The Fork in the Road That Determines Billions

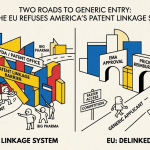
When a drug company files for FDA approval of a new medicine, it makes a strategic choice that will define the next decade of its commercial life. For small-molecule drugs, it enters a system designed in 1984 under Hatch-Waxman: a transparent, catalogued, well-litigated regime where patents get listed in a publicly searchable Orange Book and generic challengers file formal certifications to trigger predictable litigation timelines. For biologics, it enters something else entirely — a labyrinthine, multi-stage information exchange codified in the Biologics Price Competition and Innovation Act of 2010 (BPCIA), complete with a “patent dance” that critics say is more ballroom waltz than street fight, except that the music never quite starts and one partner routinely refuses to show up.
The distinction is not academic. It determines whether a $50,000-per-year adalimumab biosimilar reaches patients in 2023 or 2027. It determines whether a reference biologic manufacturer can weaponize 132 patents to build a fortress around a single drug — as AbbVie did with Humira — or whether a challenger can move straight to court, skip the dance, and force the issue. It determines who wins, who waits, and who pays.
This article examines both systems in granular detail: how they work mechanically, where they diverge strategically, how companies exploit their respective gaps, and what the case law — including the Supreme Court’s intervention in Amgen Inc. v. Sandoz Inc. — has clarified or muddied. The goal is a working map for anyone who needs to understand pharmaceutical IP competition at the highest level.
Part One: The Orange Book System — How Small-Molecule IP Linkage Works
The 1984 Hatch-Waxman Bargain
The Drug Price Competition and Patent Term Restoration Act of 1984 — universally called Hatch-Waxman — created a two-sided bargain. Brand pharmaceutical companies received something they desperately wanted: patent term restoration to compensate for years lost during FDA review. Generic companies received something they needed just as badly: an abbreviated approval pathway that allowed them to rely on the brand’s safety and efficacy data rather than repeating expensive clinical trials.
The mechanism binding these two concessions together is patent linkage. Under 21 U.S.C. § 355(b), a brand manufacturer submitting a New Drug Application (NDA) must list in that application all patents that claim the drug or a method of using the drug for which a claim of patent infringement could reasonably be asserted against an unauthorized maker, user, or seller. The FDA then publishes those patents in the Approved Drug Products with Therapeutic Equivalence Evaluations, better known as the Orange Book.
The Orange Book is, at its core, a public commitment. It tells the world: these are the patents the brand believes protect this drug. That transparency creates accountability and, as we will see, creates leverage for both sides.
The Paragraph IV Certification Mechanism
When a generic manufacturer files an Abbreviated New Drug Application (ANDA), it must certify its relationship to every Orange Book-listed patent. It has four options under 21 U.S.C. § 355(j)(2)(A)(vii):
- Paragraph I: No patent is listed in the Orange Book.
- Paragraph II: The listed patent has expired.
- Paragraph III: The applicant will wait until patent expiration before marketing.
- Paragraph IV: The listed patent is invalid, unenforceable, or will not be infringed by the generic.
Only Paragraph IV matters for competitive strategy. A Paragraph IV certification is both a legal assertion and a provocation. It tells the brand manufacturer that the generic believes it can beat — or avoid — every listed patent. Federal law then requires the generic to notify the patent holder, who has 45 days to sue.
If the brand sues within those 45 days, a 30-month stay of ANDA approval automatically triggers. The FDA cannot approve the generic for 30 months (roughly two and a half years) unless litigation resolves earlier. That automatic stay has been the most powerful, and most criticized, tool in the brand pharmaceutical arsenal for four decades.
The first company to file a Paragraph IV ANDA receives 180 days of generic market exclusivity — a reward for the litigation risk it assumes. That 180-day head start, when it actually materializes, can be worth hundreds of millions of dollars.
What Goes Into the Orange Book — And What Doesn’t
The Orange Book’s integrity depends on brands listing only patents that genuinely claim the drug or its approved uses. In practice, the line gets blurred. Courts and the FTC have repeatedly examined whether brands list patents strategically — covering devices, packaging, or minor formulation variations — to trigger additional 30-month stays and delay generic entry beyond any legitimate IP claim.
The FTC’s 2002 study of Paragraph IV litigation found that brands received an average of 2.5 30-month stays per drug through sequential ANDA filings and serial patent listings [1]. Congress responded in the 2003 Medicare Modernization Act by limiting brands to a single 30-month stay per ANDA. But the incentive to list aggressively in the Orange Book remained, because every listed patent forces a generic to certify against it and potentially litigate.
The FDA itself has limited authority to police Orange Book listings for legal correctness. In Apotex Inc. v. Thompson, 347 F.3d 1335 (Fed. Cir. 2003), the Federal Circuit held that the FDA’s role in Orange Book listings is ministerial — it lists what the brand submits. The courts, not the FDA, determine whether a listed patent is improperly listed. This gap produced what the FTC now calls “junk patents” in the Orange Book: patents covering things like the color of a tablet or the design of a bottle cap, listed not because they genuinely protect the drug but because their presence creates another litigation hurdle.
The Supreme Court’s 2023 decision in Sanofi-Aventis U.S. LLC v. Mylan Pharmaceuticals Inc. (related to listing of device patents) and the FTC’s subsequent challenge to Orange Book listings for combination inhalers pushed the issue into sharper focus. The FTC argued in 2023 that AstraZeneca, Boehringer Ingelheim, GSK, and others had improperly listed inhaler device patents in the Orange Book, threatening to create the same kind of delay architecture around inhalers that existed for oral small-molecule drugs [2].
The 30-Month Stay as Competitive Weapon
The 30-month stay has a legitimate purpose. Pharmaceutical patent litigation is complex, expensive, and slow. Allowing a generic to launch at-risk — that is, before litigation concludes — and then forcing it to recall product if the brand wins creates enormous disruption. The stay gives courts time to work.
But the stay’s value to brands depends entirely on what gets listed in the Orange Book. If a brand can list ten patents, each potentially triggering separate litigation and (pre-2003) separate stays, the cumulative effect could delay generic entry by decades. Post-2003, each ANDA still faces only one 30-month stay, but a brand can force the generic to litigate ten patents within that stay window, each adding complexity and cost.
The economics are straightforward. A brand selling $3 billion per year in a drug has an incentive to spend $30 million on patent litigation if there is any realistic chance of extending exclusivity by even a few months. For generics, the math runs the other direction: litigation costs that exceed the expected 180-day exclusivity value can make a Paragraph IV challenge economically irrational, even when the listed patents are weak.
This asymmetry — which economists call the “innovation tax” on generic challengers — is one reason that roughly 40 percent of brand drugs never face a Paragraph IV challenge at all [3].
Part Two: The BPCIA and the Patent Dance — Architecture of a Different Fight
Why Biologics Got a Different Law
Biologics are not small molecules. They are large, complex proteins, antibodies, or nucleic acid sequences produced by living cells. Their manufacture involves cell lines, fermentation processes, and purification protocols that are themselves proprietary. You cannot make an exact copy of a biologic the way you can reverse-engineer and synthesize a small molecule: the cell line, the expression system, and the manufacturing process all influence the final protein’s structure in ways that matter clinically.
This complexity meant that the pre-existing Hatch-Waxman pathway — premised on chemical equivalence — did not fit. When Congress passed the Affordable Care Act in 2010, it included the BPCIA as a stand-alone statute under 42 U.S.C. § 262. The BPCIA created a new FDA approval pathway for biosimilars: products that are “highly similar” to a licensed reference biologic notwithstanding minor differences in clinically inactive components, with no clinically meaningful differences in safety, purity, and potency.
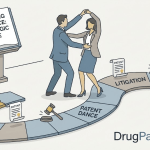
The BPCIA also created a patent dispute framework. Because biosimilar manufacturers must disclose their manufacturing processes to the FDA but not publicly, the law designed a private, bilateral information-exchange process to substitute for the public Orange Book: the patent dance.
The Patent Dance: A Step-by-Step Breakdown
The patent dance is codified at 42 U.S.C. § 262(l). Its structure is choreographed in statute:
Step 1 — The aBLA and the Initial Disclosure (Day 0 to Day 20)
When a biosimilar applicant submits a Biologics License Application to the FDA (called an aBLA), it must notify the reference product sponsor within 20 days of that submission [4]. This notification triggers the entire dance. The biosimilar applicant must simultaneously provide the reference product sponsor with a copy of the aBLA, including information describing the manufacturing processes it uses to produce the biosimilar.
This is the most structurally unusual aspect of the BPCIA. In the Hatch-Waxman context, a generic’s ANDA is confidential from the brand manufacturer. Under the BPCIA, the biosimilar applicant must affirmatively hand over its aBLA and manufacturing information to the party most motivated to use it against it. The justification is that the reference sponsor needs to evaluate whether any of its patents are at risk, since those patents are not listed in a public Orange Book.
Step 2 — The Reference Sponsor’s Patent List (Day 60)
Within 60 days of receiving the aBLA and manufacturing information, the reference product sponsor must provide a list of patents it believes could reasonably be asserted against the biosimilar, along with a statement describing, for each patent, the basis for the belief that the patent would be infringed [4]. This is the reference sponsor’s opening bid.
Step 3 — The Biosimilar Applicant’s Response (Day 120)
Within 60 days of receiving the reference sponsor’s patent list, the biosimilar applicant must provide its own list of patents it believes could be reasonably asserted (which may overlap with or differ from the reference sponsor’s list). It must also provide, for each patent on the reference sponsor’s list, either a statement that it will not engage in commercial marketing before patent expiration or a detailed statement explaining why each listed patent is invalid, unenforceable, or will not be infringed [4].
The detailed statement — sometimes called the “Section (l)(3)(B) Statement” — is essentially a pre-litigation brief. It is the biosimilar applicant’s first formal legal argument about the patent landscape it intends to challenge. Unlike a Paragraph IV certification, which is a public document, this statement is confidential and exchanged only between the parties.
Step 4 — The Reference Sponsor’s Reply (Day 180)
Within 60 days of receiving the biosimilar applicant’s response, the reference sponsor provides its reply to each detailed statement: its own analysis of why the biosimilar applicant’s invalidity and non-infringement arguments are wrong [4].
Step 5 — Patent Negotiation and the “Agreed List” (Day 240)
Within 15 days of the reference sponsor’s reply, the parties must attempt to negotiate an “agreed list” of patents to be litigated immediately [4]. This is the most negotiation-intensive phase of the dance. The parties are supposed to identify which patents are worth litigating before the biosimilar launches, understanding that other patents can be litigated later.
If the parties reach agreement, they litigate those agreed patents. If they do not, the statute provides default rules: the reference sponsor can designate up to the greater of five patents or one-half of the total number of patents in dispute; the biosimilar applicant can designate its own list [4]. The resulting litigation proceeds on both lists.
Step 6 — The 180-Day Pre-Launch Notice
Separately from the dance itself, the BPCIA requires the biosimilar applicant to provide 180 days’ notice to the reference product sponsor before it begins commercial marketing [4]. This notice requirement was at issue in Amgen v. Sandoz and remains one of the most commercially significant provisions of the statute.
The Dance’s Critical Flaw: It’s Optional
Here is what the dance’s architects may not have fully anticipated: the first step — providing the aBLA and manufacturing information — is technically mandatory, but the BPCIA’s only stated remedy for failing to provide the aBLA is that the reference sponsor may bring an action “for a declaration of infringement, validity, or enforceability of any patent” that would be at issue if the aBLA had been provided [4].
In other words, if the biosimilar applicant declines to hand over its manufacturing information, the statute does not automatically halt the approval process. The reference sponsor’s remedy is to sue. Whether that suit produces an injunction and what courts can enjoin a biosimilar applicant from doing if it skips the dance — was precisely the question that reached the Supreme Court in 2017.
Part Three: Amgen v. Sandoz — The Supreme Court Settles the Dance’s Core Questions
The Case That Defined the Rules
Amgen Inc. v. Sandoz Inc., 137 S. Ct. 1664 (2017), arose from Sandoz’s decision to develop a biosimilar of Amgen’s filgrastim product Neupogen. Sandoz filed its aBLA in May 2014, notified Amgen of the filing, but declined to provide its aBLA and manufacturing information — skipping Steps 1 through 5 of the dance. Instead, Sandoz sent Amgen a notice of commercial marketing in July 2014, nine months before FDA approval (which came in March 2015), asserting that this constituted the required 180-day pre-commercial-marketing notice.
This raised two questions the Supreme Court resolved unanimously:
Question 1: Is the aBLA disclosure mandatory?
The Court held that it is not. The BPCIA’s only stated consequence of failing to provide the aBLA is that the reference product sponsor may bring a declaratory judgment action. There is no provision authorizing a court to compel disclosure or to enjoin FDA approval as a remedy for the failure. A biosimilar applicant can therefore skip the dance entirely — accepting the consequence that the reference sponsor can immediately sue [5].
This conclusion carried enormous strategic implications. It meant that biosimilar manufacturers who wanted to accelerate litigation — rather than spending a year exchanging detailed statements — could do so. It also meant that reference sponsors who had built their entire exclusivity strategy around the dance’s information-gathering phase could not force a slow, methodical review. The dance became optional for the biosimilar applicant.
Question 2: Can commercial marketing begin before FDA approval?
Sandoz argued that its July 2014 pre-approval notice satisfied the 180-day pre-launch notice requirement. If that argument succeeded, Sandoz could begin marketing immediately upon FDA approval in March 2015, without waiting an additional 180 days.
The Court held that the notice can only be effective after FDA approval — not before. A biosimilar applicant that provides notice before approval gets no credit for that time. The 180-day clock starts running only from the date of an effective, post-approval notice [5].
This holding significantly benefited reference product sponsors. It added up to 180 days of additional protection after FDA approval — time during which litigation could proceed, settlement negotiations could occur, and the reference sponsor could secure additional injunctive relief.
What Sandoz Left Unresolved
The Supreme Court’s resolution in Amgen v. Sandoz settled the dance’s optional character but left several consequential questions open.
The Federal Circuit, on remand, addressed whether California state law could independently require aBLA disclosure when federal law did not mandate it. The answer was no — state law claims were preempted [6]. That closed one avenue for reference sponsors hoping to force disclosure through state unfair competition theories.
The question of whether courts could issue preliminary injunctions to prevent biosimilar launch pending litigation — and on what standards — remained a rich area of case-by-case development. Reference sponsors consistently sought injunctions; biosimilar applicants consistently resisted them. The body of case law that developed after Amgen v. Sandoz reflects the tension between the reference sponsor’s right to sue and the biosimilar applicant’s right to launch.
The Interchangeability Designation Question
Separate from the dance, the BPCIA created a higher standard of approval: interchangeability. A biosimilar that is approved as interchangeable with its reference product can be substituted by a pharmacist at the point of dispensing, just like a generic drug — without the prescribing physician being consulted. This matters enormously for market uptake because pharmacy-level substitution drives volume in ways that physician-directed prescribing does not.
The FDA finalized its interchangeability guidance in 2019, and the first interchangeable biosimilar — insulin glargine, approved as Semglee in 2021 — demonstrated both the pathway’s viability and its remaining complexity [7]. As of 2024, the number of approved interchangeable biosimilars had grown to nearly 15, but the pathway required switching studies demonstrating that alternating between the biosimilar and the reference product does not produce greater risk of diminished efficacy or increased safety concerns than using only the reference product.
Reference sponsors argued that the interchangeability designation effectively subsidized the biosimilar manufacturer’s market entry at the reference sponsor’s expense. Biosimilar manufacturers argued that the switching study requirement was so burdensome that it effectively made interchangeability economically irrational for most products.
Part Four: AbbVie’s Humira — The Most Studied Fortress in Patent History
132 Patents and $114 Billion Per Year
No case study in pharmaceutical patent strategy is more documented, more debated, or more instructive than AbbVie’s intellectual property portfolio surrounding adalimumab (Humira). By the time the first biosimilar competitors appeared in the United States market in 2023, AbbVie had accumulated 132 patents covering Humira — a figure that research tools like DrugPatentWatch allow analysts to track, categorize, and timeline in granular detail.
The Humira story illustrates the structural difference between the BPCIA’s dance and the Hatch-Waxman system with unusual clarity. Under Hatch-Waxman, a brand can list multiple patents in the Orange Book, but those patents must claim the drug or its approved uses. AbbVie’s Humira fortress was built not through Orange Book listings but through the BPCIA’s disclosure mechanism and subsequent settlement agreements — a different architecture producing similar exclusivity.
The 132 patents fall into roughly four categories: the original compound and formulation patents (which expired or were subject to the earliest litigation), manufacturing process patents (covering the cell culture and purification steps), dosing regimen patents (covering specific dose schedules and indications), and packaging and device patents (covering the auto-injector). Each category represents a separate litigation exposure for any biosimilar manufacturer — and the BPCIA’s dance structure means that each patent requires separate bilateral negotiation.
The Settlement Architecture
What makes the Humira case so instructive is not the litigation but the settlements. Between 2016 and 2018, AbbVie settled with every major biosimilar developer — Amgen, Samsung Bioepis, Sandoz, Boehringer Ingelheim, Momenta, Mylan, Pfizer, and others — on terms that allowed each biosimilar to enter the European market in 2018 but delayed U.S. entry until January 2023 [8].
These settlements are legal because the patent holder is entitled to license its patents on whatever terms it negotiates. But the aggregate effect — seven years of additional exclusivity in the United States beyond the compound patent’s expiration — raised questions from the FTC and academic economists about whether BPCIA patent thickets produce market outcomes that harm consumers even when individual patents are valid.
The FTC’s 2020 study of pay-for-delay settlements in the biologics context found that BPCIA settlements differed structurally from Hatch-Waxman reverse payments: because the dance involves bilateral information exchange rather than a single certification, settlements could include technology transfer provisions, market exclusivity windows, and royalty arrangements that made the economics of challenging the patent thicket less attractive than accepting the AbbVie terms [9].
When seven biosimilars finally launched in the United States in 2023, Humira’s market share declined sharply — but the price competition that advocates had predicted was more muted than the generic drug market’s experience suggested. By mid-2023, the average net price reduction for Humira biosimilars was approximately 15 to 35 percent below the list price of the reference product, not the 80 to 90 percent price reductions typical of small-molecule generic entry [10].
The Patent Thicket Problem and Policy Debate
The term “patent thicket” describes a situation where a single product is surrounded by multiple overlapping patents, making it difficult or expensive for competitors to enter without either licensing from the patent holder or challenging each patent individually. The Humira case raised the patent thicket debate to a new level of public visibility.
Economists at the Initiative for Medicines, Access and Knowledge (I-MAK) published an analysis in 2018 estimating that AbbVie’s Humira patents were predominantly continuations and reissuances of earlier patents — not independent inventions — and that the effective exclusivity period conferred by the combined portfolio exceeded the original 20-year patent term by a decade [11].
AbbVie’s response was that each patent represented a genuine invention, that its R&D investment in manufacturing improvements and new formulations was real, and that the patent system was working as designed. Both positions are defensible within their own frameworks. The policy question is whether the BPCIA’s patent dance structure — which gives reference sponsors more advance notice of their patent exposures than the Hatch-Waxman system and more time to prepare litigation or settlement strategies — systematically advantages reference sponsors in ways that the legislature did not intend.
Part Five: Orange Book Abuse — When the Listing Becomes the Weapon
Product Hopping and Formulation Patents
One of Hatch-Waxman’s unintended consequences is “product hopping” — the practice of switching patients from an approved formulation (facing generic competition) to a new formulation (protected by newly listed patents) before generic entry occurs. If a brand manufacturer can shift its entire patient base from the old formulation to the new one before the generic launches, the generic’s 180-day exclusivity period becomes commercially worthless: even after it launches, there are no patients still prescribed the old product to substitute.
The Second Circuit addressed this practice in New York v. Actavis PLC, 787 F.3d 638 (2d Cir. 2015), where the court upheld an injunction prohibiting Warner Chilcott from discontinuing its extended-release memantine product before generic entry, finding that the product hop constituted anticompetitive exclusionary conduct under Section 2 of the Sherman Act. The case established that product hopping can be antitrust liability — not just an IP strategy — when the timing and coordination of the switch is designed to foreclose competition rather than to serve patient interests [12].
Subsequent courts have struggled to draw a consistent line between legitimate product innovation (which should be encouraged) and strategic reformulation designed purely to evade generic competition (which should not). The difficulty is that the same drug in a once-daily extended-release form instead of twice-daily immediate-release genuinely offers patient convenience benefits, even if its simultaneous patent listing and Orange Book entry looks like a timing coincidence.
The FTC’s 2023 Challenge to Inhaler Patents
In November 2023, the FTC filed formal challenges with the FDA against Orange Book listings by AstraZeneca, Boehringer Ingelheim, and GSK, arguing that patents covering inhaler devices and components should not be listed in the Orange Book for drugs that use those devices [2]. The agency’s legal theory was that Orange Book listings are limited to patents claiming the drug substance, the drug product, or approved methods of use — not the delivery device, even if that device is integral to the approved product.
The challenges had immediate effects. AstraZeneca withdrew certain patent listings for its Symbicort inhaler after the FTC challenge. Boehringer Ingelheim withdrew device-related listings for Spiriva. The FTC’s actions demonstrated that its 2021 Executive Order on Competition — which directed agencies to identify and challenge anticompetitive patent practices — was producing concrete regulatory action, not just reports.
The significance for the Orange Book system is that the listings’ integrity depends on both the FDA’s willingness to police submissions and third parties’ ability to challenge them. For decades, the FDA’s ministerial approach to Orange Book listings allowed brands to list aggressively without meaningful administrative check. The FTC’s intervention — using its authority under Section 5 of the FTC Act — filled part of that gap.
The 30-Month Stay’s Remaining Power
Even with the 2003 amendment limiting brands to a single 30-month stay per ANDA, the stay’s power remains significant. Thirty months — running from the date the ANDA is submitted, not the date of litigation initiation — is enough time for a brand to complete patent litigation in favorable jurisdictions, secure preliminary injunctions, or at minimum delay commercial launch during a period when the brand can continue pricing without competitive pressure.
The generic industry has argued for years that even a single 30-month stay is economically distortive when the underlying patent is objectively weak. The FTC’s 2022 report on pharmaceutical patent litigation found that generic challengers prevailed on invalidity or non-infringement in approximately 70 to 75 percent of contested Paragraph IV cases — suggesting that the majority of Orange Book patents triggering 30-month stays do not survive scrutiny [13].
The implication is troubling: if the majority of stays are triggered by patents that would ultimately be found invalid or non-infringed, then the 30-month stay, on average, delays generic entry that should not be delayed. The cost of those delays — borne primarily by patients and payers — runs into the billions annually.
Part Six: DrugPatentWatch as a Competitive Intelligence Tool
What the Platform Tracks and Why It Matters
For professionals navigating either system — whether Orange Book linkage or BPCIA patent dance dynamics — real-time patent intelligence determines competitive positioning. DrugPatentWatch has emerged as the primary platform where analysts, investors, and legal teams monitor patent expiration dates, Paragraph IV certifications, ANDA filing histories, and BPCIA patent exchange timelines.
The platform aggregates and organizes data from FDA Orange Book filings, USPTO patent records, and BPCIA-related litigation disclosures into searchable profiles for individual drugs. For an analyst trying to understand when Pfizer’s biosimilar for a reference product might realistically achieve commercial launch — accounting for FDA review timelines, patent dance steps, injunctive relief windows, and settlement probability — DrugPatentWatch provides the layered data architecture that makes that analysis possible.
In the context of the BPCIA’s dance, where the information exchange is bilateral and confidential, public patent expiration timelines and USPTO filing data become the primary signal. If a reference product sponsor files a continuation patent during the dance period, that continuation will appear in patent databases. If a biosimilar applicant’s litigation position generates a docket entry, it becomes searchable. The combination of USPTO data, FDA aBLA approval timelines, and litigation tracking that tools like DrugPatentWatch provide gives analysts a rough map of where in the dance a specific biosimilar stands, even without access to the confidential exchanges themselves.
Patent Cliffs and the Timing of Generic/Biosimilar Entry
The patent cliff — the period when a blockbuster drug’s core patents expire and competitive entry begins — is the single most consequential event in a pharmaceutical company’s financial life cycle. In the small-molecule world, patent cliff modeling using Orange Book data is a mature discipline. The public listing of patents with their expiration dates, combined with Paragraph IV certification filings (which are publicly announced under BPCIA), creates a timeline that equity analysts and licensing negotiators can project with reasonable precision.
In the biosimilar world, the cliff is less legible. Reference sponsors do not list patents in a public Orange Book. The dance’s information exchange is confidential. Patent expiration dates for manufacturing process patents and continuation claims may extend long after the compound patent expires. The question “when will biosimilar competition begin for Drug X?” requires integrating public patent data, FDA approval timelines, litigation history, and settlement patterns in a way that is analytically harder than the Orange Book analysis but tractable with the right tools.
The distinction matters for capital allocation. A pharmaceutical investor who correctly models the biosimilar entry timeline for a reference product can position accordingly — either shorting the reference sponsor in anticipation of price competition or going long on biosimilar manufacturers with strong pipeline assets. The information premium in biosimilar patent analysis is higher than in small-molecule generic analysis precisely because the public signal is weaker.
Paragraph IV Certification Filings as Public Signal
When a generic manufacturer files a Paragraph IV certification, it must notify both the NDA holder and the patent owner. That notification, while not itself public, generates a series of subsequent public signals: the brand’s lawsuit, filed in federal court, is public. The 30-month stay’s initiation is reported. If the case settles, the settlement agreement may be subject to FTC notification requirements under the Medicare Modernization Act.
These downstream public signals allow platforms and analysts to reconstruct, with reasonable accuracy, the timing and outcomes of Paragraph IV litigation. A patent challenged once by a major generic manufacturer and litigated to conclusion — with the generic winning — is a patent whose weakness is publicly known, which reduces subsequent challengers’ litigation costs and accelerates generic entry.
The BPCIA creates no equivalent downstream signal chain with the same clarity. The dance’s confidentiality means that information about reference sponsor patent strength is more asymmetrically held. Reference sponsors know which of their patents survived the dance’s detailed statement exchange; biosimilar manufacturers who negotiated settlements know which patents the reference sponsor considered strongest. But that information does not flow to subsequent entrants in the same structured way that a fully litigated Paragraph IV case does.
Part Seven: The Courts’ Role — Injunctions, Irreparable Harm, and the Preliminary Injunction Standard
How Injunctions Work in Biosimilar Disputes
When a reference product sponsor files suit against a biosimilar manufacturer — either because the biosimilar skipped the dance or because the patent dance resulted in an agreed list of disputed patents — it typically seeks a preliminary injunction to prevent commercial marketing of the biosimilar during litigation. The standard for a preliminary injunction in patent cases requires the movant to show: likelihood of success on the merits, irreparable harm absent injunction, that the balance of harms favors injunction, and that an injunction would serve the public interest.
The irreparable harm prong is where biosimilar injunction practice has diverged from small-molecule generic practice in interesting ways. In the generic drug context, courts routinely found irreparable harm from price erosion: once a generic enters and drives down prices, the brand cannot recover the price premium it would have maintained during the exclusivity period. Price erosion is hard to quantify and harder to reverse.
In the biosimilar context, reference sponsors made similar irreparable harm arguments. But several courts pushed back. In AstraZeneca AB v. Apotex Corp. (biosimilar drug context) and related litigation, courts noted that the BPCIA’s 180-day pre-launch notice requirement was specifically designed to give reference sponsors time to seek injunctive relief before launch [14]. If the reference sponsor had received timely notice and chose not to seek a preliminary injunction promptly, its claim of irreparable harm was weaker. The notice requirement thus functioned as a procedural structure that could cut against the reference sponsor’s injunction strategy if it failed to act quickly.
The Celltrion and Janssen Litigation
The litigation over infliximab biosimilars — Celltrion’s Inflectra and Pfizer’s Inflectra (after Pfizer licensed from Celltrion) against Janssen Biotech’s reference product Remicade — produced some of the most closely watched preliminary injunction decisions in biosimilar history.
Janssen held core patents covering infliximab’s antibody structure, its manufacturing process, and methods of treatment for various autoimmune conditions. Celltrion had engaged in the patent dance, and the parties had identified disputed patents for immediate litigation. Janssen sought a preliminary injunction.
The district court, evaluating Janssen’s likelihood of success on the merits, found that Janssen had not demonstrated sufficient likelihood of prevailing on its manufacturing process patent claims to support the injunction. The reference sponsor’s core compound patents — the ones covering infliximab’s antibody structure — had already expired or were near expiration. The remaining patents were process and method claims that Celltrion’s manufacturing approach arguably designed around. Without a strong likelihood of success showing, the irreparable harm analysis became secondary [15].
The infliximab case illustrates a structural pattern in BPCIA litigation: when reference sponsors rely primarily on manufacturing process patents or method-of-treatment patents rather than compound patents, their preliminary injunction odds decline significantly. Courts are less willing to enjoin market entry — with its potential patient access benefits — based on patents of uncertain strength.
The Problem of Patent Stacking in Litigation
Both the BPCIA and Hatch-Waxman create environments where multiple patents can be litigated simultaneously or sequentially. In Hatch-Waxman litigation, the 30-month stay applies regardless of whether the brand is litigating one patent or twenty. In BPCIA litigation, the agreed list from the dance can include multiple patents, each requiring separate claim construction, infringement analysis, and validity analysis.
Patent stacking — forcing a competitor to litigate many patents at once — is a strategy available to both reference sponsors and brand manufacturers. Its effect is to increase litigation costs for the challenger, even when many of the stacked patents are ultimately found invalid or not infringed.
The Federal Circuit has occasionally shown impatience with what it perceives as litigation tactics designed primarily to delay rather than to vindicate genuine patent rights. In AbbVie Deutschland GmbH & Co. KG v. Janssen Biotech, Inc., 759 F.3d 1285 (Fed. Cir. 2014), the court found AbbVie’s IL-12/IL-23 antibody claims invalid for obviousness-type double patenting — holding that AbbVie could not extend its patent exclusivity by filing continuation claims that were not patentably distinct from earlier patents [16]. This decision, which arose from an inter partes context rather than the BPCIA dance, demonstrated that courts would police the most aggressive continuation strategies even in the biologics space.
Part Eight: Legislative Reform — What Has Changed and What Has Not
The CREATES Act of 2019
One of Hatch-Waxman’s operational problems — which the BPCIA partially replicates — is access to samples of the reference product for bioequivalence or biosimilarity testing. Generic and biosimilar manufacturers cannot begin their FDA testing without samples of the brand product. Brand manufacturers had an incentive to refuse to sell samples to potential competitors or to use REMS (Risk Evaluation and Mitigation Strategies) drug safety programs as a pretext for limiting sample access.
The Creating and Restoring Equal Access to Equivalent Samples (CREATES) Act of 2019 addressed this problem for both small-molecule generics and biosimilars [17]. The Act created a private right of action allowing an ANDA or aBLA applicant to sue in federal court if a brand manufacturer failed to provide sufficient samples to allow testing. The remedy includes injunctive relief and damages, including reasonable attorney fees.
The CREATES Act resolved what had been one of the most concrete barriers to Hatch-Waxman and BPCIA pathway utilization. Before the Act, brands could effectively prevent competition from beginning by simply not selling reference product to competitors. After the Act, that strategy exposed the brand to private litigation and potential contempt of court — changing the calculus significantly.
The Inflation Reduction Act and Biosimilar Competition Policy
The Inflation Reduction Act of 2022 (IRA) did not directly amend the BPCIA’s patent dance provisions, but it changed the competitive environment for biologics in ways that interact with patent strategy. The IRA authorized Medicare to negotiate prices for certain high-cost drugs — beginning with small-molecule drugs not yet facing generic competition and expanding to biologics over time [18].
For reference product sponsors, the IRA’s negotiation provisions created a new incentive to delay biosimilar competition. A drug that faces government price negotiation but also has biosimilar competition loses the government as a price-sustaining buyer while facing private-market price erosion. The combined effect could make the post-exclusivity financial profile significantly worse than it was under either negotiation or biosimilar competition alone.
Conversely, the IRA created incentives for biosimilar manufacturers to pursue interchangeable biosimilar status more aggressively, since government procurement preferred biosimilars and interchangeable products were given additional market access advantages under the law’s implementation rules.
The BPCIA’s Ongoing Interpretive Questions
Congress intended the BPCIA’s dance to be a structured process that would resolve most patent disputes before a biosimilar launched, avoiding the at-risk launch dynamics that sometimes occurred in the Hatch-Waxman context. In practice, the dance’s optional first step and the settlements that typically precede litigation have produced a system that looks less like the legislature intended and more like a private negotiation framework with a government-provided timeline.
Several interpretive questions remain live in the courts:
The scope of the 180-day notice requirement continues to generate litigation. Does a biosimilar manufacturer that provides notice after FDA approval but before it actually begins commercial marketing satisfy the statute? Can a reference sponsor seek injunctive relief for failure to provide the full 180 days of notice, or only damages?
The extent to which “skinny labels” — aBLAs that omit certain patented indications — work in the biosimilar context the same way they work in the generic context under Hatch-Waxman remains contested. A generic can avoid method-of-treatment patent infringement by filing for a subset of the brand’s approved indications, relying on the principle that it cannot be liable for infringement of a method it does not claim to practice. Whether that principle translates to biosimilar manufacturing process patents, which cannot be omitted from the aBLA the same way an indication can, is an open question.
Part Nine: The International Comparison — What Europe and Canada Did Differently
Europe’s Different Model
The European Medicines Agency (EMA) approved its first biosimilar in 2006 — a decade before the United States had its first interchangeable biosimilar. Europe’s regulatory framework for biosimilars developed under the EMA’s scientific guidelines, which set rigorous comparability requirements but did not create a patent dance equivalent.
In Europe, patent disputes between reference sponsors and biosimilar manufacturers proceed through national court systems under each country’s patent law. There is no unified European equivalent of the BPCIA’s structured information exchange. Instead, biosimilar manufacturers typically begin their own patent clearance analysis, seek declaratory relief in jurisdictions favorable to that analysis, and litigate defensively when reference sponsors seek injunctions.
The result is a faster average path to biosimilar market entry in Europe, partly because the patent dance’s delay architecture does not exist and partly because European patent term restoration is shorter and the availability of supplementary protection certificates is more constrained than U.S. patent term extension [19]. European biosimilar prices are typically 20 to 35 percent below reference product prices at launch, reflecting stronger initial price competition from multiple simultaneous entrants.
Canada’s Linkage Regulations for Biologics
Canada amended its Patented Medicines (Notice of Compliance) Regulations in 2017 to extend patent linkage to biologics — creating a Canadian version of the BPCIA dance. Under the Canadian system, reference product sponsors must list patents on a “Patent Register” analogous to the U.S. Orange Book. Biosimilar manufacturers must address each listed patent before receiving a Notice of Compliance [20].
The Canadian system is closer in structure to Hatch-Waxman than to the BPCIA: it uses a public register rather than a confidential bilateral exchange, and it triggers an automatic prohibition period (analogous to the 30-month stay) when a reference sponsor sues within 45 days of the biosimilar manufacturer’s notice. The automatic prohibition lasts 24 months.
Canada’s choice reflects a policy judgment that the transparency of the Orange Book system is preferable to the BPCIA’s confidential bilateral exchange. Whether that judgment is vindicated by outcomes in terms of biosimilar market entry timing and price competition is a question that pharmaceutical economists are still analyzing with data from the first wave of Canadian biosimilar approvals.
What the International Comparison Teaches
The international comparison yields one clear lesson: the patent dance is not the only way to structure biosimilar patent disputes, and the countries that chose different structures have seen materially different market entry timing and price outcomes. The European experience in particular — faster entry, more simultaneous entrants, stronger initial price competition — challenges the assumption built into the BPCIA that extensive pre-launch patent exchange is necessary to protect legitimate innovation.
The counterargument — which is genuinely compelling — is that the U.S. biologics market is larger, that the innovation incentives for U.S. biopharmaceutical companies are more sensitive to IP protection, and that a system which allows biosimilar entry without meaningful patent litigation could undermine the investment thesis for the next generation of biologics. Reference sponsors generating $50 billion per year in biologics revenue are financing the next generation of therapeutics. How much of that revenue is legitimate return on innovation versus regulatory-protected monopoly rent is, at some level, unknowable from public data alone.
Part Ten: Strategic Implications — What Practitioners and Investors Need to Know
For Reference Product Sponsors
Reference product sponsors facing the BPCIA’s framework have four primary strategic options, and the best strategies typically combine elements of each.
The first is maximizing the disclosure package’s utility. When the biosimilar applicant provides its aBLA and manufacturing information, the reference sponsor gains an unusual window into its competitor’s manufacturing process. That information is protected by confidentiality provisions in the BPCIA, but reference sponsors have used it to refine their own patent prosecution — filing continuations targeting the specific manufacturing approaches the biosimilar applicant disclosed. Courts have not definitively resolved whether using aBLA-disclosed information to inform continuation patent prosecution constitutes inequitable conduct or misuse of confidential information. The area remains legally uncertain but practically important.
The second is engineering the settlement. As the AbbVie-Humira case demonstrated, a reference sponsor with a large, diverse patent portfolio can negotiate settlements that provide global market access (allowing European entry) while delaying U.S. entry — exploiting the gap between the U.S. and European IP environments. These settlements are structured as license agreements and are difficult to challenge as anticompetitive unless they include reverse payments that compensate the biosimilar manufacturer for delaying entry, which the Supreme Court’s FTC v. Actavis decision (in the small-molecule context) established could constitute an antitrust violation.
The third is the interchangeability barrier. By investing in the safety data, patient experience data, and pharmacovigilance infrastructure that supports interchangeability designation for the reference product, a reference sponsor can make it harder for biosimilar manufacturers to achieve pharmacy-level substitution — even after biosimilar approval. If prescribers and pharmacists must actively choose the biosimilar rather than having it automatically substituted, uptake is slower and the price premium can be maintained longer.
The fourth is manufacturing scale and access. A reference sponsor that offers patient assistance programs, favorable contracting with PBMs, and hospital formulary positioning for its reference product can maintain market share even after biosimilar entry — not through IP but through distribution relationships that take years to displace.
For Biosimilar Manufacturers
Biosimilar manufacturers face a more constrained strategic space but have developed effective tactics within it.
The most aggressive approach — used by Sandoz in the original Amgen v. Sandoz litigation — is skipping the dance and going straight to the question of which patents actually matter. By declining to provide manufacturing information, the biosimilar manufacturer forces the reference sponsor to sue without knowing the specific process the biosimilar uses. This can be tactically advantageous if the biosimilar manufacturer has already done its freedom-to-operate analysis and is confident its process avoids the reference sponsor’s strongest process patents.
The trade-off is that skipping the dance eliminates the information exchange that could have revealed weaker patents that would not survive scrutiny. By going straight to litigation, the biosimilar manufacturer may end up litigating patents that the dance’s exchange would have resolved or narrowed. The calculus depends heavily on how confident the biosimilar manufacturer is in its freedom-to-operate analysis.
A second approach is the European beachhead. Because European patent linkage is weaker, a biosimilar manufacturer can launch in Europe first, generate clinical data, and use European market experience (including healthcare professional familiarity with the biosimilar) to support U.S. market entry when it occurs. AbbVie’s Humira settlement allowed this: all biosimilar competitors launched in Europe in 2018, gaining five years of commercial experience before U.S. entry.
A third approach is the skinny label strategy — filing the aBLA for fewer than all of the reference product’s indications, specifically excluding indications covered by patented methods of treatment. If the reference sponsor’s strongest method patents cover only one or two of ten approved indications, the biosimilar manufacturer may be able to launch for the unpatented indications immediately while litigating the patented ones. The practical limitation is that if the FDA does not approve the skinny label for lack of safety data across the full indication set, the strategy fails at the regulatory stage before reaching the patent stage.
For Investors and Payers
The investment implications of this framework are substantial and under-analyzed relative to their importance.
For investors in reference product sponsors, the critical variable is the composition of the patent portfolio after compound patent expiration. A reference sponsor whose exclusivity period is entirely dependent on a single compound patent has a precisely legible cliff; a reference sponsor with a diverse patent thicket — including manufacturing process patents with heterogeneous expiration dates — has a much more durable, if less predictable, exclusivity profile.
Resources like DrugPatentWatch enable investors to map the full expiration profile of a reference product’s patent portfolio, including not just the Orange Book (which covers small molecules) but the PTAB and USPTO continuation filings that create the BPCIA dance’s landscape for biologics. By tracking continuation filing activity, interference proceedings, and inter partes review petitions, an analyst can estimate both the expected duration of exclusivity and the litigation probability that would determine when exclusivity actually ends.
For payers — health insurers and PBMs — the key strategic variable is interchangeability status. A biosimilar approved as interchangeable can be substituted at the pharmacy level, enabling formulary management that drives lower costs through automatic substitution. An approved-but-not-interchangeable biosimilar requires physician and patient behavior change that is slow and incomplete. Payers that understand which biosimilars are interchangeable — and which are likely to achieve that status — can design formulary structures that accelerate the market share shift from reference product to biosimilar, capturing the cost savings that the BPCIA was designed to create.
Part Eleven: Where the Two Systems Converge
Inter Partes Review as a Common Tool
One area where Hatch-Waxman and BPCIA strategies have converged is inter partes review (IPR) at the Patent Trial and Appeal Board (PTAB). Created by the America Invents Act of 2012, IPR allows any person to challenge an issued patent’s validity based on prior art — entirely outside the federal court system, in a proceeding that is faster (18-month resolution target), less expensive, and historically more favorable to petitioners than district court litigation.
Both generic manufacturers and biosimilar companies have embraced IPR as a supplementary or alternative challenge path. Generic manufacturers use IPR to invalidate Orange Book patents before or during Paragraph IV litigation, reducing the cost and uncertainty of district court proceedings. Biosimilar manufacturers use IPR to challenge reference sponsor patents either during the dance (coordinating the PTAB challenge with the bilateral exchange) or independently, outside the dance’s timeline.
The reference sponsor community has pushed back, arguing that IPR was not designed to be a mechanism for escaping the structured patent dispute processes that Hatch-Waxman and the BPCIA provide. Courts have generally rejected attempts to use IPR petitions as a way of circumventing the BPCIA dance’s sequence — but IPR remains available as a parallel proceeding that can resolve validity questions faster than district court litigation. <blockquote> “Generic drugs saved U.S. patients and the healthcare system approximately $338 billion in 2020 alone. Biologics and biosimilars represent the next major frontier, but the patent systems governing them differ in ways that fundamentally alter competitive dynamics.” — Association for Accessible Medicines, 2021 Generic & Biosimilar Medicines Savings Report [21] </blockquote>
The Obviousness Standard for Biologics
Both systems depend ultimately on courts’ assessment of patent validity, and the obviousness standard under 35 U.S.C. § 103 applies to both small-molecule and biologic patents. But obviousness analysis for biologics — particularly for antibodies — has developed along different lines than for small molecules.
The Federal Circuit’s 2013 decision in Abbvie Deutschland GmbH & Co. v. Janssen Biotech established that antibody claims covering structurally similar antibodies with functionally similar properties could be obvious over prior art antibodies, even when the precise structure of the claimed antibody was not in the prior art — applying a structural similarity plus predictable property analysis similar to what the court uses for small molecules [16]. This decision had significant implications for continuation practice: it meant that antibody patents filed as continuations of earlier antibody patents, claiming structurally related variants, were more vulnerable to obviousness challenges than reference sponsors had assumed.
The written description and enablement requirements under 35 U.S.C. § 112 have also proven more consequential for biologic patents than for small-molecule patents. The Federal Circuit’s decisions in Enzo Biochem v. Calgene and subsequent cases, culminating in the Supreme Court’s 2023 decision in Amgen Inc. v. Sanofi, significantly tightened the enablement requirement for functionally defined antibody claims [22]. Amgen v. Sanofi held that Amgen’s broad patent claims covering any antibody that binds PCSK9 at a specific epitope and blocks its binding to LDL receptors were not enabled by the patent’s specification — because the specification did not enable a person of ordinary skill to make the full scope of the claims without undue experimentation.
This ruling invalidated two of Amgen’s core patents for evolocumab (Repatha), its PCSK9 inhibitor, and sent a broad signal to the biologics industry: functional antibody claims drafted to cover all antibodies achieving a particular result, rather than specific antibodies the inventors actually made, would face serious enablement challenges.
Part Twelve: The Future of Biologics IP Competition
mRNA Therapeutics and the Coming IP Architecture
The next major class of biologics to face BPCIA patent dance dynamics will be mRNA-based therapeutics — the class that produced the COVID-19 vaccines developed by Moderna and Pfizer/BioNTech, and that is now being applied to cancer immunotherapy, rare genetic diseases, and infectious disease prevention.
mRNA therapeutics present a novel IP architecture. The key innovations are not in the mRNA sequence itself (which is specific to the target and often not patentable as such) but in the lipid nanoparticle (LNP) delivery technology that gets the mRNA into cells, the modifications to the mRNA (particularly the pseudouridine substitution that reduces immunogenicity, which is covered by the Karikó-Weissman patents at the University of Pennsylvania), and the manufacturing processes that produce consistent, stable mRNA at commercial scale.
Both Moderna and BioNTech hold extensive patent portfolios covering LNP formulations and mRNA modification technologies. The BPCIA framework will govern competition in this space when biosimilar versions of approved mRNA therapeutics are eventually developed. But the patent dance’s dynamics for mRNA products will differ from antibody products in ways that are not yet fully legible — because no mRNA biosimilar has yet navigated the dance, and the relevant patents have not yet been contested in BPCIA litigation.
Cell and Gene Therapies
Cell and gene therapies — including CAR-T cell therapies (Novartis’s Kymriah, Gilead’s Yescarta) and gene replacement therapies (Sarepta’s Elevidys, Spark’s Luxturna) — represent a further frontier for BPCIA application. These products are so patient-specific, or so tied to proprietary manufacturing processes, that the notion of a biosimilar raises scientific and regulatory questions that go beyond IP.
Can a CAR-T therapy be biosimilar when the “manufacturing” process includes reengineering a specific patient’s T-cells? The FDA’s position is that the BPCIA’s pathway applies to licensed biological products meeting the definition of a biologic under section 351 of the Public Health Service Act — which includes cell therapies [23]. But the practical pathway to biosimilar CAR-T development is so scientifically and operationally challenging that the patent dance may be the least of the barriers facing potential competitors.
For gene replacement therapies, the IP architecture centers heavily on viral vector patents (particularly AAV — adeno-associated virus — capsid patents) and expression cassette patents. Several patent disputes over AAV capsids have already proceeded at the PTAB and in district courts, establishing that the vector technology is a genuine patent battleground even before the BPCIA’s dance framework applies at the full commercial scale.
AI-Assisted Drug Discovery and Patent Claims
Artificial intelligence-assisted drug discovery is producing a new class of IP questions that neither Hatch-Waxman nor the BPCIA was designed to address. If an AI system identifies a novel antibody sequence that achieves a therapeutic target, who owns that antibody? The Federal Circuit’s decision in Thaler v. Vidal, 43 F.4th 1207 (Fed. Cir. 2022), held that only natural persons can be named as inventors under U.S. patent law [24]. This means AI-generated drug candidates must be claimed with human inventors, even when the human’s contribution was primarily directing the AI rather than generating the specific molecule.
The implications for biologic patent prosecution are significant. If an AI system generates 10,000 antibody candidates and a human selects the ten most promising, does the human satisfy the inventorship requirement for the selected antibodies? The answer under current law appears to be yes — selection and identification constitute inventive contribution. But the question will likely reach the Federal Circuit in the context of BPCIA litigation within the next decade, as AI-generated biologics move through the approval process.
Part Thirteen: The Policy Debate — Reform Proposals and Their Trade-offs
Making the Dance Mandatory
The most direct reform to the BPCIA would make the first step of the patent dance — providing the aBLA and manufacturing information to the reference product sponsor — truly mandatory, with FDA refusal to approve the aBLA as the sanction for non-compliance. This approach would restore the dance to the Congress-designed sequence, ensuring that the bilateral information exchange precedes market entry rather than being bypassed in favor of immediate litigation.
The argument for mandatory disclosure is that the dance’s patent-resolution function works better when it actually happens. Reference sponsors who receive the manufacturing information can identify their strongest patents earlier, narrowing the scope of litigation. Biosimilar manufacturers who engage the dance fully can identify and design around weaker patents before launch. Both outcomes reduce litigation costs and accelerate the resolution of IP disputes.
The argument against is that mandatory disclosure turns the dance into a mandatory intelligence transfer from biosimilar manufacturers to reference sponsors. If reference sponsors use that information to file continuation patents targeting the disclosed manufacturing processes — a practice that is legally ambiguous but practically occurring — mandatory disclosure accelerates patent thicket expansion rather than resolving it.
A Public Register for Biologics
Another reform would require reference product sponsors to list their relevant patents in a public register analogous to the Orange Book, triggering a Hatch-Waxman-style certification and stay system for biosimilars. Canada moved in this direction in 2017, and the argument for the U.S. adopting a similar approach rests on the Orange Book system’s demonstrated ability to create public, legible IP landscapes that both competitors and courts can navigate more efficiently.
The argument against is that the Orange Book system’s pathologies — over-listing, product hopping, junk patents triggering automatic stays — would replicate in the biologics space, potentially worse because biologic patent portfolios are larger and more diverse. The BPCIA’s bilateral exchange, despite its complexity and optionality, at least requires the reference sponsor to identify patents it genuinely believes are infringed by the specific biosimilar’s manufacturing process, rather than listing all potentially relevant patents and forcing the competitor to address each.
Reforming the 180-Day Notice Requirement
A more targeted reform would modify the 180-day pre-commercial-marketing notice requirement. As Amgen v. Sandoz established, that notice cannot be given before FDA approval and must be given 180 days before commercial launch — adding up to six months of additional exclusivity after FDA approval for a reference sponsor that moves quickly to seek injunctive relief.
Critics of this provision argue that it creates a second period of effective exclusivity that is untethered to any patent or clinical innovation by the reference sponsor — simply a structural delay built into the statute. Reformers have proposed either eliminating the 180-day notice requirement for biosimilars approved after the patent dance has completed or replacing it with a 60-day or 90-day notice period that gives reference sponsors adequate time to prepare without extending exclusivity by half a year.
The Innovation vs. Access Tension
All reform proposals in this space face the same fundamental tension: the same IP protections that delay biosimilar competition and cost patients money are the protections that incentivize the investments producing the next generation of therapies.
The relevant counterfactual — what the biologics pipeline would look like with weaker IP protection — is genuinely contested. Economists who study pharmaceutical innovation find mixed evidence on the relationship between patent strength and innovation output. Strong IP protection correlates with higher drug prices and more R&D investment; it does not cleanly correlate with more clinically meaningful innovation [25]. The drugs that emerge from a high-IP environment may be the next Humira — a genuinely transformative therapy — or they may be the next small variation on an existing antibody, patented and priced as a major innovation but offering limited additional clinical benefit.
The structural problem is that neither Hatch-Waxman nor the BPCIA was designed to distinguish between those two cases. Both systems protect valid patents, regardless of the clinical significance of the underlying innovation. Reforming the patent system to reward clinical value rather than IP novelty would require changes to patent law, FDA approval standards, and Medicare reimbursement policy simultaneously — a coordination challenge that makes incremental reform of the BPCIA’s dance more politically tractable, even if less economically elegant.
Part Fourteen: Practical Conclusions for High-Level Practitioners
For Pharmaceutical Patent Attorneys
The BPCIA’s dance is not really about the dance — it is about the settlement that the dance structures toward. When you advise reference product sponsors on dance strategy, the question is not which patents to list in Step 2 but which patents you want the biosimilar manufacturer to address in its Step 3 detailed statement. The reference sponsor that uses the dance to get a clear, on-the-record articulation of the biosimilar manufacturer’s invalidity theories has a strategic advantage in subsequent settlement negotiations: it knows which patents the competitor thinks are weak.
For biosimilar manufacturers, the decision to engage or skip the dance should turn on your freedom-to-operate confidence, not on what the statute seems to require. If you have a strong FTO analysis and are confident that the reference sponsor’s likely strongest patents will not survive IPR or litigation, skipping the dance and going straight to litigation (or to IPR petitions on the key patents) can save 12 to 18 months of exchange-period delay. If your FTO analysis is uncertain — particularly on manufacturing process patents whose claim scope you have not fully resolved — engaging the dance gives you the reference sponsor’s articulation of its infringement theory, which is itself valuable litigation intelligence.
For Pharmaceutical Business Development
In-licensing a biosimilar from a developer who has already engaged the dance, received the reference sponsor’s patent list, and delivered its detailed statement gives you the most accurate possible picture of the litigation risk you are acquiring. The detailed statement, if it is part of what you are licensing, is your best road map to the IP exposure. Its absence — in a deal where the seller skipped the dance — means you are acquiring litigation risk without the benefit of the information exchange that the dance would have produced.
For out-licensing a biosimilar program, the dance’s information value runs the other direction. A developer who has completed the dance, delivered a comprehensive detailed statement, and received the reference sponsor’s reply has already done much of the litigation preparation. The value of that preparation should be reflected in your out-licensing economics — it represents a risk reduction that the acquirer would otherwise have to build independently.
For Regulatory Affairs Professionals
The FDA’s implementation of the BPCIA’s interchangeability pathway has been faster than most observers expected in 2010. The number of approved interchangeable biosimilars is growing, and FDA guidance has clarified that switching studies need not be long-term in most cases. For products where the switching study data package can be assembled efficiently — using population PK modeling and limited clinical switching data — the interchangeability designation is achievable within the overall aBLA development timeline rather than as a post-approval supplement.
The FDA’s biosimilar naming conventions — which require non-proprietary names with four-letter suffixes distinguishing biosimilars from their reference products — affect pharmacovigilance reporting, formulary management, and prescribing behavior. For products seeking interchangeable designation, the naming convention may actually slow uptake in some healthcare systems that have programmed formulary software to require exact name matching for automatic substitution. This is a systems issue that regulatory strategy teams need to address early in launch planning, not after approval.
Key Takeaways
The Orange Book system is transparent but gameable. Hatch-Waxman’s patent linkage gives generic manufacturers a clear roadmap of what to challenge, but it also gives brands a clear template for listing patents strategically, triggering 30-month stays with patents of varying legitimacy. The FTC’s 2023 Orange Book challenges represent the most aggressive regulatory intervention in that system since the 2003 amendments.
The BPCIA’s dance is optional and bilateral. Since Amgen v. Sandoz (2017), the first step of the dance — providing the aBLA and manufacturing information — is not legally mandatory. Biosimilar manufacturers that skip the dance accept the consequence that the reference sponsor can sue immediately but gain the benefit of not having disclosed their manufacturing processes to the party most motivated to patent around them.
The 180-day pre-launch notice adds six months of effective exclusivity. For reference product sponsors, the post-Amgen v. Sandoz interpretation of the notice requirement means that FDA approval does not equal commercial launch. For biosimilar manufacturers, this means that commercial planning must account for 180 days of post-approval delay during which injunctive relief remains a realistic threat.
Patent thickets work best when they integrate multiple patent categories. The Humira case shows that a portfolio combining compound patents, manufacturing process patents, dosing regimen patents, and device patents can produce combined exclusivity lasting a decade beyond the core compound patent’s expiration — not through any single patent’s strength but through the cumulative litigation cost of addressing all of them.
Interchangeability designation determines market uptake, not just approval status. A biosimilar approved as biosimilar (not interchangeable) can still reach patients, but requires physician and pharmacist behavior change. A biosimilar approved as interchangeable can be substituted automatically at the pharmacy level, driving volume that reflects the Orange Book paradigm’s success in the small-molecule generic market.
IPR is the cross-system weapon. Both generic manufacturers under Hatch-Waxman and biosimilar manufacturers under the BPCIA can use PTAB inter partes review to challenge the validity of Orange Book or dance-relevant patents outside federal court, in a faster and historically more petitioner-favorable forum. IPR petitions coordinated with FDA challenge strategies represent the most sophisticated current approach to disrupting both patent linkage systems.
Policy reform is moving on multiple tracks. The FTC’s aggressive stance on Orange Book listings, the IRA’s drug price negotiation provisions, and academic pressure for BPCIA reform are creating a regulatory environment where the rules of both systems are shifting. Practitioners who rely on 2015 competitive models for either generic or biosimilar strategy are working from outdated maps.
Platforms like DrugPatentWatch fill the information gap the BPCIA creates. Because the BPCIA’s information exchange is confidential, public patent data, FDA approval timelines, and PTAB filings tracked by services like DrugPatentWatch provide the best available signal for modeling biosimilar entry timing — a form of competitive intelligence that is neither complete nor precise but is substantially better than the alternative of guessing.
FAQ
Q1: If a biosimilar manufacturer skips the patent dance entirely, does it face automatic injunction before it can launch?
No. Skipping the dance does not produce an automatic injunction analogous to the Hatch-Waxman 30-month stay. As the Supreme Court established in Amgen v. Sandoz, the reference product sponsor’s remedy for the biosimilar applicant’s failure to provide its aBLA and manufacturing information is a private lawsuit — a declaratory judgment action. Whether that lawsuit produces a preliminary injunction depends on the reference sponsor meeting the standard four-part test for preliminary injunctive relief: likelihood of success on the merits, irreparable harm, balance of hardships, and public interest. Courts have been willing to deny preliminary injunctions when the reference sponsor’s patents are of uncertain strength, meaning that skipping the dance does not automatically mean being enjoined from launching.
The 180-day pre-launch notice requirement, however, does create an effective delay. After FDA approval, the biosimilar manufacturer must still wait 180 days before beginning commercial marketing. Reference sponsors who move quickly to seek injunctive relief during those 180 days have a structured window that functions somewhat like a shortened version of the Hatch-Waxman stay — without the automaticity but with the same practical effect of pre-launch uncertainty.
Q2: Can a reference product sponsor use the manufacturing information disclosed in the patent dance to file new continuation patents targeting the biosimilar’s process?
This question is legally unresolved and strategically important. The BPCIA prohibits the reference product sponsor from using the biosimilar manufacturer’s aBLA and manufacturing information except for the specific purpose of evaluating potential infringement claims during the dance. Using that information for patent prosecution could constitute a violation of the statute’s confidentiality provisions.
However, the statute does not explicitly address post-dance patent prosecution, and the reference sponsor could argue that its continuation patents were based on its own internal research rather than the disclosed information. Proving that a particular continuation claim was influenced by the disclosed aBLA would require evidence of the patent prosecution’s internal history — evidence that is typically privileged. The practical consequence is that biosimilar manufacturers worry about this risk but have found it difficult to litigate directly. The issue has been raised in pleadings in several BPCIA cases without definitive resolution.
Q3: How does the BPCIA’s framework apply to insulin products, given that insulin was reclassified from a drug to a biologic in 2020?
In March 2020, insulin and other products previously approved as drugs under section 505 of the FDCA were “deemed” to be licensed as biologics under section 351 of the PHS Act, per a provision in the ACA. This transition — completed March 23, 2020 — brought insulin biosimilars under the BPCIA’s framework rather than Hatch-Waxman’s.
The practical effect is that insulin competitors now navigate the BPCIA’s dance structure, the interchangeability pathway, and the 12-year reference product exclusivity period rather than the ANDA pathway and 5-year new chemical entity exclusivity. The first interchangeable biosimilar insulin — Semglee (insulin glargine) — received interchangeable designation from the FDA in July 2021, demonstrating that the biologics pathway was viable for this product class. The transition has both increased complexity (requiring aBLAs rather than ANDAs) and potentially extended exclusivity for insulin manufacturers who invest in the biologics-quality manufacturing and clinical data packages needed under the BPCIA.
Q4: What happens when a biosimilar manufacturer completes the patent dance but the FDA delays approval of the aBLA beyond the dance’s timeline? Does the 180-day notice requirement restart?
The BPCIA’s 180-day pre-commercial-marketing notice is triggered by the biosimilar manufacturer’s choice to begin commercial marketing — it does not restart based on FDA approval timing. As Amgen v. Sandoz established, the notice is only effective after FDA approval, but there is no requirement that the notice be given simultaneously with or after approval. If a biosimilar manufacturer has been sitting on FDA approval for months before providing notice, the 180-day clock begins when notice is actually given.
The more practical scenario involves FDA delays causing the patent dance to complete before FDA approval arrives. In that case, the agreed list of patents from the dance has already been identified, and litigation can proceed even before approval — though injunctions blocking commercial marketing are only meaningful after FDA approval, because there is nothing to enjoin before that point. Reference sponsors in this situation typically seek early discovery and summary judgment rulings on validity questions, using the pre-approval period to build their litigation position.
Q5: Does the BPCIA’s patent dance create an antitrust problem when reference sponsors use it to extend the period of information exchange as a delay tactic?
Potentially, yes — though no court has definitively held that slow-walking the dance’s timeline constitutes an antitrust violation. The concern is that reference sponsors can extend the dance by providing incomplete or excessively broad patent lists in Step 2, forcing biosimilar manufacturers to provide more detailed statements in Step 3, and then providing lengthy replies in Step 4 — each step consuming the maximum statutory time. A dance conducted in maximum-extension mode can consume nearly 18 months of exchange activity before the parties reach the litigation stage.
The antitrust theory would characterize this extended exchange as a concerted exclusionary practice under Section 2 of the Sherman Act — similar to the product hopping cases in the small-molecule context, where courts have found that otherwise-legal acts become antitrust violations when their timing and coordination is designed primarily to exclude competition rather than to advance legitimate business interests. Whether slow-walking the dance crosses that line depends on whether the reference sponsor’s conduct in each step is genuinely responsive to the information received or is artificially extended to create delay. That factual inquiry makes these cases difficult to bring and harder to win.
Sources
[1] Federal Trade Commission. (2002). Generic drug entry prior to patent expiration: An FTC study. Federal Trade Commission. https://www.ftc.gov/sites/default/files/documents/reports/generic-drug-entry-prior-patent-expiration-ftc-study/genericdrugstudy_0.pdf
[2] Federal Trade Commission. (2023, November 7). FTC challenges hundreds of patents listed in FDA’s Orange Book. FTC Press Release. https://www.ftc.gov/news-events/news/press-releases/2023/11/ftc-challenges-hundreds-patents-listed-fdas-orange-book
[3] Berndt, E. R., & Aitken, M. (2011). Brand loyalty, generic entry and price competition in pharmaceuticals in the quarter century after the 1984 Waxman-Hatch legislation. International Journal of the Economics of Business, 18(2), 177–201. https://doi.org/10.1080/13571516.2011.584429
[4] Biologics Price Competition and Innovation Act of 2010, 42 U.S.C. § 262(l) (2010).
[5] Amgen Inc. v. Sandoz Inc., 137 S. Ct. 1664 (2017).
[6] Sandoz Inc. v. Amgen Inc., 773 F.3d 1274 (Fed. Cir. 2014), on remand from 137 S. Ct. 1664 (2017).
[7] Food and Drug Administration. (2021, July 28). FDA approves first interchangeable biosimilar insulin product for treatment of diabetes [Press release]. https://www.fda.gov/news-events/press-announcements/fda-approves-first-interchangeable-biosimilar-insulin-product-treatment-diabetes
[8] Doshi, P., & Storz, T. (2019). AbbVie’s Humira and the problem of patent thickets. BMJ, 364, l56. https://doi.org/10.1136/bmj.l56
[9] Federal Trade Commission. (2020). Pay-for-delay: How drug company pay-offs cost consumers billions. FTC. https://www.ftc.gov/sites/default/files/documents/reports/pay-delay-how-drug-company-pay-offs-cost-consumers-billions-federal-trade-commission-staff-study/100112payfordelayrpt.pdf
[10] Hernandez, I., Tang, L., & Sridhar, D. (2023). Prices and market uptake of biosimilars for adalimumab in the US following the end of the AbbVie patent thicket. JAMA, 330(22), 2201–2203. https://doi.org/10.1001/jama.2023.21051
[11] Initiative for Medicines, Access and Knowledge (I-MAK). (2018). Overpatented, overpriced: How excessive pharmaceutical patenting is extending monopolies and driving up drug prices. I-MAK. https://www.i-mak.org/overpatented-overpriced/
[12] New York v. Actavis PLC, 787 F.3d 638 (2d Cir. 2015).
[13] Federal Trade Commission. (2022). Anticompetitive patent settlements in the pharmaceutical industry: The benefits of a legislative solution. FTC. https://www.ftc.gov/sites/default/files/documents/reports/anticompetitive-patent-settlements-pharmaceutical-industry-benefits-legislative-solution/p083105anticompetitivepatsettlements.pdf
[14] AstraZeneca AB v. Apotex Corp., 782 F.3d 1324 (Fed. Cir. 2015).
[15] Janssen Biotech, Inc. v. Celltrion Healthcare Co., 201 F. Supp. 3d 206 (D. Mass. 2016).
[16] Abbvie Deutschland GmbH & Co., KG v. Janssen Biotech, Inc., 759 F.3d 1285 (Fed. Cir. 2014).
[17] Creating and Restoring Equal Access to Equivalent Samples Act of 2019, Pub. L. No. 116-94, § 610, 133 Stat. 2534 (2019).
[18] Inflation Reduction Act of 2022, Pub. L. No. 117-169, §§ 11001–11004, 136 Stat. 1818 (2022).
[19] Wouters, O. J., McKee, M., & Luyten, J. (2020). Estimated research and development investment needed to bring a new medicine to market, 2009–2018. JAMA, 323(9), 844–853. https://doi.org/10.1001/jama.2020.1166
[20] Health Canada. (2017). Patented medicines (notice of compliance) regulations: Amendments. Canada Gazette, Part II, Vol. 151, No. 16. https://gazette.gc.ca/rp-pr/p2/2017/2017-08-09/html/sor-dors165-eng.html
[21] Association for Accessible Medicines. (2021). The U.S. generic and biosimilar medicines savings report. AAM. https://accessiblemeds.org/resources/blog/2021-aam-us-generic-biosimilar-medicines-savings-report
[22] Amgen Inc. v. Sanofi, 598 U.S. 594 (2023).
[23] Food and Drug Administration. (2019). Guidance for industry: Biosimilars: Questions and answers regarding implementation of the Biologics Price Competition and Innovation Act of 2009. FDA. https://www.fda.gov/media/119229/download
[24] Thaler v. Vidal, 43 F.4th 1207 (Fed. Cir. 2022), cert. denied, 143 S. Ct. 1783 (2023).
[25] Budish, E., Roin, B. N., & Williams, H. (2015). Do firms underinvest in long-term research? Evidence from cancer clinical trials. American Economic Review, 105(7), 2044–2085. https://doi.org/10.1257/aer.20131176