I. Drug Patent Status as a Financial Variable, Not a Legal Footnote
The Misread Signal
Most analysts treat drug patent status as binary: on-patent or off. That framing costs money. A drug’s patent position is better understood as a probability-weighted timeline of competitive exposure, shaped by the type of patents held, the regulatory exclusivity periods running concurrently, the number of filed Paragraph IV certifications, the strength of the prosecution history, and the jurisdiction-specific enforceability of each claim. Miss any one of those variables and the revenue model is wrong.
The pharmaceutical industry runs on a simple but brutal economic logic: the average cost to bring a new molecular entity to market runs above $2.6 billion when accounting for the full capitalized cost of failures, and clinical success rates for drugs entering Phase I trials sit around 12 percent. That ratio demands a period of commercial exclusivity long enough to justify the wager. The patent system provides that window, and pharmaceutical companies treat it not as a fixed period but as an actively managed asset.
The statutory patent term in the U.S. is 20 years from the priority filing date. In practice, effective market exclusivity runs 7 to 12 years for most small molecules. The gap exists because the patent clock starts ticking during preclinical development, years before the first patient is dosed. A drug filing its IND in 2010, completing a Phase III trial, and receiving FDA approval in 2018 has already consumed eight years of its 20-year term before the first commercial sale. That compression is the core economic pressure driving every lifecycle management decision a brand company makes.
What the statutory term does not capture is the layered structure of exclusivity that a sophisticated originator builds around a franchise drug. The composition-of-matter patent is the anchor, but method-of-use patents, formulation patents, process patents, and pediatric exclusivity extensions each add incremental protection. When those layers are assembled correctly, a brand company can maintain meaningful IP coverage well beyond the primary patent expiration. Keytruda (pembrolizumab), Merck’s PD-1 inhibitor, carries a patent estate with filings extending into the 2030s despite its foundational composition-of-matter claims expiring earlier. Analysts who anchor their Keytruda revenue model to a single expiration date will be wrong.
What Drug Patent Status Actually Determines
Patent status controls four things that flow directly into a financial model. First, it determines whether an ANDA filer can even submit a Paragraph IV certification or must wait for patent expiration to file a standard Paragraph III certification. Second, it sets the litigation timeline, including the 30-month stay that delays FDA final approval. Third, it calibrates the 180-day exclusivity window available to the first successful Paragraph IV challenger, which in turn determines how fast the generic cascade follows. Fourth, for biologics, patent status interacts with the 12-year data exclusivity under the Biologics Price Competition and Innovation Act (BPCIA) in ways that can render composition-of-matter patent expiration commercially irrelevant for years.
An analyst who can read the full IP stack, not just the Orange Book expiration date, can model generic entry timing with precision that moves price targets.
Key Takeaways: Section I
Drug patent status is a multi-variable probability problem, not a binary flag. Effective market exclusivity averages 7 to 12 years for small molecules, significantly below the 20-year statutory term. The relevant analytical inputs are patent type, prosecution history strength, filed Paragraph IV certifications, concurrent regulatory exclusivity, and jurisdiction-specific enforceability. Biologics require a separate analytical framework governed by the BPCIA, not Hatch-Waxman alone.
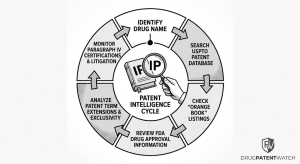
II. The FDA Orange Book: Architecture, Limitations, and Analytical Extraction
What the Orange Book Is, and What It Was Built to Do
The FDA’s ‘Approved Drug Products with Therapeutic Equivalence Evaluations,’ universally called the Orange Book, is the regulatory linchpin of the Hatch-Waxman framework. It lists every NDA-approved drug alongside the patents the NDA holder has certified as covering either the active ingredient, the formulation, or an approved method of use. The Orange Book was created to give generic applicants a structured, transparent map of the patent landscape they must navigate before filing an ANDA, and to give brand companies the mechanism to trigger automatic litigation stays when generics challenge their listed patents.
The FDA requires NDA holders to submit patent information within 30 days of patent issuance or NDA approval, whichever is later. This is not a voluntary disclosure. Submitting an improper patent, or failing to submit a valid one, carries serious consequences, including potential delisting orders and antitrust exposure. The Orange Book’s patent listings are therefore curated by brand companies with litigation strategy in mind, which means what a company lists, and what it chooses not to list, is itself analytically meaningful.
Reading an Orange Book Entry: Field-by-Field Breakdown
Each product entry in the Orange Book carries specific data fields that, read in combination, tell a sophisticated story about a drug’s competitive position.
The ‘Drug Substance’ flag (DS) indicates a patent claim on the active moiety itself, typically a composition-of-matter patent. This is the most commercially significant flag because it bars generic entry on the core compound. The ‘Drug Product’ flag (DP) indicates a formulation patent, covering the specific dosage form, excipient combination, or delivery system. The ‘Patent Use Code’ (PUC) is the most strategically complex field: it is an alphanumeric identifier (e.g., U-1402) linking a patent to a specific FDA-approved indication, dosing regimen, or patient population.
Patent Use Codes matter because they create the ‘skinny labeling’ or ‘carve-out’ mechanism. A generic applicant can omit a patented indication from its proposed label, launching with a narrower label that covers only non-patented uses. AstraZeneca’s experience with quetiapine (Seroquel) illustrates this: generics launched for schizophrenia while AstraZeneca maintained method-of-use patents on the bipolar depression indication. In practice, skinny-label carve-outs provide incomplete protection because prescribing behavior often ignores label restrictions, but they complicate enforcement and create litigation risk for both parties. The specificity of Patent Use Codes, and whether they are drafted narrowly enough to enable carve-outs, is a key analytical variable when assessing a brand company’s method-of-use patent portfolio.
The approval date field in the Orange Book, combined with the patent expiration date, lets an analyst calculate the remaining effective exclusivity directly. Subtract today’s date from the expiration date, then adjust for any patent term extension (PTE) or regulatory exclusivity running beyond the patent term.
What the Orange Book Deliberately Excludes
The Orange Book’s most important limitation is what it does not contain. Manufacturing process patents are excluded, even when those processes are proprietary and commercially significant. Metabolite patents, polymorph patents, intermediate patents, packaging patents, and patents covering unapproved uses are all outside its scope. For a drug surrounded by a dense secondary patent estate, the Orange Book expiration date is not the final barrier to generic entry.
The Orange Book also does not list patents for biologics. Biologic products approved under a Biologics License Application (BLA) fall under the BPCIA’s ‘patent dance’ procedure, not the Orange Book framework. The FDA maintains a separate ‘Purple Book’ for licensed biological products, but it does not list patents. Biosimilar applicants and reference product sponsors exchange patent lists through a statutory confidential procedure. Analysts covering biosimilar competition cannot rely on the Orange Book; they need to track the BPCIA patent exchange disclosures and subsequent litigation directly.
Third-Party Orange Book Tools: What They Add
The FDA’s native Orange Book interface is functional but limited. Third-party platforms such as DrugPatentWatch, Minesoft Orangebook, and various Bloomberg Law integrations add analytical layers that matter for professional use. These include historical patent submission and delisting tracking (critical for spotting Orange Book manipulation disputes), cross-referencing with USPTO prosecution data, and ANDA filing date tracking that flags which generics are already in the queue with Paragraph III certifications waiting for patent expiration.
DrugPatentWatch tracks patent expiration, generic entry, and litigation data across 134 countries, which is essential when assessing whether a foreign patent estate mirrors or diverges from U.S. filings. A drug with a strong Orange Book position in the U.S. but weak or expired foreign patent protection faces biosimilar or generic competition internationally that can erode the global revenue base and affect consolidated earnings ahead of U.S. patent expiry.
Investment Strategy: Using the Orange Book as a Revenue Screen
Institutional investors should run Orange Book screens quarterly as part of pharmaceutical portfolio surveillance. The key signal to watch is the emergence of first ANDA filings with Paragraph IV certifications, which trigger the 45-day litigation window for brand companies. When that window closes without a lawsuit, the generic applicant can commercially launch at risk immediately. When the brand files suit, the 30-month stay clock starts, setting the earliest possible FDA final approval date for the generic.
A Paragraph IV filing against a blockbuster with more than $1 billion in annual U.S. sales is a material event. The brand company’s stock typically drops 5 to 15 percent on the filing announcement, depending on the perceived strength of the challenged patent and the number of prior Paragraph IV filers in the queue. First-filer 180-day exclusivity status creates a binary outcome for the generic applicant: successful challenge generates a 180-day window worth hundreds of millions, while an unsuccessful challenge leaves the company exposed to significant damages.
Key Takeaways: Section II
The Orange Book maps patents to approved products within the Hatch-Waxman framework, but it excludes process, metabolite, polymorph, packaging, and unapproved-use patents. Patent Use Codes and their carve-out eligibility are analytically significant beyond the simple expiration date. Biologics are not covered by the Orange Book; BPCIA patent disclosures require separate tracking. Paragraph IV filing dates are the most commercially sensitive leading indicator of generic entry timing in the Orange Book ecosystem.
III. The USPTO: Patent Prosecution Histories as Competitive Intelligence
Why the Full Patent Document Matters
The Orange Book gives you a list of patents. The USPTO gives you the underlying documents, which is where the real analytical work happens. A patent number means nothing without its claims. The scope of protection, the validity vulnerabilities, and the prosecution history estoppel that limits claim interpretation all live in the USPTO record, not in any regulatory database.
The USPTO’s Patent Public Search tool (ppubs.uspto.gov) replaced the legacy Full-Text and Image Database (PatFT) and the Patent Application Full-Text Database (AppFT) systems. It supports Boolean operators, proximity operators, and field-specific searching through defined field codes, including .clm. for claims text, .ab. for abstract, .as. for assignee name, .pn. for patent number, and .pd. for publication date. For pharmaceutical patent searches, the claims field is the primary target. The abstract summarizes the invention, but the claims define the legal scope of protection, and independent claims vs. dependent claims have meaningfully different scopes.
Reading Pharmaceutical Patent Claims Analytically
A composition-of-matter claim for a small molecule typically reads as a Markush structure claim, covering a defined class of chemical compounds by specifying a core scaffold with variable substituents. The broader the Markush group, the harder the patent is to design around, but also the more vulnerable it is to prior art challenges because broader claims must meet a higher bar of enablement under 35 U.S.C. 112. When Teva filed Paragraph IV challenges against Warner Chilcott’s Asacol HD (mesalamine), part of the challenge targeted enablement, arguing the specification did not sufficiently support the full scope of the formulation claims. That case illustrates how prosecution history details translate into litigation strategy.
The prosecution history, accessible through the USPTO’s Patent Center portal, contains the full record of examiner rejections, applicant arguments, and claim amendments made during examination. When an applicant narrows a claim to overcome a prior art rejection, prosecution history estoppel limits how broadly the patent can be interpreted in infringement litigation. A claim that was narrowed to distinguish a prior art compound cannot later be asserted as covering that compound under the doctrine of equivalents. Pharmaceutical IP teams use prosecution history analysis to identify the ceiling of a patent’s enforceability, and generic applicants use it to spot opportunities to design around claims without litigation exposure.
Assignee Tracking and Patent Ownership Chains
The USPTO Patent Assignment Search database (assignment.uspto.gov) tracks patent ownership transfers. In pharmaceutical M&A, patent assignments are often filed months after deal close, creating a lag between the transaction and the public record. Tracking assignment histories reveals the full provenance of a patent estate, including whether patents were originally filed by an acquired company, licensed in from an academic institution, or obtained through a patent purchase agreement.
Ownership chain gaps matter. A patent with a broken chain of title, where an intermediate assignment was never recorded, can create an unenforceability argument. Generic manufacturers that invest in prosecution history analysis routinely check assignment records as part of patent challenge preparation. Institutional investors evaluating pharmaceutical M&A transactions should do the same; acquiring a company with unrecorded patent assignments exposes the acquirer to title challenges that reduce the IP asset value.
Global Dossier: Mapping the International Patent Family
The USPTO’s Global Dossier connects U.S. applications to related applications at the IP5 offices: the European Patent Office (EPO), the Japan Patent Office (JPO), the Korean Intellectual Property Office (KIPO), and the China National Intellectual Property Administration (CNIPA). Patent families matter because a successful invalidity challenge in one jurisdiction can undermine the legal position in others, and because geographic gaps in patent coverage define where generics can manufacture or market freely.
A drug with strong U.S. and European composition-of-matter patents but no equivalent filing in India or Brazil faces authorized generic or compulsory licensing exposure in those markets. For multinational originator companies, patent family mapping is a prerequisite for accurate revenue forecasting by geography. For generic manufacturers planning international launches, family gaps are commercially actionable.
Key Takeaways: Section III
Patent claims, not patent numbers, define the scope of protection. Prosecution history estoppel constrains claim interpretation and is the primary tool generic manufacturers use to design around or challenge a patent’s enforceability. Assignment records must be verified for completeness; broken ownership chains create validity exposure. Global Dossier analysis identifies geographic gaps in patent families that determine where competition can legally enter first.
IV. Pharmaceutical Patent Types: A Technical Field Guide
Composition of Matter Patents: The Highest-Value IP Asset
A composition-of-matter patent protects the active pharmaceutical ingredient (API) as a chemical entity. For new molecular entities, this is the foundational IP filing and carries the highest commercial value because it bars any competing product using the same API, regardless of formulation, indication, or dosing regimen. For biologics, the equivalent protection comes through patents on the specific amino acid sequence or structural configuration of the biologic molecule.
To obtain a composition-of-matter patent, the claimed compound must satisfy novelty (not previously disclosed in the prior art, including prior publications, patents, and public use), non-obviousness (not an obvious modification of known compounds to a person having ordinary skill in the relevant field), and utility (a specific, credible, and substantial use). The non-obviousness requirement is the most frequently litigated. In the 2007 KSR International Co. v. Teleflex Inc. Supreme Court decision, the Court rejected a rigid approach to non-obviousness and ruled that combining known elements using known methods to yield predictable results is obvious. Post-KSR, pharmaceutical patent applicants must provide more rigorous data, often pharmacological activity data, to demonstrate that a claimed compound’s properties were not predictably derivable from known analogs.
The IP valuation implication of composition-of-matter patents is significant. An unexpired composition-of-matter patent with broad claim coverage and remaining exclusivity of eight or more years is typically valued using a risk-adjusted net present value (rNPV) model that discounts future cash flows by the probability of patent survival through litigation. A patent that has survived a Paragraph IV challenge without invalidation carries a substantial premium over one that has never been tested, because legal uncertainty is partially resolved.
Method-of-Use Patents: Durable but Carveable
A method-of-use patent covers a specific therapeutic application of a compound, including a particular disease indication, a dosing schedule, a patient subpopulation, or a combination therapy protocol. These patents are filed after initial approval and can extend effective exclusivity substantially beyond the composition-of-matter expiration. Pfizer’s sildenafil (Viagra) provides a textbook example: the composition-of-matter patent expired in 2012, but Pfizer held method-of-use patents on erectile dysfunction treatment that continued to generate royalty disputes and licensing negotiations well beyond that date.
The strategic limitation of method-of-use patents is the skinny label carve-out. Generic applicants can exclude a patented indication from their proposed label and certify Paragraph III (or not certify at all) against method-of-use patents whose use codes do not apply to the carved-out indication. The adequacy of carve-outs has generated substantial litigation. In GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc., the Federal Circuit held that Teva induced infringement of GSK’s carvedilol heart failure method-of-use patent even though Teva’s label nominally carved out that indication, because Teva’s marketing materials and labeling as a whole directed physicians to use the drug for the patented indication. The lesson for generic applicants: a technically compliant skinny label is necessary but not sufficient protection against method-of-use infringement claims.
Formulation Patents: Lucrative but Vulnerability-Prone
Formulation patents protect specific combinations of API with excipients, particular dosage forms, delivery systems, or release profiles. Extended-release, delayed-release, and modified-release formulation patents are among the most commercially important because they can justify a distinct NDA filing and obtain a separate Orange Book listing from the immediate-release version of the same drug. AstraZeneca used this approach extensively with esomeprazole (Nexium), protecting the S-enantiomer of omeprazole with both composition-of-matter claims and formulation patents on specific pellet coating systems designed to achieve the desired acid stability and release profile.
Formulation patents are vulnerable to obviousness challenges grounded in prior art showing similar excipient combinations or release mechanisms in existing literature. The patentee must typically demonstrate ‘unexpected results’ relative to obvious alternatives. Unexpected results require comparative data showing that the claimed formulation outperforms or behaves differently from what a skilled formulator would have predicted. Without that data, a formulation patent faces elevated invalidation risk in Paragraph IV litigation and inter partes review (IPR) proceedings.
Formulation patents also face specific vulnerability through the ‘product-by-process’ doctrine. A formulation defined by a processing step can be anticipatable by prior art that achieves the same physical result through a different process, which courts have found sufficient for anticipation in some circumstances. IP teams assessing formulation patent portfolios should flag any claim that defines the product primarily by how it is made rather than by structural or functional characteristics of the final dosage form.
Process Patents: Strategic Role in Biologics Manufacturing
Process patents protect specific manufacturing methods for a drug compound, rather than the compound itself. In small molecule pharmaceuticals, process patents matter primarily when a generic API synthesizer uses a process that reads on the brand’s patented synthesis route. In biologics, manufacturing process patents have enormous strategic importance because the biologic molecule’s structure is inseparable from its manufacturing process.
A monoclonal antibody produced in Chinese Hamster Ovary (CHO) cells under specific culture conditions will have a glycosylation pattern that differs from the same antibody expressed in a different cell line or under different conditions. Originators file process patents covering cell culture conditions, fermentation parameters, purification chromatography sequences, and formulation compositions, partly to protect their manufacturing investment and partly to create IP barriers for biosimilar developers who must demonstrate that their manufacturing process produces a product sufficiently similar to the reference biologic to meet the FDA’s totality of the evidence standard for biosimilarity.
Abbvie’s defense of adalimumab (Humira) illustrates the scale of process-patent deployment. Abbvie maintained over 100 U.S. patents covering adalimumab, including numerous process and formulation patents that extended well beyond the composition-of-matter expiration. Biosimilar developers including Amgen, Sandoz, and Boehringer Ingelheim negotiated licensing deals rather than litigating the full patent estate, effectively conceding years of market exclusivity to Abbvie in exchange for settlement certainty. The commercial result: adalimumab generated approximately $21 billion in global sales in 2022, its last full year as the unchallenged market leader in the U.S., despite the composition-of-matter patent having expired years earlier.
Humira as an IP Valuation Case Study
Abbvie’s adalimumab patent estate is the most-studied example of pharmaceutical patent layering. At peak, Abbvie held over 100 U.S. patents and over 250 patents globally covering the antibody composition, the manufacturing process, the citrate-free formulation (which reduced injection-site pain and was filed as a separate indication), and methods of use across more than a dozen indications including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, plaque psoriasis, Crohn’s disease, and ulcerative colitis.
From an IP valuation standpoint, the citrate-free formulation patent (covering Humira’s high-concentration, citrate-free pen formulation) deserves specific attention. Abbvie filed a supplemental NDA for this formulation, obtained a separate Orange Book listing, and then argued that biosimilar developers who had obtained interchangeability designations for the original citrate-containing formulation needed to conduct separate interchangeability studies for the citrate-free version. That filing extended the effective commercial relevance of the formulation IP by creating regulatory uncertainty for biosimilar switchers, independent of the underlying litigation outcome. The citrate-free formulation patent’s discounted cash flow value, using a conservative 70 percent probability of survival and a 3-year exclusivity extension at a blended price that biosimilar competition would otherwise have eroded, equates to several billion dollars in retained revenue. That is what a single formulation patent is worth when it protects a $20 billion annual franchise.
Key Takeaways: Section IV
Composition-of-matter patents carry the highest IP value and are the first analytical priority. Method-of-use patents are durable but subject to skinny-label carve-outs and induced infringement risk. Formulation patents are vulnerability-prone absent ‘unexpected results’ data, but they are commercially powerful for modified-release and biologic delivery systems. Process patents are strategically decisive in biologics manufacturing, where the product and process are biologically inseparable. The Humira estate demonstrates that a layered portfolio of secondary patents can generate billions in retained revenue beyond composition-of-matter expiration.
V. Patent Duration, PTE, PTA, and Regulatory Exclusivity: The Math Behind Effective Market Life
The 20-Year Term: Starting Point, Not Endpoint
All U.S. utility patents, including pharmaceutical patents, receive a statutory 20-year term measured from the earliest effective U.S. filing date. That date is typically the priority date of the U.S. application or, for PCT applications, the international filing date. The 20-year term is not extended by continuation or continuation-in-part filings, which inherit the priority date of the parent application for overlapping claim subject matter.
For pharmaceutical patents, the 20-year term represents a ceiling that is almost never reached in practice as an effective commercial exclusivity period. By the time a drug completes Phase I, II, and III development and obtains FDA approval, 8 to 14 years have typically elapsed since the patent priority date. The remaining patent life at launch, commonly called ‘patent runway,’ directly determines how aggressively a company can price the drug and how much revenue it can capture before generic exposure.
Patent Term Extension Under 35 U.S.C. 156
The Hatch-Waxman Act created patent term extension (PTE) under 35 U.S.C. 156 to compensate originators for the regulatory review time consumed between initial patent filing and FDA approval. PTE adds back a portion of the patent term consumed during the FDA review process, calculated as half of the ‘testing phase’ (from IND filing to NDA submission) plus the full ‘approval phase’ (from NDA submission to approval), minus any time the applicant failed to act with ‘due diligence.’
The PTE calculation is capped at five years of added term, and the total effective patent term, including PTE, cannot exceed 14 years from the date of FDA product approval. Only one patent per drug product can receive PTE, and it must be the patent most relevant to the approved product. The strategic implication of the single-patent-per-product rule is that IP teams must carefully select which patent to extend. Selecting the composition-of-matter patent over a method-of-use patent maximizes breadth but may leave the formulation unprotected at expiration. Selecting a formulation patent preserves manufacturing exclusivity but allows API-based composition challenges.
PTE applications must be filed within 60 days of FDA approval. Missing that window forfeits the extension entirely. The USPTO reviews PTE applications independently, and PTE calculations are litigated, primarily in Federal Circuit appeals. The 2022 Federal Circuit decision in Acadia Pharmaceuticals Inc. v. Aurobindo Pharma Ltd. addressed PTE calculation methodology in the context of reissue patents, reaffirming that PTE attachments follow strict eligibility criteria tied to the specific regulatory review pathway.
Patent Term Adjustment Under 35 U.S.C. 154(b)
Patent term adjustment (PTA) compensates for delays at the USPTO during patent examination, distinct from PTE, which compensates for FDA review delays. PTA accrues when the USPTO fails to meet statutory examination timeline benchmarks: issuing a first office action within 14 months of filing, responding to applicant replies within 4 months, and issuing a final decision within 3 years of filing. PTA is calculated as A-delay plus B-delay plus C-delay, minus any applicant-caused delays, with adjustments for overlapping periods.
For pharmaceutical patents, PTA can add meaningful term. A patent that experienced 18 months of USPTO prosecution delay receives 18 months of PTA added to the 20-year term (minus any applicant delay). Unlike PTE, PTA is not capped, and multiple patents can receive PTA. This creates a situation where secondary patents filed later in the development cycle, which experience less prosecution delay (because USPTO examination timelines have improved), may receive less PTA than earlier-filed patents that sat in the queue during periods of USPTO backlog.
PTA calculation disputes are litigated regularly. Gilead’s tenofovir alafenamide (TAF) and rilpivirine combination patents illustrate the commercial stakes: PTA disputes over several months of additional term, when applied to a drug generating several billion in annual sales, translate directly into hundreds of millions of dollars in additional revenue.
FDA Regulatory Exclusivity: Running Independently of Patent Protection
Regulatory exclusivity is granted by the FDA upon drug approval and operates entirely independently of the patent system. A drug can have robust patent protection but no exclusivity, or it can have exclusivity but no relevant patents remaining. The distinctions matter because exclusivity bars FDA from accepting or approving competing ANDA or 505(b)(2) applications, while patents are enforced through civil litigation after FDA approval.
New Chemical Entity (NCE) exclusivity runs five years from the date of NDA approval for drugs containing an active moiety not previously approved in the U.S. During the first four years of NCE exclusivity, the FDA will not accept an ANDA at all. Starting in the fifth year (or from the four-year mark if the ANDA includes a Paragraph IV certification), FDA can accept but not finally approve an ANDA. This timing creates a specific litigation window: a generic filing a Paragraph IV at the four-year mark triggers the 30-month stay, which then runs through year 7 or beyond, effectively providing brand companies with automatic litigation time even before any court activity.
Three-year exclusivity applies to drugs with a new indication, new dosage form, or new strength requiring new clinical studies for approval. This is a narrower protection than NCE exclusivity; it bars FDA from approving ANDAs that rely on the new clinical information but does not prevent the FDA from accepting ANDAs altogether. Generic applicants can sometimes work around three-year exclusivity through independent clinical data development, which is why this form of exclusivity is generally viewed as weaker than NCE.
Orphan Drug Exclusivity (ODE) runs seven years from approval of a drug for a designated orphan disease indication, defined as a condition affecting fewer than 200,000 patients in the U.S. at the time of orphan designation. ODE bars competing applications for the same drug in the same orphan indication but does not bar competing drugs with different APIs for the same condition, and it does not bar the same drug for different indications. Sarepta Therapeutics’ Exondys 51 (eteplirsen) and its subsequent exon-skipping DMD drug portfolio illustrate the strategic use of overlapping orphan exclusivity periods to maintain exclusivity in rare pediatric neuromuscular diseases across multiple product iterations.
Pediatric Exclusivity (PED) adds six months to existing patents and exclusivity periods when the sponsor completes FDA-requested pediatric studies. It applies broadly, extending all listed Orange Book patents and all existing exclusivity periods for the drug by six months. For a blockbuster with $3 billion in annual U.S. sales, six months of extended exclusivity is worth approximately $1.5 billion at full price, making pediatric study completion one of the highest-ROI regulatory activities a company can pursue.
GAIN Act exclusivity adds five years to existing exclusivity for Qualified Infectious Disease Products, or QIDPs, targeting serious bacterial or fungal pathogens. This provision was designed to incentivize antibiotic development, where the commercial model has been historically unattractive due to the short treatment durations and the clinical imperative to reserve new antibiotics for last-resort use. Antibiotics designated as QIDPs can achieve 10 years of combined NCE and GAIN exclusivity, meaningfully improving the commercial case for development investment.
The Concurrence Problem: Modeling Overlapping Protections
The analytical challenge for IP teams and investors is that patents and exclusivities run concurrently, not sequentially. The correct model stacks all relevant protection layers chronologically and identifies the last expiring relevant protection for each approved use, then evaluates the probability and timing of Paragraph IV challenges against each remaining layer. The output is not a single expiration date but a probability distribution of generic entry dates, weighted by litigation outcomes.
For example: a drug approved in 2019 for an NCE indication might carry NCE exclusivity through 2024, a composition-of-matter patent through 2027 (with PTE), a formulation patent through 2029, a method-of-use patent on a second indication approved in 2021 (with its own three-year exclusivity through 2024) through 2031, and pediatric exclusivity tacked onto the composition-of-matter patent through mid-2027. The generic manufacturer’s actual entry window depends on which of those layers it can successfully challenge, and in what sequence.
Key Takeaways: Section V
PTE and PTA extend effective patent life beyond the 20-year statutory term but are subject to calculation disputes and litigation. Regulatory exclusivities are independent of patents; NCE exclusivity is the most commercially powerful, while orphan and pediatric exclusivities serve specific strategic roles. Modeling effective market life requires stacking all concurrent protections and probability-weighting each layer against Paragraph IV challenge risk. The output is a distribution, not a single date.
Investment Strategy: Section V
Investors should request IP counsel’s complete exclusivity model for any pharmaceutical company under coverage. A company disclosing a single ‘patent expiration date’ without specifying which patent and without accounting for regulatory exclusivity, additional patents, and pediatric extension eligibility is providing an incomplete picture. Compare the disclosed exclusivity model against an independently constructed Orange Book and USPTO analysis. Significant gaps between the company’s disclosed model and the independent model indicate either a conservative company or an incomplete one, and the direction of the gap matters for revenue modeling.
VI. Evergreening and Patent Thickets: The Full Tactical Playbook
What Evergreening Actually Is
Evergreening is the systematic extension of effective market exclusivity through the sequential filing of secondary patents on modifications of an approved drug. The term is used pejoratively in policy discussions and neutrally in IP strategy discussions, but the mechanism is the same either way: a brand company extends the IP protection around a commercial drug franchise by obtaining patents on new salt forms, new polymorphs, new enantiomers, new formulations, new delivery systems, new dosing regimens, or new indications, and then listing as many of those patents in the Orange Book as the FDA’s listing criteria permit.
The empirical record on evergreening is consistent. A study published in JAMA Internal Medicine found that 78 percent of new patents associated with drugs approved between 2005 and 2015 protected existing drugs rather than novel treatments. For 2016-approved drugs, the median number of Orange Book patents per drug was three, but for top-selling biologics the figure ran substantially higher. Humira, as noted above, carried over 100 U.S. patents. Revlimid (lenalidomide), Celgene’s multiple myeloma drug, was protected by a dense thicket of composition-of-matter, formulation, and method-of-use patents that Bristol-Myers Squibb inherited through its acquisition of Celgene in 2019. Generics launched in 2022 after negotiated settlement agreements, not after patent expiration in the traditional sense.
The Patent Thicket Taxonomy
A full pharmaceutical patent thicket contains multiple distinct layers, each with a specific strategic role.
The composition-of-matter patent is the core. The first evergreening layer typically targets the specific approved salt form (e.g., a hydrochloride salt vs. a free base) or the specific polymorph (crystal form) of the API. Polymorph patents are among the most commercially valuable secondary patents because they are difficult to design around, a generic manufacturer cannot simply use the free base if the only commercially viable synthesis route produces the patented crystal form, yet they face heightened obviousness scrutiny under post-KSR analysis. The litigation over Bristol-Myers Squibb’s clopidogrel (Plavix) bisulfate salt patent, filed after the composition-of-matter patent, generated one of the largest settlements in generic litigation history. Apotex launched at risk in 2006 before the court resolved the dispute; BMS and Sanofi ultimately obtained a $1.1 billion payment from Apotex for lost profits and damages.
The second evergreening layer covers delivery system improvements. An immediate-release formulation is replaced by an extended-release, a transdermal patch, or a subcutaneous auto-injector. The new formulation generates a new NDA filing, a separate Orange Book listing, separate patent protection, and potentially a new regulatory exclusivity period if new clinical studies supported the approval. AbbVie’s evolution of the Humira formulation from a glass vial to a citrate-free auto-injector pen directly followed this model, creating distinct IP protection for the patient-preferred delivery format and forcing biosimilar developers to demonstrate interchangeability separately for each formulation.
The third evergreening layer pursues label expansion. A drug approved for a single indication gains additional approved indications through supplemental NDA filings, each supported by new clinical data, each generating a new Patent Use Code, each potentially generating new method-of-use patent filings with their own 20-year terms. When those supplemental approvals qualify for three-year exclusivity, the brand company gains both regulatory and IP protection for the expanded use. For drugs with multiple-indication labels, the resulting Patent Use Code matrix can become extremely complex, requiring generic applicants to file Paragraph IV certifications against multiple patents with different expiration dates and different use codes.
The fourth layer is pediatric evergreening. If the FDA issues a Written Request for pediatric studies, the brand company’s completion of those studies earns a six-month PED extension across all listed patents and exclusivities. For drugs with pediatric-appropriate formulations and indications, the six-month extension is essentially free money if the company was going to conduct the pediatric studies for clinical or commercial reasons anyway.
Inter Partes Review: The Patent Thicket’s Structural Weak Point
The America Invents Act of 2011 created inter partes review (IPR) as an administrative proceeding before the USPTO’s Patent Trial and Appeal Board (PTAB), allowing challengers to request cancellation of issued patent claims on grounds of anticipation or obviousness based on patents and printed publications. IPR has become a preferred tool for generic and biosimilar manufacturers because it is faster and cheaper than district court litigation, PTAB applies a lower claim construction standard (ordinary meaning rather than narrowest reasonable construction), and IPR petition grant rates for pharmaceutical patents have historically run above 60 percent.
For patent thickets, IPR creates a scalable invalidation tool. A generic manufacturer facing 30 Orange Book-listed patents cannot efficiently litigate all 30 in district court within the 30-month stay window. IPR allows parallel challenges across multiple patents simultaneously, with a lower cost per proceeding and a faster statutory timeline (final written decisions within 12 months of institution). The combination of ANDA litigation plus parallel IPR petitions against the secondary patent estate is now standard practice for generics targeting high-value franchises.
PTAB’s 2016 decision in Coalition for Affordable Drugs v. various patent holders established that IPR petitioners have standing even without an ANDA filing, allowing hedge funds and advocacy organizations to petition for cancellation of pharmaceutical patents. While PTAB subsequently tightened its approach to hedge fund-driven petitions, the decision confirmed that the IPR mechanism is accessible beyond the traditional ANDA litigation context.
Product Hopping: A Related Evergreening Strategy
Product hopping occurs when a brand manufacturer discontinues the original drug formulation and converts the market to a new formulation with fresh patent protection, just before generic entry on the original formulation. The strategy exploits FDA substitution rules: generic pharmacists can automatically substitute an AB-rated generic for the original formulation but cannot substitute it for a new, non-bioequivalent formulation with a different dosage form. By shifting the commercial market to the new formulation, the brand company makes the incoming generic commercially irrelevant even after it obtains FDA approval.
Warner Chilcott’s extended-release mesalamine (Asacol to Asacol HD) and its subsequent switch to Delzicol (mesalamine capsules) demonstrate the tactic in sequence. Each formulation switch created a new NDA, a new Orange Book listing, and several additional years of effective market protection, even as generic versions of the earlier formulations received approval and attempted commercial launch. Antitrust scrutiny of product hopping has intensified: the Second Circuit’s 2016 ruling in New York ex rel. Schneiderman v. Actavis held that a product hop designed primarily to obstruct generic competition can constitute unlawful monopolization under the Sherman Act. That ruling is the leading case shaping brand company decisions on when a formulation switch crosses from legitimate lifecycle management into anticompetitive product hopping.
Regulatory Reform Pressures on Patent Thickets
Congressional attention to pharmaceutical patent thickets has grown since 2020. The Affordable Prescriptions for Patients Act, which has been introduced in multiple sessions, would cap the number of patents a brand company can assert in Hatch-Waxman litigation and bar Orange Book listing of late-filed secondary patents that the FDA determines do not meet the applicable listing criteria. The bill has not passed but reflects the direction of legislative pressure.
The FTC has ramped up Orange Book delisting demands under its authority, focusing on improperly listed device patents and process patents that do not meet FDA’s statutory listing criteria. In September 2023, the FTC issued hundreds of delisting demand letters to pharmaceutical companies, targeting patents it determined were improperly listed in the Orange Book for inhaler and auto-injector drugs, where manufacturers had listed device patents alongside drug patents to trigger additional 30-month stays.
The FTC’s delisting campaign is an analytical variable for IP teams: any Orange Book patent that does not squarely cover the drug substance, drug product, or an approved method of use is vulnerable to an FTC challenge, and an FTC-ordered delisting removes it from the stay-triggering framework immediately. Investors in companies with device-heavy Orange Book listings should treat those listings as elevated-risk assets and consider discounting the exclusivity runway associated with potentially improperly listed patents.
Key Takeaways: Section VI
Evergreening is a structured strategy with four primary tactical layers: salt/polymorph patents, delivery system improvements, label expansion, and pediatric exclusivity. Patent thickets face structural challenges through IPR proceedings at PTAB, which provide generic manufacturers a faster and cheaper invalidation pathway than district court litigation. Product hopping extends market exclusivity but carries antitrust risk post-Actavis. Regulatory pressure on Orange Book listings, particularly the FTC’s 2023 delisting campaign, introduces new uncertainty around improperly listed device patents.
Investment Strategy: Section VI
Analysts covering brand companies with large secondary patent estates should model two scenarios: a ‘thicket survives’ scenario in which the secondary patents hold through their full terms, and a ‘thicket erodes’ scenario in which IPR proceedings and Orange Book delistings accelerate generic entry by two to three years. The revenue delta between those scenarios, discounted at an appropriate rate, represents the IP risk premium embedded in the stock price. Companies trading at premiums that implicitly assume full thicket survival in a high-regulatory-scrutiny environment are carrying unpriced downside.
VII. The Biologics IP Landscape: A Separate Rulebook
Why Biologics Require a Different Analytical Framework
Biologics, including monoclonal antibodies, fusion proteins, recombinant enzymes, and cell therapies, are fundamentally different from small molecule drugs in terms of manufacturing complexity, regulatory approval pathway, and IP framework. The Biologics Price Competition and Innovation Act of 2009 (BPCIA), enacted as part of the Affordable Care Act, created the U.S. biosimilar approval pathway and established a distinct IP regime that operates in parallel to, but differently from, Hatch-Waxman.
The key structural difference is that biologics receive 12 years of reference product exclusivity from the date of first licensure of the reference biologic. No biosimilar application can be approved by the FDA until that 12-year period expires. This is distinct from and additive to any patent protection: even if all relevant biologic patents expired on day one of approval, the 12-year exclusivity bars biosimilar approval entirely. The Hatch-Waxman 5-year NCE exclusivity is commercially significant but shorter; the BPCIA’s 12-year period is categorically longer and reflects the regulatory judgment that biologic development requires a longer period to justify R&D investment given manufacturing complexity.
The BPCIA ‘Patent Dance’: A Parallel Litigation Choreography
The BPCIA created a formal, multi-step patent information exchange procedure between the biosimilar applicant and the reference product sponsor, colloquially called the ‘patent dance.’ When a biosimilar manufacturer files a 351(k) application, it must notify the reference product sponsor within 20 days of FDA acceptance of the application. The reference product sponsor then has 60 days to identify patents it would assert against the biosimilar applicant. The biosimilar applicant responds within 60 days with its contentions on infringement and validity. The parties then negotiate a list of patents for immediate litigation, with any unresolved patents addressed in a second phase of litigation after biosimilar commercial launch.
The patent dance is optional, not mandatory, for biosimilar applicants. The Federal Circuit ruled in Sandoz Inc. v. Amgen Inc. (2017) that failure to participate in the patent dance does not bar the biosimilar applicant from launching, though it may affect the timing of the reference product sponsor’s ability to seek a preliminary injunction. Many biosimilar manufacturers elect not to participate in the patent dance to preserve strategic flexibility and avoid early disclosure of their invalidity contentions.
Biosimilar Interchangeability: The Regulatory Standard That Reshapes IP Value
The BPCIA created two tiers of biosimilar approval: ‘biosimilar’ and ‘interchangeable.’ A biosimilar demonstrates it is highly similar to the reference product with no clinically meaningful differences in safety, purity, and potency. An interchangeable biologic meets the additional standard of demonstrating it produces the same clinical result as the reference product in any given patient and, for products administered more than once, that the risk in terms of safety and diminished efficacy of alternating or switching is not greater than the risk of using the reference product without switching.
Interchangeability is commercially decisive because only interchangeable biologics can be automatically substituted by a pharmacist without physician intervention, the functional equivalent of the AB-rating in the small molecule world. Without interchangeability, biosimilars must be prescribed and dispensed specifically, which limits uptake because it requires active physician prescribing behavior change.
The interchangeability designation interacts with IP strategy in a specific way. The first biosimilar to achieve interchangeability for a given reference product receives one year of exclusivity against other interchangeable biosimilars for that product. This ‘first interchangeable’ exclusivity creates a race among biosimilar manufacturers to demonstrate switching data through clinical trials, with the winner capturing a 12-month window of reduced competition at the more commercially viable tier.
The Humira biosimilar market after 2023 illustrates the interchangeability economics. Amgen’s Amjevita (adalimumab-atto) launched in January 2023 as the first Humira biosimilar in the U.S. Hadlima (adalimumab-bwwd, Samsung Bioepis/Organon) received the first interchangeability designation among Humira biosimilars. The market dynamics post-launch have been driven as much by PBM formulary positioning and rebate negotiations as by interchangeability status, but the interchangeability designation remained a key purchasing committee and P&T committee consideration for hospital systems establishing automatic substitution protocols.
The Biologic IP Valuation Problem: Process Defines Product
Valuing a biologic patent estate requires different inputs than valuing a small molecule estate. The primary valuation drivers for biologic IP are: the breadth of the amino acid sequence claims (a sequence patent covering a broader range of structural variants is more valuable than a narrow patent on a specific sequence), the coverage of manufacturing process patents (including cell line patents, upstream culture conditions, and downstream purification processes), the formulation patent estate (particularly subcutaneous formulations, which offer patient convenience over intravenous administration and carry separate IP), and the method-of-use patent estate across approved indications.
For next-generation biologics, bispecific antibodies and antibody-drug conjugates (ADCs) represent the most active patent-filing categories. Companies including AstraZeneca, Daiichi Sankyo, Pfizer, and Genentech have built ADC patent estates covering linker-payload combinations, conjugation chemistry, drug-to-antibody ratios, and methods of use in specific tumor types. The ADC linker-payload combination is structurally analogous to the small molecule composition-of-matter patent: it is the most fundamental IP protection and typically carries the highest rNPV weight in a valuation model.
Cell therapy and gene therapy represent the frontier of biologic IP strategy. CAR-T therapies from Novartis (Kymriah/tisagenlecleucel) and Gilead/Kite (Yescarta/axicabtagene ciloleucel) are protected by patent estates covering lentiviral vector constructs, CAR receptor design, T-cell activation and expansion protocols, and manufacturing processes for patient-specific cell products. The exclusivity duration for these therapies is effectively indeterminate because the manufacturing process for an autologous cell therapy, which requires using the specific patient’s own cells, is inherently difficult to replicate and biosimilarity standards for personalized cell therapies have not been fully established by the FDA.
Key Takeaways: Section VII
Biologics operate under the BPCIA, not Hatch-Waxman, and receive 12 years of reference product exclusivity independent of patent protection. The BPCIA patent dance is optional but strategically significant. Biosimilar interchangeability is the commercial threshold that enables automatic substitution, and the first interchangeable product earns a 12-month exclusivity window. Biologic IP valuation requires modeling sequence claims, process patents, formulation patents, and method-of-use patents as distinct but interacting asset classes. ADC linker-payload patents and CAR-T manufacturing process patents represent the highest-complexity frontier of biologic IP strategy.
VIII. Global Patent Search: Jurisdiction-by-Jurisdiction Execution Guide
Why Patent Protection Is Territorial
A U.S. patent creates no rights outside the United States. A European Patent granted by the EPO creates rights only in the European Patent Convention member states where the patent is validated. A PCT application establishes a priority date and extends the window for national phase entry, but it does not create granted patent rights in any jurisdiction until the applicant pursues national phase examination and grant. For pharmaceutical companies operating globally, the patent map for a single drug product can look entirely different in Germany, Japan, India, Brazil, and South Africa, with implications for where manufacturing can occur, where competition can enter, and how revenue forecasts should be structured by geography.
WIPO PATENTSCOPE: The PCT Application Database
PATENTSCOPE provides free access to international patent applications filed under the PCT system and to national collections from over 50 participating countries. For pharmaceutical patent searches, PATENTSCOPE’s PCT application database is the most efficient starting point for international patent family analysis because PCT filings represent the applicant’s intent to seek protection in multiple jurisdictions from a single application.
A PCT application does not guarantee grant in any specific country, but it establishes the priority date and extends the national phase entry deadline to 30 months from priority for most jurisdictions (31 months for EPO, 32 months for some national offices). Analysts tracking a drug’s international patent position should identify all PCT applications claiming the relevant priority date, note the international filing classification codes (IPC or CPC codes), and track which national phases have been entered by reviewing the national phase entry entries in PATENTSCOPE’s legal status data.
PATENTSCOPE’s chemical structure search functionality is particularly useful for pharmaceutical analysis: it allows searching by International Nonproprietary Name (INN), CAS number, or structural formula, returning all PCT applications that explicitly claim or disclose the compound. This is useful for identifying patent applications that may not appear in a keyword search because they describe the compound using IUPAC nomenclature or Markush structure language without using the INN.
EPO Espacenet and the European Patent Register
Espacenet provides free access to over 150 million patent documents from jurisdictions worldwide, with particular depth in European patent applications and grants. For pharmaceutical companies, the European Patent Register, accessible within Espacenet, is the definitive source of legal status data for European patents, including grant status, opposition filings, appeal decisions, and national validation status.
The European post-grant opposition procedure, available within nine months of grant, is a cost-effective mechanism for competitors to challenge European pharmaceutical patents. Unlike U.S. IPR, European oppositions are heard by the EPO Opposition Division and can result in revocation or amendment of the patent on grounds of lack of novelty, inventive step, or insufficient disclosure. Opposition decisions can be appealed to the EPO Technical Boards of Appeal. Pharmaceutical patent oppositions at the EPO are common and have resulted in some high-profile revocations, including partial revocations of biologics patents that narrowed the scope of protection substantially.
Supplementary Protection Certificates (SPCs) are a European-specific mechanism that extends patent protection for up to five years beyond patent expiration for medicinal products that received regulatory approval after the patent was granted. SPCs are granted at the national level in each EU member state and the UK separately, and their validity and coverage have been the subject of extensive CJEU and UK court litigation. For pharmaceutical analysts, SPCs are the European equivalent of U.S. PTE and must be tracked separately per jurisdiction.
Japan Patent Office (JPO) J-PlatPat: Asia-Pacific Intelligence
Japan has the third-largest pharmaceutical market globally by revenue, with distinct patent prosecution practices that create analytical nuance absent from U.S. analysis. J-PlatPat provides free access to Japanese patent documents including prosecution histories and machine translations. Japan historically restricted ‘multi-multi’ claims (claims combining multiple diseases with multiple dosing regimens in a single claim), though JPO practice has evolved. Japanese courts have also addressed extraterritorial enforcement in ways that differ from U.S. Federal Circuit jurisprudence, particularly regarding method claims for drugs manufactured in Japan for export.
Japanese pharmaceutical patent examination requires ‘working examples,’ meaning that broad claims not supported by actual experimental data in the specification are vulnerable to enablement objections. A Markush claim covering 10,000 compounds may not survive examination without working examples for a representative sample of structurally distinct compounds. This means that Japanese pharmaceutical patents often have narrower practical claim scope than their U.S. counterparts, which is analytically important when assessing the territorial breadth of a global patent estate.
Health Canada Patent Register: The North American Picture
Health Canada’s Patent Register covers human and veterinary drug products and is updated nightly. For drugs marketed in Canada, the Register lists medicinally active ingredients with their associated patents, patent expiry dates, and patent linkage information under Canada’s Patented Medicines (Notice of Compliance) Regulations, which are Canada’s analog to the Hatch-Waxman framework.
The Canadian patent linkage system has important differences from the U.S. system. Canada allows a generic manufacturer to challenge listed patents before the Minister of Health through a Notice of Allegation (NOA), which triggers a 24-month prohibition period (shorter than the U.S. 30-month stay). Canadian courts apply a two-step test for patent validity that is distinct from the U.S. framework, and Canadian obviousness jurisprudence differs from post-KSR U.S. standards. Companies with significant Canadian revenue exposure, including those with market-leading positions in major therapeutic categories in Canada, should model Canadian patent expiry separately from U.S. expiry.
Emerging Markets: India, Brazil, and Compulsory Licensing
India and Brazil represent the most commercially significant emerging market pharmaceutical patent jurisdictions because both countries manufacture and export generic drugs at scale and both have used their patent systems to limit originator market exclusivity for drugs of public health importance.
India’s Patents Act, amended in 2005 to comply with TRIPS, includes Section 3(d), which bars patenting of new forms of known substances, including polymorphs, salts, esters, and isomers, unless they demonstrate ‘significantly enhanced efficacy’ relative to the known form. Section 3(d) effectively bars polymorph and salt form evergreening that would be permissible in the U.S. Novartis’s challenge to the Indian patent office’s rejection of its imatinib mesylate (Gleevec) polymorph patent under Section 3(d) was denied by the Indian Supreme Court in 2013 in Novartis AG v. Union of India, establishing that Section 3(d) is constitutional and substantive. That decision remains the governing precedent for secondary pharmaceutical patent applications in India.
Brazil’s IP framework allows compulsory licensing for drugs of public health interest. Brazil has issued compulsory licenses for efavirenz (HIV) and, most recently, has threatened compulsory licensing for high-cost oncology drugs when originator companies declined to negotiate government pricing. For companies with significant Brazilian revenue, the compulsory licensing risk is a distinct and material IP risk that does not appear in the Orange Book or USPTO databases but requires active tracking through ANVISA (Brazil’s FDA) and INPI (Brazil’s patent office) filings.
Specialized Databases for Global Patent Surveillance
Commercial databases including Derwent Innovation (Clarivate), PatSnap, and LexisNexis TotalPatent One provide pharmaceutical patent analytics that go beyond what national patent office databases offer natively. These platforms aggregate patent families across jurisdictions, apply machine learning to identify claim scope similarities, visualize patent citation networks to map competitive white spaces, and track legal status changes across multiple patent offices simultaneously.
Derwent Innovation’s Derwent World Patents Index (DWPI) is the industry standard for patent analytics in pharma; it enhances patent records with standardized abstracts and manually assigned Derwent Classification codes that group patents by therapeutic area and mechanism of action, independent of the original IPC codes assigned by patent offices. For pharmaceutical competitive intelligence, DWPI-classified searches identify competitor patent activity in a therapeutic area with greater precision than raw keyword or IPC searches.
The Medicines Patent Pool (MPP) operates Medspal, a database of the patent and licensing status of essential medicines in low- and middle-income countries (LMICs), covering HIV, hepatitis C, tuberculosis, and a growing portfolio of other conditions. For companies with licensing obligations to generic manufacturers in LMICs, Medspal provides a centralized reference for the status of voluntary licensing agreements and the scope of geographic coverage.
Key Takeaways: Section VIII
Patent protection is territorial; a U.S. patent creates no rights outside U.S. borders. PCT applications establish priority but require national phase entry in each target jurisdiction. European SPCs are the analog of U.S. PTE and must be tracked per member state. Japanese prosecution practice requires working examples, often narrowing claim scope relative to U.S. equivalents. India’s Section 3(d) bars secondary pharmaceutical patents on known compounds without enhanced efficacy evidence, making polymorph and salt form evergreening unavailable in the world’s largest generic drug producer. Commercial patent analytics platforms are essential for global patent family surveillance at scale.
IX. Paragraph IV Certification: How Generic Manufacturers Weaponize Patent Challenges
The Mechanics of a Paragraph IV Filing
When a generic manufacturer files an ANDA, it must certify to the FDA its position on each Orange Book patent listed for the reference listed drug. Paragraph I certifies that no relevant patent has been filed with the FDA. Paragraph II certifies that the relevant patent has expired. Paragraph III certifies that the ANDA will not be marketed before the patent expires. Paragraph IV certifies that the listed patent ‘is invalid or will not be infringed’ by the proposed generic drug.
A Paragraph IV certification is a deliberate legal challenge to an Orange Book patent, and the ANDA filer must simultaneously send detailed notice to the NDA holder and patent owner explaining the factual and legal basis for the invalidity or non-infringement contention. This notice letter is a commercial signal: it tells the brand company that a specific generic manufacturer has committed resources to challenging specific listed patents, and it starts the 45-day clock during which the brand can file a patent infringement suit and trigger the automatic 30-month stay.
The strategic calculus behind a Paragraph IV filing is straightforward. If the generic applicant is the first to file on a given drug, it earns 180-day exclusivity upon a successful challenge, worth hundreds of millions of dollars for major drugs. The filing costs, including legal fees for the challenge preparation and ANDA chemistry, manufacturing, and controls (CMC) work, are typically a fraction of the potential upside. Teva’s Paragraph IV challenge against BMS’s clopidogrel (Plavix) salt form patent, which ultimately failed, cost tens of millions in legal fees and launch-at-risk inventory; Apotex’s at-risk launch, which also failed, resulted in the $1.1 billion damages judgment mentioned above.
At-Risk Launches: The $1 Billion Gamble
A generic manufacturer that has won its ANDA approval but is still in litigation with the brand over Paragraph IV patents can choose to launch its product commercially before the litigation is resolved. This is an ‘at-risk launch.’ If the brand’s patent is ultimately upheld, the generic manufacturer is liable for damages calculated as a reasonable royalty or, in cases where the patent is found willfully infringed, up to treble damages. At-risk launches have been commercially catastrophic in some cases, including the Apotex/Plavix situation, and enormously profitable in others, where the generic won the litigation or reached a favorable settlement after capturing months of first-mover market share.
The decision to launch at risk requires a probability-weighted analysis of litigation outcome, the expected duration of any remaining litigation, the revenue opportunity during the at-risk period, and the damages exposure if the patent is upheld. Generic manufacturers with strong patent challenge teams and experienced litigation counsel can make this assessment with reasonable precision. Those without that capability should not consider at-risk launches on major drugs.
Pay-for-Delay Settlements: The FTC Battleground
Brand companies frequently resolve Paragraph IV litigation through settlement agreements that include compensation to the generic challenger in exchange for the generic’s agreement to delay commercial launch. These agreements, historically called ‘reverse payment’ or ‘pay-for-delay’ settlements, were common practice until the Supreme Court’s 2013 decision in FTC v. Actavis, Inc., which held that large, unjustified reverse payments from a brand company to a generic challenger can raise antitrust concerns and must be evaluated under a ‘rule of reason’ analysis.
Post-Actavis, settlement negotiations have become more complex. Direct cash transfers are now more difficult to justify. Brand companies have shifted to alternative consideration structures, including authorized generic agreements (where the brand grants the settling generic the right to sell an authorized generic, which counts as the generic’s 180-day exclusivity use), supply agreements, licenses to other products, and co-promotion arrangements. The FTC reviews these settlements and has challenged several post-Actavis agreements that it views as functionally equivalent to direct cash payments.
For investors, pay-for-delay settlements that delay generic entry by two to three years are worth modeling explicitly. A negotiated delay of two years on a drug with $2 billion in annual U.S. sales, at a settlement cost of $100 to $200 million in authorized generic rights, represents an economically rational transaction for the brand company and a recoverable investment for the settling generic. The settlement removes litigation uncertainty for both parties and is financially superior to continued litigation for most scenarios where patent invalidity risk is material.
Key Takeaways: Section IX
Paragraph IV certifications are deliberate patent challenges with a structured legal timeline and significant commercial stakes. The 180-day first-filer exclusivity is the core financial incentive driving Paragraph IV filings. At-risk launches amplify both potential returns and damages exposure and require disciplined probability-weighted analysis. Post-Actavis pay-for-delay settlement structures have evolved toward non-cash consideration but remain subject to FTC antitrust scrutiny. Analysts should model Paragraph IV filing dates as lead indicators of generic entry timing, not lag indicators.
Investment Strategy: Section IX
Monitor FDA’s Paragraph IV certification database, which is publicly available through the FDA’s ANDA list and confirmable through court docketing systems (PACER). When a first Paragraph IV certification appears against a drug in your coverage universe, immediately assess: which specific patents are being challenged, whether the brand has filed suit and triggered the 30-month stay, the strength of the challenged patents based on prosecution history and prior IPR outcomes, and whether the generic applicant has a history of successful Paragraph IV litigation. A first Paragraph IV filing against a drug with weak secondary patents and a strong generic challenger is a materially different signal than a first filing against a drug with a broad composition-of-matter patent that has survived prior IPR.
X. IP Valuation: Pricing a Pharmaceutical Patent Portfolio
Why Standard Financial Metrics Miss Patent Value
Pharmaceutical patent portfolios do not appear as discrete assets on most balance sheets; they are embedded in the broader category of intangibles. Yet patent protection, its remaining duration, its legal strength, and its competitive moat are the primary drivers of pharmaceutical revenue forecasting. A drug with 8 years of remaining composition-of-matter protection plus a dense secondary patent estate and no pending Paragraph IV challenges is a fundamentally different asset from a drug with 8 years of remaining protection on a formulation patent only, with three Paragraph IV filers in queue and two IPR petitions pending at PTAB.
Quantitative IP valuation methods used by pharmaceutical M&A teams and institutional investors fall into three primary categories: income-based, market-based, and cost-based approaches.
Income-Based Valuation: Risk-Adjusted Net Present Value
The risk-adjusted net present value (rNPV) method is the industry standard for pharmaceutical IP valuation. It projects the expected cash flows attributable to a drug’s patent protection during each future period, discounts those cash flows at an appropriate rate reflecting the cost of capital and the risk of generic competition, and multiplies each period’s discounted cash flow by a probability factor reflecting the likelihood that the IP protection survives patent challenges and regulatory changes during that period.
The probability factors applied to each period are the most analytically sensitive inputs. A composition-of-matter patent that has survived a Paragraph IV challenge without amendment carries a higher survival probability than one that has never been tested. A formulation patent that relies entirely on unexpected results data carries a lower survival probability than one with extensive comparative clinical data. IPR petition grant rates by therapeutic area and technology type, available from PTAB’s published statistics, provide empirical base rates for adjusting survival probabilities by patent category.
The discount rate applied should reflect both the time value of money and the systematic risk of pharmaceutical cash flows, which are correlated with regulatory decisions and patent court outcomes that are largely uncorrelated with broad market movements. Rates used by pharmaceutical M&A teams typically range from 8 to 15 percent, with higher rates applied to drugs with earlier-stage clinical support for their patent claims and lower rates for drugs with full clinical validation and commercial track records.
Market-Based Valuation: Comparable Transactions and Royalty Rates
Market-based valuation uses comparable royalty rates from licensing transactions and comparable patent sale prices to establish a market-derived value for the patent estate. The Royalty Range and ktMINE databases aggregate pharmaceutical royalty rates from license agreements disclosed in SEC filings, providing empirical data on royalty rates by therapeutic area, stage of development, and patent type.
Royalty rates for composition-of-matter patents on commercial-stage drugs in major therapeutic areas have historically ranged from 8 to 15 percent of net sales for leading commercial products with multiple indications and strong clinical differentiation. Formulation patents licensing rates are typically lower, in the 2 to 5 percent range, reflecting the narrower scope of protection and higher substitution risk. Method-of-use patents covering a single approved indication command rates in the 3 to 8 percent range, depending on the commercial importance of that indication and the carve-out risk.
For M&A due diligence, market-based comparables serve as a cross-check on rNPV analysis. When the two methods diverge significantly, the divergence reflects either a misestimated discount rate, an incorrect survival probability, or a market mispricing, each of which is worth investigating before transaction close.
The Patent Cliff Discount: Modeling Revenue Step-Downs
Patent expiration creates non-linear revenue erosion. The ‘patent cliff’ is not a cliff in the sense of instantaneous revenue elimination; it is a sequence of step-downs as generic competition enters and intensifies. The typical pattern for a small molecule drug in the U.S. runs as follows: in the first six months after generic launch, the first generic filer captures 20 to 30 percent market share at a price approximately 20 to 25 percent below the brand, generating significant revenue for the generic while leaving the brand relatively intact. After 180-day exclusivity expires, a wave of additional generic entrants drives prices down to 10 to 20 percent of the original brand price within 12 to 18 months. The brand retains a residual share, typically 10 to 20 percent, at the brand price among patients and payers who prefer it or for whom it remains formulary-preferred.
For biologics, the erosion pattern is slower, reflecting the higher cost and complexity of biosimilar manufacturing, the interchangeability threshold for automatic substitution, the role of formulary positioning and PBM rebate negotiations, and physician inertia around switching established biologic patients. Empirical data from the first years of adalimumab biosimilar competition in the U.S. shows brand erosion running at a slower pace than small molecule generics, with Humira retaining substantial market share through negotiated rebate arrangements, particularly in the private payer market. The Medicare Part D provisions of the Inflation Reduction Act, which began requiring rebates on certain drugs and may affect biologic pricing dynamics, add additional uncertainty to biologic patent cliff modeling.
Key Takeaways: Section X
rNPV is the standard pharmaceutical IP valuation method, with patent survival probability as the most analytically sensitive input. Market-based royalty rate comparables provide a cross-check, with rates varying by patent type (composition-of-matter, formulation, method-of-use) and therapeutic area. Patent cliff modeling requires a step-down structure, not a single drop-off, with the 180-day generic exclusivity period acting as a commercial buffer that affects both brand and first-generic pricing dynamics. Biologic patent cliffs erode more slowly than small molecule patent cliffs due to manufacturing barriers and interchangeability thresholds.
Investment Strategy: Section X
For pharmaceutical equity coverage, build a patent life matrix for each drug in the covered company’s portfolio. The matrix should list every Orange Book patent by type (DS, DP, PUC), its expiration date including any PTE or PED, its litigation status (pending Paragraph IV, active district court case, pending IPR), and an assigned survival probability based on prosecution history analysis and comparable litigation outcomes. Map the matrix against annual revenue by product to calculate the ‘IP-adjusted revenue’ for each forward year. Companies where the market-implied revenue forecast assumes no patent erosion in years where significant Paragraph IV activity is pending are screened candidates for short positions. Companies where the market is pricing in patent expiration that is actually protected by strong secondary IP that the market has not modeled offer long opportunities.
XI. Global Drug Patent Checking: A Step-by-Step Operational Workflow
Building the Multi-Database Search Protocol
Checking whether a drug is patented is not a single database query. It is a structured multi-database protocol that produces a defensible, jurisdiction-specific patent status determination. The following workflow covers the primary search sequence for a U.S.-focused analysis with international supplementation.
Start with the FDA Orange Book to identify the ANDA/NDA application number, all listed patents with their expiration dates, drug substance and drug product flags, and patent use codes. Note which patents carry DS flags (composition-of-matter), which carry DP flags (formulation), and which are method-of-use patents linked to specific use codes. Record the relevant exclusivity periods: NCE, 3-year, orphan, pediatric, and GAIN.
Proceed to the USPTO Patent Public Search. For each Orange Book patent number, pull the full patent document and read the independent claims. Note claim scope, Markush structure breadth for composition-of-matter claims, and any functional language in method-of-use or formulation claims that could broaden or narrow scope. Pull the prosecution history through Patent Center and identify any office actions, claim amendments, or statements made to distinguish prior art, as these create prosecution history estoppel.
Search the USPTO assignment database for each relevant patent to confirm current ownership and identify any licensing or security interest encumbrances recorded against the patent.
For additional patents not listed in the Orange Book (process patents, polymorph patents, metabolite patents), conduct an assignee-name search in USPTO Patent Public Search using the NDA holder’s name and related subsidiary names as assignees, filtered to the relevant IPC or CPC classification codes for pharmaceuticals. Supplement with a commercial database search in Derwent Innovation or PatSnap using the INN and related compound names to catch any patents that may not yet appear in the USPTO database due to publication delays.
For the international dimension, search WIPO PATENTSCOPE using the PCT application numbers identified from the Global Dossier, note national phase entry status in target jurisdictions, and supplement with Espacenet for European legal status and SPC filings. Check J-PlatPat for Japanese equivalent applications and Health Canada’s Patent Register for Canadian linkage information.
Compile the results into a jurisdiction-specific patent matrix with expiration dates, current legal status, and litigation flags. This matrix is the analytical deliverable; everything that follows in terms of generic entry timing, IP valuation, or competitive strategy analysis flows from this structured foundation.
Key Takeaways: Section XI
A complete drug patent status determination requires a sequential multi-database protocol: Orange Book for regulatory linkage, USPTO for claims analysis and prosecution history, patent assignment records for ownership verification, commercial databases for secondary patent identification, and international databases for jurisdiction-specific legal status. The output is a patent matrix, not a single expiration date. The matrix enables downstream analysis of generic entry timing, IP valuation, and litigation risk.
Conclusion
Determining whether a drug is patented, and what that patent status actually means commercially, requires a level of analytical depth that the phrase ‘check the Orange Book’ does not capture. The Orange Book is the starting point. The USPTO prosecution history is the substance. The global patent family is the competitive map. The regulatory exclusivity stack is the timeline modifier. The Paragraph IV litigation docket is the real-time competitive signal. The rNPV model is the financial output.
For pharmaceutical IP teams, this multi-database, multi-layer analytical framework is the minimum viable approach for any lifecycle management or competitive intelligence project. For R&D strategy leads assessing whether a target indication is protected, the framework applies at the earliest stage of business development evaluation. For institutional investors, understanding the IP stack behind a pharmaceutical company’s revenue forecast is not due diligence enhancement; it is due diligence itself.
The drugs that generate the highest returns over time are not simply the most efficacious; they are the ones whose IP estates are managed with the greatest precision, with the right patents filed at the right time, listed correctly in the Orange Book, defended aggressively against Paragraph IV challenges, and supplemented with regulatory exclusivities that extend effective market life well beyond what the composition-of-matter patent alone would provide. The analysts and investors who understand that architecture, in its full technical and legal complexity, make better decisions. That is the value of knowing how to read a drug patent.
This analysis was prepared for informational purposes only and does not constitute legal advice. Patent status determinations for commercial or litigation purposes require qualified legal counsel with expertise in pharmaceutical intellectual property.