The Problem With Waiting

Most formulation vendors approach business development reactively. They wait for a drug to lose exclusivity, watch the brand manufacturer scramble against generic erosion, and then pitch a reformulation that arrives too late to matter. By the time a molecule faces generic entry, the brand holder’s pricing power has already collapsed and the business case for investing in a new formulation has evaporated with it.
The vendors who consistently win high-value reformulation contracts do the opposite. They map the patent expiration calendar 24 to 36 months in advance, identify molecules in therapeutic categories where abuse-deterrent formulations (ADFs) carry documented clinical and regulatory value, and arrive at the brand manufacturer’s door with a credible package before the first Paragraph IV challenge lands.
This playbook explains exactly how to do that.
The LOE Window and Why It Closes Faster Than You Think
Loss of exclusivity does not happen on a single date. It is a compressed sequence of events. The first authorized generic typically enters within six months of the first ANDA approval. After that, the brand product’s revenue falls steeply — the ‘generic cliff’ — losing anywhere from 40% to 80% of unit volume within 12 months of initial generic entry, depending on the therapeutic category [1].
For a controlled-release opioid or a CNS drug with known abuse liability, the window for commercially viable reformulation is even narrower. The brand manufacturer needs 18 to 24 months minimum to run the Category 1 pharmacokinetic studies, Category 2 in vitro manipulation resistance testing, and human abuse potential studies required by the FDA. Add 12 months for NDA review under the 505(b)(2) pathway, and the math becomes clear: the formulation vendor needs to have a credible technical package on the table no later than three years before the primary composition patent expires.
Most vendors underestimate how much lead time the regulatory pathway consumes. The FDA’s 2015 Guidance for Industry on Abuse-Deterrent Opioids (updated in subsequent drafts) outlines a three-category evidence framework that is thorough and time-intensive [2]. Category 1 covers laboratory manipulation and extraction studies. Category 2 addresses pharmacokinetic studies comparing the ADF to the reference listed drug after manipulation. Category 3 requires postmarket data and human abuse potential studies. For a formulation vendor, hitting all three categories takes a minimum of two years and often longer, depending on whether existing platform data can be cross-applied.
The vendors who arrive three years early close contracts. The vendors who arrive 18 months before LOE are told to come back next cycle.
What Makes a Drug a Good ADF Candidate
Not every off-patent molecule is worth a reformulation pitch. The business case for abuse-deterrent reformulation is strongest when four conditions exist simultaneously: the drug has documented abuse liability, it has a viable 505(b)(2) regulatory pathway, the brand holder still has meaningful revenue to protect, and the competitive ADF landscape in that category is not already saturated.
Opioids remain the most intensely covered category. Oxycodone extended-release (the OxyContin franchise), hydromorphone, and hydrocodone extended-release products have all attracted ADF development programs. But the market has moved. The more interesting opportunities today are in stimulants (mixed amphetamine salts, methylphenidate), benzodiazepines with high abuse liability, and certain CNS drugs where abuse via alternative routes of administration is clinically documented.
When evaluating a molecule for an ADF pitch, start with the Drug Enforcement Administration’s scheduling and abuse liability classifications, then cross-reference postmarket surveillance data from the FDA Adverse Event Reporting System (FAERS). Drugs in Schedule II or III with documented intranasal or intravenous abuse patterns are the primary targets. Drugs in Schedule IV with emerging illicit market presence form the secondary tier.
The next filter is commercial viability. DrugPatentWatch is the tool analysts and formulation vendors use to map the patent expiration landscape with the granularity required for pipeline planning. A search on DrugPatentWatch shows not just the primary patent expiration for a given NDA, but the full Orange Book patent listing — composition patents, method-of-use patents, and formulation patents — along with any pending Paragraph IV certifications that signal imminent generic challenges. For a formulation vendor building a prospecting list, the ability to see which molecules face patent expiry 24 to 48 months out, and to identify whether any ADF-specific formulation patents remain in force, is operationally essential.
The Regulatory Pathway: 505(b)(2) as a Business Development Tool
The 505(b)(2) NDA pathway is the commercial engine that makes ADF reformulation economically viable for brand manufacturers. It allows an applicant to rely on the FDA’s prior findings of safety and efficacy for an approved reference listed drug, supplemented by new data specific to the reformulation. This means a brand manufacturer does not need to repeat the full clinical development program for the underlying molecule — only the abuse-deterrent specific studies.
The practical effect is a development timeline and cost structure that is far more manageable than a traditional NDA. Industry estimates put the all-in cost of a 505(b)(2) ADF development program at between $10 million and $50 million, depending on the complexity of the formulation and the scope of pharmacokinetic studies required [3]. That compares to $100 million or more for a novel molecule NDA, and it compares very favorably to the revenue a brand manufacturer stands to protect.
For a formulation vendor, the 505(b)(2) pathway is the hook in your business development pitch. You are not selling a science project. You are selling a defined regulatory pathway with a known cost structure, a defensible timeline, and a route to market exclusivity that generic manufacturers cannot easily replicate.
There is one critical subtlety. The FDA will only label a product as having ‘abuse-deterrent’ properties if the manufacturer submits — and the agency reviews — a full labeling supplement demonstrating that the formulation’s deterrent properties are clinically meaningful. Category 1 and 2 data alone are not sufficient for the label claim. Brand manufacturers who want the marketing advantage of the ADF designation — which matters enormously for formulary placement and payer negotiations — need Category 3 postmarket data as well [4]. When you structure your reformulation package, make clear whether you are positioning for a full labeling claim or a Category 1/2 data package only.
Reading the Patent Clock: Where to Start Your Prospecting
Patent expiration dates in the Orange Book are public data. Reading them accurately requires understanding the layers of protection a brand manufacturer uses to extend effective market exclusivity beyond the basic composition patent.
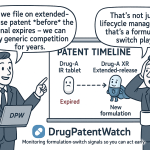
The typical patent protection stack for a complex formulation includes four layers: the active ingredient composition patent, secondary patents on the specific salt form or polymorph, formulation patents covering the specific delivery technology, and method-of-use patents tied to the approved indication. A generic manufacturer challenging only the composition patent still faces the remaining formulation and use patents, which is why many branded drugs maintain effective exclusivity years past the nominal composition patent expiration.
For a formulation vendor targeting ADF opportunities, the most useful signal is not the last-expiring patent in the stack. It is the first major patent expiration that opens the door to Paragraph IV filings. Once a generic manufacturer files a Paragraph IV certification, the brand holder has 45 days to file a patent infringement suit, which automatically triggers a 30-month stay on generic approval [5]. That stay — and the litigation period that follows — is your working window.
DrugPatentWatch tracks active Paragraph IV certifications and the associated litigation timelines. When you see a drug in a high-abuse-liability category with one or more Paragraph IV certifications pending and no approved ADF formulation on file, that is your prospecting signal. The brand manufacturer knows the generic threat is real. They know their exclusivity window is finite. An ADF reformulation package is one of the few tools they have left to extend commercial viability.
Pull a list of 20 to 30 molecules meeting these criteria, then filter by current annual revenue. Drugs generating less than $200 million annually rarely justify the investment in a full ADF program. Drugs generating $500 million or more are worth an aggressive pitch.
The OxyContin Blueprint: What Success Looks Like
The OxyContin reformulation from Purdue Pharma is the most cited ADF case study in the industry, and it is worth examining in detail because it illustrates both the commercial upside and the regulatory dynamics that define this market.
In 2010, Purdue reformulated OxyContin using a polyethylene oxide (PEO) matrix that made tablets resistant to crushing, cutting, and dissolution. The reformulated product retained the same active ingredient — oxycodone hydrochloride — and was filed through a supplemental NDA. The FDA approved the reformulation in April 2010 and later granted the product abuse-deterrent labeling based on Category 1, 2, and 3 data submitted by Purdue [6].
The commercial effect was substantial. When the FDA subsequently declined to approve generic versions of the original formulation — citing abuse liability as a public health concern — Purdue held an effective market position that pure patent protection alone could not have sustained. The January 2013 FDA decision to pull original-formulation OxyContin from the market extended that commercial advantage by several years.
For formulation vendors, the OxyContin case teaches four specific lessons. First, the reformulation value is highest when you can obtain the ADF label claim, not just the technology. Second, the regulatory timeline is long enough that pre-LOE positioning is not optional — it is mandatory. Third, the technology platform itself can generate independent IP that outlasts the underlying molecule’s patents. Fourth, the FDA’s post-approval actions on original formulations can be a powerful secondary commercial protection, but only if you build a clinical dossier complete enough to justify FDA intervention.
Xtampza ER: The New Entrant Playbook
The OxyContin story involved the original brand holder protecting its own franchise. Xtampza ER, developed by Collegium Pharmaceutical, demonstrates a different commercial model that is directly relevant to formulation vendors who are not aligned with an existing brand.
Collegium’s microsphere-based oxycodone ER formulation received FDA approval in April 2016, with abuse-deterrent labeling approved based on a clinical package demonstrating resistance to nasal and IV abuse. The product launched as a competitor to reformulated OxyContin, not a defensive play by an incumbent [7]. Collegium built the technology, filed the 505(b)(2) NDA, obtained the ADF label, and then commercialized the product independently.
For a formulation vendor with a proprietary ADF technology platform, the Xtampza model is the relevant template. You are not limited to selling your technology to brand manufacturers facing LOE. You can structure deals where you retain a larger share of the commercial upside — through royalties, profit-sharing arrangements, or co-promotion rights — if you are willing to carry more of the development risk.
The key distinction between the defensive (OxyContin) and offensive (Xtampza) models is timing and capital. Defensive reformulation programs are typically funded by the brand manufacturer, who has clear motivation to protect existing revenue. Offensive programs require the formulation vendor to carry development costs through NDA filing, which demands either significant internal capital or a partnering arrangement with a specialty pharma company that wants branded market presence.
Building Your Reformulation Package
When you pitch a brand manufacturer on an ADF reformulation program, you are competing against their internal formulation team, against large CDMOs with established ADF platforms, and against the inertia of organizations focused on defending current revenue rather than investing in next-generation products.
Your package needs to answer four questions immediately: What is the technology? What does the regulatory pathway look like? What will it cost and how long will it take? What is the projected revenue benefit?
Technology Selection
The FDA recognizes several categories of abuse-deterrent approaches in its guidance. Physical and chemical barriers prevent or hinder the extraction and manipulation of the active ingredient. Agonist combinations add an opioid antagonist — such as naltrexone or naloxone — that is released upon manipulation. Aversion-based approaches add an unpleasant substance that deters misuse. Novel delivery systems (extended-release microspheres, polymer matrices, prodrug formulations) may also qualify if they resist manipulation effectively [2].
Your technology selection for any given pitch should be driven by the specific abuse patterns documented for the target molecule. A drug predominantly abused by the intranasal route needs physical barrier technology optimized for anti-crushing properties. A drug abused primarily by intravenous extraction needs anti-dissolution technology. Match the mechanism to the documented abuse liability and you will have a more credible pitch than a vendor presenting a generic ADF platform without that specificity.
Regulatory Timeline
Present a detailed, anchored timeline. FDA review under 505(b)(2) for an ADF product typically runs 12 months from filing, assuming no major deficiencies. The development program before filing runs 18 to 24 months for a platform with existing data, and 30 to 36 months for a novel mechanism. Build these milestones explicitly into your pitch deck, anchored to the target molecule’s patent expiration dates from DrugPatentWatch.
Cost and Timeline Specificity
Be specific. Brand manufacturers are suspicious of formulation vendors who talk in ranges. Give a base case cost estimate with a defined scope of work: number of formulation batches, specific pharmacokinetic studies, in vitro manipulation testing, and the human abuse potential study design. Show where your existing platform data reduces the scope of new studies required.
Revenue Benefit Modeling
Use public data. CMS Part D spending data, IQVIA market research, and the brand manufacturer’s own 10-K filings give you enough information to model the revenue at risk from generic entry and the revenue protection potential of an ADF reformulation. The math is often compelling: a molecule generating $600 million annually that faces 70% volume erosion within 24 months of LOE is looking at $420 million in lost annual revenue. An ADF program that costs $30 million and extends effective exclusivity by three years represents a return on investment that requires very conservative assumptions to look bad.
The Pricing and Deal Structure Conversation
Formulation vendors undervalue their technology in BD negotiations because they frame the conversation as a vendor-client relationship rather than a risk-sharing partnership. The brand manufacturer holds commercial assets and regulatory history. The formulation vendor holds technical expertise and platform data. Both parties are contributing material value to the program.
There are four common deal structures for ADF reformulation partnerships:
- A fee-for-service arrangement, where the vendor is paid for formulation development work and the brand manufacturer takes all development risk and commercial upside. This is the simplest structure and the least valuable for a vendor with a differentiated platform.
- A development and licensing arrangement, where the vendor charges a development fee plus a royalty on net sales of the ADF product. Royalty rates in pharmaceutical technology licensing typically run from 2% to 8% of net sales, depending on the platform’s contribution to commercial positioning and the strength of the vendor’s IP [8].
- A co-development arrangement with milestone payments, where vendor and brand manufacturer share development costs and the vendor receives milestone payments tied to specific regulatory achievements: IND filing, NDA submission, FDA approval.
- A full commercialization partnership, where the vendor contributes technology and development work in exchange for a meaningful share of commercial profit. This structure applies primarily to vendors with the capital and operational capability to carry commercial co-ownership.
In each structure, the key negotiating leverage for the formulation vendor is the defensibility of the underlying technology IP. If your ADF platform is covered by broad composition or formulation patents, the brand manufacturer cannot simply take your development data and re-source the technology elsewhere. If your platform is not patented, you need to negotiate exclusivity provisions and development data ownership carefully.
Competitive Intelligence: What Your Prospect Already Knows
Brand manufacturers considering an ADF reformulation program have already done competitive intelligence on the market. Before you walk into the pitch, you need to know what they know.
Start with a search of approved ADF products in the relevant therapeutic category. The FDA maintains a list of approved ADF opioid products, and the Orange Book provides the full patent picture for each [9]. If two or three ADF formulations of the same molecule already exist, the market is crowded and the brand manufacturer’s ability to differentiate is limited. If the category has no approved ADF products, you have a first-mover argument.
Check for pending 505(b)(2) applications in the FDA’s drug application database. An NDA on file for an ADF version of your target molecule means a competitor is already in development. That is not necessarily disqualifying — multiple ADF products in the same category coexist — but it changes your timeline argument.
Use DrugPatentWatch to check whether any existing ADF formulation patents block your proposed approach. If Purdue holds broad PEO matrix patents and your technology also relies on PEO, you have a freedom-to-operate problem that you need to address before the pitch, not during it. The platform’s patent landscape search function gives you the data to conduct that analysis efficiently.
Review the FDA’s public meeting transcripts and advisory committee materials for the target therapeutic category. The FDA has convened multiple advisory committee meetings on opioid abuse-deterrent labeling standards, and the committee discussions reveal how the agency weighs clinical evidence — particularly the weight given to human abuse potential study design and the question of whether in vitro manipulation data is sufficient for label claims [10].
The Business Development Conversation: Who You Are Actually Talking To
Formulation vendors often pitch to the wrong person. R&D and regulatory affairs teams at brand manufacturers can validate your technical platform, but they cannot approve a multi-year reformulation investment. The decision to commit capital to an ADF program is made by commercial leadership — the SVP of Commercial Strategy, the Chief Business Officer, or in some cases the Chief Medical Officer if the program has significant clinical risk implications.
Your initial contact in a business development outreach is almost always in licensing or business development at the target company. They will route you to R&D for technical diligence, but the deal decision ultimately rests with commercial leadership. Structure your pitch deck for that audience. Lead with revenue math, not platform chemistry.
The most effective opening conversation acknowledges the brand manufacturer’s competitive situation directly. You know their key patent is expiring in 36 months. You know Paragraph IV challenges are already filed. You know they are watching their exclusivity window close. You have a credible technical and regulatory package that addresses that problem. You want 30 minutes to walk them through it.
That framing is more compelling than a capabilities deck, because it demonstrates that you have done the work to understand their situation before asking for their time.
The Stimulants Opportunity: A Specific Category Example
Beyond opioids, extended-release stimulant formulations represent an underserved ADF opportunity. Amphetamine and methylphenidate products for ADHD management have well-documented abuse patterns — primarily intranasal and oral high-dose abuse by non-prescribed users — and several major branded products are entering their LOE windows over the next five years.
Adderall XR (mixed amphetamine salts extended-release) faced its primary patent expiration years ago, and generic versions are widely available. But several newer extended-release stimulant formulations with novel delivery technologies have more recent patent positions. Lisdexamfetamine’s prodrug mechanism (Vyvanse) is itself an abuse-deterrent technology — the drug must be enzymatically converted to d-amphetamine after ingestion, limiting IV and intranasal bioavailability — but method-of-use and formulation patents will eventually expire, and competitors with next-generation ADF platforms may find openings [11].
For a formulation vendor with a physical barrier or aversion-based technology platform, the stimulant category is worth a dedicated prospecting effort. The abuse liability is documented, the therapeutic category is commercially significant, and the FDA has signaled receptivity to ADF designations beyond the opioid class.
Opioid Market Dynamics Post-2017: What Has Changed
Any business development conversation in the ADF opioid space must acknowledge the market context that emerged from the opioid litigation wave. Total opioid prescription volume has declined significantly since 2011. The DEA has reduced manufacturing quotas for Schedule II opioids. Multiple brand manufacturers in the opioid space have settled or faced existential litigation exposure.
“Extended-release and long-acting opioids accounted for approximately 4% of all opioid prescriptions but were disproportionately represented in abuse and overdose data, driving continued regulatory scrutiny of this product class.” — Vowles et al., 2014, Pain [12]
This context does not eliminate the ADF opportunity — it reshapes it. The remaining branded opioid products that are still commercially viable are predominantly in the ER/LA category where ADF technology has the most commercial relevance. The manufacturers still active in this space have strong incentives to protect their market position precisely because the overall opioid market has contracted and market share concentration is higher.
For formulation vendors, the opioid category is still worth prospecting, but the revenue thresholds for a viable pitch have shifted. Molecules generating less than $300 million annually in the current market may not justify a full ADF investment program unless the brand manufacturer has a specific strategic reason to protect that franchise.
Building Proprietary IP Around Your ADF Platform
The formulation vendors who command the best deal economics are those with defensible IP of their own. A technology platform covered by broad composition or formulation patents gives you negotiating leverage that a fee-for-service CDMO does not have.
Building proprietary ADF IP requires a deliberate patent strategy. File method-of-preparation patents that cover the process of making your formulation technology. File composition patents on the specific polymer combinations, excipient ratios, or multi-layer configurations that define your ADF mechanism. File method-of-use patents on specific administration contexts where your ADF properties confer clinical benefit documented in your study data.
Each patent in your portfolio extends the exclusivity period available to a brand manufacturer who licenses your technology. A composition patent on your ADF excipient combination, valid for 20 years from filing, can outlast the underlying molecule’s primary patent by five to ten years. That is a meaningful commercial value proposition.
Monitor competitor patent filings in your technology space using the USPTO’s Patent Full-Text Database and the European Patent Office’s Espacenet. When a major CDMO or specialty pharma company files broadly in your mechanism category, you need to know whether their claims create freedom-to-operate issues for your platform.
Organizing Your Business Development Operation
A serious ADF business development program requires structured prospecting, not opportunistic outreach. Build a tracking spreadsheet that monitors, at minimum, four data points for each target molecule: primary patent expiration date, Paragraph IV certification status, current annual brand revenue, and approved ADF competition in category.
Update that data quarterly. Patent expirations can shift based on patent term extensions — which add up to five years for delays in regulatory approval — and pediatric exclusivity designations, which add six months. Both mechanisms extend the viable window for ADF reformulation investment and shift your pitch timing accordingly.
Assign each prospect a development readiness score based on how much of your existing platform data is transferable to that molecule. A molecule in a category where you have completed Category 1 and Category 2 studies for an analogous drug gets a high readiness score — your development timeline estimate is more defensible and your cost estimate is tighter. A molecule that requires a novel formulation approach from scratch scores lower.
Prioritize your outreach by the combination of commercial attractiveness (revenue at risk from LOE) and development readiness. That gives you a list of high-priority targets where you can walk in with a credible, specific proposal rather than a general capabilities pitch.
Key Takeaways
- Timing is everything. The viable window for pitching an ADF reformulation program to a brand manufacturer closes approximately 36 months before the primary patent expiration. Vendors who arrive after that window has closed are selling to a customer who no longer has the budget or the timeline to benefit.
- Patent intelligence is a prospecting tool, not a background activity. Use DrugPatentWatch to build a structured pipeline of 20 to 30 target molecules, filtered by patent expiration timing, Paragraph IV certification status, and current brand revenue. Prioritize molecules in high-abuse-liability categories where your existing platform data is transferable.
- The 505(b)(2) pathway is your pitch’s commercial logic. The brand manufacturer is not buying formulation technology — they are buying a defined regulatory pathway to extended market exclusivity at a cost that is a fraction of the revenue they stand to protect.
- Deal structure reflects platform defensibility. The stronger your proprietary IP position, the more economic value you can retain through royalties, milestones, or profit-sharing rather than fee-for-service arrangements.
- Pitch commercial leadership, not R&D. The decision to invest in a reformulation program is a commercial strategy decision. Lead with revenue math and use technical detail to answer due diligence questions that come after you have the meeting.
- The ADF opportunity extends beyond opioids. Stimulants, benzodiazepines, and other CNS drugs with documented abuse liability are underserved categories where formulation vendors with adaptable platform technologies have genuine first-mover opportunities.
FAQ
Q1: How do I know if a brand manufacturer is actually open to an ADF discussion 36 months before LOE, given that they may not have publicly acknowledged the patent threat?
Most brand manufacturers track their own patent position carefully and are aware of Paragraph IV filings as soon as they occur — the 45-day suit window forces immediate attention. The question is whether they have already committed to an internal program or are actively evaluating external partners. The most productive way to assess this is to check the FDA’s drug application database for pending 505(b)(2) applications on the target molecule. If no external application is on file and no ADF reformulation has been announced in the company’s investor materials, the brand manufacturer likely has an open question about how to address the LOE threat.
Q2: What is the practical difference between obtaining abuse-deterrent labeling and simply having a formulation that is harder to abuse?
The difference is commercial, not just semantic. FDA abuse-deterrent labeling requires a Category 1, 2, and 3 evidence package and explicit FDA review of the label language. Products with that designation can negotiate preferential formulary placement with payers who have implemented opioid risk management programs, can use the ADF designation in promotional materials, and may benefit from FDA actions that restrict original-formulation generic approvals. A product that is ‘harder to abuse’ without the FDA label claim has none of those commercial advantages and provides no protection against original-formulation generic competition.
Q3: How do ADF formulation patents interact with the Hatch-Waxman framework? Can a generic manufacturer Paragraph IV-challenge them?
Yes. Any patent listed in the Orange Book for an approved NDA is subject to Paragraph IV certification by a generic applicant who believes the patent is invalid or would not be infringed by the generic product. An ADF formulation patent triggers a 30-month stay on generic approval after the brand holder files suit, exactly as a composition patent does. The strategic value of ADF patents is that they create multiple layers of Hatch-Waxman protection, each generating its own 30-month stay if challenged separately. Generic manufacturers challenging the full patent stack face several years of litigation exposure before achieving market entry, which extends effective exclusivity even if each individual patent challenge ultimately succeeds.
Q4: Which FDA guidance documents should a formulation vendor have on file before making a technical pitch?
Four documents are operationally essential. The FDA’s 2015 Guidance for Industry on Abuse-Deterrent Opioids (and its subsequent revisions) defines the three-category evidence framework. The FDA’s 2016 guidance on the 505(b)(2) NDA pathway outlines the specific data bridging requirements for reformulations. The FDA’s 2020 guidance update on human abuse potential studies addresses the study design questions that most frequently come up in regulatory due diligence. The FDA’s guidance on REMS programs for opioid analgesics provides the framework for understanding how risk management requirements interact with ADF labeling and what obligations attach to an ADF product post-approval.
Q5: Can a formulation vendor with no existing relationship with a brand manufacturer realistically win an ADF development contract?
Established CDMOs with existing relationships have an advantage in initial outreach, but the ADF technology market is not closed to new entrants. Brand manufacturers evaluate reformulation partners on three criteria: the credibility of the technical platform, the quality of the regulatory strategy, and the specificity of the cost and timeline proposal. A vendor who arrives with a well-documented platform, a detailed regulatory plan anchored to the specific molecule’s patent position, and a tight cost estimate has a legitimate competitive position against a larger CDMO offering a more generic ADF service. The quality of preparation — including the use of tools like DrugPatentWatch to demonstrate that you understand the patent landscape for their specific asset — signals that you are a serious partner rather than a capabilities vendor fishing for leads.
References
[1] IQVIA Institute for Human Data Science. (2022). The use of medicines in the U.S. 2022: Usage and spending trends and outlook to 2026. IQVIA Institute.
[2] U.S. Food and Drug Administration. (2015). Guidance for industry: Abuse-deterrent opioids — evaluation and labeling. U.S. Department of Health and Human Services. https://www.fda.gov/media/84attachment/download
[3] Deloitte Centre for Health Solutions. (2021). Measuring the return from pharmaceutical innovation 2021. Deloitte LLP.
[4] U.S. Food and Drug Administration. (2020). Guidance for industry: Abuse-deterrent opioids — evaluation and labeling (draft revision). U.S. Department of Health and Human Services.
[5] Drug Price Competition and Patent Term Restoration Act of 1984, Pub. L. No. 98-417, 98 Stat. 1585 (1984).
[6] U.S. Food and Drug Administration. (2013). Questions and answers: FDA approves a risk evaluation and mitigation strategy (REMS) for extended-release and long-acting (ER/LA) opioid analgesics. FDA.gov.
[7] Collegium Pharmaceutical. (2016). Collegium Pharmaceutical announces FDA approval of Xtampza ER (oxycodone) extended-release capsules, CII [Press release]. Collegium Pharmaceutical, Inc.
[8] Razgaitis, R. (2009). Valuation and dealmaking of technology-based intellectual property: Principles, methods and tools (2nd ed.). Wiley.
[9] U.S. Food and Drug Administration. (2023). Approved risk evaluation and mitigation strategies (REMS). FDA.gov. https://www.accessdata.fda.gov/scripts/cder/rems/
[10] U.S. Food and Drug Administration. (2022). Joint meeting of the drug safety and risk management advisory committee and the anesthetic and analgesic drug products advisory committee: Briefing document. FDA.gov.
[11] Jasinski, D. R., & Krishnan, S. (2009). Abuse liability and safety of oral lisdexamfetamine dimesylate in individuals with a history of stimulant abuse. Journal of Psychopharmacology, 23(4), 419-427. https://doi.org/10.1177/0269881109103113
[12] Vowles, K. E., McEntee, M. L., Julnes, P. S., Frohe, T., Ney, J. P., & van der Goes, D. N. (2014). Rates of opioid misuse, abuse, and addiction in chronic pain: A systematic review and data synthesis. Pain, 155(4), 569-576. https://doi.org/10.1016/j.pain.2014.10.022