1. The 20-Year Myth: Why Nominal Term Is the Wrong Starting Number
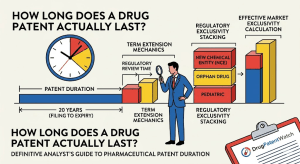
Ask most people in the pharmaceutical business how long a drug patent lasts and they say 20 years. That number is technically correct and commercially useless. The 20-year term under 35 U.S.C. § 154(a)(2) is measured from the earliest effective filing date of the patent application, which is typically the date the patent attorney files the composition-of-matter application on a newly discovered molecule. At that point, the molecule has almost certainly not completed a single Phase 1 trial. It may not even have a clinical development plan.
The patent clock runs through all of that. It runs through the Investigational New Drug (IND) application, through Phase 1 safety studies, through Phase 2 dose-finding, through Phase 3 pivotal trials, through FDA review, and through any Complete Response Letters that require additional data. By the time an NDA is approved and the drug reaches commercial distribution, a typical New Chemical Entity has consumed 10 to 13 years of its 20-year nominal term, leaving between 7 and 10 years of patent-protected commercial life. That is the real number, and it is the one that drives every revenue forecast, every DCF valuation, and every lifecycle management decision.
The shift from the old ’17 years from grant’ system to the current ’20 years from filing’ rule, which occurred as part of the U.S. implementation of the GATT/TRIPS agreements in 1995, made this problem structurally worse. Under the prior system, the 17-year commercial clock did not start until the patent was granted, which meant that long prosecution periods at the USPTO did not eat into the commercial monopoly. Under the current system, every month of prosecution delay, every office action round-trip, every continuation filing extending prosecution consumes a month of the eventual commercial term. The America Invents Act introduced Patent Term Adjustment to partially address this, but the core problem remains: pharmaceutical patents are filed early and consumed slowly by a development process that is expensive, mandatory, and takes years.
The gap between the nominal 20-year term and the actual commercial exclusivity period is not a technicality. It is the economic problem that the entire regulatory exclusivity and patent term extension system exists to solve. Understanding the statutory instruments available to narrow that gap, and their precise limits, is the foundational analytical skill for pharmaceutical IP teams, R&D finance functions, and biotech investors.
Key Takeaways: Section 1
The 20-year nominal patent term is commercially misleading. Effective patent life for a typical New Chemical Entity averages 7 to 10 years after FDA approval, because development and regulatory review consume 10 to 13 years of the nominal term before a product reaches the market. The shift from ’17 years from grant’ to ’20 years from filing’ under GATT/TRIPS structurally penalizes long development programs. Every revenue model and DCF valuation of a pharmaceutical asset should use effective patent life, not nominal term.

2. Filing Date Erosion: How the Patent Clock Starts Before the Molecule Has a Name
The filing date problem in pharmaceuticals is more severe than in most other technology sectors, because the lead time between invention and commercialization is longer. A software company files patents close to product launch. A pharmaceutical company files foundational composition-of-matter patents during early discovery, sometimes before the molecule being patented has been screened in a single animal model.
This happens for rational reasons. The moment a chemist synthesizes a novel compound with an interesting pharmacological profile, there is competitive pressure to file. Competitor programs in the same target class may be months behind. Filing early establishes priority, prevents subsequent inventors from patenting the same structure, and begins building the portfolio that will eventually define the commercial franchise. The cost of filing early is years of patent term. For a blockbuster drug that takes 12 years to reach approval after the foundational patent is filed, the effective remaining patent life at launch might be 8 years. For a drug that takes 14 years due to difficult Phase 3 trials or safety issues requiring additional studies, it might be 6 years.
Average patent pendency compounds this. Grabowski and Vernon’s foundational analysis of effective patent life in pharmaceuticals calculated an average patent pendency of 3.8 years from filing to grant for pharmaceutical applications. The top quartile of complex applications, where claim scope disputes with the USPTO require multiple office action rounds, averaged 8.49 years of pendency. This is time during which the innovator has no issued patent to assert and during which the 20-year clock is running without the patent having been granted yet. Patent Term Adjustment was created specifically to address this USPTO-side delay, but it cannot restore time lost to the development timeline itself.
The practical consequence for IP teams is that the filing strategy for a pharmaceutical program must be calibrated against the expected development timeline from the outset. A molecule expected to require a 12-year development program should be analyzed for continuation-in-part filings and divisional applications that can push some patent coverage to later filing dates, even if the composition-of-matter patent is filed early. Later-filed secondary patents on specific formulations, dosing regimens, or methods of use for confirmed indications will have later expiry dates and may, in some cases, define the operative end of the commercial protection window.
Key Takeaways: Section 2
Early filing of composition-of-matter patents is commercially rational but structurally expensive in terms of patent life consumed. Average pharmaceutical patent pendency of 3.8 years removes nearly four years of the 20-year term before the patent is even granted. Secondary patent strategies with later filing dates can and should be used to push the operative terminal protection date forward, because the composition-of-matter patent is often not the last patent to expire on a major drug.
3. Patent Pendency and PTA Under 35 U.S.C. § 154: The USPTO Delay Dividend
Patent Term Adjustment, governed by 35 U.S.C. § 154(b), was introduced by the American Inventors Protection Act of 1999 to compensate patent applicants for prosecution delays attributable to the USPTO. PTA adds days directly to the 20-year nominal term and is calculated at the time of patent issuance. It has no statutory maximum, unlike Patent Term Extension.
3a. The Three PTA Delay Categories
PTA accrues under three categories of USPTO failure, each with a specific performance standard. An ‘A delay’ occurs when the USPTO fails to issue a first office action within 14 months of the application filing date, fails to respond to a reply by the applicant within four months, or fails to act on a notice of appeal within four months. A ‘B delay’ occurs when the total prosecution period from filing to grant exceeds three years (the three-year pendency guarantee), excluding time consumed by continuations, requests for continued examination (RCEs), appeals, or applicant-requested delays. A ‘C delay’ covers administrative and appellate review delays, including secrecy orders and Patent Trial and Appeal Board proceedings.
PTA days accumulate throughout prosecution. The USPTO tracks them and lists the final PTA calculation on the issued patent. Applicants who believe the USPTO has undercounted their PTA have the right to petition for recalculation within two months of patent issue and can challenge the calculation in federal district court if the petition fails.
3b. PTA Offset: Applicant Delay and Its Calculation
PTA days accrued through USPTO delay are reduced by days of applicant delay. Applicant delay includes any day the applicant failed to respond to a USPTO action within three months. If the USPTO issues an office action and the applicant takes five months to respond, two months of applicant delay are subtracted from the accumulated PTA. This offset rule is significant for pharmaceutical prosecution strategy: applicants who allow office actions to go unanswered for extended periods to manage workload or costs forfeit the PTA days that accumulate during that delay.
3c. PTA and Its Interaction With PTE
PTA is not merely a technical adjustment. It is a material variable in LOE date calculation. A pharmaceutical patent that receives 18 months of PTA has an effective expiry date 18 months later than its 20-year nominal expiry. If that patent is subsequently selected for a Patent Term Extension under § 156, the PTE calculation starts from the PTA-adjusted expiry date. The PTE is then added on top of that extended term, potentially pushing the final LOE date further out. An innovator company with a drug that earned a 24-month PTA on its foundational patent and qualifies for a 48-month PTE under § 156 reaches an effective expiry nearly six years beyond the nominal 20-year term.
Pharmaceutical IP teams should treat PTA as a recoverable asset during prosecution, not an administrative footnote. Aggressive monitoring of USPTO compliance with 14-month and 4-month response deadlines, prompt petition filing when delays are identified, and careful management of applicant response timing to avoid offset deductions can materially affect the PTA balance and, by extension, the eventual LOE date.
Investment Strategy: PTA as Undervalued LOE Extension
Buy-side analysts frequently calculate LOE dates using the patent’s nominal 20-year expiry without adjusting for PTA. For pharmaceutical patents with complex prosecution histories, PTA adjustments of 12 to 36 months are common. A 24-month PTA on the last operative patent protecting a blockbuster drug represents hundreds of millions to billions of dollars in protected revenue that will not appear in LOE calculations that ignore PTA. Checking issued pharmaceutical patents for their listed PTA amounts and incorporating those adjustments into LOE models is a straightforward source of analytical edge.
Key Takeaways: Section 3
PTA adds days to the 20-year patent term based on documented USPTO delays and has no statutory maximum. It is calculated at issuance and directly extends the patent’s expiry date. PTA is offset by applicant delay days, making prosecution response timing a material financial variable. When PTE is subsequently applied, it starts from the PTA-adjusted date, compounding the extension. Analysts who ignore PTA in LOE modeling systematically underestimate protected revenue duration.
4. The Hatch-Waxman Framework: The Legislative Architecture That Built the Modern Generic Market
The Drug Price Competition and Patent Term Restoration Act of 1984, passed as a compromise between the generic and brand pharmaceutical industries, is the statutory foundation on which virtually all U.S. pharmaceutical patent strategy rests. Its operational logic is a quid pro quo: generic manufacturers receive an expedited approval pathway that lets them reference innovator clinical data, and innovators receive patent term restoration to compensate for development time lost to mandatory FDA review.
4a. The ANDA Pathway and Bioequivalence Standard
Before Hatch-Waxman, a generic manufacturer had to conduct its own full clinical development program to prove safety and efficacy, even though the innovator had already proven both. The Abbreviated New Drug Application (ANDA) under 505(j) of the Federal Food, Drug, and Cosmetic Act eliminated that requirement. Generic applicants now demonstrate bioequivalence: the generic must deliver the same active pharmaceutical ingredient to the systemic circulation at the same rate and to the same extent as the reference listed drug, with the 90% confidence interval for AUC and Cmax ratios falling within the 80-to-125% window.
This bioequivalence standard reduced generic development costs from hundreds of millions of dollars to a range of roughly $1 million to $4 million and compressed development timelines to 18 to 36 months. The cost reduction is the reason generic markets are competitive. If generic development required the same investment as innovator development, only a few manufacturers would attempt it, and prices would not fall to commodity levels even after patent expiry.
4b. The Orange Book: Patent Registry and Competitive Intelligence Tool
The FDA publishes ‘Approved Drug Products with Therapeutic Equivalence Evaluations,’ universally called the Orange Book, as the registry of approved drug products and the patents that brand manufacturers certify as covering each product. Brand manufacturers must list any patent claiming the drug, a method of using the drug, or a method of manufacturing the drug within 30 days of patent issuance or NDA approval, whichever is later. The Orange Book lists the patent number and its expiry date, including any PTE.
Generic manufacturers must certify against every patent listed in the Orange Book for the reference listed drug (RLD). The four certification options are Paragraph I (no relevant patent information submitted), Paragraph II (patent already expired), Paragraph III (wait for patent expiry), and Paragraph IV (patent invalid, unenforceable, or not infringed). The Paragraph IV certification is the mechanism that drives pre-expiry patent challenges and the associated 30-month automatic stay.
4c. The 30-Month Automatic Stay: How Patent Listing Creates Delay
When an ANDA applicant files a Paragraph IV certification and the brand manufacturer sues for patent infringement within 45 days of receiving formal notice, the FDA is automatically prohibited from approving the ANDA for 30 months, or until the court enters final judgment of invalidity or non-infringement, whichever comes first. No court finding is required. The mere act of filing suit within the 45-day window triggers the stay.
The 30-month stay is the single most immediately valuable legal tool available to an innovator after a Paragraph IV filing. It provides 30 months of continued market exclusivity during which the innovator can execute lifecycle management transitions, negotiate settlement terms, or simply collect branded revenue while the legal process runs its course. The Medicare Modernization Act of 2003 limited the stay to one 30-month period per ANDA, preventing the ‘stay stacking’ that had been achieved by listing new patents after an ANDA was already on file.
Key Takeaways: Section 4
Hatch-Waxman’s ANDA pathway reduced generic development costs by approximately 95%, enabling the competitive generic markets that now account for 92% of U.S. dispensed prescriptions. The Orange Book is both a regulatory registry and a strategic weapon: every patent listing activates the Paragraph IV certification requirement and, if challenged, can trigger the 30-month automatic stay. Understanding which patents are listable, when to list newly issued secondary patents, and how to sequence listings to maximize stay duration is core pharmaceutical IP strategy.
5. PTE Under 35 U.S.C. § 156: Eligibility, Statutory Limits, and the 14-Year Ceiling
Patent Term Extension under 35 U.S.C. § 156 is the primary mechanism for restoring patent life consumed by mandatory FDA regulatory review. It is specific to regulatory products, applies to pharmaceuticals, veterinary drugs, animal biologics, food additives, color additives, and medical devices, and operates through a formal joint process involving both the USPTO and the FDA.
5a. Eligibility Requirements
Four conditions must be met for PTE eligibility. The patent must claim the approved product, a method of using the approved product, or a method of manufacturing the approved product. The drug product must have obtained regulatory review permission (an IND approval, which allows clinical trials to begin) prior to the patent’s issuance. The product must have received its first permission for commercial marketing or use (NDA, BLA, or equivalent). The patent must not have previously been extended, and the application for extension must be filed within 60 days of product approval.
The last requirement, the 60-day filing deadline, is absolute and cannot be waived. Missing the 60-day window forfeits PTE eligibility for that patent entirely. For a blockbuster drug where a PTE of even 12 months could be worth hundreds of millions of dollars in protected branded revenue, a missed filing deadline represents a material financial loss attributable to administrative failure.
The patent selected for PTE should generally be the patent with the longest remaining nominal term among those eligible, to maximize the absolute duration of the extended term. This is not always the composition-of-matter patent; if the composition-of-matter patent was filed early and has already been eroded by a long development timeline, a later-filed secondary patent may have more nominal life remaining and produce a longer effective exclusivity period when extended.
5b. The Two Statutory Ceilings
PTE is bounded by two independent limitations, both of which must be satisfied. The absolute maximum extension is five years, regardless of how long the regulatory review period was. If the FDA took eight years to approve a drug, the maximum PTE is still five years. The second limitation is the 14-year effective patent life cap: the extension cannot result in a post-approval patent term exceeding 14 years. If the patent would still have 13 years remaining as of the date of FDA approval even without any PTE, the extension is limited to one year. The 14-year cap reflects Congress’s judgment that 14 years of post-approval exclusivity is sufficient to justify the R&D investment, and that extensions beyond this level are unwarranted.
The practical effect of the 14-year cap is asymmetric. Drugs with long development timelines (12 or more years from patent filing to approval) will have short remaining patent lives at approval, often 7 to 8 years, and will qualify for extensions approaching the 5-year maximum. Drugs approved relatively quickly (8 or 9 years from filing to approval) will have more residual patent life and may hit the 14-year cap at a smaller extension. Drugs approved within 6 years of patent filing will have so much patent life remaining that they receive no PTE at all.
5c. The One-Patent Rule and Its Strategic Implications
Only one patent per approved drug product can receive a PTE. The innovator selects which patent to extend, subject to the eligibility requirements. This restriction creates a selection problem: the innovator must identify which among several eligible patents will, when extended by the calculated PTE, produce the latest terminal expiry date and thus the longest effective commercial exclusivity. In a portfolio where a composition-of-matter patent expires in 2029 and a formulation patent expires in 2031, a PTE calculated at 3 years applied to the composition-of-matter patent produces a 2032 expiry. A 3-year PTE applied to the formulation patent produces a 2034 expiry. The selection decision is worth two years of protected revenue.
The one-patent rule also creates an interaction with the innovator’s secondary patent strategy. If the innovator has built a dense secondary patent portfolio with multiple patents expiring after the composition-of-matter patent, the PTE may be less strategically critical than the secondary portfolio itself. Conversely, if the secondary portfolio is thin and the composition-of-matter patent is the last line of defense, maximizing the PTE calculation through scrupulous documentation of the regulatory review period becomes the most important IP activity in the drug’s commercial life.
Key Takeaways: Section 5
PTE eligibility requires four conditions, and the 60-day filing deadline after FDA approval is absolute. The two statutory ceilings (5-year maximum extension and 14-year effective life cap) operate independently, and both must be satisfied. The one-patent rule makes patent selection for PTE a material financial decision that can be worth years of protected revenue. For drugs with long development timelines, PTE extensions approach the 5-year maximum. For drugs approved quickly relative to their patent filing date, the 14-year cap limits the extension substantially or eliminates it.
6. PTE Calculation Mechanics: Testing Period, Approval Period, and the Due Diligence Trap
The statutory formula for PTE under 35 U.S.C. § 156(c) involves calculating a Regulatory Review Period (RRP) that consists of two components: a testing period and an approval period. The formula then applies deductions for time that occurred before the patent was granted and for time attributable to applicant lack of due diligence.
6a. The Testing Period
The testing period begins on the effective date of the IND application, which is the date on which clinical trials are authorized to begin, and ends on the date of initial submission of the NDA or BLA. For a drug where the IND was effective January 1, 2005, and the NDA was submitted July 1, 2017, the testing period is 12.5 years.
The testing period is a relatively straightforward calculation when the clinical development program proceeds linearly. It becomes more complex for drugs that received multiple INDs for different indications, had INDs transferred between companies through licensing or acquisition, or had clinical holds imposed by the FDA that paused the development clock. FDA regulations specify how these scenarios are handled in the RRP determination, but they require careful documentation to defend.
6b. The Approval Period
The approval period begins on the initial NDA/BLA submission date and ends on the date of FDA approval. For the NDA submitted July 1, 2017, and approved December 1, 2019, the approval period is 2.5 years. A critical statutory nuance is that the NDA submission date is counted in both the testing period and the approval period. The submission date is the endpoint of the testing period and the starting point of the approval period simultaneously, which prevents the submission date from being ‘lost’ in the calculation.
For drugs that received Complete Response Letters requiring additional data and resubmission, the approval period includes the full time from initial submission to final approval, regardless of how many submission cycles occurred. This can significantly extend the approval period component of the RRP and increase the calculated PTE.
6c. The PTE Calculation Formula
The USPTO calculates PTE using the following components. The gross RRP equals the testing period plus the approval period. From this, two deductions are made: the pre-grant regulatory review period (PGRRP), which is the portion of the RRP that occurred before the patent was issued; and the due diligence deduction (DD), which reflects time attributable to the applicant’s failure to act with due diligence during the RRP.
The gross PTE equals one-half of the testing period plus the full approval period, minus the PGRRP and DD. This formula compensates for only half the clinical development time, recognizing that the innovator has at least partial use of the patent during clinical development even though the product is not yet marketed. The full approval period is compensated because the innovator cannot begin marketing during FDA review and derives no commercial benefit from the patent during that time.
A simplified numerical example: testing period of 12 years, approval period of 2 years, PGRRP of 2 years (patent issued 2 years before approval), no due diligence issues. Gross PTE = (0.5 x 12) + 2 – 2 – 0 = 6 years. Statutory maximum is 5 years. PTE granted = 5 years (maximum applies).
6d. The Due Diligence Trap
The due diligence requirement is a source of significant legal risk that operating pharmaceutical companies frequently underestimate. During the FDA’s determination of the RRP, the agency reviews the sponsor’s responsiveness to FDA information requests, the timing of NDA amendments and responses to FDA queries, and whether any periods of sponsor inactivity cannot be explained by bona fide development activity.
Generic manufacturers, in Paragraph IV litigation, routinely review FDA review files obtained through Freedom of Information Act requests, searching for periods during which the NDA sponsor was slow to respond to FDA queries or failed to provide required information promptly. If a generic challenger successfully argues that a significant period of the RRP was attributable to sponsor lack of due diligence, the USPTO will deduct those days from the calculated PTE. For a drug where a 90-day period of sponsor delay is identified, the PTE is reduced by 45 days (because only half the testing period is compensated). For a drug where a 12-month delay is identified during the approval period, the full 12 months are deducted from the PTE.
The financial exposure from due diligence challenges is real. A successful 6-month due diligence challenge against the PTE of a drug generating $5 billion annually reduces the protected monopoly period by six months, potentially costing the innovator $2.5 billion in branded revenue. This makes thorough, contemporaneous documentation of every FDA interaction, every amendment submission timeline, and every due diligence period the most commercially valuable record-keeping function in a pharmaceutical company’s regulatory affairs operation.
Key Takeaways: Section 6
The PTE formula compensates for half the testing period plus the full approval period, with deductions for pre-grant RRP time and applicant due diligence failures. The NDA submission date counts in both the testing period and the approval period. Due diligence challenges by generic manufacturers during Paragraph IV litigation can reduce the calculated PTE, and contemporaneous FDA documentation is the primary defense. Six months of documented due diligence failure can cost an innovator billions in reduced protected revenue on a major drug.
7. The One-Patent Rule: Its Statutory Basis and Its Limits
The restriction that only one patent per approved drug product can receive a PTE is codified in 35 U.S.C. § 156(c)(4). The legal rationale is that the RRP for a single drug product generates one compensable period of delay, and compensating multiple patents from the same single delay event would over-reward the innovator. The statute’s use of the term ‘drug product’ as the unit of analysis is what creates the strategic opportunity for multi-PTE strategies.
7a. What Constitutes a Separate ‘Drug Product’?
The USPTO and FDA have interpreted ‘drug product’ in the context of § 156 to refer to the approved product in the form submitted for regulatory review. This interpretation means that a single active moiety can potentially qualify for multiple PTEs if it appears in multiple distinct regulatory submissions that each have their own RRP. The critical question is whether each NDA constitutes a separate ‘drug product’ eligible for its own RRP and its own PTE.
In practice, the FDA’s determination of the RRP for each NDA submission creates the basis for a separate PTE application. If the same active pharmaceutical ingredient is approved as a monotherapy under NDA-A and as part of a fixed-dose combination under NDA-B, each NDA has its own regulatory submission date, its own review period, and its own approval date. Each constitutes a separate RRP. Each can support a separate PTE application for a different patent claiming the product.
7b. Why the One-Patent-Per-Product Rule Does Not Prevent Multiple PTEs for One Molecule
The statutory language says one patent per approved product, not one patent per active moiety. An innovator that simultaneously obtains approval for three products containing the same active moiety, each under a distinct NDA, can apply for three separate PTEs, each against a different patent, each anchored to a different product’s RRP. The restriction that only one patent can be extended per product does not prevent the same molecule from providing the basis for PTEs on three separate patents, as long as each patent is tied to a distinct approved product.
This statutory gap is not a loophole that was accidentally overlooked. It reflects the underlying design of § 156, which compensates regulatory review time on a product-by-product basis. The consequence is that innovators who time their combination product NDA submissions to coincide with their monotherapy approval can legally secure multiple PTEs for the same active ingredient, extending multiple patents by the relevant regulatory review periods.
Key Takeaways: Section 7
The one-patent rule restricts PTEs to one per ‘drug product,’ not one per active moiety. A single active pharmaceutical ingredient appearing in multiple separate NDA approvals, each with its own RRP, can support multiple PTE applications, each against a different patent. The statutory interpretation of ‘drug product’ as the NDA-level approval unit is the legal basis for the multi-PTE strategies discussed in the Nesina case study below.
8. The Nesina Multi-PTE Strategy: IP Valuation of a Same-Day NDA Approval Tactic
The simultaneous approval of alogliptin and two alogliptin-containing combination products on the same day in 2013, and the subsequent successful prosecution of three separate PTE applications arising from those three distinct NDA approvals, is the most comprehensively documented example of multi-PTE execution in the U.S. patent system.
8a. The Regulatory Architecture of the Nesina Approval
Takeda Pharmaceutical Company filed three separate NDAs for alogliptin-containing products: NDA 022271 for Nesina (alogliptin benzoate, the monotherapy), NDA 022426 for Kazano (alogliptin and metformin hydrochloride, fixed-dose combination), and NDA 022424 for Oseni (alogliptin and pioglitazone, fixed-dose combination). The FDA approved all three on January 25, 2013.
Each NDA had its own review history, its own regulatory submission date, and its own FDA approval date (which happened to be the same calendar date). Each constituted a separate ‘drug product’ under the § 156 framework. Each generated a distinct RRP. Takeda filed three separate PTE applications, each citing a different patent, each anchored to a different NDA’s RRP.
8b. PTE Grants and Their Financial Impact
The USPTO granted all three PTE applications. The three extended patents covered different aspects of the alogliptin franchise: the compound itself, specific formulation aspects, and methods of use for treating type 2 diabetes. By extending three separate patents on three separate legal bases, Takeda secured protection for the alogliptin franchise through multiple independent patent layers, any one of which could block generic entry regardless of whether the others were challenged or expired.
The financial value of this strategy is quantifiable. Alogliptin-containing products generated hundreds of millions of dollars in annual revenue. Each additional year of patent protection on any one of the three extended patents, if that patent was the last operative protection against generic entry in any year, represented the full annual revenue that would otherwise have been lost to generic competition. The legal fees associated with prosecuting three PTE applications are trivial compared to even one additional quarter of protected branded revenue.
8c. Lyrica and the Two-Indication Same-Day Approval
A similar strategy executed by Pfizer for pregabalin (Lyrica) produced two PTEs from two same-day approvals for different indications (epilepsy and fibromyalgia), each submitted under distinct NDA supplements with independent regulatory histories. The strategy worked on the same principle as the Nesina approach: two separate regulatory review periods, two separate PTE applications, two separate patents extended.
Lyrica’s combined annual revenue exceeded $5 billion at peak, making the value of even a modest PTE extension worth hundreds of millions of dollars per year. The Lyrica multi-PTE strategy, like Nesina’s, was not challenged as legally impermissible because it complied precisely with the statutory language.
8d. Organizational Requirements for Multi-PTE Execution
The Nesina and Lyrica strategies required precise coordination between Regulatory Affairs and IP Counsel beginning years before the NDA submissions were filed. The regulatory team had to structure the NDA submissions to ensure that each product was filed under a distinct NDA (not as a supplement to the same NDA), that each NDA had its own clinical data package sufficient to support a complete approval, and that the filings were timed to produce simultaneous or near-simultaneous approvals.
The IP team had to identify in advance which patents would be selected for PTE under each NDA’s RRP, ensure those patents were eligible (i.e., not previously extended, filed before IND approval, claiming the product), and file the PTE applications within the 60-day window after each product’s approval. This requires a coordinated project plan across two disciplines that historically operate with significant information barriers. Companies that lack this coordination mechanism cannot execute multi-PTE strategies even when the regulatory conditions permit them.
Investment Strategy: Multi-PTE as Portfolio Valuation Signal
When a pharmaceutical company simultaneously files multiple NDA submissions for the same active moiety in combination products and achieves same-day or near-simultaneous approvals, this is a public regulatory signal that the company may be positioning for a multi-PTE application. Tracking FDA approval calendars for simultaneous approvals across NDA filings for the same active ingredient, cross-referenced with the Orange Book patent listings for each NDA, allows analysts to identify when an innovator has successfully stacked PTEs and adjust LOE date estimates accordingly.
Key Takeaways: Section 8
The Nesina case produced three separate PTE grants for the same active ingredient by securing three distinct NDA approvals with three independent regulatory review periods. The Lyrica case replicated the strategy with two same-day indication approvals. Multi-PTE execution requires tight coordination between Regulatory Affairs and IP Counsel, beginning years before NDA submission. Public FDA approval records and Orange Book PTE listings allow analysts to identify when this strategy has been successfully deployed.
9. PTA and PTE Interaction: How the Two Mechanisms Compound
PTA and PTE operate independently but their outputs are additive in a specific sequential manner. Understanding the mechanics of this interaction is necessary for accurate LOE date calculation.
PTA is calculated and applied at the time of patent issuance. It adds days to the nominal 20-year expiry date, producing a PTA-adjusted expiry date. This is the actual statutory expiry of the patent before any PTE. When PTE is subsequently granted under § 156, the extension is added to the PTA-adjusted expiry date, not the nominal 20-year date. The sequence matters: PTA first, then PTE on top.
The 14-year effective life cap under § 156 applies to the PTE component only. It is calculated as the number of years from FDA approval to the proposed extended expiry date. If the PTA adjustment has already pushed the expiry date beyond 14 years from FDA approval, the PTE may be limited or entirely foreclosed by the cap. Conversely, if the patent has very little PTA, the PTE has more room to operate before hitting the 14-year ceiling.
A practical calculation illustrates the compounding. Patent filing date: January 2002. Patent issue date: February 2007 (patent pendency of 5 years, of which 14 months was USPTO delay, generating approximately 14 months of PTA). Nominal expiry: January 2022. PTA-adjusted expiry: March 2023. NDA approved: December 2018 (16.9 years after filing). PTE calculated at (0.5 x 11.8 years testing period) + 2.1 years approval period = 8.0 years gross PTE, but PGRRP adjustment reduces it. Final PTE: 48 months (5-year maximum applies, but the 14-year cap calculation from December 2018 + 48 months = December 2022 is within 14 years of the December 2018 approval date, so both ceilings are satisfied). Final effective expiry: March 2023 + 48 months = March 2027. This drug has 8.25 years of commercial patent life (December 2018 to March 2027), versus only 3.3 years (December 2018 to March 2023) without PTE.
Key Takeaways: Section 9
PTA extends the nominal expiry date, and PTE is applied on top of the PTA-adjusted date. The 14-year effective life cap applies only to the PTE component and uses the PTA-adjusted date as the starting point. Significant PTA can either reduce the available PTE (by putting the patent closer to the 14-year ceiling already) or expand it (by creating a later adjusted expiry date from which the PTE calculation is applied). Accurate LOE date modeling requires explicit calculation of both components in sequence.
10. FDA Regulatory Exclusivities: The Parallel Protection System
FDA regulatory exclusivities are legally distinct from patent rights. They are granted by the FDA as a condition of NDA or BLA approval and prohibit the FDA from accepting or approving competing applications for a defined period, regardless of the patent status of the drug. A drug can have both patent protection and regulatory exclusivity, either alone, or neither. They run on independent clocks and can stack, overlap, or extend protection beyond the terminal patent date.
The regulatory exclusivity system exists because patents are imperfect barriers in pharmaceuticals. Not every drug receives a broad composition-of-matter patent. Some drugs are based on naturally occurring molecules that may be difficult to patent. Some drugs reach approval late in their patent term, leaving insufficient remaining exclusivity to justify the commercial investment without additional protection. Regulatory exclusivities fill these gaps and supplement patent protection for all others.
For LOE date calculation, the operative terminal protection date is the later of: (a) the last relevant patent’s expiry date (including any PTE), and (b) the expiry of the last relevant FDA regulatory exclusivity. In cases where regulatory exclusivity extends beyond the last patent, the LOE date is determined by the exclusivity, not the patent calendar.
Key Takeaways: Section 10
FDA regulatory exclusivities and patent rights operate on independent legal bases and must both be tracked to determine the correct LOE date. The later of the last patent expiry and the last regulatory exclusivity expiry is the operative LOE date. Failure to account for unexpired regulatory exclusivities when calculating LOE dates produces systematically early LOE forecasts.
11. NCE Exclusivity and the NCE-1 Litigation Trigger: Four Years of Certainty, Not Five
New Chemical Entity exclusivity is granted for five years when the FDA approves an NDA for a drug containing an active moiety never previously approved in any other NDA or application. The five-year period prohibits the FDA from accepting any ANDA or 505(b)(2) application referencing the innovator’s drug during that period.
11a. Active Moiety Definition and Its Strategic Implications
The ‘active moiety’ standard for NCE eligibility is more restrictive than it appears. A salt, ester, or other non-covalent derivative of a previously approved active moiety does not qualify as a new active moiety under FDA regulations. If esomeprazole (the S-enantiomer of omeprazole) had been classified as a salt or derivative of omeprazole rather than as a distinct active moiety, it would not have qualified for NCE exclusivity. The FDA determined that the individual enantiomer was in fact a distinct active moiety, qualifying for NCE exclusivity.
This determination creates significant incentives for chiral switch programs and for development of structurally distinct molecules rather than derivatives of existing drugs. An active moiety determination that grants NCE exclusivity adds five years of regulatory protection from the approval date, independently of any patent protection, and represents a substantial financial benefit relative to a product that qualifies only for the three-year new clinical investigation exclusivity.
11b. The NCE-1 Date: When ‘Five Years’ Becomes ‘Four Years’
The practical ceiling of NCE exclusivity is four years, not five, for any drug that has patents listed in the Orange Book. The exception under 21 C.F.R. § 314.108(b)(2) allows a generic applicant to submit an ANDA with a Paragraph IV certification one year before the five-year NCE exclusivity expires, at the four-year mark. This ‘NCE-1’ date is when generic manufacturers can initiate their formal legal challenge, even though they cannot receive approval until the five-year period expires.
Filing at the NCE-1 date starts the 45-day clock for the innovator to file a patent infringement suit and trigger the 30-month stay. If the brand files suit promptly, the 30-month stay runs from the filing date of the ANDA (year 4 of NCE exclusivity) and expires 30 months later (approximately year 6.5 post-approval). By this point, the five-year NCE exclusivity has long since expired, and the generic’s path to approval depends entirely on the outcome of the patent litigation. The practical result is that NCE exclusivity provides four years of complete regulatory certainty, followed by at least 30 months of litigation-dependent protection if the brand files suit.
11c. NCE Exclusivity in IP Valuation
For drugs approved with NCE exclusivity, the four-year certainty period (before the NCE-1 trigger) is the highest-confidence component of the exclusivity stack. It cannot be shortened by patent challenge outcomes or PTAB decisions. It is a regulatory fact, not a legal probability. Analysts should treat the NCE period as near-certain revenue (subject only to safety withdrawals and commercial execution failures), while the post-NCE period, dependent on patent survival, should be probability-weighted.
Key Takeaways: Section 11
NCE exclusivity provides five calendar years of FDA registration protection, but the NCE-1 provision allows generic ANDA filing with a Paragraph IV certification at year four, initiating patent litigation. For drugs with Orange Book-listed patents, NCE exclusivity provides four years of full certainty and then converts into patent litigation territory. Active moiety determinations are binary events that have significant IP valuation implications, particularly for chiral switches and enantiomer-based programs.
12. Orphan Drug Exclusivity: Seven Years, No Paragraph IV Override
Orphan Drug Exclusivity (ODE) under the Orphan Drug Act provides seven years of market protection for drugs designated to treat rare diseases (fewer than 200,000 U.S. patients) or diseases affecting more than 200,000 patients when there is no reasonable expectation that development costs will be recovered from U.S. sales.
ODE is the most defensively durable form of pharmaceutical market exclusivity in the U.S. system. It cannot be overcome through the Paragraph IV certification process. During the seven-year ODE period, the FDA will not approve any application for the ‘same drug’ for the ‘same disease or condition.’ Generic challengers cannot file an ANDA or 505(b)(2) and have it approved during the ODE period, regardless of how strong their patent challenge might be. The ODE is a pure regulatory block, not a patent block, and it bypasses the litigation-dependent mechanisms that govern patent exclusivity.
12a. Same Drug, Same Indication: The ODE Blocking Standard
ODE protection is limited to the specific drug for the specific orphan designation. A biosimilar of a biologic that holds ODE for Crohn’s disease is blocked from approval in Crohn’s disease, but it is not blocked from approval in rheumatoid arthritis or plaque psoriasis if ODE does not cover those indications. Innovators seeking to maximize ODE protection across their biologic’s approved label must secure separate orphan designations for each qualifying indication.
AbbVie held multiple orphan designations for adalimumab (Humira) covering specific inflammatory conditions with small patient populations. Each designation potentially generated its own ODE period running from the approval date for that specific indication. The interaction between these indication-specific ODE periods and Humira’s patent thicket created a multi-layered exclusivity structure that biosimilar developers had to analyze indication by indication.
12b. ODE in IP Valuation: The ‘Unbreakable Wall’ Premium
Because ODE cannot be challenged through Paragraph IV, it carries a higher certainty premium than patent-based exclusivity in NPV models. A drug with four years of ODE remaining on its key indication should be valued with near-100% probability of protection for those four years, versus a drug with four years of patent protection remaining that faces active Paragraph IV challenges, which should be valued at a probability-weighted fraction of that period. Analysts who apply the same discount rate to patent-based exclusivity and ODE-based exclusivity systematically undervalue the ODE component.
Key Takeaways: Section 12
ODE is the most legally durable form of U.S. pharmaceutical market exclusivity. It cannot be shortened by Paragraph IV patent challenges, PTAB decisions, or district court patent invalidity findings. ODE is indication-specific and must be analyzed separately for each orphan designation. In NPV models, ODE-protected revenue should carry a higher certainty weighting than patent-protected revenue of equivalent duration.
13. New Clinical Investigation Exclusivity: The Lifecycle Management Workhorse
New Clinical Investigation (NCI) exclusivity, sometimes called three-year or ‘clinical investigation’ exclusivity, provides three years of protection for approved changes to a previously approved drug, including new indications, new dosage forms, new routes of administration, and switches from prescription to over-the-counter status, when the approval required the conduct of new clinical studies essential to the approval.
NCI exclusivity protects the change, not the underlying active ingredient. When a drug’s NCI exclusivity period expires, generic manufacturers can reference the innovator’s original safety and efficacy data for the original indication, but they are blocked from referencing the new clinical studies that supported the new indication, dosage form, or route until the three-year period ends.
13a. NCI as a Lifecycle Management Tool
For innovators managing products approaching patent cliff, NCI exclusivity is the most accessible and reliably executable protective mechanism. A new controlled-release formulation that requires clinical studies to demonstrate bioequivalence under changed release conditions, a new pediatric indication requiring studies under a Pediatric Research Equity Act requirement, or a new indication for an existing drug requiring a full Phase 3 program: each of these, if approved by the FDA, generates three years of NCI exclusivity running from the approval date of the change.
NCI exclusivity is also additive to unexpired patent protection and other regulatory exclusivities. A drug with two years of NCE exclusivity remaining that also receives a new indication approval with NCI exclusivity does not simply extend the protection from year two to year five. The NCI exclusivity runs independently, potentially extending protection for the new indication beyond the NCE period by years.
13b. NCI and the Secondary Patent Synergy
NCI exclusivity is most powerful when combined with secondary patent protection on the same formulation or indication it covers. A new controlled-release formulation protected by both a formulation patent expiring in 2031 and NCI exclusivity running until 2030 creates redundant barriers: if the formulation patent is invalidated in Paragraph IV litigation, the NCI exclusivity continues to block the generic’s new formulation application until its own expiry date.
Key Takeaways: Section 13
NCI exclusivity grants three years of protection for approved changes requiring new clinical studies. It protects the change rather than the underlying molecule, making it compatible with simultaneous patent and exclusivity strategies. NCI exclusivity is the primary regulatory tool for late-stage lifecycle management and is additive to other exclusivities and patent protections.
14. Pediatric Exclusivity: The Six-Month Extension Multiplier and Its ROI
Pediatric Exclusivity under the Best Pharmaceuticals for Children Act (BPCA) adds six months to all existing patents and regulatory exclusivities for a drug product when the innovator voluntarily conducts pediatric studies according to an FDA-approved Written Request. Unlike most exclusivity mechanisms, which protect a specific product or indication, Pediatric Exclusivity attaches to the drug as a whole and extends every form of existing protection simultaneously.
14a. The Multiplier Mechanism
The six months of Pediatric Exclusivity does not create a new period of protection following the last expiring exclusivity or patent. It extends each individual existing protection by six months. A drug with a composition-of-matter patent expiring December 2027 and a formulation patent expiring June 2028 that earns Pediatric Exclusivity will see both patents extended: the composition-of-matter patent to June 2028, and the formulation patent to December 2028. The NCE exclusivity, if any, is also extended by six months. Any ODE protection is extended by six months.
This ‘extension of all protections’ design means the financial impact of Pediatric Exclusivity scales with the revenue being protected in the six months following the last protection’s expiry. For a drug generating $10 billion annually, six months of Pediatric Exclusivity is worth approximately $5 billion in protected revenue. The cost of conducting the pediatric studies required to earn this exclusivity is typically $10 million to $50 million. The return on investment is 100:1 to 500:1, making Pediatric Exclusivity among the most financially efficient investments in pharmaceutical IP strategy.
14b. Written Request Compliance and Timing
The FDA issues a Written Request specifying the studies required to earn Pediatric Exclusivity. The innovator then conducts those studies and submits the data. If the FDA determines the studies were conducted according to the Written Request, Pediatric Exclusivity is granted regardless of whether the studies showed benefit in the pediatric population. The exclusivity is a reward for conducting the studies, not for proving pediatric efficacy.
Timing of the Written Request response relative to the drug’s existing exclusivity and patent timeline matters. Pediatric studies typically take 3 to 5 years to complete. An innovator should initiate discussions with the FDA about a Written Request early enough that studies can be completed and submitted before the last existing exclusivity expires, to ensure the six-month extension has something to attach to. If all existing protections have already expired before Pediatric Exclusivity is granted, there is nothing left to extend.
Key Takeaways: Section 14
Pediatric Exclusivity extends all existing patents and regulatory exclusivities simultaneously by six months. For major drugs, the return on investment from conducting pediatric studies is 100:1 to 500:1. Timing of Written Request initiation and study completion must be coordinated against the expiry schedule of existing protections to ensure the exclusivity has operational extensions to apply. Pediatric Exclusivity is among the highest-ROI activities in pharmaceutical lifecycle management.
15. GAIN Exclusivity and Qualified Infectious Disease Product Strategy
The Generating Antibiotic Incentives Now (GAIN) Act, enacted as part of the Food and Drug Administration Safety and Innovation Act of 2012, added five years to the exclusivity periods available for Qualified Infectious Disease Products (QIDPs) designated to treat serious or life-threatening infections, including those caused by drug-resistant pathogens.
For a drug designated as a QDIP that also qualifies for NCE exclusivity, the five-year GAIN addition produces a total of 10 years of NCE-equivalent exclusivity (five from NCE, five from GAIN) running from FDA approval. For a QDIP with Orphan Drug Exclusivity, the GAIN addition produces 12 years of protection. A QDIP that qualifies for both NCE exclusivity and ODE, with GAIN added to both, has exclusivity running to the later of those totals.
GAIN exclusivity is an underappreciated strategic asset for companies developing antibiotics, antifungals, and antivirals targeting resistant pathogens. The antibiotics pipeline has historically been thin relative to other therapeutic areas precisely because the patent life consumed by development often leaves insufficient exclusivity to justify the investment. GAIN’s 5-year addition partially addresses this and has contributed to increased investment in antibacterial programs since 2012, though the fundamental economic challenge of antibiotics development (low prices, short treatment courses, resistance concerns) remains.
Key Takeaways: Section 15
GAIN exclusivity adds five years to all existing exclusivities for Qualified Infectious Disease Products. A QDIP with NCE exclusivity receives 10 years of total NCE-type protection from FDA approval. GAIN exclusivity is additive to ODE and can produce 12 years of total protection for drugs with both designations. For companies with infectious disease programs targeting resistant pathogens, GAIN is a material factor in LOE date calculation.
16. The 180-Day First-Filer Exclusivity: How Generic Manufacturers Price the Paragraph IV Gamble
The 180-day generic drug market exclusivity, granted to the first applicant to file an ANDA with a Paragraph IV certification, is the primary economic incentive for generic manufacturers to challenge brand-name patents. During the 180-day period, the FDA cannot approve any subsequent generic ANDA for the same drug product, creating a temporary duopoly between the brand and the first generic that typically produces higher generic pricing and larger revenue capture than the fully competitive post-exclusivity market.
16a. The Financial Logic of Paragraph IV Investment
Generic manufacturers evaluate Paragraph IV challenges using expected value analysis. The costs of a Paragraph IV challenge include ANDA preparation and filing fees, attorney fees for the invalidity and non-infringement case, potential litigation costs if the brand sues and the case goes to trial, and opportunity costs of capital and development resources committed to the challenge.
The expected revenue from a successful challenge includes the revenues earned during the 180-day exclusivity period, calculated as the expected generic market price (typically 20-40% below brand during the exclusivity period, before full competition drives prices lower) multiplied by the expected volume capture. For a drug with $5 billion in annual U.S. revenues, the 180-day period at 30% market share and a 25% discount to brand pricing generates approximately $375 million in gross revenue for the first-filer.
16b. Forfeiture Provisions and Their Strategic Significance
The MMA of 2003 introduced forfeiture provisions that strip the 180-day exclusivity from a first-filer under specific circumstances. A first-filer forfeits the exclusivity if it fails to market within 75 days of a final court decision finding the patent invalid or not infringed, fails to market within 30 months of ANDA filing if the brand did not sue, or enters into an agreement with the brand company that delays commercial marketing beyond a defined trigger date.
The forfeiture provision for agreements that delay marketing is the statutory basis for the FTC’s anti-pay-for-delay enforcement program. An agreement in which the brand company pays the first-filer (in cash, patent licenses, or other valuable consideration) to delay its commercial launch beyond the forfeiture trigger triggers FTC scrutiny under the rule-of-reason standard established in FTC v. Actavis (2013). Companies that structure reverse payment settlements must now analyze whether the settlement agreement will be characterized as anticompetitive under Actavis.
Key Takeaways: Section 16
The 180-day first-filer exclusivity is the primary financial incentive for Paragraph IV patent challenges. For major drugs, the 180-day exclusivity period generates hundreds of millions to over a billion dollars in revenue for the first filer. Forfeiture provisions under the MMA can strip this exclusivity if the first-filer enters anticompetitive agreements or fails to market promptly after winning litigation. The FTC’s Actavis doctrine subjects reverse payment settlements to antitrust scrutiny.
17. Secondary Patent Strategy: From Polymorphs to Auto-Injector Device Patents
The composition-of-matter patent covering the active pharmaceutical ingredient is rarely the last patent to expire on a major commercial drug. Secondary patents covering formulations, polymorphic crystalline forms, salts, esters, prodrugs, dosage regimens, specific indications, drug delivery devices, and manufacturing processes are routinely filed after the original compound patent and, by virtue of their later filing dates, expire later.
17a. Polymorph and Salt Form Patents
Active pharmaceutical ingredients frequently exist in multiple polymorphic forms, each with potentially different physical properties including solubility, stability, hygroscopicity, and compressibility. A polymorph patent claiming a specific crystalline form of an API does not prevent generic manufacturers from using a different polymorph of the same API; it prevents them from using the specific patented form. In theory, a generic manufacturer can design around a polymorph patent by selecting a different crystalline form. In practice, the commercially optimal polymorph for a given drug’s formulation (highest bioavailability, best stability, easiest manufacturing) is often the one that has been patented, making the design-around economically or technically difficult.
Salt form patents operate on a similar principle. A drug approved as a besylate salt might have a later-expiring patent on that specific salt form. Generic manufacturers using the free acid or a different salt form must conduct their own bioequivalence studies and demonstrate that their product is therapeutically equivalent to the reference listed drug.
17b. Controlled-Release and Drug Delivery Patents
Formulation patents covering extended-release, delayed-release, or controlled-release delivery mechanisms are among the most commercially valuable secondary patents in pharmaceutical portfolios. An extended-release formulation that reduces dosing from twice daily to once daily improves patient adherence, reduces side effect frequency, and can often be priced at a premium to the immediate-release product. The formulation patent protects this commercial advantage, and a generic manufacturer wishing to market the extended-release form must either design around the patent (developing a different release mechanism that achieves similar performance) or challenge it through Paragraph IV.
Controlled-release formulation patents are also harder to design around than composition-of-matter patents for simple small molecules, because the release mechanism is a physical system rather than a defined chemical structure. A generic that achieves the same clinical bioequivalence performance through a different physical mechanism still faces the challenge of demonstrating that its release profile satisfies the FDA’s bioequivalence standard, which adds technical complexity to the generic development program.
17c. Drug-Device Combination Product Patents
Biologics administered by injection increasingly come to market as pre-filled syringes or auto-injector pen systems. The device components of these combination products can receive separate USPTO patents, listed in the Purple Book as device-related intellectual property. A biosimilar developer must either develop its own compatible delivery device (requiring human factors studies and usability testing, both time-consuming and expensive) or license the brand’s device technology.
AbbVie’s auto-injector patents for Humira, covering the specific plunger mechanism, needle shield geometry, and dosing indicator design, required every adalimumab biosimilar developer to design and validate an independent auto-injector system. This added 12 to 24 months to biosimilar development timelines and several tens of millions of dollars in human factors development and validation costs. The device patent strategy is particularly effective for combination products because the FDA’s human factors requirements for drug-device combinations are rigorous and the clinical equivalence standard for the device component is separate from the biosimilarity standard for the biologic.
17d. Method-of-Use Patents
Method-of-use patents claim specific therapeutic applications of a drug, often covering indications approved after the original NDA. A drug approved initially for condition A, with a later approval for condition B secured under a method-of-use patent filed at the time of the Phase 3 program for condition B, has patent protection for condition B that runs from the method-of-use patent’s filing date, which may be several years later than the composition-of-matter patent’s filing date.
Generic manufacturers can sometimes use ‘skinny labels,’ omitting the patented indication from their ANDA label, to market a generic version of the drug for non-patented uses while ostensibly not infringing the method-of-use patent for the patented use. Courts have grappled extensively with when skinny labeling constitutes non-infringement and when it constitutes induced infringement of the method-of-use patent through promotion of off-label use that effectively covers the patented indication.
Key Takeaways: Section 17
Secondary patents on polymorphs, salt forms, controlled-release formulations, device components, and methods of use routinely expire after the composition-of-matter patent and can define the operative LOE date. Device patents on auto-injector systems for biologics impose significant development cost and timeline burdens on biosimilar manufacturers. Method-of-use patents interact with skinny label strategies in ways that remain contested in patent litigation, making them valuable but legally uncertain components of the secondary patent portfolio.
18. Patent Thickets: Quantitative Assessment, Legislative Pushback, and the ETHIC Act
A patent thicket is a dense web of overlapping patents surrounding a single drug product, covering multiple aspects of the molecule, formulation, process, and use, such that a generic challenger must address dozens or hundreds of patents simultaneously to clear a path to market entry. The thicket strategy’s primary commercial function is to maximize the time and cost a challenger must incur before reaching the market, regardless of whether each individual patent in the thicket would withstand scrutiny.
18a. Quantitative Thicket Analysis
Academic analysis of the relationship between patent thicket density and generic entry delay has produced measurable correlations. A Yale Law & Policy Review study of top-selling branded drugs found that 91% of drugs that obtained PTEs subsequently maintained their monopolies beyond the PTE expiration date through secondary patents. EvaluatePharma and academic researchers have documented that drugs with 10 or more Orange Book-listed patents delay generic entry by an average of 3.4 years beyond the composition-of-matter expiry. Drugs with more than 20 listed patents show average delays exceeding five years.
AbbVie’s Humira thicket, with more than 250 patents and approximately 90% of them filed after the drug’s initial 2002 approval, produced a seven-year delay beyond the composition-of-matter patent expiry, translating to an estimated $60 to $80 billion in protected U.S. revenues that would have been eroded by biosimilar competition if the thicket had failed or not been built.
The financial exposure to healthcare systems from patent thickets is substantial. A study cited in Yale Law & Policy Review estimated the conservative cost of secondary patent-extended monopolies to the U.S. healthcare system at $53.6 billion. This figure represents the excess cost paid for branded drugs relative to what generic prices would have been if only composition-of-matter patents had been used to define the LOE date.
18b. The ETHIC Act and Legislative Response
The Eliminating Thickets to Increase Competition (ETHIC) Act, introduced with bipartisan support in the Senate by Senators Cornyn and Blumenthal, targets the secondary patent strategy by requiring that patents listed in the Orange Book cover ‘real innovation’ that adds value to patients rather than simply delaying competition. The Act would impose ‘reasonable limits’ on the number of patents an innovator can assert against a generic ANDA or 505(b)(2) application.
The ETHIC Act has not yet been enacted as of March 2026, but it represents a legislative trend that pharmaceutical IP teams must treat as a material risk to their secondary patent strategies. The FTC has separately pursued enforcement actions challenging certain Orange Book patent listings as improper, particularly listings of device patents in the context of drug-device combination products where the device component was arguable as not ‘claiming the drug’ within the meaning of the Hatch-Waxman listing requirements.
18c. PTAB as Thicket Pruning Mechanism
The Patent Trial and Appeal Board’s Inter Partes Review (IPR) procedure allows generic manufacturers and biosimilar developers to petition for administrative invalidation of issued pharmaceutical patents on grounds of prior art. The PTAB institution rate for pharmaceutical patent petitions has historically been 60-70%, and petitions that are instituted result in partial or full invalidation in approximately 75% of final written decisions.
IPR petitions are faster and cheaper than district court litigation for the challenger. A well-constructed IPR petition against a formulation patent or polymorph patent with weak prior art distinguishment can reach a final written decision in 12 to 18 months, compared to 3 to 5 years for district court patent litigation. For innovators, PTAB is the mechanism through which the value of secondary patent portfolios can be reduced materially between the filing date and the LOE date, and this risk must appear in IP portfolio valuations.
Investment Strategy: PTAB Discount
Every secondary pharmaceutical patent faces a non-trivial probability of IPR-based invalidation. For formulation patents and method-of-use patents with obvious claim scope, IPR institution probability approaches 70%. For these patents, a full NPV credit for the years of protection they nominally provide overstates their value. Apply a probability discount based on IPR exposure: high-novelty composition-of-matter patents warrant 80-90% probability weights; obvious formulation or polymorph patents warrant 30-50% weights. PTAB docket monitoring, including recently filed petitions and institution decisions on comparable pharmaceutical patents, is available through the USPTO’s PTAB e-discovery portal and provides real-time inputs for adjusting these probability weights.
Key Takeaways: Section 18
Patent thickets delay generic entry by measurable average durations (3.4 years for drugs with 10+ Orange Book patents; 5+ years for 20+ patents). The ETHIC Act represents a legislative threat to secondary patent strategies that IP teams must track as a scenario risk. PTAB IPR procedures impose a litigation discount on formulation, polymorph, and method-of-use patents that must appear as probability weights in IP valuation models.
19. IP Valuation Framework: Pricing Pharmaceutical Exclusivity as a Discounted Cash Flow Asset
A pharmaceutical patent portfolio is a cash flow-generating asset, and its value can be modeled using standard DCF methodology with pharmaceutical-specific probability adjustments.
19a. The Revenue-Duration Model
The foundational input for pharmaceutical patent NPV is the product of expected annual revenue and the probability-weighted duration of protection. For each year from commercial launch to the terminal LOE date, the analyst must estimate annual revenue (based on peak sales forecasts, market penetration curves, and competitive dynamics) and multiply by the probability that the drug remains protected in that year.
For years within the NCE exclusivity period or the ODE period, the probability of protection is near-100%, subject only to safety withdrawal risk. For years protected only by secondary patents facing active Paragraph IV challenges or PTAB petitions, the probability should reflect the expected outcome distribution of the patent litigation. For years beyond the last active patent but within an NCI or Pediatric Exclusivity period, the protection probability depends on whether the regulatory exclusivity is being challenged (unlikely but possible for NCI exclusivity, which can be contested on the grounds that the underlying clinical studies were not actually essential to approval).
19b. Terminal LOE Probability Distribution
Rather than modeling a single LOE date, sophisticated IP valuation models use a probability distribution over possible LOE dates. The distribution is constructed from scenario analysis: a ‘thicket holds’ scenario where secondary patents survive litigation and define the LOE date at the last secondary patent’s expiry; a ‘core patent only’ scenario where secondary patents are invalidated and LOE occurs at the composition-of-matter patent’s expiry; and an ‘at-risk entry’ scenario where a generic challenger launches before all patent challenges are resolved, imposing partial revenue erosion at an earlier date.
Each scenario is assigned a probability weight based on the patent portfolio’s characteristics. A strong composition-of-matter patent with tight secondary patents that closely track the marketed product’s formulation and no pending PTAB petitions: high weight to the ‘thicket holds’ scenario. A formulation-patent-dependent portfolio where the composition-of-matter patent has already expired and three PTAB petitions are pending against the formulation patents: high weight to the ‘core patent expired, secondary patents vulnerable’ scenario.
19c. Comparing PTE NPV to Secondary Patent NPV
For drugs still within the 60-day PTE filing window or currently in PTE prosecution, the value of the PTE in isolation can be calculated by discounting the brand revenue expected during the PTE extension period, probability-weighted for the risk that the PTE calculation is challenged (due diligence challenges by generic manufacturers) or that the extended patent is invalidated during the extension period. The expected value of a 4-year PTE on a drug generating $3 billion annually, with a 10% discount rate and an 80% probability that the PTE is not materially challenged, is approximately $7.7 billion.
The secondary patent portfolio value is calculated similarly but requires individual patent analysis. Each secondary patent’s remaining term, its probability of surviving Paragraph IV litigation, and its contribution to the operative LOE date (i.e., whether it is the last patent standing or whether its invalidation merely reveals the next secondary patent as the new last line of defense) must be assessed independently.
Key Takeaways: Section 19
Pharmaceutical patent portfolios should be valued using probability-weighted DCF models with scenario analysis across terminal LOE date distributions. NCE and ODE exclusivity years should receive near-100% probability weights; secondary patent years should receive discounted weights based on litigation vulnerability. The terminal LOE probability distribution is more analytically accurate than a single LOE point estimate.
20. The PTAB Litigation Discount: Inter Partes Review and Its Effect on Effective Term Modeling
The America Invents Act of 2011 created the Patent Trial and Appeal Board as an alternative forum for patent validity challenges. IPR petitions allow any party to challenge an issued patent on grounds of prior art (novelty and obviousness), with a 12-to-18-month proceeding timeline versus the 3-to-5-year timeline for district court patent litigation.
20a. Pharmaceutical Patent IPR Statistics
Pharmaceutical patents have been among the most frequently petitioned technology areas at the PTAB since its creation. The pharmaceutical sector accounts for 12-15% of all IPR petitions filed annually. Institution rates for pharmaceutical patent petitions have historically ranged from 60-70%. Among instituted petitions reaching final written decisions, partial or full invalidation occurs in approximately 75% of cases. These statistics imply that a pharmaceutical patent facing an IPR petition should be modeled with approximately a 45-50% probability of full or partial invalidation in a final written decision, before accounting for settlement, claim narrowing, or prosecution history estoppel effects.
Composition-of-matter patents fare better than secondary patents in IPR proceedings. The prior art landscape for a genuinely novel molecular structure is generally thinner, and obviousness arguments are harder to make for a molecule that no one had previously synthesized. Formulation patents, polymorph patents, and method-of-use patents with claim scope that can be mapped to combinations of published prior art are most vulnerable to IPR invalidity.
20b. IPR and the Orange Book Listing Strategy
Generic manufacturers increasingly file IPR petitions alongside Paragraph IV certifications as a parallel track litigation strategy. IPR is faster and cheaper than district court litigation, and an IPR final written decision invaliding a patent can trigger forfeiture of the first-filer’s 180-day exclusivity if the first-filer has not marketed within 75 days of the decision. This creates a mechanism whereby later-filed generic applicants can ‘free-ride’ on the first-filer’s IPR victory, potentially sharing the post-exclusivity market without having incurred the costs of the patent challenge.
Key Takeaways: Section 20
PTAB IPR petitions face 60-70% institution rates and result in partial or full patent invalidation in approximately 75% of instituted cases, implying a 45-50% invalidation probability per petition for pharmaceutical patents. Secondary patents are substantially more vulnerable to IPR invalidity than composition-of-matter patents. IPR institution decisions are real-time signals that should trigger immediate adjustments to the probability weights on the affected patents in LOE models.
21. The EU Supplementary Protection Certificate: Mechanics, the Bolar Exemption, and 2024 SPC Reform
The European Union’s equivalent to U.S. Patent Term Extension is the Supplementary Protection Certificate (SPC), governed by EU Regulation (EC) No. 469/2009 for medicinal products. The SPC is a sui generis intellectual property right that extends the protection of a basic patent for a medicinal product after the patent’s nominal expiry date.
21a. SPC Term Calculation
The SPC term is calculated as the period between the patent filing date and the date of the first Marketing Authorisation (MA) in the European Economic Area, minus five years. The statutory maximum SPC term is five years. If the MA was granted more than ten years after the patent filing date (a common situation for complex biologics with long development timelines), the SPC receives the full five-year maximum. If the MA was granted less than five years after the patent filing date (very uncommon in pharmaceuticals), no SPC is available. If the MA was granted between five and ten years after the patent filing date, the SPC term is the difference minus five years.
For a patent filed January 2004 with a first EU MA granted March 2017, the SPC term is 13 years and 2 months (between filing and MA) minus 5 years = 8 years and 2 months, but the 5-year statutory maximum applies, so the SPC grants 5 years. The patent plus SPC runs to March 2022 + 5 years = March 2027.
21b. The Paediatric Extension: Six Months Added to the SPC
The EU pediatric extension, granted under Regulation (EC) No. 1901/2006 when the SPC holder completes an agreed Paediatric Investigation Plan (PIP), adds six months to the SPC term, not the underlying patent. If the SPC term is at its 5-year maximum, the paediatric extension adds six months to produce a maximum SPC term of 5.5 years. The total maximum market exclusivity period protected by patent plus SPC plus paediatric extension is the original patent term (to nominal expiry) plus 5.5 years, which for a 20-year patent filed at discovery equals up to 25.5 years from filing, or approximately 13.5 to 15.5 years of post-approval market protection depending on how long development took.
21c. SPC Reform: The 2024 Unitary SPC and Manufacturing Waiver
A major reform of the EU SPC system entered into force with the creation of the unitary patent system in 2023 and the concurrent development of the Unitary SPC framework. Previously, SPCs were national rights, granted separately by each EU member state’s patent office and subject to different national validity standards and court interpretations. A single drug could hold 27 separate national SPCs with different expiry dates and validity histories.
The Unitary SPC, which came into operation with the Unified Patent Court in 2023, allows a single SPC application based on a unitary patent to provide protection across participating EU member states. This simplifies the SPC landscape substantially for innovators and reduces the administrative burden of maintaining 27 separate SPCs.
The EU also finalized a manufacturing waiver provision for SPCs, which allows generic and biosimilar manufacturers to produce patented and SPC-protected products within the EU for export to non-EU markets where the patent or SPC has expired, during the SPC term. This waiver addresses the competitive disadvantage that EU-based manufacturers faced relative to manufacturers in countries with no equivalent SPC protection, who could produce at-scale during the SPC term and enter the EU market the moment the SPC expired with an existing commercial-scale manufacturing operation.
Key Takeaways: Section 21
The EU SPC provides up to 5 years of post-patent protection, with a 6-month paediatric extension available to produce a 5.5-year maximum SPC term. The SPC calculation formula is the gap between patent filing and first EU MA, minus 5 years, subject to the 5-year maximum. The Unitary SPC simplifies the previously fragmented national SPC system across participating EU member states. The manufacturing waiver introduced in 2023-2024 allows EU-based generic and biosimilar manufacturers to produce for export markets during the SPC term.
22. Japan, Canada, South Korea, and Other Key Markets: Global Patent Extension Equivalents
22a. Japan: Re-Examination Period and Patent Term Extension
Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) administers both a patent term extension system and a re-examination period that functions as a data exclusivity analog. Japan’s patent term extension compensates for regulatory review time, with a maximum of 5 years, similar to the U.S. PTE. The calculation uses the period between the filing date of the patent and the date of pharmaceutical approval in Japan, minus a statutory baseline period.
The re-examination period provides a separate regulatory data protection mechanism. New drugs receive a re-examination period of 8 to 10 years during which the MHLW will not approve a generic application referencing the innovator’s clinical data. This functions similarly to U.S. NCE exclusivity but with a longer default duration. Japan also employs administrative patent linkage, where the MHLW considers the patent status of the reference drug before approving a generic, functioning similarly to the Orange Book/Paragraph IV system despite the absence of a formal statutory linkage.
22b. Canada: Certificate of Supplementary Protection
Canada implemented its Certificate of Supplementary Protection (CSP) system in 2017 to comply with the Comprehensive Economic and Trade Agreement (CETA) with the EU. The CSP provides up to two years of additional patent-like protection for pharmaceutical products after the patent expires, subject to the condition that the patent was filed before Health Canada’s approval of the first drug containing the active ingredient. Canada’s 2-year maximum CSP term is notably shorter than the EU’s 5-year maximum SPC, which has been a source of criticism from innovator companies seeking regulatory harmonization.
22c. South Korea: SPC-Equivalent Regulation and Patent Linkage
South Korea introduced a patent term extension system similar to the U.S. PTE in 1987, allowing extensions of up to 5 years for regulatory review time. South Korea also operates a formal patent linkage system under the Korea-U.S. Free Trade Agreement (KORUS), requiring the Korean MFDS (Ministry of Food and Drug Safety) to notify patent holders when a generic application is filed for a patented drug and prohibiting approval of the generic for up to 9 months while the patent dispute is resolved.
22d. China: Patent Linkage and Pilot SPC System
China implemented a formal patent linkage system in 2021 as part of regulatory reforms aligned with the Patent Law amendments of 2020. The linkage system requires generic applicants to certify against relevant pharmaceutical patents, and NDA holders have 45 days to initiate patent infringement proceedings to trigger a 9-month stay on the generic’s approval. China also established a pilot SPC-equivalent system in Hainan Free Trade Port and several other designated zones, though a comprehensive national SPC system similar to the EU or U.S. had not been enacted as of March 2026.
Global Patent Extension Comparison Table
| Jurisdiction | System | Maximum Extension | Key Limitation | Data Exclusivity Equivalent |
|---|---|---|---|---|
| United States | PTE (35 U.S.C. § 156) | 5 years | 14-year effective life cap; one patent per product | NCE: 5 years; ODE: 7 years; Biologics: 12 years |
| European Union | SPC (Reg. 469/2009) | 5 years (+6 months paediatric) | Based on first EU MA date | 8 years data + 2 years market = 10 years |
| Japan | Patent Term Extension | 5 years | Based on first Japan approval | Re-examination period: 8-10 years |
| Canada | CSP (2017) | 2 years | Must be filed within 30 days of approval | Data protection: 8 years |
| South Korea | Patent Term Extension | 5 years | Based on first Korea approval | Data exclusivity: 6 years (10 years for new biologics) |
| China | Pilot SPC (selective) | 5 years (pilot zones) | National system incomplete as of 2026 | Data protection: 6 years |
| Australia | Extended Innovation Patent / SPC-equivalent | 5 years | Based on first Australian approval | Data protection: 5 years (8 for biologics) |
Key Takeaways: Section 22
Japan’s 8-to-10-year re-examination period provides substantially longer regulatory data protection than U.S. NCE exclusivity. Canada’s 2-year CSP maximum is significantly shorter than the EU’s 5-year SPC maximum. China’s patent linkage system, enacted in 2021, creates a formal analog to the U.S. Hatch-Waxman linkage framework but with a 9-month stay rather than 30 months. A complete global LOE model requires jurisdiction-specific inputs for each of these systems, because the operative terminal protection date in each market may differ by multiple years from the U.S. LOE date.
23. Effective Market Exclusivity: Building the LOE Date Calendar
The LOE date is not a single number that can be looked up. It is the output of a structured analytical process that integrates patent expiry data, PTE status, PTA adjustments, regulatory exclusivity expiry, pending Paragraph IV litigation, active PTAB IPR petitions, and settlement agreement terms.
23a. The Seven-Step LOE Date Construction Process
The process for constructing a defensible LOE date for a given drug product involves the following analytical steps.
First, identify all patents listed in the Orange Book (for small molecules) or Purple Book (for biologics) for the reference listed drug. Extract each patent’s nominal expiry date, PTE status (including the PTE grant amount), and any PTA listed on the face of the patent. Calculate the PTA-adjusted nominal expiry for each patent and then add any PTE to produce the final extended expiry date.
Second, identify all FDA regulatory exclusivities applicable to the NDA or BLA, including NCE, ODE, NCI, PED, and GAIN exclusivities. Determine the expiry date of each. For drugs with multiple NDA supplements, each supplement may carry its own NCI exclusivity with a distinct expiry date.
Third, determine the operative protection date for each year from commercial launch by identifying whether a patent, regulatory exclusivity, or combination is still active in that year. The terminal year in which any protection remains is the provisional LOE date.
Fourth, identify all pending Paragraph IV certifications and their litigation status. For each challenged patent, apply a probability weight based on the strength of the patent’s claims relative to the prior art, the history of PTAB petitions against the same patent, and the track record of the asserting innovator in analogous litigation.
Fifth, identify all pending PTAB IPR petitions against listed patents. Apply institution probability and invalidation probability to each petition. Adjust the probability weight on the affected patents accordingly.
Sixth, examine publicly available settlement agreements (disclosed in SEC filings and litigation dockets) between the innovator and generic or biosimilar challengers. Settlements with authorized entry dates earlier than the nominal patent expiry define the effective LOE date for that market entry, regardless of the patent’s formal expiry date.
Seventh, construct a probability-weighted LOE date distribution using the scenario framework (thicket holds, core patent only, at-risk entry) and calculate the expected LOE date as the probability-weighted mean of the distribution.
Key Takeaways: Section 23
Accurate LOE date construction requires integrating seven distinct data categories: Orange Book / Purple Book patent listings with PTA and PTE adjustments, FDA regulatory exclusivity expiry dates, Paragraph IV litigation status and probability weights, PTAB IPR petition status, settlement agreement terms, authorized generic agreements, and scenario probability weights. Single-number LOE date estimates are inferior to probability distributions for financial modeling purposes.
24. Investment Strategy: Patent Duration Data as Alpha Signal
Patent expiry and exclusivity data is systematically under-analyzed in pharmaceutical equity research. The data is public, filed with the USPTO and FDA, and accessible through the Orange Book, Purple Book, USPTO patent database, and PTAB docket portal. Yet most buy-side LOE models use simplified single-date estimates that ignore PTA, treat all secondary patents as equally survivable, and fail to adjust for pending PTAB petitions.
24a. Identifying PTA-Based LOE Underestimates
PTA adjustments on pharmaceutical patents of 12 to 36 months are common for applications with complex prosecution histories. A 24-month PTA adjustment on the last operative patent protecting a $5 billion drug represents approximately $2.5 billion in protected revenue that will not appear in LOE models ignoring PTA. Checking every commercially relevant pharmaceutical patent’s PTA balance through the USPTO Patent Center database and incorporating those adjustments into LOE models produces measurably more accurate estimates.
24b. PTAB Petition Monitoring as Bearish Signal
An IPR petition filed against a secondary formulation patent on a major drug is a public event, visible on the PTAB docket within days of filing. If that patent is the last operative protection for the drug (i.e., the composition-of-matter patent has already expired or will soon), an IPR petition with strong prior art is a material negative signal for the branded drug’s revenue duration. Systematic monitoring of PTAB petitions cross-referenced with Orange Book listings allows analysts to identify when a drug’s secondary patent thicket is under credible administrative attack.
24c. Multi-PTE Approval Monitoring as Bullish Signal
Simultaneous or near-simultaneous NDA approvals for an active moiety across monotherapy and fixed-dose combination products is a public FDA event that signals potential multi-PTE execution. Tracking FDA approval databases for these simultaneous approvals, cross-referencing them with PTE applications filed at the USPTO within 60 days, and identifying the patents selected for extension allows analysts to update their LOE date models in real time as the multi-PTE strategy unfolds.
24d. LOE Model Sensitivity to Exclusivity Type
In financial models for drugs within ODE protection, reducing the probability weight on the ODE period meaningfully increases the probability of early revenue erosion. For drugs in NCE exclusivity, the four-year certainty period should produce minimal revenue uncertainty from patent challenges. For drugs with only secondary patent protection remaining, the probability distribution of LOE dates should be wider and more sensitive to litigation outcomes. Analysts should calibrate their confidence intervals on projected revenues according to the type of protection in force, not just the duration remaining.
Key Takeaways: Section 24
PTA-adjusted LOE date modeling captures protected revenue that simplified models miss. PTAB petition monitoring against operative secondary patents is a real-time bearish signal for branded drug revenue duration. Multi-PTE approval calendar monitoring is a real-time bullish signal. The confidence interval on projected branded revenues should scale with the type of protection in force: narrow for ODE years, moderate for NCE years, wide for secondary-patent-only years.
25. The $236 Billion Patent Cliff: Drugs at Risk, 2025-2030
GlobalData estimates that approximately $236 billion in annual branded pharmaceutical sales face LOE in the U.S. market between 2025 and 2030. Approximately 70 products are classified as blockbusters in this window. The aggregate figure is the largest patent cliff in pharmaceutical history by absolute revenue.
25a. The Keytruda Problem: $25 Billion at Risk Starting 2028
Merck’s pembrolizumab (Keytruda) generated approximately $25 billion in global revenue in 2024 and is the world’s best-selling drug. Its core U.S. composition-of-matter patents begin their expiry window in 2028. Biosimilar applicants have already initiated development programs. The biosimilar development challenge for a PD-1 monoclonal antibody with 16 tumor types and more than 30 distinct indications involves characterizing a large, complex antibody against a reference product with well-documented structural complexity, conducting comparative clinical pharmacokinetic studies, and navigating a method-of-use patent landscape covering individual tumor types that may sustain independent 3-year NCI exclusivity periods beyond the composition-of-matter expiry.
Merck’s secondary patent strategy for Keytruda includes formulation patents covering its 25 mg/mL concentration, patents covering specific dosing regimens (flat dosing at 200 mg every three weeks and 400 mg every six weeks), and method-of-use patents for combination therapy with chemotherapy in first-line non-small cell lung cancer. Each of these creates an independent patent layer that biosimilar developers must analyze and either invalidate or design around.
25b. Eliquis (Apixaban): Small-Molecule Cliff with Multiple Generic Challengers
BMS and Pfizer’s apixaban (Eliquis) generated approximately $12 billion in U.S. revenues in 2024. Its core U.S. patents are expected to expire in 2026 to 2028, with the exact operative LOE date depending on the outcome of ongoing Paragraph IV litigation and the terms of any settlement agreements. The FTC has maintained active scrutiny of Eliquis settlement negotiations, and any reverse payment settlements in this space face Actavis antitrust analysis.
As a small molecule, apixaban faces a potentially rapid and steep post-LOE price decline. Multiple ANDA filers have already submitted applications. If the FDA approves several ANDAs simultaneously at the operative LOE date, the day-one competitive structure will resemble Plavix’s 2012 LOE more than Lipitor’s more graduated erosion.
25c. Stelara (Ustekinumab): Active Biosimilar Competition Since 2024
Johnson & Johnson’s ustekinumab (Stelara) entered the active phase of its LOE in early 2024 with U.S. biosimilar approvals and commercial launches. Stelara generated approximately $10 billion at peak. With a thinner patent thicket than Humira and fewer biosimilar settlement agreements restricting early launch, the early Stelara biosimilar experience is generating the second major data set on biologic LOE market dynamics in U.S. immunology. Preliminary market data from 2024 and early 2025 indicates faster biosimilar penetration than Humira’s first year, though PBM formulary dynamics continue to moderate the speed of transition.
25d. Drugs at Risk: 2025-2030 Reference Table
| Drug | Active Ingredient | Approx. Peak Annual Revenue | Expected U.S. LOE Window | Molecule Type |
|---|---|---|---|---|
| Keytruda | Pembrolizumab | ~$25B | 2028-2029 | Biologic (PD-1 mAb) |
| Eliquis | Apixaban | ~$12B (U.S.) | 2026-2028 | Small molecule |
| Stelara | Ustekinumab | ~$10B | 2024 (active) | Biologic (IL-12/23 mAb) |
| Ozempic/Wegovy | Semaglutide | ~$21B+ | 2031+ (contested) | Small molecule / GLP-1 peptide |
| Xarelto | Rivaroxaban | ~$5B (U.S.) | 2024 (active) | Small molecule |
| Revlimid | Lenalidomide | ~$7B | 2022-2026 (graduating) | Small molecule |
| Dupixent | Dupilumab | ~$13B | 2033+ (estimated) | Biologic (IL-4/IL-13 mAb) |
| Skyrizi | Risankizumab | ~$8B+ | ~2035+ (estimated) | Biologic (IL-23 mAb) |
| Darzalex | Daratumumab | ~$9B | ~2030-2032 (estimated) | Biologic (CD38 mAb) |
| Opdivo | Nivolumab | ~$9B | ~2028-2031 (estimated) | Biologic (PD-1 mAb) |
Revenue figures approximate, based on reported 2023-2024 financials. LOE windows are estimates based on published Orange Book/Purple Book data and litigation status as of March 2026; actual LOE dates depend on ongoing patent litigation outcomes and settlement agreements.
Key Takeaways: Section 25
The 2025-2030 patent cliff is the largest in pharmaceutical history at $236 billion in at-risk U.S. branded revenues. Keytruda’s impending LOE represents the largest single drug revenue exposure. Apixaban’s small-molecule LOE will follow the steep, rapid erosion pattern of previous oral anticoagulants and antiplatelet agents. Stelara’s active biosimilar competition is producing the second major real-world data point on biologic LOE dynamics in U.S. immunology after Humira.
26. Master Key Takeaways and Practitioner Checklist
The 20-year nominal patent term is analytically misleading. Effective patent life for a New Chemical Entity averages 7 to 10 years of commercial exclusivity after FDA approval, because development consumes 10 to 13 years of the nominal term. Revenue models should use effective patent life, not nominal term.
PTA and PTE are separate, additive mechanisms. PTA compensates for USPTO prosecution delays (no maximum duration). PTE compensates for FDA regulatory review time (5-year maximum, 14-year effective life cap). PTE is applied to the PTA-adjusted expiry date, compounding the two mechanisms. Both must be tracked independently and combined sequentially for accurate LOE date calculation.
The 60-day PTE filing deadline is absolute. Missing it forfeits the extension entirely. For a major drug, this is a multi-billion-dollar administrative failure.
Multi-PTE strategies are legally executable for drugs approved in multiple NDAs. The one-patent rule restricts PTEs to one per ‘drug product,’ not one per active moiety. Same-day or near-simultaneous NDA approvals for fixed-dose combinations containing the same active ingredient can support separate PTE applications. The Nesina and Lyrica cases are the documented templates.
Regulatory exclusivities are independent of patent protection. ODE cannot be shortened by Paragraph IV challenges. NCE exclusivity provides four years of full certainty before the NCE-1 litigation trigger. Pediatric Exclusivity extends all protections simultaneously at a 100:1+ ROI on study costs.
Patent thickets have quantifiable delay effects. Drugs with 10+ Orange Book patents delay generic entry by an average of 3.4 years beyond composition-of-matter expiry. The ETHIC Act represents a legislative risk to secondary patent strategies that must be modeled as a scenario.
PTAB IPR petitions impose probability discounts on secondary patents. The 45-50% composite invalidation probability per petition requires explicit probability weighting of secondary patent years in NPV models. Formulation and polymorph patents are most vulnerable.
The EU SPC provides up to 5.5 years of post-patent protection. The paediatric extension adds 6 months to the SPC maximum. The Unitary SPC simplifies the previously fragmented national SPC system. The manufacturing waiver changes the competitive landscape for generic and biosimilar manufacturers in the EU.
Global LOE dates differ by jurisdiction. Japan’s re-examination period (8-10 years), Canada’s 2-year CSP, South Korea’s 5-year extension and 9-month patent linkage stay, and China’s nascent linkage system all produce jurisdiction-specific LOE dates that must be modeled independently.
LOE date construction is a seven-step analytical process. It requires integrating Orange Book / Purple Book listings with PTA and PTE, FDA regulatory exclusivity data, Paragraph IV litigation status, PTAB petition outcomes, settlement agreement terms, and scenario probability weights. Single-point LOE estimates are inadequate for financial modeling.
Practitioner LOE Date Checklist
- [ ] Identify all Orange Book / Purple Book patent listings for the RLD. Extract nominal expiry, PTA balance (from patent face), and any PTE grant amount from the USPTO PTE database.
- [ ] Calculate PTA-adjusted expiry for each patent. Add PTE to PTA-adjusted date to get final extended expiry.
- [ ] Identify all applicable FDA regulatory exclusivities (NCE, ODE, NCI, PED, GAIN) and their expiry dates. Flag any indication-specific ODE periods.
- [ ] Check for pending Paragraph IV certifications in the FDA’s ANDA database. For each challenged patent, obtain the litigation docket, assess the prior art vulnerability, and assign a survival probability.
- [ ] Check the PTAB docket for all IPR petitions filed against listed patents. Flag instituted petitions and apply composite invalidation probability.
- [ ] Review SEC 10-K, 10-Q, and 8-K filings for disclosure of patent settlement agreements with generic or biosimilar applicants that define authorized commercial entry dates.
- [ ] Check FDA approval calendar for simultaneous NDA approvals involving the same active moiety (signal for multi-PTE strategy). Verify PTE applications at the USPTO within 60 days of those approvals.
- [ ] Check for any multi-PTE grants already on record in the Orange Book.
- [ ] Construct terminal LOE probability distribution using three scenarios: thicket holds, core patent only, at-risk launch. Assign probability weights. Calculate expected LOE date.
- [ ] For global assets, construct jurisdiction-specific LOE dates for EU (SPC + paediatric extension), Japan (patent extension + re-examination period), Canada (CSP), South Korea (extension + linkage stay), and China (linkage stay) separately.
This analysis is written for pharmaceutical IP counsel, R&D finance and strategy teams, business development professionals, and institutional investors in biopharmaceutical equities. It reflects publicly available statutes, regulations, and patent data. Patent expiry dates, PTE grants, PTA balances, and regulatory exclusivity periods are subject to change based on ongoing USPTO proceedings, FDA administrative actions, and court decisions. Nothing in this analysis constitutes legal advice, and LOE date determinations for specific products should be confirmed with qualified patent counsel with access to the complete prosecution history and litigation record for each relevant patent.