibercept (EYLEA) biosimilar litigation involves multiple sequential trials scheduled through October 2026.
The absence of any first-filer exclusivity for biosimilar challengers — covered in Section 11 — makes aggressive patent challenge strategies economically unattractive for all but the largest biosimilar manufacturers with the balance sheets to absorb multi-year litigation costs.
Health Canada’s 2025 Biosimilar Regulatory Reforms
On June 10, 2025, Health Canada launched a consultation on revisions to its Guidance Document for biosimilar submissions. The most commercially significant proposed change eliminates the requirement that biosimilar submissions include comparative clinical efficacy trials in most cases, accepting comparative analytical and pharmacokinetic/pharmacodynamic data as sufficient for approval. The consultation closed September 8, 2025.
If finalized, this change will reduce the cost and timeline of biosimilar development programs, particularly for monoclonal antibody biosimilars where comparative clinical trials in therapeutic indication populations are expensive and lengthy. A reduced clinical data burden accelerates the filing timeline, compressing the window between patent expiry or CSP expiry and first biosimilar approval. Innovators with Canadian biologic portfolios should treat the finalization of this guidance as a trigger for Patent Register audit — any formulation, dosage form, or use patent that might not survive a balance-of-probabilities invalidity challenge should be assessed for its realistic stay value.
The November 2024 Agile Licensing amendments to the Food and Drug Regulations took effect July 1, 2025, for Schedule D Drugs (which include all biosimilars). These amendments replace Part C, Division 4 requirements with a modernized, adaptive framework that streamlines post-authorization change management for biologics — reducing the regulatory overhead of manufacturing changes and presentation updates that previously required separate NDS supplements, each of which had independent Patent Register implications.
Provincial Biosimilar Transition Policies: The Market Access Overlay
By August 1, 2024, all Canadian provinces had implemented policies requiring patients receiving public drug plan funding for certain reference biologic drugs to transition to biosimilars. Manitoba was the last to act, effective August 1, 2024. These policies do not interact directly with the PMNOC patent linkage framework — they operate at the reimbursement level, not the approval level — but they fundamentally reshape the commercial value of biologic patent estates in Canada.
The practical effect: even where a patentee successfully defends listed patents and prevents a biosimilar from receiving an NOC, provincial formulary policies accelerate biosimilar uptake once that NOC eventually issues. Smart & Biggar data shows biosimilar price discounts to reference products ranging from 15% to 50% in Canadian public formularies. For biologics where biosimilars have achieved high formulary penetration — filgrastim biosimilars hold 94.5% prescription share, pegfilgrastim biosimilars hold 99.9% — the residual commercial value of a patent estate that delays a biosimilar by 24 months via stay is substantially lower than an equivalent stay calculation for a small-molecule blockbuster.
Live Biologic Patent Disputes: 2025 Snapshot
Aflibercept (Bayer/Regeneron’s EYLEA) is the most actively litigated biologic in the Canadian PMNOC system. As of mid-2025, multiple sequential trials are underway or scheduled: the Bayer/Regeneron v. Amgen (ABP938) trial completed in May-June 2025; a second trial between the same parties on different patents was scheduled for August 2025; Bayer/Regeneron v. Sandoz (AFQLIR) trials are scheduled commencing October 2026. Biocon Biologics’ YESAFILI actions were discontinued pre-trial.
Ustekinumab (Janssen’s STELARA) generated the first application under the Competition Act’s abuse-of-dominance provisions by a pharmaceutical company in Canada. JAMP Pharma sought leave under section 79 of the Competition Act against Janssen relating to STELARA’s Canadian patent strategy. The Competition Tribunal dismissed JAMP’s application November 20, 2024 — but the attempt itself signals a broadening toolkit for generic and biosimilar challengers that extends beyond Federal Court patent invalidity actions.
Pertuzumab (Hoffmann-La Roche/Genentech’s PERJETA) drew a PMNOC action in May 2025 against Organon and Shanghai Henlius Biotech for their POHERDY biosimilar, with no trial date yet set.
Key Takeaways: Section Overview
- Biosimilars are nominally within the PMNOC framework but lack the structural protections — notably first-filer exclusivity and comprehensive data exclusivity — that make aggressive patent challenges economically rational.
- Health Canada’s 2025 proposed removal of comparative clinical trial requirements for biosimilar approval will compress filing timelines, increasing urgency for Patent Register audits on biologic portfolios.
- Provincial biosimilar transition mandates have been implemented Canada-wide as of August 2024, reducing the commercial payoff of patent-derived delays for biologic franchises.
- The JAMP/Janssen Competition Act application — the first of its kind — signals that competition law is becoming a parallel channel for biosimilar market access challenges.
Case Studies: Adalimumab, Ustekinumab, Aflibercept, and Eculizumab
Adalimumab (AbbVie’s HUMIRA): The Biosimilar Patent Thicket in Practice
AbbVie’s adalimumab franchise is the global archetype for biologic evergreening through patent portfolio accumulation. In Canada, the PMNOC litigation against JAMP Pharma Corporation produced the most instructive post-2017 biologic trial decision to date. The Federal Court analyzed multiple adalimumab patents across a consolidated trial. A formulation patent — specifically the third patent claiming an aqueous adalimumab formulation — was found valid and JAMP conceded infringement. The Court declined to issue a permanent injunction on public interest grounds, noting that forcing patients to switch to other adalimumab biosimilars could cause increased injection site pain due to citrate-containing formulations.
The injection site pain rationale for denying a permanent injunction on a valid, infringed patent is a notable departure from standard patent enforcement logic and directly relevant to IP valuation. A patent team modeling the enforcement value of AbbVie’s Canadian formulation patents should incorporate the probability that a Canadian court applies a similar public-interest override on the injunction, reducing the practical exclusivity yield below what the patent’s validity would suggest.
AbbVie’s appeal and JAMP’s cross-appeal of the trial decision were pending as of year-end 2025. AbbVie’s separate judicial review of Health Canada’s determination that JAMP was not a ‘second person’ for two of the four consolidated actions was dismissed. The determination of second-person status — which controls whether the PMNOC Regulations apply at all to a given filing — is itself a source of strategic litigation.
Ustekinumab (Janssen’s STELARA): Competition Law Meets Patent Linkage
Janssen’s Canadian ustekinumab portfolio illustrates how a dense, well-maintained Patent Register listing strategy can generate multiple sequential NOA obligations for biosimilar sponsors. Samsung Bioepis commenced a section 8.1 pre-filing invalidity action against a key ustekinumab patent before filing its PYZCHIVA regulatory submission — an offensive move designed to neutralize a listed patent before it could trigger a stay. Janssen’s motion for leave to counterclaim for infringement against PYZCHIVA after it received its NOC was dismissed (Samsung Bioepis v. Janssen, 2024 FC 1715), meaning Janssen cannot use the section 8.2 infringement counterclaim mechanism against a product that has already cleared regulatory approval.
JAMP’s Competition Act challenge, while ultimately dismissed, raised substantive questions about whether maintaining a dense patent listing strategy for the purpose of delaying biosimilar entry constitutes abuse of dominance. The Tribunal’s dismissal on procedural grounds did not resolve those substantive questions. Future challengers — better resourced or with stronger factual records — may revisit the Competition Act pathway.
Aflibercept (Bayer/Regeneron’s EYLEA): Sequential Litigation as Portfolio Defense
EYLEA’s Canadian patent litigation demonstrates how a multi-patent PMNOC strategy translates into years of sequential market exclusivity defense. The Federal Court of Appeal’s August 2025 decision — affirming the Federal Court’s determination that a patent added to the Register 8 days after submission was listed before Biocon Biologics’ YESAFILI filing — confirms that the timing rules for Register listing are precise and litigatable. A patent listed 8 days earlier than an competitor’s regulatory submission carries full stay rights; the same patent listed 1 day after does not.
The sequential trial calendar for EYLEA — Amgen trial in May-June 2025, second Amgen trial August 2025, Sandoz trial October 2026 — illustrates the compounding effect of a multi-patent portfolio with staggered expiry dates and multiple biosimilar challengers. Each trial consumes 24 months of stay time on the specific patents at issue, but patents with later expiry dates continue to apply against later-filed biosimilar submissions.
Eculizumab (Alexion/AstraZeneca’s SOLIRIS): The Permanent Injunction Precedent
The April 2025 Federal Court decision granting Alexion a declaration of infringement and a permanent injunction against Amgen’s BEKEMV — a SOLIRIS biosimilar — is the only post-2017 merits decision on a biologic that produced an injunction. The decision establishes, at the Federal Court level, that the PMNOC framework supports injunctive relief on valid biologic patents. Amgen’s discontinuance of its appeal means the decision stands without Federal Court of Appeal review, limiting its precedential scope, but confirming the practical availability of injunctions for biologic patent holders in PMNOC proceedings.
Key Takeaways: Section Overview
- The AbbVie/JAMP adalimumab decision shows that Canadian courts may override permanent injunctions on public interest grounds even for valid, infringed patents — a discount factor in enforcement value modeling.
- JAMP’s Competition Act challenge against Janssen signals a broadening toolkit for biosimilar challengers beyond traditional PMNOC invalidity actions.
- The EYLEA litigation demonstrates how a multi-patent, multi-challenger PMNOC strategy can extend exclusivity defense through sequential stays.
- The Alexion/Amgen eculizumab permanent injunction (April 2025) confirms injunctive relief is available for biologic patents in PMNOC actions.
Evergreening Tactics and Their Limits Under the PMNOC Framework
What Evergreening Looks Like in the Canadian Context
Evergreening — the practice of filing successive patents on minor modifications of a drug to extend effective market exclusivity beyond the original composition patent — is structurally constrained by the PM(NOC) Regulations’ eligibility criteria, but not eliminated. The key evergreening vectors in the Canadian system are: formulation patents (new salt forms, polymorphs, or delivery systems that qualify as ‘formulation’ claims), dosage form patents (new solid oral dosage forms with distinct pharmacokinetics), and indication-specific use claims.
Method-of-manufacture patents, which are among the most common evergreening instruments globally, are categorically excluded from Canada’s Patent Register. This exclusion eliminates the broadest category of weak lifecycle patents that inflate the U.S. Orange Book and directly limits the number of stays a patentee can trigger on marginal IP.
Polymorph patents occupy a contested space. Canadian courts have found some polymorph patents obvious when the prior art disclosed the amorphous form and routine experimentation would have identified the crystalline form. Formulation patents for improved delivery systems — extended release, co-crystal formulations, subcutaneous vs. intravenous presentations — are a more productive evergreening tool, particularly for biologics where the SC formulation may have a clinically distinct profile from the IV reference product.
The 30-Day Form IV Window as a Lifecycle Tool
The 30-day Form IV window after patent grant is the primary lifecycle management instrument for adding patents to the Register on already-approved drugs. A company running a standard innovation cascade — composition patent at filing, formulation patent on solid oral dosage form issued two years post-approval, dosing regimen use patent issued five years post-approval — can add each of these patents to the Register as they issue, provided the 30-day window is respected. Each new listing creates a new NOA obligation for biosimilar and generic sponsors.
The strategic constraint is the 2021 Federal Court of Appeal timing rule: a patent added after a second person has already filed its regulatory submission creates no obligation for that specific filer. This means the value of Form IV evergreening is entirely front-loaded — it protects against future filers but cannot recapture the competitive position against a competitor already in the queue.
The AbbVie Adalimumab Model: Formulation Patents as the Last Line of Defense
AbbVie’s Canadian HUMIRA patent strategy illustrates the ceiling of evergreening. The composition-of-matter patent on adalimumab expired years before the PMNOC litigation concluded. What remained in the Register were formulation patents — specifically aqueous formulation patents covering low-citrate and high-concentration presentations. The Federal Court found one of these valid and infringed by JAMP’s biosimilar, but declined to issue a permanent injunction because the public interest in maintaining supply of a non-pain-inducing formulation outweighed enforcement of the patent monopoly.
The lesson for IP teams managing biologic patent portfolios: formulation patents can survive validity challenges and produce infringement findings, but the Canadian courts retain equitable discretion to deny injunctions when enforcement would harm patients. That equitable override is not a speculative risk — it materialized in the most commercially significant biologic patent case in Canadian legal history.
Key Takeaways: Section Overview
- Method-of-manufacture patents are categorically excluded from Patent Register listing, blocking the most common evergreening instrument used in the U.S.
- Formulation and dosage form patents remain viable evergreening tools but are subject to obviousness challenges on the Canadian balance-of-probabilities standard.
- The 30-day Form IV window value is entirely front-loaded; it cannot be deployed against a second person already in the regulatory queue.
- The AbbVie adalimumab case establishes that even valid, infringed formulation patents may not produce permanent injunctions if enforcement conflicts with patient welfare.
The First-Filer Problem: Canada’s Outlier Status on Generic Exclusivity
Why Canada Is the Last Hold-Out
Among countries with patent linkage regimes, Canada is the only one that does not provide temporary market exclusivity to the first generic or biosimilar manufacturer that successfully challenges a listed patent. The U.S. provides 180 days of exclusivity to the first Paragraph IV ANDA filer that successfully defends a patent challenge. Taiwan provides 12 months of exclusivity to the first generic applicant if the generic is found non-infringing or the patent is found invalid. China provides 12 months of exclusivity, available only where the generic achieves invalidation of the patent through inter partes proceedings.
Canada’s alternative to first-filer exclusivity is section 8 damages — the right to claim lost profits from the patentee if the stay is found to have been wrongfully triggered. This is not functionally equivalent to first-filer exclusivity. Section 8 damages are uncertain in amount (no post-2017 merits precedent), payable by the patentee rather than earned from the market, and contingent on winning the PMNOC action. The incentive to be first to file a challenge is largely absent when the upside of being first is only the possibility of suing the losing patentee for damages.
The consequence, analyzed in Aidan Hollis’s 2025 assessment in the Journal of Pharmaceutical Policy and Practice, is asymmetric litigation incentives. Patentees find it profitable to file section 6(1) actions on weak patents because the 24-month stay generates revenue regardless of eventual outcome. Generics, conversely, have weak incentives to challenge even patents they expect to win against, because any market reward accrues to all generic competitors simultaneously once the patent is cleared — the classic free-rider problem.
Policy Proposals on the Table
As of early 2026, no legislation has been introduced to create Canadian first-filer exclusivity. Industry observers expect the issue to resurface in the context of a future trade agreement negotiation or as part of a comprehensive PMNOC review. The most straightforward reform — a 12-month first-filer exclusivity period modeled on Taiwan’s system, applicable to the first second person that files and successfully wins a PMNOC action — would materially change generic challenge incentives without requiring a structural overhaul of the Regulations.
For portfolio managers, the absence of first-filer exclusivity affects the competitive value of Canadian generic and biosimilar launch strategies. A generic manufacturer that wins a Canadian PMNOC action captures no period of market exclusivity; it shares the market with every competitor that received a pending NOC. The launch economics for a first Canadian PMNOC challenger therefore approximate a day-one competitive launch, not the six-month quasi-monopoly that makes U.S. Paragraph IV challenges financially attractive even for molecules with modest Canadian revenues.
Key Takeaways: Section Overview
- Canada is the only country with a pharmaceutical patent linkage regime that provides no first-filer market exclusivity for successful patent challengers.
- Section 8 damages do not replicate the economic function of first-filer exclusivity; they are uncertain, litigation-contingent, and payable by the patentee, not earned from the market.
- The asymmetric incentive structure — profitable to litigate for patentees, unattractive to challenge for generics — contributes to Canada’s high rate of pre-merits settlement.
- First-filer exclusivity reform is a credible near-term policy development; IP teams should model its introduction as a scenario that would materially increase generic challenge activity.
Investment Strategy: What the Litigation Data Tells Portfolio Managers
Valuing Canadian Patent Portfolio Positions
For pharma M&A teams, licensing analysts, and institutional investors modeling a Canadian pharmaceutical asset, the PMNOC framework generates a set of quantifiable inputs that belong in every discounted cash flow model.
Stay value. For each patent listed on the Register, calculate the probability the innovator files a section 6(1) action within 45 days (historically close to 100% for commercially significant products), the probability the 24-month stay runs to completion without a merits loss, and the annual Canadian revenue at risk. The 2024-2025 data showing 33 new actions filed against a backdrop of declining filings suggests patentees are becoming more selective — a signal that older, weaker patents are being voluntarily cleared from Register listings as their stay value fails to justify litigation cost.
CSP layer. Add the CSP term to the base patent expiry for any NCE or new combination product with a CSP. The CSP’s own listing on the Register and its separate NOA obligation provides a second stay cycle covering the post-base-patent period. Treat CSP validity as a separate risk variable — currently under-litigated but increasingly contested as CSP expirations accumulate.
Biosimilar switching risk discount. For biologic assets, discount cash flows in the post-patent period more aggressively than for small molecules. Provincial formulary transition mandates now cover all of Canada; once a biosimilar receives its NOC, rapid formulary substitution is the default, not the exception. The 15-50% price discount range observed on Canadian formularies translates directly into revenue haircuts that arrive faster than in markets without mandatory switching policies.
Litigation outcome uncertainty. The 51% innovator win rate on merits decisions since 2017 is a starting prior, not a prediction for any specific patent. Adjust upward for composition patents with dense prior art differentiation and clinical data-backed claims. Adjust downward for formulation and polymorph patents with thin obviousness defenses. Adjust dramatically downward for any method-of-treatment claim pending the SCC’s Pharmascience decision.
SCC methods-of-treatment contingency. The single highest-impact scenario for Canadian patent portfolio valuations in 2026 is a broad SCC ruling on patentable subject matter in the Pharmascience appeal. A ruling that narrows methods-of-treatment patentability would invalidate a category of use claims listed on the Patent Register, eliminating stay rights for every affected patent simultaneously. IP teams should review all listed use claims and model their portfolio value under a scenario where these claims are struck.
Generic and Biosimilar Entry Timing Models
For payers, benefit managers, and generics analysts, the PMNOC system creates a predictable — if legally uncertain — timeline between first biosimilar or generic regulatory submission and market entry. The practical steps:
Identify all patents listed on the Register against the CRP or reference product. Estimate the probability and timing of an NOA for each listed patent, based on the competitive landscape and comparable filings on similar products. Model a 24-month stay from NOA service date for each patent where a section 6(1) action is filed. Discount for the ~80% probability that the case resolves before merits (historical base rate), which means commercial agreements — not court decisions — set the actual entry date in most cases. Layer in CSP expiry if applicable.
The resulting entry timeline probability distribution is the key output. For products with dense patent portfolios and CSP coverage, the P50 entry date is typically 24-36 months after the first commercial biosimilar NOA. For products with a single composition patent near expiry and no CSP, the P50 is closer to 12-18 months from first NOA.
Key Takeaways: Section Overview
- Patent Register position and CSP status are direct quantifiable inputs to Canadian pharmaceutical asset NPV models.
- The SCC’s Pharmascience methods-of-treatment case is the highest-impact contingency in Canadian patent portfolio valuation for 2026.
- Provincial mandatory biosimilar switching policies create faster post-NOC revenue erosion for biologic assets than historical analogs suggest.
- Commercial resolution (settlement/discontinuance) at ~80% rate means deal-making skill, not courtroom skill, determines entry timing in most PMNOC disputes.
Regulatory Roadmap: 2024-2026 Changes That Reshape Market Entry Calculus
Agile Licensing (November 2024, Effective July 1, 2025)
The Agile Licensing amendments, registered November 2024 and effective July 1, 2025, for Schedule D Drugs (biosimilars), replace Part C, Division 4 of the Food and Drug Regulations with a modernized post-authorization change management framework. The operational impact: biosimilar manufacturers can implement certain manufacturing process changes and updated presentations without filing full supplemental NDS submissions, which previously created independent Patent Register obligations. Under the new framework, the scope of Register-triggering submissions narrows, reducing the volume of NOA obligations generated by manufacturing optimization changes.
For innovators, Agile Licensing also means that reference product updates — new presentations, delivery devices, formulation improvements — may proceed through faster regulatory pathways that complicate the timing of corresponding Form IV filings. IP teams should audit the new post-authorization change categories to determine which updates require new Form IV submissions and plan filing timelines accordingly.
PMPRB Guidelines: January 1, 2026
The Patented Medicine Prices Review Board published its new Guidelines for PMPRB Staff on June 30, 2025, effective January 1, 2026. The guidelines introduce a two-step pricing review: an initial review assesses whether the Canadian price exceeds the highest international price among the PMPRB11 comparator countries; if it does, an annual review follows. This framework replaces prior Excessive Price Guidelines and directly affects the revenue modeling for patented products during their PMNOC-protected period.
The PMPRB11 international comparator basket includes France, Germany, Italy, Spain, Sweden, Switzerland, the UK, Japan, Australia, Belgium, and the Netherlands. For a product priced above the maximum of this basket, the new guidelines create a mechanism for mandatory price reduction regardless of patent status. For portfolio managers, this means that Canadian revenue projections during a PMNOC stay period are no longer simply a function of the branded price; they are subject to a downward adjustment risk tied to international pricing benchmarks that change as the product’s global price evolves.
Health Canada’s Biosimilar Clinical Trial Guidance Revision (Consultation Closed September 2025)
The proposed elimination of comparative clinical efficacy trial requirements for most biosimilar submissions, consulted on through September 8, 2025, is expected to be finalized in 2026. When implemented, the guidance change will reduce the cost of a Canadian biosimilar development program by eliminating Phase III-scale comparative clinical studies for molecules where analytical and PK/PD comparability is sufficient. The direct effect on the PMNOC system: faster first regulatory submissions, shorter windows for Form IV filings to preempt competitor entries, and increased filing volume as economic barriers to biosimilar development fall.
SCC Pharmascience Appeal Decision (Expected 2026)
Pharmascience’s appeal on the scope of patentable methods of medical treatment was heard October 9, 2025. The SCC’s decision will either affirm the current scope — which permits claims to specific dosing regimens, patient populations, and therapeutic endpoints — or narrow it by carving out ‘methods of medical treatment’ as non-patentable subject matter. A narrow ruling would invalidate a class of use claims currently generating stay rights on the Patent Register.
IP teams across the Canadian pharmaceutical market should treat the SCC decision as a binary event risk requiring scenario planning. The base case (no change in patentable subject matter scope) preserves the current portfolio value of use claims. The alternate scenario (narrowed scope) requires immediate Patent Register audit, re-evaluation of which listed use claims survive, identification of whether underlying composition or formulation claims on the same patents provide independent grounds for listing, and revision of licensing and co-promotion agreements whose royalty structures depend on Canadian use claim coverage.
Key Takeaways: Section Overview
- Agile Licensing reduces Register-triggering supplemental submissions for biosimilars from July 2025, narrowing the universe of NOA obligations from manufacturing optimization changes.
- The new PMPRB two-step pricing guidelines (effective January 2026) apply downward price pressure during PMNOC stay periods if Canadian prices exceed PMPRB11 international benchmarks.
- Finalizing the biosimilar clinical trial guidance revision will compress biosimilar development timelines and increase Register filing urgency for biologic innovators.
- The SCC Pharmascience decision is a binary event risk for all Canadian pharmaceutical patent portfolios with listed use claims; scenario planning is required now.
Key Takeaways by Segment
For Pharma/Biotech IP Teams
The Patent Register is the practical instrument of Canadian patent protection — not the patent itself. A patent that is not listed, filed on time, and maintained against the correct DIN-specific product has no stay value. Conduct regular Register audits against your active portfolio, particularly when manufacturing changes, new presentations, or new indications generate supplemental NOC submissions that could trigger new listing obligations or create timing windows for Form IV filings.
The 30-day Form IV window after patent grant is your primary lifecycle tool. Build patent grant monitoring into the IP operations workflow so that every eligible patent receives a Form IV assessment within days of grant, not weeks.
The SCC methods-of-treatment case requires immediate action regardless of outcome: if you have use claims listed on the Register, model two scenarios. Do not wait for the decision to begin the analysis.
For Portfolio Managers and Institutional Investors
Canadian patent portfolio value is a function of Register position, CSP coverage, the litigation calendar (filed actions and pending stays), and the new PMPRB pricing framework. None of these inputs are static. Build dynamic tracking into your monitoring workflow.
The EYLEA, STELARA, and HUMIRA biosimilar litigations collectively represent the practical ceiling of Canadian biologic exclusivity defense under current law. Study them as models for what maximum-effort patent portfolio defense looks like in the Canadian system, and calibrate your expectations for comparable assets accordingly.
The absence of first-filer exclusivity means that generic and biosimilar challenge economics in Canada are weaker than in the U.S. Products with large Canadian revenue exposure and dense patent portfolios that have not yet attracted PMNOC actions are not safe — they are simply waiting for a challenger with the balance sheet to absorb multi-year litigation without a first-mover reward.
For R&D Leads and Business Development Teams
The Health Canada biosimilar guidance consultation, if finalized as proposed, will reduce the development cost and timeline for your biosimilar programs targeting Canadian approvals. Build the revised clinical package requirements into your regulatory strategy planning now, rather than after finalization.
Agile Licensing’s reduced requirement for supplemental NDS filings changes the manufacturing change management workflow for biologics. Fewer supplemental filings means fewer Patent Register implications from routine manufacturing optimization — but also fewer opportunities to add newly issued patents to the Register through supplemental filing pathways.
The provincial mandatory biosimilar switching programs are now universal across Canada. For any biologic franchise entering the Canadian market, model the post-exclusivity period as a rapid biosimilar transition market, not a gradual erosion market.
Sources: PM(NOC) Regulations (SOR/93-133 as amended); CETA Article 20.28; Health Canada Statistical Report FY 2024-2025; Smart & Biggar PMNOC Anniversary Reports (Year 7, Year 8, 2025); Smart & Biggar Biosimilar Update June 2025; Hollis (2025) Journal of Pharmaceutical Policy and Practice; PMPRB New Guidelines effective January 1, 2026; Federal Court decisions: Samsung Bioepis v Janssen (2024 FC 1715); Alexion v Amgen (eculizumab, April 2025 FC); BLG, Cassels, Osler, and McCarthy Tetrault pharmaceutical practice group publications.