Part I — The Foundational Divide
Why Biologic IP is a Different Discipline Entirely
Before building any patent strategy, a team must understand why small-molecule IP logic breaks down when applied to biologics. The divergence is not legal convention — it’s non-negotiable science.
A small-molecule drug like paroxetine (Paxil) has a molecular weight of roughly 329 g/mol. It’s a well-defined, reproducible chemical structure that can be synthesized precisely and described completely. A monoclonal antibody tips the scales at approximately 150,000 Daltons. You don’t synthesize it in a flask; you grow it inside living cells — typically genetically engineered Chinese Hamster Ovary (CHO) cells — under conditions that introduce inherent batch-to-batch variation. That variation is not a manufacturing defect; it is a scientific reality baked into every biologic ever produced.
The technical term for this is “inherent heterogeneity.” A batch of a biologic is a population of highly similar, but not structurally identical, molecules. The most consequential source of that variability is glycosylation — the complex sugar-coating patterns attached to the protein backbone. Glycoforms are not decorative. They determine a biologic’s safety profile, receptor binding efficacy, serum half-life, and immunogenicity. Change the glycosylation pattern and you have, in a meaningful scientific sense, a different drug.
“Because of inherent heterogeneity, it is scientifically impossible to create an exact replica of an innovator’s biologic. That scientific fact is the legal bedrock of the entire biosimilar regime — and the engine that powers the patent thicket.”
This is why regulatory frameworks created an entirely separate approval pathway. The Biologics Price Competition and Innovation Act of 2009 (BPCIA) governs biosimilar approvals in the U.S. A biosimilar is not a “generic” — it must demonstrate it is “highly similar” to the reference product with “no clinically meaningful differences” in safety, purity, and potency. That is a materially higher bar than the “same active ingredient” standard applied to generic small-molecule drugs under Hatch-Waxman.
The Orange Book vs. The Purple Book: An Asymmetry That Works for Innovators
Hatch-Waxman requires an innovator to proactively list all relevant patents in the FDA’s Orange Book, giving any would-be generic manufacturer complete, early visibility into the patent estate they will need to challenge. The BPCIA has no equivalent mandate. The FDA’s Purple Book does not require patent listing. A biosimilar developer often only learns the full scope of an innovator’s patent portfolio after they have already spent hundreds of millions of dollars on biosimilarity development — and have formally initiated the BPCIA “patent dance” process.
This asymmetry is structural, not accidental. It creates a “fog of war” for biosimilar developers and a material information advantage for innovators. For R&D and legal teams on the originator side, it is an underappreciated strategic asset.
| Feature | Small-Molecule (Hatch-Waxman) | Biologic (BPCIA) |
|---|---|---|
| Molecular scale | <900 Daltons; fully characterizable | 10,000-150,000+ Daltons; inherently heterogeneous |
| Competitor product | Generic (identical) | Biosimilar (highly similar, not identical) |
| Patent disclosure | Proactive listing in Orange Book | Reactive disclosure after biosimilar aBLA filing initiates the dance |
| Base regulatory exclusivity | 5 years (New Chemical Entity) | 12 years market / 4 years data |
| Core patent strategy | Single composition-of-matter claim on active ingredient | Layered patent thicket: molecule, cell line, process, formulation, device, dosing, diagnostics |
| Manufacturing relevance | Synthesis process rarely determines product identity | “Process is the product” — manufacturing pathway is an FDA-approved part of the product definition |
The 12-Year Exclusivity Myth
Congress designed the BPCIA’s 12-year market exclusivity assuming that biologic patents were inherently weaker and easier to design around than small-molecule patents — and therefore biologics needed a longer regulatory head-start to justify their higher development costs. That assumption has been proven spectacularly wrong. The 12-year clock is the opening bid. The Humira and Enbrel case studies demonstrate that the real monopoly, which can stretch to 30 years, is not maintained by FDA-granted exclusivity. It is maintained by the patent thicket.
Part II — The Amgen Earthquake
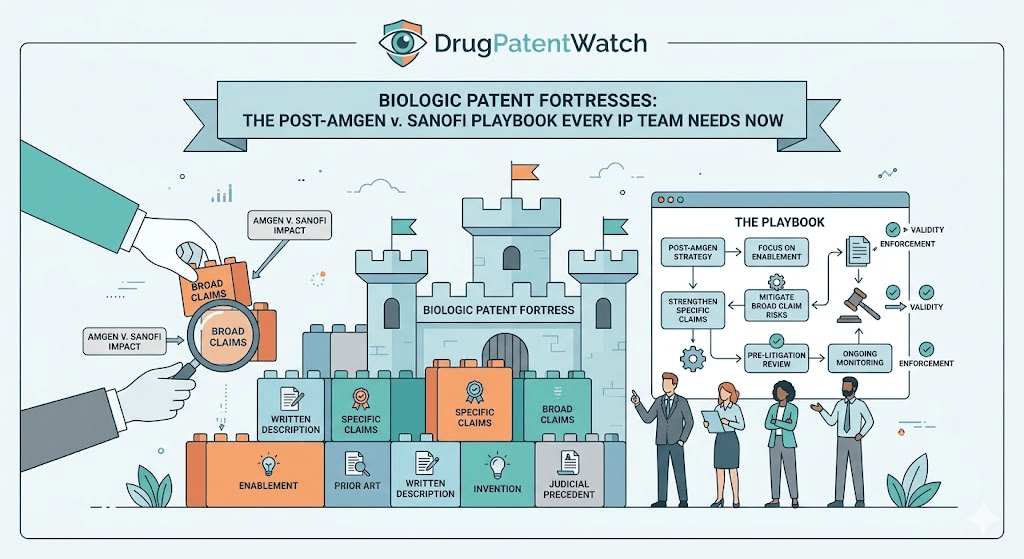
Amgen v. Sanofi: What the Supreme Court Actually Held, and Why It Reshapes Everything
On May 18, 2023, the Supreme Court issued a unanimous decision in Amgen Inc. v. Sanofi that invalidated the central patent estate protecting Amgen’s Repatha (evolocumab). The case was a direct confrontation between two blockbuster PCSK9-targeting monoclonal antibodies: Amgen’s Repatha and Sanofi/Regeneron’s Praluent (alirocumab). Both drugs work by binding to the PCSK9 protein and preventing it from degrading LDL receptors — which lowers LDL-cholesterol.
Amgen had not patented just the specific antibody sequences it discovered. It had patented the entire functional class of antibodies that: (1) bind to a specific epitope region on PCSK9, and (2) block PCSK9 from binding to LDL receptors. The claims were written to cover any antibody that performed those two functions — potentially millions of undiscovered sequences. Praluent, being a different antibody that performed identical functions, infringed those claims.
Sanofi’s defense was straightforward: the claims were invalid for failure to satisfy the enablement requirement of 35 U.S.C. § 112(a). Amgen’s specification described only 26 specific antibody examples. It could not teach a person of ordinary skill in the art how to make and use the millions of antibodies it claimed to own without engaging in a massive, open-ended research program. The Supreme Court agreed unanimously.
Enablement vs. Written Description: A Distinction IP Teams Must Internalize
35 U.S.C. § 112(a) imposes two distinct requirements that are frequently conflated in litigation strategy. Written Description (WD) is backward-looking and evidentiary: it asks whether the patent application demonstrates that the inventor was in possession of the claimed invention at the time of filing. Enablement is forward-looking and instructive: it asks whether the specification teaches a person of ordinary skill in the art (POSA) how to make and use the invention across its full scope without undue experimentation.
Amgen v. Sanofi was decided entirely on enablement grounds. Amgen may have possessed its 26 antibodies — meeting the written description requirement — but it did not teach the world how to make and use the enormous genus it claimed. Justice Gorsuch, writing for the Court, distilled the holding to a maxim that every IP drafter must now treat as a hard constraint: “The more one claims, the more one must enable.”
The Wands Factors — Now Supercharged
Courts assess enablement through the eight-factor “Wands test,” which asks whether a POSA would need “undue experimentation” to practice the full scope of the claim. The eight factors include breadth of the claims, level of predictability in the art, existence of working examples, and quantity of experimentation needed. Amgen did not overturn the Wands framework, but it dramatically increased its bite in “unpredictable arts” like antibody engineering. Where changing a single amino acid can destroy binding affinity, a “roadmap” that amounts to “make candidates and test them” is a research assignment — not enablement.
The Structural Pivot: CDRs Over Functional Genus Claims
The practical consequence for patent prosecution is immediate. The strategy of “an antibody that binds to epitope X and inhibits function Y” — the classic broad functional genus claim — is no longer viable as a primary claiming strategy in antibody patents. Patent prosecutors must pivot to structural claims, specifically the amino acid sequences of the six Complementarity-Determining Regions (CDRs) that define the antibody’s binding interface. These claims are narrower: a competitor could theoretically substitute an amino acid and design around them. But they are exceptionally strong against direct biosimilar challenge, and they actually pass muster under the new enablement standard.
Supplementary strategies include claims to specific nucleic acid sequences encoding the antibody, and — where a broad genus is still attempted — specifications that disclose a “general quality running through the class” that gives it a “peculiar fitness” and genuinely enables a POSA to identify and produce all claimed members without extensive trial-and-error.
The Double-Edged Consequence: Functional Claims Fall, Thicket Rises
The most counterintuitive strategic consequence of Amgen is this: by constraining the scope of the core composition-of-matter (CoM) patent, the ruling made the rest of the patent portfolio more valuable, not less. Innovators who relied on a single powerful genus patent as their primary moat now have a narrower front gate. The perimeter wall — the hundreds of secondary patents on formulations, manufacturing processes, delivery devices, and dosing regimens — becomes the primary line of defense. The thicket is not a fallback; it is now the primary strategy.
| Requirement | Statutory Basis | Core Question | Relevance Post-Amgen |
|---|---|---|---|
| Written Description | 35 U.S.C. § 112(a) | “Did you possess the invention at filing?” | Backward-looking; evidentiary. Usually met if 26 concrete antibodies are disclosed. |
| Enablement | 35 U.S.C. § 112(a) | “Can a POSA make and use the full scope of your claim?” | Forward-looking; instructive. Now the decisive battleground for broad genus claims. Wands factors supercharged. |
Investment Strategy Note — Post-Amgen Portfolio Assessment
For portfolio managers and analysts, the first-order question after Amgen is whether any asset in a biologic portfolio has a core CoM patent drafted primarily as a broad functional genus claim. Those patents face elevated invalidity risk and should be stress-tested against the Wands factors. Assets whose primary protection relies on process patents, formulation patents, or CDR-level structural claims are better positioned. The ruling creates a bifurcation in portfolio value that is not yet fully priced into the market.
Part III — Claiming the Core Asset
Beyond the Sequence: Structural Claims, Glycosylation, and the Obviousness Trap
CDR-Level Sequence Claims as the New Gold Standard
Post-Amgen, the most defensible primary claim on a novel antibody is a structural claim on the six CDR amino acid sequences. CDRs are the “fingers” of the antibody — the hypervariable loops that physically contact the target epitope. Two antibodies can have nearly identical framework regions but entirely different CDRs, producing entirely different binding profiles. A patent that specifies all six CDRs provides genuine specificity, passes the enablement test because the full scope of the claim is what you’ve actually characterized, and maps cleanly to the physical structure a biosimilar developer must copy.
Nucleic acid sequence claims — covering the DNA sequences encoding the heavy and light chains — provide complementary structural protection and can be relevant when a biosimilar developer uses a different but functionally equivalent production gene sequence. These two claiming layers, CDR amino acid plus encoding nucleic acid, now form the structural core of any defensible biologic patent application.
Bio-betters, Fusion Proteins, and the Half-Life Extension Obviousness Problem
Lifecycle management via “bio-better” development — taking an existing approved biologic and engineering improvements into it — is a legitimate and commercially powerful strategy. The most common approach is half-life extension through fusion with a long-lasting carrier protein. Fusing the biologic payload to the Fc region of an IgG antibody (as in etanercept’s structure) or to human serum albumin (HSA) extends circulating half-life, reduces dosing frequency, and creates a new patentable entity.
The problem is that this approach has become a victim of its own ubiquity. Patent examiners now treat Fc-fusion and HSA-fusion as “obvious approaches to take” for any biologic where extended half-life would be beneficial. Claiming a fusion protein and asserting that its longer half-life constitutes the inventive step will fail. Overcoming the obviousness rejection requires data demonstrating unexpected results — not just a quantitatively longer half-life, but a qualitatively different or surprisingly superior outcome. For example: a fusion that exhibits an entirely new biological activity not present in either component, an unexpectedly superior safety profile, or a novel and beneficial change in tissue distribution. Without that data, the application is a sitting target for a 35 U.S.C. § 103 rejection.
The Glycosylation Fortress: Three-Walled IP Around Post-Translational Modifications
Glycosylation — the attachment of complex sugar molecules (glycans) to specific asparagine residues on the antibody backbone — is perhaps the most fertile and underutilized territory for biologic patent strategy. Because glycosylation patterns are determined by the host cell’s enzymatic machinery and the culture conditions, they are inherently difficult for a biosimilar developer to replicate precisely. A sophisticated innovator can build a three-layer fortress around a proprietary glycoform.
The first layer covers the specific desirable glycoform itself: a patent claiming an antibody having a defined glycosylation pattern correlated with superior efficacy, stability, or reduced immunogenicity. The second layer covers the manufacturing method that produces that glycoform: specific cell culture conditions, feeding strategies, or bioreactor parameters that reliably generate the target sugar pattern. The third layer covers the analytical assay used to characterize and confirm that glycoform: a novel mass spectrometry or liquid chromatography method that constitutes the QC “ruler” for the product.
This three-walled structure traps biosimilar developers at every stage. They cannot reliably replicate the glycoform. They cannot patent-freely use the manufacturing method to produce it. And if they attempt to characterize their comparability to the reference product using the most advanced available methods, they may infringe the QC assay patent — the “Catch-22” discussed in detail in the process patents section below.
The U.S./EPO Divide on Non-Obviousness: A Global Strategy Must Account for Both
There is a material difference in how the USPTO and the European Patent Office (EPO) assess the obviousness of antibody inventions — and ignoring it produces costly, avoidable failures in European prosecution.
At the USPTO, a novel amino acid sequence is generally treated as structurally non-obvious. The examination practice holds that creating a new sequence is, absent prior art that specifically pointed to that structure, inventive on its face. At the EPO, this is explicitly not the case. The EPO’s Guidelines for Examination state that “the fact that an antibody’s structure is not predictable is not a reason for considering the antibody to be non-obvious.” A patent application filed with the EPO must demonstrate an inventive step by showing either that the antibody overcomes a specific technical difficulty in its generation, or that it possesses a new and unexpected functional property.
The practical consequence: a U.S.-centric prosecution strategy that relies solely on structural novelty of the CDR sequences will survive USPTO examination but fail at the EPO. Every biologic patent application intended for global filing must be loaded with data showing unexpected functional results — superior affinity, unexpected selectivity, reduced toxicity relative to structurally similar prior art candidates — from day one. The EPO won’t allow you to add this data post-filing.
Key Takeaways — Core Asset Claiming
CDR-level structural claims are now the defensible post-Amgen standard. Half-life extension via Fc or HSA fusion requires unexpected-results data to overcome obviousness. Glycosylation creates a three-layer fortress across product, process, and assay patents. Global prosecution requires EPO-specific functional data from the application filing date — structural novelty alone fails in Europe.
Part IV — Process is the Product
The Manufacturing Patent Stack: Building the Unseen Fortress
The “process is the product” doctrine is the conceptual engine of biologic patent strategy. Because the FDA-approved manufacturing pathway defines a biologic’s identity — its glycoforms, aggregation profile, impurity signature — every step in that pathway is simultaneously a regulatory requirement and a patentable invention. Process patents in this space are not a secondary defense; they are a primary offense, particularly once you understand how the BPCIA “patent dance” weaponizes them.
Host Cell Line Patents: Patenting the Factory
Your biologic is manufactured in a living “factory” — most commonly a genetically engineered CHO cell line that has been selected, adapted, and optimized over years to produce your specific protein at high yield and with the correct glycosylation pattern. That master cell bank has extraordinary IP value, and it can be protected directly.
The core strategy is to patent the host cell line itself — the specific genetic modifications, the stable integration locus for the expression cassette, and the phenotypic characteristics (growth rate, productivity, glycosylation capacity) that make it a “super-producer.” More advanced strategies cover the proprietary method for creating such cell lines, for example a novel cell fusion technique, a specific selection pressure protocol, or a method for selecting cells with elevated density of the endoplasmic reticulum machinery required for high-fidelity protein folding.
Cell line patents are among the strongest in the biologic arsenal because they are genuinely difficult to design around without starting the years-long CHO development process from scratch — and because a different cell line will, by definition, produce a product with a different glycosylation profile, potentially requiring a biosimilar developer to run entirely new clinical comparability studies.
Upstream Process Patents: The Growth Medium and Culture Conditions
Upstream biomanufacturing covers everything that happens before the cells start secreting your protein at scale: the composition of the growth medium, the culture conditions (temperature, pH, dissolved oxygen, agitation), and the feeding strategy used to keep cells productive during extended culture runs. Each of these parameters has been individually optimized over years of development work, and each is individually patentable if the resulting technical effect is novel and non-obvious.
Proprietary media compositions — particularly complex, chemically-defined, serum-free formulations that support high cell density and maximize specific productivity — are valuable and defensible subject matter. A patent on the medium used to produce a blockbuster biologic forces any biosimilar developer either to use a different medium (potentially changing the glycoform and triggering new comparability studies) or to license the medium patent.
Downstream Purification Patents: The Refinery
Downstream processing is the “refinery” that converts a bioreactor harvest — a complex mixture of the target protein alongside millions of host cell proteins (HCPs), DNA fragments, and process-related impurities — into a pharmaceutical-grade product at 99.999%+ purity. This multi-step chromatographic purification train is highly specific to each molecule and represents years of process development.
The core elements of the purification train are individually patentable: the specific affinity chromatography step using a novel Protein A resin variant; the ion-exchange polishing step configured to resolve the target product from its charge variants; the hydrophobic interaction chromatography (HIC) step optimized for aggregate removal; and the specific tangential flow filtration (TFF) protocol used for final concentration and buffer exchange. Each novel element — a new resin ligand, a new elution buffer composition, a new step sequence that achieves a previously unattainable purity level — is a distinct patentable invention.
Analytical QC Assay Patents: The Catch-22 Weapon
This is the most strategically elegant element of the process patent stack, and the most underappreciated. An innovator can patent the analytical methods — mass spectrometry protocols, glycan analysis workflows, potency assays, charge variant profiling methods — used to characterize the biologic and demonstrate that it meets its quality specifications.
The strategic consequence is a genuine dilemma for the biosimilar applicant. To obtain FDA approval, a biosimilar must conduct extensive analytical characterization studies demonstrating “highly similar” quality attributes to the reference product. The FDA expects state-of-the-art analytical methods. If the innovator has patented the only high-resolution method capable of producing the comparative data FDA reviewers will find convincing, the biosimilar developer faces two unattractive options: avoid the patented assay and submit inferior analytical data that may slow or prevent approval, or use the assay, produce compelling data, obtain FDA approval, and immediately face a patent infringement claim.
The BPCIA Dance Weaponizes Process Patents
What transforms these process patents from defensive shields into offensive spears is the BPCIA’s information exchange mechanism. When a biosimilar developer chooses to engage in the patent dance, one of the first obligations is to provide the innovator with a copy of their abbreviated Biologics License Application (aBLA) — including their confidential manufacturing process. In any other industry, proving infringement of a competitor’s internal manufacturing process requires extraordinary effort: litigation discovery, inspections, expert testimony. In the BPCIA, the competitor hands over their blueprints at the start of the litigation process.
An innovator’s legal team can then methodically compare those confidential manufacturing process documents against the cell line patent, the medium patent, the purification train patents, and the QC assay patents. Points of infringement are identified before Phase I litigation has fully commenced. The information asymmetry is extraordinary.
Part V — Thicket Architecture
Type I vs. Type II: The Framework That Determines How to Build and How to Attack
The debate over biologic “patent thickets” — dense webs of overlapping patents protecting a single drug — generates more heat than light, largely because the participants on both sides are describing different things. A 2018 legal analysis proposed a conceptual distinction that is, in practice, the single most useful framework for any IP team assessing either how to build a portfolio or how to challenge one.
Type I Thicket — Inherent Complexity
- Non-overlapping patents covering distinct, legitimate aspects of the drug
- Natural result of the “process is the product” doctrine
- Includes: molecule (CDR sequences), host cell line, purification process, formulation, delivery device, dosing regimen, QC assays
- High-quality, inventive, difficult to invalidate
- Challenger’s best response: design around (alternative formulation, different purification step)
- Amgen’s Enbrel protection is the archetype: two key Type I patents blocked an FDA-approved biosimilar for years
Type II Thicket — Strategic Duplication
- Overlapping or non-inventive patents on the same aspect of the drug with trivial variation
- Artificially constructed via continuation application strategy
- Approximately 80% of Humira’s post-approval U.S. patents were classified as “duplicative” in independent analysis
- Lower individual quality; purpose is to create litigation friction and a “numbers game”
- Challenger’s best response: PTAB inter partes review (IPR) — faster, cheaper, lower standard of proof
- AbbVie’s Humira strategy is the archetype: sheer volume forced settlements without a single trial victory required
This distinction drives the entire litigation calculus. A biosimilar developer facing a large patent portfolio should not evaluate it as a monolith. The correct approach is systematic classification: identify which patents are Type I (inventive, difficult to invalidate, requiring design-around or licensing) and which are Type II (weak, duplicative, suitable for PTAB invalidation). Each category demands a different response. Treating a portfolio as uniformly strong wastes settlement leverage. Treating it as uniformly weak invites a surprise loss on a genuine Type I patent.
Secondary Patents as Primary Defenses: Formulations, Devices, and Dosing
The term “secondary patents” is a misnomer that has persisted long past its usefulness. For a modern biologic franchise, formulation patents, delivery device patents, and method-of-use patents are often the most commercially durable protection available — especially post-Amgen, when the core molecule patent is both narrower and more vulnerable to enablement challenge.
The formulation patent strategy often involves a “product hop.” AbbVie patented a novel high-concentration, citrate-free formulation of adalimumab years before Humira’s core patent expired. The new formulation reduced injection-site pain — a genuine clinical improvement that also, by design, shifted the entire prescribing market toward a dosage form biosimilar developers had not optimized for. When biosimilars arrived, they were approved against the old formulation that physicians were no longer prescribing. A patent on a novel excipient combination that enables high-concentration subcutaneous delivery can have more commercial value than the molecule patent it nominally “supplements.”
Delivery device patents operate on a related logic. Biologics are typically administered by injection. Patenting the autoinjector pen — its spring mechanism, ergonomic casing, needle-shield design, and the Human Factors engineering that ensures patient usability — creates a new 20-year monopoly entirely independent of the drug patent. A biosimilar developer may hold a fully approved drug but be unable to deliver it in the format physicians and patients have come to expect, which is commercially equivalent to not having a product.
Method-of-use and dosing regimen patents extend the timeline through indication expansion and precision medicine claims. A patent on “using Molecule X to treat Crohn’s disease” or “administering Molecule X at 100 mg every three weeks to patients positive for Biomarker Y” can outlast the core composition-of-matter patent by five to ten years. The 2025 Federal Circuit decision in ImmunoGen v. Stewart provides an important constraint: the “objective reach” of a dosing claim matters, not the patentee’s specific motivation. A court will ask whether a POSA would have been motivated to try that dosing regimen for any reason — not just the specific technical problem the innovator was solving. Dosing patents must be supported by unexpected-results data demonstrating that the specific regimen, not just the general range, produces outcomes no skilled artisan could have predicted.
Key Takeaways — Thicket Architecture
The Type I/Type II distinction is the foundational framework for both portfolio construction and challenge strategy. Type I patents (inventive, distinct) require design-around or licensing responses. Type II patents (duplicative, continuation-based) are efficiently attacked via PTAB IPR. Formulation, device, and dosing patents are not secondary to the molecule patent — they are often its most durable successors. Product hops (shifting the market to a new, patented formulation before core patent expiry) are among the most effective commercial plays in lifecycle management.
Part VI — Case Studies in IP Warfare
Humira and Enbrel: Two Multi-Billion-Dollar Playbooks, One Underlying Goal
AbbVie’s Humira (Adalimumab): Quantity as Strategy
AbbVie’s approach to Humira is the canonical reference for a Type II-dominant portfolio strategy. The core composition-of-matter patent for adalimumab expired in the United States in 2016. By that date, the drug should have faced immediate biosimilar competition. It did not — for seven more years.
AbbVie filed approximately 250 patent applications for Humira in the U.S., 90% of them after the drug’s 2002 approval. Of the 130 that were granted, independent analysis concluded roughly 80% were “duplicative” — overlapping, non-inventive Type II continuation patents covering minor variations on the same aspects of the drug. The thicket’s purpose was not to protect distinct inventions. It was to manufacture uncertainty. Faced with 130 asserted patents and the prospect of multi-year, multi-hundred-million-dollar litigation on each, every biosimilar challenger settled. Amgen, Sandoz, Pfizer, Boehringer Ingelheim, and others all agreed to delay their U.S. launches until January 2023.
The financial calculus is stark. Humira’s U.S. net revenue in 2020 was approximately $16 billion. AbbVie sustained near-monopoly U.S. pricing for seven years after the core patent expired — a period during which European patients had already gained access to biosimilar competition. The asymmetry in market dynamics between the U.S. and Europe during this period is almost entirely attributable to the difference in how patent linkage operates in each jurisdiction.
Adalimumab’s IP Valuation as a Core Asset
At peak, Humira was the world’s best-selling drug, generating approximately $21 billion in global annual revenues. The asset’s IP estate — the thicket of 130 U.S. patents plus corresponding foreign filings — functioned as the primary value-preservation mechanism long after the CoM patent expired. From an IP valuation standpoint, the portfolio’s commercial value was not the sum of individual patents assessed at face value. Its value was the collective settlement leverage it generated: the certainty that any biosimilar developer would face litigation costs and delays that exceeded the discounted present value of early market entry. The thicket’s deterrent effect, not any individual patent’s validity, was the asset. That is the correct analytical frame for evaluating a Type II-dominant portfolio in M&A or licensing due diligence.
| Metric | Humira (AbbVie) — Type II Strategy | Enbrel (Amgen) — Type I Strategy |
|---|---|---|
| Core molecule | Adalimumab (anti-TNF-alpha mAb) | Etanercept (TNF receptor-Fc fusion) |
| Core patent expiry (U.S.) | 2016 | Mid-2010s (structure/method patents later) |
| Portfolio size (U.S.) | ~130 granted patents (~250 applications) | 2 key patents litigated (8,063,182 and 8,163,522) |
| Dominant patent type | Type II: continuation-based, duplicative | Type I: core fusion protein and manufacturing method |
| Legal tactic | Assert volume; force settlement via litigation cost burden | Defend validity of two key patents at district court and Federal Circuit |
| Litigation outcome | 100% settlement rate; biosimilars launched Jan 2023 | Federal Circuit affirmed patent validity; Sandoz’s Erelzi still off U.S. market |
| Effective exclusivity extension | ~7 years beyond core patent expiry | Potentially ~30 years total effective exclusivity |
Amgen’s Enbrel (Etanercept): Quality Over Quantity
Amgen’s Enbrel strategy is the surgical counterpoint to AbbVie’s mass approach. Sandoz received FDA approval for its etanercept biosimilar, Erelzi, in 2016. It has still not launched in the United States as of 2025.
The blockade rests on two patents: U.S. Patent Nos. 8,063,182 and 8,163,522, covering the etanercept fusion protein structure and the method of making it. Sandoz (with its licensor Hoffmann-La Roche) argued both patents were invalid for obviousness-type double patenting and lack of written description. The District Court and the U.S. Court of Appeals for the Federal Circuit both upheld Amgen’s patents.
From an IP valuation standpoint, these two Type I patents — a single legal victory on a single appeal — have maintained Amgen’s U.S. Enbrel monopoly for nearly a decade past what basic CoM patent math would have suggested. The asset’s IP value proposition was entirely concentrated in the quality and defensibility of those two claims, not in portfolio volume. For analysts assessing biologic assets in M&A or licensing contexts, the Enbrel case is a clear demonstration that a small number of genuinely inventive, properly prosecuted patents can outperform a much larger portfolio of weaker ones.
Investment Strategy Note — Reading Portfolio Quality
When evaluating a biologic asset’s IP estate for M&A or licensing purposes, the patent count is nearly useless as a standalone metric. The relevant variables are: (1) the proportion of Type I to Type II patents in the portfolio; (2) the expiry spread of the Type I patents; (3) the existence of any blocking patents on the manufacturing process or delivery device that outlast the molecule CoM; and (4) whether the innovator has executed a product-hop to a new, separately patented formulation. A portfolio with 10 strong Type I patents and a post-CoM product hop is worth substantially more than a portfolio with 100 Type II continuation patents that terminate at the same date.
Part VII — The BPCIA Battlefield
The Patent Dance, the PTAB Hammer, and the EU Divergence
The BPCIA “Patent Dance”: A Structured Confrontation with Strategic Optionality
The BPCIA’s most operationally complex feature is its pre-launch patent dispute resolution process — colloquially called the “patent dance.” Following a biosimilar applicant’s filing of an aBLA, the BPCIA provides a structured multi-step information exchange designed to identify and litigate the relevant patents before the biosimilar reaches market.
The timeline runs roughly as follows: the biosimilar applicant sends its aBLA and manufacturing process to the innovator within 20 days of FDA acceptance; the innovator discloses all patents it believes are infringed within 60 days; the biosimilar counters with invalidity and non-infringement contentions within 60 days; the parties then negotiate which patents to litigate first. Phase I litigation covers the agreed list. The biosimilar applicant must provide 180 days’ commercial marketing notice before launch, which triggers a Phase II litigation wave covering any patents excluded from Phase I.
The Supreme Court’s 2017 decision in Sandoz v. Amgen established that participation in the dance is optional. A biosimilar applicant may choose to forgo the information exchange entirely. This creates a binary first-move strategic decision with different risk/reward profiles.
Dance (Engage the Process)
- Forces the innovator to identify all asserted patents up front
- Allows the biosimilar to litigate and “clear the thicket” before commercial launch
- Provides legal certainty and reduces at-risk launch exposure
- Cost: biosimilar must disclose its confidential manufacturing process to the innovator at the outset
- Best when: biosimilar has high confidence in its FTO and wants to resolve the patent estate definitively pre-launch
Opt Out (Bypass the Process)
- Preserves manufacturing process confidentiality — innovator does not receive blueprints
- Denies innovator visibility into the biosimilar’s invalidity and non-infringement arguments
- Forces the innovator to file a “blind” infringement suit with no process disclosure
- Typically means launching at risk (before patents are cleared)
- Best when: biosimilar’s manufacturing process is its primary competitive advantage, or when the at-risk launch economics are favorable
The PTAB as the Biosimilar’s Hammer
No sophisticated biosimilar developer fights on a single front. Parallel to the BPCIA “dance” in district court, the PTAB — the Patent Trial and Appeal Board — provides a faster, cheaper, and structurally more favorable venue for invalidating weak patents in an innovator’s thicket.
Inter Partes Review (IPR) proceedings at the PTAB typically resolve in 12 to 18 months, compared to multi-year district court timelines. The cost is an order of magnitude lower. The standard of proof to invalidate is “preponderance of the evidence” — more likely than not — versus the “clear and convincing evidence” standard required in district court. And PTAB panels have historically been more receptive to invalidity arguments based on obviousness and anticipation.
The best-in-class biosimilar strategy is to engage the dance in district court for the most significant Type I patents — where a proper legal ruling on infringement and validity is needed — while simultaneously filing a targeted barrage of IPRs against the Type II continuation patents in the thicket. IPRs efficiently kill the weaker patents, narrow the innovator’s litigation position, and create settlement pressure without requiring the full expense of district court litigation on each weak patent.
The European Alternative: No Patent Linkage, Very Different Market Dynamics
The European Medicines Agency (EMA) operates under a fundamentally different philosophy. The EMA grants marketing authorization for biosimilars based purely on a scientific assessment of quality, safety, and efficacy. The innovator’s patent estate is entirely irrelevant to that regulatory decision. There is no European equivalent of the BPCIA patent dance, no Orange Book or Purple Book, and no pre-approval patent dispute mechanism.
The EU operates on an “8+2+1” exclusivity model: eight years of data exclusivity (no application filing), plus two years of market exclusivity (no marketing), plus a one-year extension available if the innovator secures approval for a significant new therapeutic indication during the initial exclusivity period. Total maximum regulatory exclusivity is 11 years — shorter than the U.S. 12-year term, and critically, completely decoupled from the patent estate.
A biosimilar can receive full EMA marketing authorization while the innovator’s European patents are still in force. It then launches “at risk,” forcing the innovator to pursue post-launch patent infringement litigation in each relevant national court system — a fragmented, expensive, and slow process that creates a very different deterrent dynamic than the U.S. pre-launch patent dance. The result is a European biosimilar market that is materially more competitive and accessible than the U.S. market, as demonstrated by the 7.7-year average head-start European patients had on Humira biosimilars.
| Feature | United States (BPCIA) | European Union (EMA) |
|---|---|---|
| Base exclusivity | 12 years market / 4 years data | 10 years (8+2) with potential +1 year extension |
| Patent linkage | Yes — patent dance is pre-approval mechanism | No — regulatory approval is fully decoupled from patent status |
| Dispute timing | Pre-launch (BPCIA dance) or concurrent | Post-launch (national court litigation) |
| Biosimilar market maturity | Delayed by patent thicket; meaningful competition began 2023 for Humira | Mature, competitive; Humira biosimilars available since 2018 |
| Innovator strategic advantage | Patent dance forces early disclosure; process patents weaponized pre-approval | Regulatory speed advantage; must pursue infringement post-launch in each member state |
Part VIII — Data as Competitive Advantage
CI, FTO, and Patent Landscaping: Turning Patent Data into Market Position
Competitive Intelligence: Reading a Competitor’s R&D Pipeline Through Patent Filings
A patent application is the earliest public signal of a competitor’s research direction — often filed years before clinical trial data appears in a registry and long before a press release is issued. Systematic monitoring of patent filings in relevant therapeutic areas gives R&D and business development teams a forward-looking view of the competitive landscape that no other public data source can match.
The correct approach is not to “search” for competitor patents reactively. It is to monitor continuously and to analyze claims systematically. When a competitor files a cluster of new formulation patents, that signals a product hop is in preparation — a specific dosage form is being protected in anticipation of the core molecule patent’s expiry. A cluster of new manufacturing process patents signals either cost reduction efforts or a productivity improvement initiative. A cluster of combination therapy patents reveals the shape of their next-generation clinical trial program. These signals arrive one to two years before the clinical evidence surfaces.
For innovators defending an existing franchise, patent CI enables proactive “war-gaming” of the biosimilar threat. By mapping all known biosimilar developers’ IP estates and tracking their PTAB filings, a defense team can identify which competitors are approaching launch readiness, anticipate their legal arguments before the patent dance begins, and initiate pre-litigation preparation for the most vulnerable patents in the thicket.
Freedom-to-Operate (FTO) as a Continuous R&D Process
FTO analysis answers the question every development team needs answered before committing significant capital: “Can we develop and sell this product without infringing an existing patent?” For a biologic, this question is not answerable at a single point in time. It must be answered continuously, because the product evolves, the patent landscape evolves, and the cost of discovering a blocking patent late in development is orders of magnitude higher than discovering it early.
Best-in-class FTO practice is staged and iterative. At feasibility, FTO covers the active protein sequence, the target indication, and the candidate formulation approach — establishing whether there is a clear path to market and identifying any blockers that require design-around at the earliest, cheapest point. As the manufacturing process is defined, FTO expands to cover the host cell line, the upstream culture conditions, and the downstream purification steps. Before clinical investment scales, FTO must clear the device and dosing regimen. Before launch, a final formal written opinion from patent counsel provides the “insurance policy” against willful infringement findings, which can trigger treble damages in the U.S.
When FTO analysis identifies a blocking patent, the team faces three substantive options: design around it by changing a specific technical parameter (the best option when caught early); negotiate a license if the patent is valid and the blocking position is unavoidable; or challenge validity proactively at the PTAB before the patent can be asserted, removing the obstacle rather than accommodating it. Each choice has different cost, timeline, and risk implications that must be evaluated in context of the asset’s overall commercial timeline.
Patent Landscaping: Identifying White Space and M&A Targets
A patent landscape is a structured, comprehensive map of who owns what in a given technology area — which companies, which institutions, what types of claims, and at what filing rates. For R&D strategy, a landscape identifies “white space”: commercially relevant technical areas where competitors are not patenting, suggesting opportunity for novel IP position with lower risk of blocking patents. For business development, the same landscape is a directory of potential licensing partners or acquisition targets, identified by the specific technology gaps they fill.
In mature biologic technology areas, landscapes reveal which platform technologies are heavily contested (making new patent position expensive and difficult) and which specific applications of those technologies remain open. In emerging areas — novel modalities, new target classes, early AI-designed antibody platforms — landscapes help a team identify whether a proposed R&D investment will result in proprietary IP or will immediately encounter a dense thicket owned by incumbents.
Part IX — The Next Frontier
AI-Designed Biologics, CAR-T, and the Patent vs. Trade Secret Dilemma
AI Inventorship: The Thaler v. Vidal Problem and the 2024 USPTO Guidance
AI and machine learning platforms have moved beyond screening to design. Systems can now generate de novo antibody sequences optimized for specific epitope targets, predict off-target binding risks, and propose structural modifications to improve manufacturability — tasks that previously required years of expert wet-lab iteration. This capability is commercially powerful and legally unresolved.
Patent law is built on human inventors. The Federal Circuit’s decision in Thaler v. Vidal confirmed that an “inventor” must be a natural person. An AI system, regardless of its contribution to the conception of an invention, cannot be named as an inventor on a U.S. patent application. The consequence is that any patent application for an AI-designed molecule that does not clearly establish a human’s “significant contribution” to each claim is vulnerable to an inventorship challenge that would invalidate the patent entirely.
The USPTO’s February 2024 guidance provides the current standard. An invention is patentable if a natural person “significantly contributed” to the conception of each claim. Presenting a problem to an AI (“find me an antibody that binds to PCSK9”) is explicitly insufficient — that is the AI’s assignment, not a human’s conception. A human scientist must have contributed to how the invention was conceptualized: the specific design constraints built into the AI model, the curation of training data in a novel and inventive way, the specific and technically substantive modifications made to the AI’s output in the wet lab, or the particular and unexpected combination of AI-generated structural features selected and validated by a human expert.
This creates a meticulous documentation obligation. Lab notebooks, computational notebooks, model training records, and validation reports must all capture not just the AI’s output but the specific human decisions and contributions at each stage of the design process. The documentation habit must be built into the R&D workflow from day one — retroactive reconstruction of inventive contribution will not survive legal scrutiny.
The collision of Amgen and Thaler creates a particularly sharp dilemma. AI platforms can generate millions of candidate antibodies within a defined functional space. Amgen says you cannot claim that functional space without enabling all of it. Thaler says you cannot claim individual candidates without proving human conception. The only navigable path is a focused, well-documented human-AI collaboration model: humans define specific structural constraints, humans evaluate and select specific outputs for specific technical reasons, and humans materially modify the AI’s candidates in the wet lab. The lab notebook, in this environment, is as strategically important as the AI model itself.
35 U.S.C. § 101 and the Gene Therapy Eligibility Problem
The most advanced therapies in biopharmaceutical development — CAR-T cell therapies, ex vivo gene-edited cell products, and viral vector-based gene therapies — face a different existential legal risk: subject matter ineligibility under 35 U.S.C. § 101. The Supreme Court’s trilogy of Mayo Collaborative Services v. Prometheus Laboratories, Association for Molecular Pathology v. Myriad Genetics, and Alice Corp. v. CLS Bank International established that laws of nature, natural phenomena, and abstract ideas are not patentable, regardless of their novelty or utility. An invention derived from a natural product must be “markedly different” from what exists in nature to qualify for patent protection.
The January 2024 federal district court decision in REGENXBIO v. Sarepta Therapeutics brought this risk into direct focus for gene therapy. The court invalidated patents covering an AAV-based gene therapy vector, finding that the combination of natural AAV sequences with a non-AAV therapeutic gene, expressed in a host cell, was not “markedly different” from naturally occurring biology and therefore patent-ineligible under § 101. The patents were detailed, technically sophisticated, and unquestionably novel. They were still invalid.
The Patent vs. Trade Secret Decision for CGT Manufacturing
The REGENXBIO decision forces every cell and gene therapy company to confront a strategic choice that carries the stakes of a bet-the-company decision.
The classic “patent bargain” — disclose your invention, receive a 20-year monopoly — rests on the assumption that the patent will be valid. For complex CGT manufacturing processes, that assumption may no longer hold. A company that spends a decade and several hundred million dollars developing a proprietary viral vector manufacturing process — a specific cell line, a specific transduction protocol, a specific downstream purification and characterization method — faces a genuine risk that the resulting patent will be found ineligible under § 101. If that happens, the patent publishes, the manufacturing process is disclosed to every competitor in the world, and the company receives nothing in exchange.
The trade secret alternative avoids § 101 and § 112 risk entirely. Trade secret protection is theoretically indefinite — it lasts as long as the information remains confidential. But it provides zero protection against independent discovery or reverse engineering by a competitor. If a biosimilar or competing CGT developer independently develops the same process, the trade secret holder has no legal recourse.
The emerging consensus among leading CGT companies is a hybrid model: patent the front-end product elements (the novel CAR construct, the specific transgene sequence, the vector capsid engineering) where patent eligibility is clearer and the claims map to well-defined structural features, while protecting the back-end manufacturing know-how (the transduction conditions, the expansion protocol, the release testing methods) as a closely guarded trade secret. The tradeoff is that the manufacturing moat is harder to enforce but potentially more durable than a patent that could be invalidated in a single legal proceeding.
Investment Strategy Note — CGT Asset Valuation Under § 101 Risk
For analysts and portfolio managers evaluating cell and gene therapy assets, the patent/trade secret composition of the IP estate is a material valuation variable that is rarely disclosed with sufficient granularity. An asset whose manufacturing moat rests entirely on patents faces § 101 invalidation risk that could simultaneously destroy IP protection and publicly disclose the manufacturing process. An asset with a hybrid patent/trade secret structure, where the manufacturing “secret sauce” is protected as confidential know-how, has a different and potentially more durable moat. Due diligence on CGT acquisitions should specifically probe the trade secret inventory, the information security practices protecting it, and any prior publication or disclosure events that could have eroded trade secret status.
Key Takeaways for IP Teams and Portfolio Managers
01The Amgen ruling ended broad functional genus claims as a primary patenting strategy. CDR-level structural claims on specific amino acid sequences are now the defensible standard. The ruling simultaneously narrowed the front gate and raised the value of the perimeter — process, formulation, device, and dosing patents.
02Process patents are not secondary protection — they are primary offense. The BPCIA dance’s mandatory manufacturing process disclosure weaponizes process patents in a way that has no equivalent in any other industry. Host cell, purification, and QC assay patents become informational asymmetry tools before Phase I litigation begins.
03The Type I / Type II distinction determines both how to build and how to attack a portfolio. Challengers should not evaluate a thicket as a monolith. Classify each patent: Type I gets design-around or licensing; Type II gets targeted PTAB IPR. Treating the whole thicket as uniformly strong or uniformly weak is strategically costly.
04The patent dance is optional. Engaging provides pre-launch certainty; opting out preserves manufacturing process confidentiality and litigation strategy. The correct choice depends on whether the biosimilar’s manufacturing approach is more sensitive to disclosure than its patent exposure is to uncertainty.
05The U.S. and EU are different IP battlefields requiring different strategies. Patent linkage in the U.S. makes the BPCIA dance the central mechanism. The EU’s complete decoupling of regulatory approval from patent status produces a fundamentally different market dynamic — more competitive, faster-moving, and requiring a post-launch litigation posture rather than a pre-approval one.
06AI inventorship requires meticulous documentation of human contribution at every design stage. The 2024 USPTO guidance demands “significant human contribution” to each claim. Presenting a problem to an AI is not sufficient. Human decisions about model design, training data curation, specific structural modifications, and experimental validation must be documented in real time.
07The REGENXBIO ruling makes the patent vs. trade secret decision existential for CGT manufacturing processes. If a gene therapy manufacturing patent is invalidated under § 101, the disclosure is permanent and unrecoverable. Hybrid models — patent the front-end product, protect back-end manufacturing know-how as trade secret — are the emerging best practice.
08Glycosylation patents are underutilized and exceptionally defensible. A three-layer fortress covering the specific glycoform, the manufacturing method to produce it, and the analytical assay to confirm it creates a barrier that traps biosimilar developers at every stage of development and regulatory approval.
09EPO prosecution requires unexpected-results data that USPTO prosecution does not. Structural novelty alone fails the EPO’s inventive step requirement for antibodies. Every global filing strategy must load functional advantage data into the specification from the original application date — post-filing data additions are not permitted at the EPO.
Frequently Asked Questions
Practical Questions from IP Teams and Analysts
After Amgen v. Sanofi, are broad functional genus claims completely dead?
Not legally dead, but practically non-viable as a primary claiming strategy in antibody patents. The Supreme Court left a narrow path: a specification that discloses a “general quality running through the class” giving it “peculiar fitness” and reliably enabling a POSA to make and use every member without extensive trial-and-error. In antibody engineering — an inherently unpredictable art — meeting this standard requires a specification robustly populated with diverse working examples across the claimed genus, not 26 antibodies as a stand-in for millions. Prosecutors should treat broad functional genus claims as high-risk supplementary claims at best, and should build the core patentable subject matter around CDR-level structural claims from the outset.
What is the single most strategically powerful secondary patent type for a new biologic?
Delivery device patents and manufacturing process patents are both candidates, and the “best” answer depends on the asset’s competitive context. Device patents create a completely independent 20-year monopoly on the user interface between drug and patient — a biosimilar that cannot legally put its approved drug in a patented autoinjector pen is commercially blocked regardless of what the drug patent situation looks like. Process patents derive their unique power from the BPCIA dance: the mandatory manufacturing process disclosure at the start of biosimilar litigation allows an innovator to identify infringement of process patents — cell line, purification train, QC assays — using documents the competitor is legally required to provide. No other IP regime creates this kind of informational asymmetry.
When should a biosimilar developer opt out of the BPCIA patent dance?
The opt-out is most advantageous when the biosimilar’s manufacturing process is itself a competitive differentiator or when the confidentiality of the biosimilar’s legal strategy provides more value than pre-launch patent certainty. Dancing forces disclosure of manufacturing blueprints at the outset — which is a significant gift to an innovator with a strong process patent portfolio. Opting out denies that disclosure, forces the innovator to litigate “blind,” and shifts the timing and framing of litigation to the biosimilar’s advantage. The tradeoff is that an opt-out strategy typically means launching at commercial risk before all blocking patents are cleared. The correct choice is case-specific and depends on the strength of the innovator’s process patent position relative to the biosimilar’s manufacturing process confidentiality interest.
Should a CAR-T or gene therapy company patent its manufacturing process or protect it as a trade secret?
The REGENXBIO decision has shifted the default answer toward trade secret for complex back-end manufacturing processes. If a patent covering a viral vector manufacturing method is challenged under § 101 and found to be a “natural phenomenon” combined with routine steps — a real risk given current case law — the patent is invalid and the manufacturing process is permanently disclosed. That outcome is worse than not patenting at all. For front-end elements (novel CAR construct, specific transgene sequences, engineered vector capsid variants that are “markedly different” from nature), patent protection remains the correct choice. The practical framework is: patent what you can defend under § 101 and § 112, and protect everything else as trade secret with rigorous information security practices, access controls, and confidentiality agreements.
How should analysts use patent filings to track competitor biologic pipelines?
The leverage is in trend analysis across filing clusters, not individual patent reviews. A single filing tells you what a competitor did 18 months ago when the application was drafted. A cluster of filings — multiple applications on closely related subject matter filed within a short window — reveals the shape of a strategic initiative. Formulation clusters signal a product hop in preparation. Manufacturing process clusters signal a cost or purity improvement program. Combination therapy clusters reveal the next-generation clinical program design. Monitoring tools that track filing activity by therapeutic area and assignee, and flag clusters for analyst review, convert patent data from a rearview mirror into a forward-looking strategic signal. The signal typically leads the clinical evidence by 18 to 36 months.
The New Strategic Imperative
The era of the single, broad CoM patent as a biologic’s primary fortress is over. What replaced it is more complex, more expensive to build, and — executed correctly — far more durable. The thicket, the process stack, the glycosylation fortress, the device monopoly, and the product hop are not defensive fallbacks; they are the strategy. Teams that integrate IP, R&D, regulatory, and competitive intelligence into a single, continuous function will build portfolios that hold. Teams that rely on any single patent to carry the weight will not.
Based on public patent records, court decisions, regulatory filings, and published industry analysis. Not legal advice. For informational and strategic planning purposes only.