KSR killed your obviousness defense. Amgen killed your functional claims. Here is the complete IP architecture for winning anyway, from R&D program design through Patent Term Extension, with real drugs, real litigation, and the PMOA decision that determines whether you get the Orange Book’s automatic 30-month stay or the BPCIA’s patent dance.
The Market Forcing the Strategy: Why Combinations Are Now the Default
The lung cancer market tells the story cleanly. It is projected to reach $64.53 billion by 2032, expanding at a 12.6% CAGR from an estimated $28.12 billion in 2025. What drives that number is not the old cytotoxic chemotherapy franchise. Biologics, specifically immunotherapies, antibody-drug conjugates (ADCs), and targeted monoclonal antibodies, are on track to hold 65.1% of the entire segment by 2025. They command that share because they deliver superior survival outcomes in advanced and metastatic non-small cell lung cancer (NSCLC) by engaging the immune system rather than carpeting the patient with non-selective cytotoxicity.
The clinical argument for combinations is now settled. Johnson & Johnson’s RYBREVANT (amivantamab-vmjw) plus LAZCLUZE (lazertinib) is the proof-of-concept that the entire industry is now racing to replicate. The FDA approved this regimen in August 2024 for first-line treatment of EGFR-mutated advanced NSCLC, making it the first chemotherapy-free regimen in its class. The Phase 3 MARIPOSA trial data, which showed the 30% reduction in progression risk over osimertinib along with a nine-month improvement in median duration of response, is exactly the kind of quantitative clinical delta that both regulators and patent examiners need to see.
Amivantamab is a bispecific antibody targeting EGFR and MET simultaneously. Lazertinib is a third-generation, brain-penetrant, EGFR-mutant-selective tyrosine kinase inhibitor (TKI). Individually, both are already established molecules. Together, they produce a clinical outcome that neither achieves alone. That gap, between what each component does individually and what the regimen achieves in combination, is the entire foundation of your IP argument. And it is more legally fragile than most R&D teams appreciate.
Amivantamab is protected by a multi-family patent portfolio spanning the biologic structure itself (antibody sequence and CDR-specific claims), the EGFR/MET bispecific mechanism, the MARIPOSA-defined dosage regimen, and the co-administration method with lazertinib. The composition of matter protection for the bispecific antibody issued in the early 2020s; the combination-specific method of use patents, tied directly to the FDA label indication, extend exclusivity significantly further. The IP valuation of this franchise is not located in any single patent. It is distributed across the thicket, which is precisely the architecture this article will build.
Lazertinib itself was originally developed by Yuhan Corporation in South Korea and licensed to J&J’s Janssen unit in 2018 in a deal worth up to $1.26 billion including milestones. The in-licensing of a structurally-defined, late-stage small molecule TKI gave J&J a clean second component with its own existing IP estate, reducing the enablement risk on that side of the combination while concentrating IP effort on the combination method and the bispecific antibody’s structural claims.
The commercial outcome of the amivantamab/lazertinib regimen matters because it sets pricing and licensing reference points for every combination program that follows. When a regimen displaces osimertinib, which is itself a $5+ billion/year product for AstraZeneca, it does so only if it commands a premium that the IP estate can defend. Without a durable, multi-pillar patent portfolio, the combination’s clinical superiority translates directly into a generic and biosimilar target the moment exclusivity gaps appear. That is the dynamic driving every strategic decision in this analysis.
‘The clinical ‘no-brainer’ is often the IP ‘nightmare.’ The same obviousness that makes a combination attractive to prescribers is exactly what a patent examiner will cite to reject your application.’
Part 01 — The KSR Gauntlet
Proving Your Combination Is an Invention, Not a Predictable Mix
35 U.S.C. §103 • KSR v. Teleflex • Isobolographic Analysis • R&D Design for Patentability
KSR International Co. v. Teleflex Inc.: The Decision That Changed Everything
In 2007, the Supreme Court’s unanimous ruling in KSR International Co. v. Teleflex Inc. rewrote the legal standard for obviousness under 35 U.S.C. §103. Before KSR, an examiner had to find a specific “Teaching, Suggestion, or Motivation” (TSM) in the prior art to combine elements into your claimed invention. The TSM test created a relatively predictable bar; if no prior art explicitly suggested combining Drug A and Drug B, your combination was patentable almost by default.
KSR eliminated that predictability. The Court replaced the rigid TSM test with a flexible, “common sense” inquiry that redefines the Person Having Ordinary Skill in the Art (PHOSITA) as a person of “ordinary creativity” rather than an automaton executing predefined steps. The practical effect: a patent examiner now has legal authority to reject your billion-dollar combination patent by arguing that combining two known cancer drugs to treat cancer is obvious, based on intuition backed by multiple references rather than a single explicit teaching.
For combination therapy applications, this created a presumption of obviousness that sits at the start of every prosecution. The examiner’s rejection structure is almost formulaic. Prior Art Reference 1 establishes that your small molecule (Drug A) inhibits a specific kinase relevant to the indication. Prior Art Reference 2 establishes that your biologic (Drug B) targets an immune checkpoint or surface antigen in the same indication. The rejection then concludes that a PHOSITA, seeing two drugs effective against the same disease, would have had both motivation to combine them and a reasonable expectation of additive or better efficacy. The resulting effect is called a “predictable result,” and predictable results are not patentable.
The burden has inverted. Your combination is presumed obvious until you prove it is inventive. Your data is the only path to that proof.
The Quantitative Synergy Standard: What ‘Better’ Actually Means to the USPTO
Most R&D teams conflate “it works better” with “it’s synergistic.” The USPTO does not. A qualitative assertion that your combination outperforms monotherapy is worthless in prosecution. Synergy, in the legal sense required to rebut an obviousness rejection, is a mathematically demonstrated result where the combined effect of Drug A and Drug B exceeds the predicted additive contribution of each component measured independently across a full dose-response curve.
The standard methodology is isobolographic analysis. You construct a dose-response surface for Drug A alone, Drug B alone, and then calculate the theoretical additive isobologram. You then overlay the actual measured combination response. If the combination data points fall below the additive isobologram, you have documented synergy. If they fall on it, you have documented additivity. If they fall above it, you have documented antagonism, which is commercially catastrophic and must never appear in a patent record without an explanation of how it was addressed.
The “side-by-side comparison” the USPTO wants to see is not a simple bar chart showing the combination outperforms monotherapy. It is the complete isobolographic dataset showing that the combination outperforms the mathematically predicted additive effect. This comparison must appear in the patent specification, not buried in an appendix. It must be accompanied by the underlying dose-response data for each component, the statistical analysis confirming significance, and, ideally, a mechanistic rationale explaining why the synergy occurs at the molecular level.
The MARIPOSA trial’s 30% reduction in progression risk vs. osimertinib is not, by itself, a synergy argument for patent purposes. It is a comparative efficacy argument against a different molecule (osimertinib) rather than an isobolographic comparison of amivantamab and lazertinib individually vs. in combination. Your patent specification must contain the latter, not just the former.
The Four Paths to Non-Obviousness Beyond Efficacy Synergy
Efficacy synergy is the most powerful argument but also the most demanding to demonstrate. Three other evidentiary categories can each independently, or in combination, support a finding of non-obviousness, and your R&D program should be designed to generate data across all of them.
Reduced toxicity or improved safety is the most frequently underused and commercially powerful argument available. In pharmacology, efficacy and toxicity are mechanistically linked; a drug that produces off-target toxicity at a therapeutically effective dose creates a ceiling on its clinical utility. If your combination achieves equivalent or superior efficacy at a dose of Drug A that is, say, 40% lower than the monotherapy dose required for the same effect, and if that lower dose eliminates a known dose-limiting toxicity, you have documented a “surprisingly low dose” finding that is directly patentable. A PHOSITA would have expected that combining two drugs with overlapping toxicity profiles would produce additive or multiplicative toxicity. If your combination shows the opposite, you have turned the examiner’s logic against itself.
A novel mechanism of action is the most elegant argument and, if available, essentially ends the obviousness inquiry. If your combination produces a biological effect through a mechanism that neither component produces alone, you have demonstrated that combining the two drugs did not produce a predictable result. It produced an unexpected one. This requires mechanistic characterization at the molecular level: receptor occupancy studies, pathway signaling analysis, transcriptomic profiling of combination-treated vs. monotherapy-treated cells. Expensive, but conclusive.
Long-felt but unresolved need is a historical argument supported by literature review. If the prior art documents a decade of failed attempts to achieve the clinical outcome you achieved, your success is non-obvious by definition. Collect every failed clinical trial, every abandoned IND, every paper concluding that combinations of this class produced “unacceptable toxicity” or “insufficient clinical benefit.” That record of failure, assembled as an exhibit in your patent specification, directly contradicts the examiner’s claim that success was predictable.
Teaching away is the strongest single argument you can make and the rarest. If prior art does not merely fail to suggest your combination but actively warns against it, citing, for example, documented hepatotoxicity when combining PD-1 inhibitors with this specific class of TKI, your decision to proceed despite that warning and your demonstration that you solved the problem is the definition of non-obvious innovation. Every paper and regulatory communication recommending against the combination becomes evidence in your favor.
Key Takeaways — Part 01: The KSR Gauntlet
- Post-KSR, combination therapy applications carry a presumption of obviousness. Your R&D program must be pre-designed to generate rebuttal data, not retrofitted to patent counsel’s needs after the FDA submission is filed.
- Isobolographic analysis comparing the combination against the predicted additive effect is the evidentiary standard. Comparative efficacy against a different standard of care does not satisfy the USPTO’s synergy requirement.
- Reduced toxicity, novel mechanism, long-felt need, and teaching away are each independently sufficient grounds for non-obviousness. Design experiments to capture all four data types, not just efficacy endpoints.
- The control arms required for patent prosecution (full dose-response curves for each component individually, plus the mathematical additive prediction) are not standard FDA submission arms. Your clinical operations and IP teams must build the trial design together.
For analysts assessing a pipeline combination asset, ask whether the company has published or disclosed isobolographic synergy data, not just head-to-head clinical comparisons against a standard of care. Assets backed by robust pre-clinical synergy datasets with mechanistic rationale carry materially lower IPR invalidation risk at the PTAB. Assets where the combination argument is “our biologic plus the SoC small molecule works better than the SoC alone” carry the full weight of a KSR obviousness presumption and should be discounted accordingly in patent-adjusted NPV models.
Also assess whether the company has designed clinical control arms for patent purposes, specifically whether monotherapy arms for each component exist in the trial design. Their absence in a registrational study is a structural weakness in the future patent estate.
Part 02 — The Amgen Shadow
Disarming the Enablement Threat to Your Biologic Claims
35 U.S.C. §112 • Amgen v. Sanofi • Structural vs. Functional Claiming • CDR Sequences • Epitope Claims
Amgen Inc. v. Sanofi (2023): The Decision That Killed Functional Genus Claims
On May 18, 2023, the Supreme Court ruled unanimously in favor of Sanofi in Amgen Inc. v. Sanofi, invalidating Amgen’s patents covering its blockbuster cholesterol drug Repatha (evolocumab). The ruling did not turn on obviousness. It turned on 35 U.S.C. §112, specifically the enablement requirement: the statutory obligation that a patent specification must teach a PHOSITA how to make and use the full scope of the claimed invention, without undue experimentation.
Amgen’s patents did not claim evolocumab by its specific amino acid sequence. They claimed a genus of antibodies defined by function: any antibody that (a) binds to a specific epitope on PCSK9 and (b) blocks PCSK9 from binding to LDL receptors. This “functional claiming” strategy had been common in the antibody space for decades. The theory was that the inventor who identifies the therapeutic target and demonstrates the mechanism deserves to own all molecules that operate through that mechanism, not just the specific one they synthesized first.
The Court rejected this theory on practical grounds. Amgen had disclosed roughly two dozen antibody examples in its specification. The functional genus it claimed potentially covered millions of structurally distinct antibodies that Amgen had never made and could not predict how to make. The Court concluded that Amgen’s specification provided a “roadmap” for discovery, not a teaching sufficient to practice the full claimed scope. The patent was, in the Court’s phrase, a “research assignment” to the public rather than the required disclosure in exchange for exclusivity.
The USPTO followed within months with a memorandum to examiners, clarifying that identifying a newly characterized antigen alone cannot constitute adequate written description for a claimed antibody to that antigen. Functional genus claims for biologics are now presumptively insufficient. The examiner will ask: how many structurally distinct antibodies does this genus encompass, and have you enabled making and using all of them?
The Amgen/Sanofi conflict over PCSK9 antibody patents was one of the most commercially significant patent disputes in recent pharma history. Both Repatha (evolocumab, Amgen) and Praluent (alirocumab, Sanofi/Regeneron) target PCSK9 and achieved LDL-C reductions of 50-60% on top of statin therapy. The patent dispute centered on whether Amgen’s broadly-claimed genus of PCSK9-inhibiting antibodies covered Praluent’s alirocumab, which has a structurally distinct CDR sequence from evolocumab but performs the same functional blocking mechanism.
Sanofi and Regeneron spent approximately $650 million in R&D developing alirocumab. Amgen argued its genus claim entitled it to reach into that investment. The Supreme Court’s ruling in Sanofi’s favor effectively ended the claim that a first-to-target-identify innovator can preempt all structurally distinct antibodies against that target. For your combination product’s biologic component, the practical consequence is that you own what you built and described, not the broader functional category your drug exemplifies.
IP Valuation Note
Amgen’s market capitalization dropped approximately $4 billion in the days following the Supreme Court ruling. The case illustrates that biologic patent risk is not a legal abstraction. It is a direct input to equity valuation. For portfolio managers modeling pharma IP risk, post-Amgen enablement risk on functional genus claims is now a line item, not a footnote.
The New Structural Claiming Playbook for Biologic Components
The post-Amgen mandate is a pivot from function to structure. Your biologic component in a combination patent must be defined with sufficient structural specificity that the examiner can confirm: every antibody within the claimed scope has been made and tested, or can be made and tested without undue experimentation, based on the specification’s disclosure.
The safest claim structure defines the antibody by its six Complementarity-Determining Regions (CDRs), the amino acid loops that physically contact the antigen. A CDR-based claim is narrow by design. It covers the specific antibody you discovered, and potentially very close structural analogs with trivially different CDR sequences. This claim will survive an enablement challenge because the number of antibodies within the CDR-defined scope is finite and definitionally small. The vulnerability is that a competitor can design around it by producing an antibody with different CDR sequences that achieves the same functional effect. If their antibody does not fall within your CDR sequence claims, they are not infringing, even if their molecule is functionally identical to yours.
Epitope claims offer a structurally-grounded alternative with broader commercial reach. Rather than claiming the antibody sequence, you claim the specific binding site on the target antigen (the epitope) that your antibody contacts. A well-crafted epitope claim might read: “An antibody that competitively inhibits binding of [your reference antibody] to human [target protein], wherein the antibody binds to a region comprising residues [specific residue list].” This is a structural limitation on the target, not the antibody, and it can encompass multiple structurally distinct antibodies that all happen to contact the same epitope. This approach was the basis for the Amgen PCSK9 antibody patents that survived in Japan even as the US claims failed. The Japanese claims specifically recited the contact residues on PCSK9 rather than the antibody structure alone.
If you intend to support a genus claim at all, the specification must contain a wealth of representative structural examples, not a single lead antibody plus a promise that others can be found by following the same screening protocol. Five or ten structurally diverse antibodies that all share the claimed property, with complete sequence data and experimental demonstration of activity for each, is the minimum. Twenty or more is better. The cost of generating this disclosure in your discovery phase is substantial. The cost of losing a genus claim post-filing, as Amgen demonstrated, is existential.
How Amgen and KSR Interact: Turning a Narrowing Decision Into a Competitive Moat
The intuition most IP teams have is that Amgen is a setback: it forces narrower claims. That framing misses a strategic opportunity. When both the KSR obviousness hurdle and the Amgen enablement standard are applied together to a combination product, they create a claim structure that is simultaneously harder to invalidate and harder to design around.
Here is why. A competitor trying to launch a competing combination product now faces two constraints. First, under post-KSR standards, they need their own synergy data for their specific combination, not just an argument that the general approach is obvious. Second, under post-Amgen standards, swapping your specific biologic for a functionally equivalent antibody with a different sequence does not necessarily escape infringement of a combination method-of-use claim if your claim recites the combination in structurally-specific terms and the competitor’s alternative combination lacks its own independent clinical data demonstrating equivalent outcomes.
The winning patent argument in this environment is not: “This class of TKIs combined with this class of PCSK9 antibodies is synergistic.” It is: “This specific small molecule, when co-administered with an antibody defined by CDR sequences [X, Y, Z], reduces disease progression by 30% vs. monotherapy. This interaction is a non-obvious biological phenomenon tied to the specific structural features of the antibody that determine its binding kinetics on the target.” You have anchored your non-obviousness argument to the structural specificity that Amgen now requires. A competitor cannot replicate your synergy claim without replicating your specific structural antibody, which is your composition of matter patent. The thicket begins here.
Key Takeaways — Part 02: The Amgen Shadow
- Functional genus claims for biologic components are presumptively insufficient post-Amgen. CDR sequence claims are the safest harbor; epitope claims with explicit residue specification provide broader coverage with manageable risk.
- Supporting a genus claim requires multiple structurally diverse, experimentally characterized representative examples in the specification, not a single lead antibody plus screening instructions.
- The Amgen ruling creates a competitive moat when used offensively: by tying combination non-obviousness arguments to structurally-specific biologic claims, you force competitors to run their own independent clinical programs rather than substituting a functionally equivalent biosimilar into your regimen.
- The Repatha ($4B market cap loss post-ruling) and Praluent case history is the reference point for quantifying enablement risk in biologic IP valuations.
Part 03 — The Patent Thicket
Building the Four-Pillar Architecture That Survives a KSR/IPR Attack on Your Core Patent
Composition of Matter • Method of Use • Dosage Regimen • Formulation • Kit Claims • Induced Infringement
Why the Fortress Model Is Obsolete
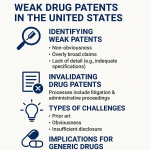
Pharmaceutical IP teams still routinely talk about their “core patent” as the asset to protect. The metaphor implies a single, defensible position. This is structurally wrong. Inter Partes Review at the PTAB has a grant rate consistently above 60% and an invalidation rate on instituted claims that makes even well-drafted patents vulnerable. Any competitor with $2-5 million in legal budget and a credible prior art argument can put your single most valuable patent under review within nine months of it issuing.
The correct model is a thicket: a portfolio of overlapping, mutually reinforcing patents, each covering a different legally recognized dimension of your commercial product, such that invalidating one leaves the others intact and still blocking. Studies of IP portfolios covering the top-selling pharmaceutical products consistently show that composition of matter patents on the active ingredient represent a minority of the total portfolio. The majority is built on formulation, method of use, dosage regimen, and delivery system patents, each of which requires independent litigation and presents independent invalidity arguments. A competitor attacking your franchise must file separate IPRs against each pillar. You have just multiplied their cost by four or five and given yourself four or five independent chances to win on the merits.
Pillar One: Composition of Matter Claims
Your foundation is the combination composition of matter (COM) patent: a claim covering a composition comprising a therapeutically effective amount of your small molecule and your biologic, optionally in a co-formulated delivery vehicle. This is where your isobolographic synergy data from Part 1 and your CDR-specific structural claims from Part 2 converge. The claim is strong because it is supported by both non-obviousness data and structurally-defined, Amgen-compliant biologic specification.
You should also maintain separate COM patent families on each component. The biologic gets its own patent with CDR sequence claims and epitope claims. The small molecule gets its own patent covering the specific crystalline polymorph in the commercial product, the pharmaceutically acceptable salts used in manufacture, and any novel prodrug forms or metabolites with their own clinical activity. These component patents serve a specific portfolio function: if a competitor biosimilarizes your biologic (which they will eventually attempt), the separate biologic composition patent gives you a cause of action that is independent of your combination patent. The same logic applies if a generic attempts the small molecule component alone.
Pillar Two: Method of Use and Dosage Regimen Claims
Method of use (MOU) patents are frequently the most durable assets in a pharmaceutical portfolio because they are structurally tied to the FDA-approved label. A generic or biosimilar competitor’s label must include the same indication as the reference product. If your MOU patent claims the exact indication on the label, the competitor infringes by necessity of the regulatory approval they seek.
Draft your MOU claims with the specificity of the MARIPOSA trial indication rather than a generic “treating cancer” claim. “A method for treating EGFR-mutated advanced non-small cell lung cancer in a treatment-naive adult patient, comprising administering [amivantamab] at a dose of [X] mg/kg and [lazertinib] at [Y] mg, concomitantly or in sequence” is both harder to design around and more directly tied to the approved label than any generic oncology MOU claim.
Dosage regimen patents represent the most underutilized, high-value IP play in the combination product space. The key insight is that a dosage regimen can be independently inventive, separate from the drug itself. If your clinical program discovers that a twice-weekly injection schedule of the biologic produces equivalent efficacy with dramatically reduced injection site reactions compared to the “obvious” weekly or daily dosing, that finding may be non-obvious if the prior art suggested the opposite schedule. You then patent the dosage, not just the drug. A competitor cannot copy the FDA-approved schedule without infringing your dosage regimen patent. To avoid infringement, they must run their own clinical trial with a different schedule, demonstrating bioequivalence or non-inferiority. That is a billion-dollar proposition that most generics and biosimilar companies will not undertake for a single indication.
AstraZeneca’s Nexium (esomeprazole) portfolio is the reference case for dosage regimen value. After the composition of matter patent expired, AstraZeneca maintained market position partly through dosage and formulation patents. The more instructive recent case is the Humira (adalimumab) portfolio, where AbbVie’s thicket included over 130 U.S. patents covering the antibody composition, formulation (the citrate-free, high-concentration formulation that reduced injection pain), dosing schedules, and prefilled syringe device. Biosimilar entrants faced the full thicket rather than a single COM patent; most settled for delayed entry licenses rather than litigating through all 130+ patents.
For your combination product, the lesson is that the biologic component’s dosage regimen and the co-administration schedule of the combination are each potential patent families worth developing independently, not merely including as dependent claims in your core combination patent.
Pillar Three: Formulation and Delivery System Claims
For a biologic-small molecule combination, the formulation challenge is genuinely technically difficult, and that difficulty is your IP opportunity. Biologics are large, structurally complex molecules that are highly sensitive to pH, temperature, ionic strength, and the presence of small molecules that can act as denaturants, chelators, or competitors for excipient binding. Engineering a stable co-formulation, or even a stable lyophilized biologic formulation that maintains activity after reconstitution in the presence of a small molecule diluent, requires significant formulation science that is not predictable from first principles.
If your team develops a novel buffer system, a specific excipient combination, or a particular pH range that stabilizes the biologic in the presence of the small molecule, that formulation is patentable independently of the drug itself. Formulation patents survive long after COM patents expire and provide a second layer of protection against biosimilar entry because a biosimilar manufacturer must not only match the biologic’s structural characterization but also must not infringe your formulation if they use the same stabilizing system.
Delivery system patents cover the physical device or dosage form. For a combination product, this often means a dual-chamber auto-injector (one chamber for the biologic, one for the small molecule, mixed at time of administration), a co-packaged kit with two separate pre-filled syringes, or, in the oral-biologic combination space, a novel oral delivery mechanism for the biologic component. Each device configuration is separately patentable and should be filed as a separate patent family coordinated through your device development partner.
Pillar Four: Kit Claims and the Induced Infringement Doctrine
Kit claims are the combination patent practitioner’s most elegant weapon against biosimilar competitors who believe they can safely enter the market with only one component of your regimen. A kit claim covers the as-sold product in its commercial packaging, specifically: a first container holding the biologic, a second container holding the small molecule (or a third container if the two must be kept separate), and a package insert with instructions for co-administration according to the approved regimen.
The legal power of the kit claim derives from 35 U.S.C. §271(b), the induced infringement doctrine. When a biosimilar manufacturer obtains approval to sell a biosimilar version of your biologic and their FDA-approved label instructs the physician to administer the biosimilar “in combination with” the small molecule (which it must, to match your approved indication), they are actively inducing the physician to perform the co-administration method that your MOU patent covers. They are also inducing the pharmacist and patient to assemble the functional equivalent of your patented kit. You have a cause of action for induced infringement against a competitor who may not be directly infringing any single component patent.
This is the thicket functioning as designed. A competitor who invalidates Pillar 1 in an IPR still cannot launch because Pillars 2 and 4 independently block them. A competitor who avoids Pillar 1 entirely by producing the biosimilar alone, without the small molecule, still cannot carry the approved label indication without inducing infringement of Pillar 2. There is no clean path through a properly constructed four-pillar thicket.
Key Takeaways — Part 03: The Patent Thicket
- Any combination patent strategy built around a single core COM patent is structurally vulnerable to PTAB IPR, where grant rates exceed 60%. The thicket model distributes risk across four independently sufficient pillars.
- Dosage regimen patents tied to the FDA-approved schedule are among the most commercially durable assets in a pharmaceutical portfolio. A competitor cannot copy the approved dosing without infringing them.
- Formulation patents for biologic-small molecule combinations are frequently undervalued because the technical challenge of stabilizing a biologic in the presence of a small molecule is both genuinely difficult and genuinely patentable.
- Kit claims enable induced infringement actions against biosimilar competitors who sell only the biologic component but whose label instructs co-administration with the small molecule. This closes the most common design-around path for biosimilar entrants.
For analysts benchmarking a company’s IP estate around a combination product, count patent families across all four pillars, not just composition of matter patents. A company with one strong COM patent and no dosage, formulation, or kit patents is structurally exposed at the PTAB. A company with patents spread across all four pillars, each with independent claims and separate prosecution histories, is meaningfully more defensible. The IPR filing cost per patent is approximately $500K-$1M through a PTAB decision. A four-pillar thicket costs a competitor $2-4M to attack in full, before considering district court litigation on the remaining valid patents. This is the correct metric for estimating the economic deterrence value of a pharmaceutical IP portfolio.
The Humira thicket (130+ patents, $21B peak sales, biosimilar entry delayed until 2023 in the U.S.) is the outer limit of what a well-executed thicket achieves. For most combination products, a portfolio of 8-15 patent families across the four pillars is a realistic and meaningful defensive position.
Part 04 — The FDA Fork in the Road
PMOA, the Orange Book’s 30-Month Stay, and Why the Patent Dance Is a Trap
Primary Mode of Action • NDA vs. BLA • Hatch-Waxman Act • BPCIA • Orange Book vs. Purple Book • Paragraph IV
The Office of Combination Products: The Decision Nobody Talks About
Your small molecule-biologic combination is a “combination product” under 21 CFR 3.2(e) by definition. The FDA’s Office of Combination Products (OCP) will assign a lead center (CDER or CBER) based on what it determines to be the “Primary Mode of Action” (PMOA) of your product, meaning the therapeutic mechanism most responsible for the intended effect. If the OCP decides the small molecule’s pharmacology drives the primary outcome, your product goes to CDER as a New Drug Application (NDA). If the biologic’s mechanism drives the primary outcome, your product goes as a Biologics License Application (BLA).
This is the most commercially consequential regulatory decision your product will face, and most management teams treat it as an administrative formality. It is not. The NDA vs. BLA designation determines which statutory exclusivity and patent linkage regime governs your product’s entire commercial life. It determines whether you get the Orange Book’s automatic 30-month stay against generic entry or are forced into the BPCIA’s convoluted and court-contested “patent dance.” The financial value of these two pathways is not comparable. They are orders of magnitude different in the commercial certainty they provide.
The PMOA determination is not a passive judgment. Your regulatory, R&D, and legal teams must build a scientific dossier, proactively and years before submission, that argues for the PMOA characterization you want. This means structuring your mechanism of action studies, your dose-response experiments, and your clinical trial design to generate data that supports your preferred PMOA narrative. If the biologic is your most structurally novel component and you have the broadest patent protection around it, you might prefer BLA designation. If you want the Orange Book’s 30-month stay, you must argue convincingly that the small molecule is the primary driver.
The Orange Book Path: Hatch-Waxman’s Patent Linkage and the Automatic 30-Month Stay
Under the NDA pathway, the Hatch-Waxman Act of 1984 creates a direct statutory link between your FDA-listed patents and the generic approval process. As the NDA holder, you submit a list of all patents meeting the Orange Book listing criteria (composition of matter, drug product/formulation, and method of use patents) to the FDA with your application. The FDA publishes them. Every subsequent ANDA filer for your drug must address each listed patent with one of four certifications. A Paragraph I certification states the patent does not exist. A Paragraph II certification states the patent has expired. A Paragraph III certification states the filer will not launch before the patent expires. A Paragraph IV certification states the patent is invalid, unenforceable, or not infringed by the proposed generic product.
A Paragraph IV certification is a statutory act of patent infringement under 35 U.S.C. §271(e)(2). If you file a patent infringement lawsuit within 45 days of receiving notice of the Paragraph IV certification, the FDA is automatically prohibited from approving the ANDA for 30 months, or until the patent dispute resolves in court, whichever comes first. This 30-month stay is not discretionary and does not require you to demonstrate likelihood of success on the merits. It is automatic.
The commercial value of this mechanism is enormous. You have 2.5 years of market exclusivity from the moment the generic files, regardless of whether your patent ultimately holds. This window allows you to prepare lifecycle management products, negotiate favorable settlements, and plan the commercial transition at a pace that the market rather than litigation urgency dictates. It is the most powerful commercial weapon in pharmaceutical IP, and it is available only on the NDA pathway.
The BPCIA Patent Dance: An Uncertain Alternative to the 30-Month Stay
On the BLA pathway, the Biologics Price Competition and Innovation Act (BPCIA) governs the biosimilar approval process. The BPCIA does not include an automatic 30-month stay. It replaces it with the “patent dance,” a structured information-exchange process between the innovator and the biosimilar applicant that is simultaneously more transparent, more expensive, and far less commercially certain than the Orange Book mechanism.
The sequence runs roughly as follows. The biosimilar applicant files an abbreviated BLA (aBLA). The BPCIA contemplates, but does not mandate, that the applicant provide the innovator with a copy of the aBLA and full manufacturing process details within 20 days of FDA acceptance. The innovator then has 60 days to identify all patents that could reasonably be asserted against the biosimilar. The applicant provides detailed invalidity and non-infringement contentions. The innovator responds. The two sides negotiate to a “list 2” of patents for first-wave litigation, with any remaining innovator patents reserved for a second wave.
The Supreme Court’s 2017 ruling in Sandoz Inc. v. Amgen Inc. determined that the patent dance is not mandatory. A biosimilar applicant can decline to provide the aBLA and manufacturing process information at all. If they do, the innovator cannot obtain an injunction forcing compliance. The innovator’s only remedy is to file a declaratory judgment action, removing the ordered structure of the dance and placing both parties into standard litigation without the benefit of the aBLA information exchange. The dance’s value to the innovator depends entirely on the biosimilar applicant’s voluntary participation, which they have every strategic reason to decline.
Manufacturing and process patents, which are not listable in the Orange Book under the NDA pathway, are squarely in scope in the BPCIA framework. Biologics are inherently process-dependent: the cell line, fermentation conditions, purification steps, and post-translational modification profile are all functions of the specific manufacturing process, and differences in process can produce structurally and functionally distinguishable biologics even when targeting the same antigen. If your patent portfolio includes robust manufacturing process claims, the BPCIA framework may actually give you more weapons than the Orange Book mechanism. This is one of the few genuine advantages of the BLA pathway for an innovator with sophisticated process IP.
| Feature | NDA Path — Orange Book (Hatch-Waxman) | BLA Path — Purple Book / BPCIA |
|---|---|---|
| Governing statute | Hatch-Waxman Act (1984) | Biologics Price Competition and Innovation Act (2010) |
| Patent listing | Mandatory pre-approval listing in Orange Book for all eligible patents | No global pre-litigation listing. Limited Purple Book listing after dance initiation |
| Listable patent types | Drug substance, drug product (formulation), method of use | All patents identified in the dance, including manufacturing process patents |
| Litigation trigger | Generic’s Paragraph IV certification | Patent dance exchange or declaratory judgment if applicant declines to dance |
| Automatic stay | Yes. 30 months from notice of Paragraph IV certification | None. No automatic stay mechanism |
| Dance mandatory? | N/A | No. Sandoz v. Amgen (2017) held dance is optional for biosimilar applicants |
| Key offensive asset | Method of use patents timed to label; automatic 30-month stay | Manufacturing process patents; total patent count to maximize litigation cost |
Table 1: Patent Linkage Dual-Track. Source: Hatch-Waxman Act, BPCIA, Sandoz v. Amgen (2017), USPTO/FDA guidance.
Key Takeaways — Part 04: The FDA Fork
- The PMOA designation by the FDA’s Office of Combination Products determines whether you access the Orange Book’s automatic 30-month stay or the BPCIA’s patent dance. This decision must be actively managed, not passively accepted.
- The 30-month stay provides guaranteed market exclusivity from the Paragraph IV filing date regardless of patent strength. It is the most commercially valuable single feature of the Hatch-Waxman framework and is available only on the NDA pathway.
- The BPCIA patent dance is not mandatory under Sandoz v. Amgen. A biosimilar applicant who declines to dance removes the structured information exchange that benefits innovators and forces the dispute into standard litigation without aBLA access.
- Manufacturing process patents are not Orange Book-eligible but are fully in scope in the BPCIA framework. A biologic-rich patent portfolio covering process claims may perform better on the BLA pathway than a portfolio focused only on composition and method claims.
For portfolio managers, the NDA vs. BLA designation is a direct input to exclusivity duration modeling. A product on the NDA pathway with Orange Book-listed patents and a well-timed Paragraph IV filing strategy can reliably add 2.5 years of protected revenue per patent challenge cycle. A product on the BLA pathway, absent the automatic stay, relies on injunctive relief from district courts, which requires a showing of irreparable harm and likelihood of success on the merits, a meaningfully higher bar. Model the two pathways separately in your NPV analysis, with the BLA pathway carrying a 15-25% discount to the NDA pathway’s exclusivity certainty absent a robust manufacturing patent estate.
Part 05 — The 20-Year Plan
Lifecycle Management: FTO, PTAB Defense, and the Patent Term Extension Decision
Inter Partes Review • Freedom-to-Operate • Patent Term Extension • PTE Strategy • Lifecycle Management
Freedom-to-Operate: Why FTO Is a Continuous Process, Not a Pre-Launch Checkbox
Your combination product’s commercial trajectory begins well before FDA approval, and the patent landscape you must navigate includes not just your own portfolio but your competitors’ growing estates in the same target space. A Freedom-to-Operate (FTO) analysis conducted once at IND filing and not revisited is a liability, not an asset. Competitor patent filings in your indication continue during your clinical program. A bispecific antibody competitor may file combination method claims that could block your regimen’s expansion into a new indication. A formulation competitor may patent a stabilization technology that you are independently developing. Without continuous monitoring, you discover these conflicts at the worst possible time, when you are inside a Phase 3 trial that would have been designed differently had you known the IP landscape six months earlier.
The intelligence infrastructure for continuous FTO monitoring requires coverage across the USPTO’s published application database, global patent offices (EPO, JPO, CNIPA), and the PTAB’s IPR filing docket. For your target antigen and your small molecule scaffold, you need automated alerts on new filings, newly issued claims, and IPR petitions against competitor patents that you might want to rely on for FTO clearance. When a competitor’s blocking patent is successfully challenged in IPR, that development changes your FTO picture in your favor and may open design space you previously had to avoid. When a competitor files a new combination method claim, you need to assess its scope before your next clinical milestone is locked in, not after.
Surviving Inter Partes Review at the PTAB
The Patent Trial and Appeal Board is the primary battlefield on which your patents will be challenged after issuance. IPR proceedings are decided by three Administrative Patent Judges, all with technical and legal credentials, who apply the preponderance of evidence standard rather than the clear and convincing evidence standard used in district court. This lower evidentiary threshold, combined with the technical sophistication of the judges, produces a highly efficient claim-elimination mechanism that generic and biosimilar manufacturers have used aggressively since the PTAB’s establishment under the America Invents Act in 2012.
The PTAB’s institution rate on IPR petitions has historically been above 60%, meaning that if a well-funded competitor files a petition, they have better than even odds of getting a full trial. Institution does not guarantee invalidation, but the invalidation rate on instituted claims has remained high enough that any patent facing a credible obviousness or enablement attack should be treated as potentially vulnerable.
Your thicket is the PTAB defense. If your core combination COM patent is challenged and invalidated, your dosage regimen patent must be independently sufficient to block a commercial launch. If your dosage regimen patent is also challenged, your formulation patent must independently block the competitor’s specific commercial product. Each pillar must be drafted to stand alone as a complete blocking position, not merely as a dependent claim on the core COM patent’s validity. This means each pillar patent needs its own independent claim set with its own non-obviousness support in the specification, not a cross-reference to the combination patent’s synergy data.
A competitor attacking your entire thicket must file separate IPRs against each patent family, each requiring its own petition fee, its own prior art search, its own legal team, and its own 12-18 month PTAB proceeding. At $500K-$1M per petition, a four-pillar thicket represents $2-4M in PTAB costs before district court litigation begins on whichever patents the PTAB declines to invalidate. This is genuine economic deterrence. It is not impenetrable, but it raises the entry price for any competitor from a nuisance-level legal skirmish to a serious capital commitment that must be justified by expected commercial returns.
Patent Term Extension: The Final Strategic Decision
The Hatch-Waxman Act allows the holder of an NDA or BLA-approved product to apply for a Patent Term Extension (PTE) to compensate for regulatory review time lost while the product was pending FDA approval. The rules are specific: the extension is capped at five years; the total effective patent life remaining after approval cannot exceed 14 years; only one patent per approved product is eligible; and the USPTO application must be filed within 60 days of FDA approval.
For a single-molecule drug, this decision is usually straightforward: extend the core composition of matter patent. For your combination product, the decision is genuinely strategic and requires analysis of the entire patent portfolio’s expiration landscape relative to the expected competitive threat timeline.
The eligible candidates are the combination COM patent, the individual component COM patents (which are separately eligible if they claim an active ingredient in the approved combination, regardless of synergy), the dosage regimen MOU patent, and potentially the formulation patent. The law does not require you to extend the most valuable patent, only the one whose extended term provides the most durable protection against the most probable competitive threat.
Make this decision only after you know which patents have survived PTAB challenges, because extending a patent that subsequently gets invalidated in IPR produces no commercial benefit. Make it after assessing which patent, with the extension, expires latest relative to the expected biosimilar or generic launch date. If your combination COM patent expires in 2031 and your dosage regimen patent expires in 2033, but the combination COM is already under IPR attack and the dosage regimen patent is not, extending the dosage regimen patent may provide more reliable long-term coverage even at an earlier nominal expiration date.
Key Takeaways — Part 05: The 20-Year Plan
- FTO is not a point-in-time analysis. It is a continuous intelligence function that must track competitor filings, PTAB IPR outcomes affecting competitor patents, and new combination method claims in your indication throughout the clinical development period.
- Each pillar in your thicket must be drafted with independent claims that can block a competitor’s commercial launch without relying on any other pillar’s validity. The thicket fails if the pillars are merely dependent on the core COM’s survival.
- Patent Term Extension allows only one patent per approved product. This decision must be made after assessing PTAB outcomes, expiration timelines across the portfolio, and the expected timing and nature of the competitive threat. Extending the most recently challenged patent is rarely the right choice.
- Individual component patents (biologic-only or small molecule-only) are PTE-eligible if they claim an active ingredient in the approved combination product, giving you optionality beyond the combination COM patent alone.
Frequently Asked Questions
Our combination shows synergy only at a 1:1 ratio. How broadly can we claim that without triggering an Amgen enablement problem?
This is the direct collision between KSR (where broader coverage is more commercially valuable) and Amgen (which punishes unsupported breadth). The answer is a tiered or “picture frame” claim structure. Your broadest claim asserts “a synergistic combination” without specifying ratio, backed by whatever mechanistic rationale supports the claim that synergy is not ratio-limited. A dependent claim narrows to a range of ratios (e.g., 1:5 to 5:1) supported by a handful of experimental data points beyond the 1:1 center. A further dependent claim specifies “about 1:1,” which your primary data directly supports. In prosecution or litigation, you fall back to the narrowest claim that your data fully enables. Draft for the broadest claim you can honestly support; expect to defend at the narrowest that your experiments actually demonstrate.
We are developing a combination where the two components are sold in separate devices but co-packaged in one box. Does the PMOA analysis still apply, and which claims protect us?
The PMOA analysis still applies. The OCP evaluates the primary therapeutic mechanism of the combination’s intended effect, not the physical configuration of the packaging. Your regulatory team must build the scientific record arguing for whichever PMOA serves your Orange Book vs. BPCIA strategy. On the patent side, the separate-device, co-packaged configuration is precisely what kit claims (Pillar 4) are designed to cover. Your kit claim enumerates the first device, the second device, and the package insert with co-administration instructions. The induced infringement doctrine then reaches any competitor whose biosimilar label instructs co-administration with the other component.
How does the Amgen enablement standard apply to CAR-T cell combinations, not just antibodies?
The Amgen ruling applies to all “unpredictable arts” under 35 U.S.C. §112, not just antibodies. CAR-T cells are at least as structurally complex as monoclonal antibodies and arguably more unpredictable in their performance characteristics. A claim to “all CAR-T cells targeting CD19” defined purely by the target antigen, without structural specificity in the single-chain variable fragment (scFv), the costimulatory domain configuration, or the hinge and transmembrane domain, faces the same Amgen objection that Amgen’s PCSK9 genus claim faced. The specification must disclose sufficient structurally-characterized CAR-T constructs to enable the full scope of the claim without undue experimentation. For combination products pairing CAR-T cells with small molecule checkpoint inhibitors, the scFv sequence and costimulatory domain structure (4-1BB vs. CD28, for example) must be specified in the biologic component claims.
Our core combination COM patent was just invalidated in IPR. What are the immediate steps to preserve commercial exclusivity?
Convene a franchise-level war room within 48 hours: IP litigation, regulatory, commercial, and the CEO. The first question is whether an appeal to the Federal Circuit is viable based on the grounds of the PTAB decision. A procedural error in the IPR institution decision or a clear claim construction error is worth appealing. If the invalidity finding was on the merits and the prior art is strong, redirect resources. Immediately assert Pillars 2, 3, and 4 in district court. The invalidated Pillar 1 does not affect the validity of your dosage regimen, formulation, or kit patents, and the competitor who “won” the IPR still cannot launch a commercial product without infringing the remaining valid patents. If you are on the NDA pathway, confirm that each still-valid Orange Book patent has received a Paragraph IV certification from the competitor; any missed certification represents a gap in their freedom to launch and triggers a fresh 30-month stay opportunity if you file suit within 45 days.
We are a biosimilar/generic developer targeting an innovator combination product. What is the most efficient attack strategy?
Start with the regulatory pathway determination. If the innovator is on the NDA pathway (Orange Book-listed patents), the 30-month stay is your primary obstacle and your strategy must account for 2.5 years of blocked approval regardless of IPR outcomes. Target the core combination COM patent first at the PTAB with an obviousness argument rooted in the KSR “obvious to try” doctrine, using prior art references showing that combining these drug classes was the logical extension of the existing literature. Simultaneously assess the biologic component COM patent for Amgen-style enablement weakness: did the innovator claim a functional genus that their specification’s exemplars do not fully enable? A successful IPR on the biologic COM combined with a declaratory judgment of non-infringement of the combination COM, even before the 30-month stay expires, can secure a favorable settlement. If the product is on the BLA pathway, the patent dance’s optionality under Sandoz v. Amgen gives you the choice of engaging the dance (gaining access to the aBLA’s manufacturing details for your litigation) or declining it and launching at risk after approval, forcing the innovator to seek a preliminary injunction without the benefit of the dance’s information exchange. Declining the dance is more aggressive but removes a significant discovery advantage from the innovator’s side.
The Blueprint: What You Should Do Next Week
Every section of this analysis points to the same structural conclusion. Winning in the combination patent era requires integration across functions that pharmaceutical organizations typically keep separate. R&D, IP, regulatory, and commercial teams each have a role in building the patent estate, and the critical decisions, from trial design to PMOA argumentation to PTE selection, cannot be made by any one function in isolation.
The specific actions worth prioritizing now: audit your current R&D program design against the control arms required for KSR obviousness rebuttal. If you do not have a planned full dose-response curve for each component individually, plus an isobolographic analysis protocol, your patent attorney has nothing to work with when the first obviousness rejection arrives. Assess your biologic component claims for functional vs. structural characterization. If your current draft specification relies on functional genus language to claim the biologic broadly, commission the additional structural exemplar work required to survive an Amgen challenge. Begin building the PMOA scientific dossier now, not at pre-NDA/pre-BLA meeting stage. The OCP responds to proactive scientific arguments, and waiting until late-stage submission to make your PMOA case is waiting too long. Audit your patent portfolio for all four thicket pillars: if you have only COM claims, your estate is a single wall. Identify the FTO monitoring gaps in your current intelligence process and build the continuous tracking function that combination-era competition requires.
The combination drug era is not coming. It is here. The companies that will define the next decade of pharmaceutical returns are the ones that treat patent strategy as an R&D discipline, not a legal afterthought.