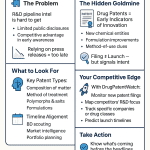
1. Why Patent Database Literacy Is a Competitive Asset
Patent information does not merely describe a drug’s legal status. It prices the asset. For every branded pharmaceutical product, the gap between current revenue and the net present value of that revenue stream narrows as patent expiration dates approach and exclusivity periods erode. Generic manufacturers build entire pipeline strategies around the dates published in the FDA Orange Book. Biosimilar developers track Purple Book exclusivity expiration windows years before filing their abbreviated Biologics License Applications (aBLAs). Institutional investors with long or short positions in pharma equities need the same data, read the same way.
The problem is that most practitioners treat the Orange Book and Purple Book as lookup tools rather than intelligence assets. They search a brand name, note an expiration date, and move on. That is a category error. These two databases, used together with Paragraph IV litigation dockets, the FDA’s exclusivity tables, and external patent prosecution records, generate a full picture of when a drug’s IP moat breaks, how deep it was to begin with, and which generic or biosimilar filer is positioned to cross first.
This guide covers every layer of that picture: the statutory frameworks, the mechanics of patent listing and exclusivity grants, the strategic use of patent use codes and biosimilar interchangeability designations, IP valuation implications, and a complete technology roadmap for evergreening and biologic lifecycle management.
2. The Legislative Architecture Behind the Books
The Hatch-Waxman Act (1984) and the Orange Book
The Drug Price Competition and Patent Term Restoration Act of 1984, universally called the Hatch-Waxman Act, created the modern generic drug approval pathway and made the Orange Book what it is today. Before 1984, generic manufacturers had to conduct their own full clinical trials to secure approval, a cost burden that effectively barred market entry for most compounds. Hatch-Waxman introduced the Abbreviated New Drug Application (ANDA) process, which allows a generic applicant to demonstrate bioequivalence to a previously approved reference listed drug (RLD) rather than repeating safety and efficacy studies. In exchange for this streamlined approval mechanism, the law established a structured system for patent notification and litigation, with the Orange Book as the registry at its center.
Section 505(b)(1) NDAs and certain 505(b)(2) applications submitted by brand manufacturers are required to disclose all patents that claim the approved drug substance, drug product, or approved method of use. The FDA publishes those disclosures in the Orange Book, and ANDA applicants must then certify their position on each listed patent through one of four certifications: Paragraph I (patent not applicable), Paragraph II (patent expired), Paragraph III (applicant will wait for patent expiration), or Paragraph IV (patent is invalid or will not be infringed by the generic). A Paragraph IV certification triggers the litigation clock, a 45-day window in which the NDA holder can file a patent infringement suit and automatically receive a 30-month stay on FDA final approval of the ANDA.
The BPCIA (2010) and the Purple Book
The Biologics Price Competition and Innovation Act, enacted as Title VII of the Affordable Care Act in 2010, did for biologics what Hatch-Waxman did for small molecules, though with considerably more complexity. It created the 351(k) abbreviated pathway for biosimilar and interchangeable applications and established the 12-year reference product exclusivity period that anchors Purple Book listings. Unlike the streamlined Paragraph IV process, the BPCIA established the ‘patent dance,’ a multi-step, sequenced exchange of product and manufacturing information between the biosimilar applicant and the reference product sponsor, with specific timelines governing each step.
Recent Amendments: Orange Book Transparency Act and Purple Book Continuity Act
The Orange Book Transparency Act of 2020, enacted as part of the Consolidated Appropriations Act of 2021, strengthened requirements for NDA holders to delist patents that courts have found invalid or not infringed, reducing the ability of brand companies to use stale listings as a litigation delay mechanism. The Purple Book Continuity Act of 2019 (also enacted in 2021) mandated that reference product sponsors submit to the FDA any patent list exchanged during the patent dance, and required the FDA to publish those lists in the searchable Purple Book database within 30 days of receipt. Both acts increased transparency, though both also left gaps that experienced litigants continue to exploit.
3. Decoding the FDA Orange Book: Patents, Use Codes, and IP Valuation
Patent Listing Requirements and Submission Mechanics
The NDA holder’s patent submission obligation is triggered at two discrete points. First, at the time of NDA submission, Form FDA 3542a must accompany any patent that claims the drug substance, drug product, or an approved method of use. Second, within 30 days of NDA approval, those same patents must be resubmitted on Form FDA 3542, and any patent issued after approval must be listed within 30 days of issuance.
The FDA’s role in this process is ministerial. The agency does not adjudicate whether a patent is properly listed, does not evaluate claim scope, and does not independently verify expiration dates. It publishes what NDA holders submit. That is why the Orange Book contains errors, intentional and otherwise, and why a sophisticated generic developer never relies on Orange Book listings as a substitute for independent freedom-to-operate analysis.
Certain patents are categorically ineligible for Orange Book listing: process patents covering manufacturing, packaging patents, patents claiming metabolites, synthesis intermediates, and methods of use that have not been approved by the FDA. Polymorph patents occupy a nuanced middle ground. A polymorph patent is listable if the NDA holder provides certification with supporting analytical data demonstrating that the patented polymorph is present in and performs identically to the approved drug product. That data burden is non-trivial, but companies with mature IP strategy teams have used it to list additional patents and extend the Orange Book’s effective blocking power by several years.
Supplemental NDAs (sNDAs) that add a new dosage form, new route of administration, new strength, or an Rx-to-OTC switch also carry listing obligations for patents claiming those new features. This is where sophisticated life-cycle management begins: each approved change to the product can pull in new Orange Book-listed patents with fresh expiration dates.
Patent Use Codes: The Skinny-Label Playbook
Patent use codes are alphanumeric identifiers, formatted as ‘U-‘ followed by a number, that describe the specific FDA-approved method of use covered by a method-of-use patent. Each code corresponds to a description published in the FDA’s Patent Use Code database, accessible through the Orange Book interface.
For generic developers, patent use codes are the primary tool for executing a ‘skinny label’ strategy (technically, a section viii carve-out under 21 CFR 314.94(a)(12)(iii)). If a drug has three approved indications and only one is covered by a listed method-of-use patent, a generic applicant can seek approval only for the two non-patented indications by filing a section viii statement, carving out the patented use from the proposed labeling. If the FDA accepts the carve-out, the generic can launch before the method-of-use patent expires, generating revenue on the unpatented indications while the brand retains market protection for the patented one.
The risks of skinny-label launches are real and have increased. GlaxoSmithKline v. Teva Pharmaceuticals USA (Fed. Cir. 2021) held that a generic manufacturer could face induced infringement liability even when its label carved out a patented indication, if prescribing patterns and promotional conduct effectively directed physicians toward the patented use. That ruling produced a significant recalibration of skinny-label risk analysis. Generic companies now evaluate not just label text but the real-world prescribing environment, market research data on indication mix, and the brand company’s likely litigation posture before committing to a carve-out launch.
The strategic implication for brand companies is the mirror image: carefully drafted method-of-use patent claims, combined with patents timed to cover the highest-revenue indication, can force any skinny-label generic into the lowest-value segment of the market even when the drug substance patent has expired.
Evergreening Tactics and the Orange Book as an IP Roadmap
Evergreening is the practice of layering new patents onto an approved drug product to extend the effective period of market exclusivity beyond the original composition-of-matter (COM) patent. The Orange Book is both the tool that enables evergreening and the public record that documents it. A trained analyst reading a drug’s Orange Book entry can reconstruct the brand company’s entire lifecycle strategy.
The standard evergreening toolkit includes:
Formulation patents cover specific dosage forms, controlled-release mechanisms, co-crystal forms, or delivery systems. Purdue Pharma’s suite of OxyContin formulation patents is the canonical case. After the original oxycodone COM patent expired, Purdue listed formulation patents covering the abuse-deterrent matrix that extended Orange Book protection and created a new barrier to generic entry requiring bioequivalence to the reformulated product, not the original.
Method-of-use patents cover new indications, patient subpopulations, or dosing regimens for existing molecules. AstraZeneca’s esomeprazole (Nexium) lifecycle illustrates this: after omeprazole generics flooded the market, AstraZeneca launched the S-enantiomer with new method-of-use patents covering specific GERD indications, buying several additional years of premium pricing.
Combination product patents cover fixed-dose combinations of a branded molecule with another agent, often a generic, creating a new Orange Book entry with fresh exclusivities and patents. Pfizer’s Eliquis co-development with Bristol-Myers Squibb demonstrates the commercial logic: a combination patent portfolio covering apixaban’s specific dosing regimens added years of IP runway beyond the base compound’s expiration.
Pediatric extension patents are not patents at all but represent the most reliable way to add 6 months to every existing Orange Book-listed patent and exclusivity simultaneously. If the FDA issues a Written Request for pediatric studies and the NDA holder completes them, all listed patents and exclusivities extend by 6 months, regardless of whether the pediatric results support the underlying claims.
Polymorph and salt form patents require more analytical work to list (the NDA holder must submit performance data), but once listed, they present a legitimate barrier to ANDA filers who must either challenge the patent via Paragraph IV or demonstrate that their generic uses a different polymorph or salt that does not infringe.
Enantiomer patents cover the pharmacologically active stereoisomer of a racemate that is going off-patent. The Forest Laboratories/citalopram-to-escitalopram transition (Celexa to Lexapro) is the defining example. The racemic citalopram patent was expiring; Forest obtained patents on the S-enantiomer escitalopram, launched Lexapro, and aggressively migrated patients before generics could fill the citalopram market. The result was a years-long revenue bridge funded entirely by enantiomer IP.
IP Valuation: How Orange Book Patents Are Priced Into Drug Assets
For pharma IP teams and investors conducting discounted cash flow (DCF) analysis on a drug asset, Orange Book data is the primary input for modeling the revenue cliff. The key variables are patent expiration dates (by type), regulatory exclusivity expiration, the number and quality of Paragraph IV filers, litigation outcome probabilities, and the timing of 180-day exclusivity periods for the first generic filer.
A basic framework for IP-adjusted revenue modeling proceeds as follows. Start with the latest Orange Book patent expiration date across all listed patents and stack any applicable exclusivities on top. That gives you the outer bound of full market exclusivity, assuming no successful Paragraph IV challenge. Then assess Paragraph IV risk by identifying current ANDA filers from the FDA’s ANDA pending list and their Paragraph IV certification dates. The 30-month stay means that even a validly filed Paragraph IV challenge cannot produce a final FDA approval until the stay expires, giving brand companies a predictable litigation runway. If litigation resolves in the brand’s favor, the stay extended the protected period. If the generic wins, the entry date collapses to the date of approval plus any remaining 180-day exclusivity held by the first filer.
The 180-day first-filer exclusivity is itself a valuation input for generic companies. A drug with a strong Paragraph IV case, a large branded market, and no other authorized generics can be worth hundreds of millions of dollars in exclusivity period revenue, essentially a temporary monopoly on the generic supply. Mylan’s launch of a generic version of Pfizer’s Lipitor (atorvastatin) in 2011 generated an estimated $600 million in revenue during its 180-day exclusivity window, a figure that reflects the commercial ceiling of first-to-file strategy executed correctly.
For analysts building sum-of-the-parts models, each Orange Book entry on a pipeline asset contributes separately to the NPV stack. A new formulation patent that extends exclusivity by 3 years on a drug generating $2 billion annually is worth roughly $4-6 billion in present value, depending on the discount rate and the probability that the patent survives a challenge. That calculation drives M&A premiums: acquirers price in the patent portfolio, not just the molecule.
Regulatory Exclusivities: Full Technical Breakdown
Regulatory exclusivities operate independently from patents. A drug with all patents expired but an active exclusivity period still blocks ANDA or 505(b)(2) approval. Conversely, a patent can be challenged and invalidated while exclusivity continues uninterrupted. The interplay between the two systems is where lifecycle management strategy gets sophisticated.
Five-Year New Chemical Entity (NCE) Exclusivity
Granted to drugs containing an active moiety never before approved by the FDA under any NDA. The 5-year period runs from the date of NDA approval and generally prohibits submission of an ANDA or 505(b)(2) application. There is one exception: a Paragraph IV ANDA can be submitted one year early, at the 4-year mark, but cannot receive final approval before the NCE period expires unless the patent is found invalid or not infringed before that date. NCE exclusivity is the most commercially significant exclusivity type for truly novel compounds because it covers the entire first commercial period without needing any Orange Book patent to be valid.
Three-Year New Clinical Study Exclusivity
Unlike NCE exclusivity, this 3-year period does not block ANDA submission. It blocks final ANDA approval for the specific condition of approval supported by the new clinical studies. This means a generic company can submit its ANDA during the 3-year period, build its technical package, navigate the Orange Book patents, and be positioned for immediate launch the day the exclusivity expires. Brand companies use this exclusivity primarily for lifecycle extensions: a new dosage form, a new route of administration, or an expanded indication can each earn a 3-year period, provided the approval relied on new clinical investigations (not just bioavailability studies) conducted or sponsored by the NDA holder.
Seven-Year Orphan Drug Exclusivity (ODE)
Drugs designated as orphan drugs by the FDA’s Office of Orphan Products Development and approved for the orphan indication earn 7 years of exclusivity that blocks any same-drug approval for the same orphan indication. The ‘same drug’ standard is compound-specific and has been the subject of significant litigation. ODE can coexist with NCE exclusivity; the longer of the two periods controls. Drugs targeting rare diseases with limited generic development incentive often rely more on ODE than on Orange Book patents because the patient population is too small to attract a Paragraph IV filer willing to absorb litigation costs.
Six-Month Pediatric Exclusivity
Awarded when an NDA holder completes pediatric studies in response to a Written Request from the FDA under 21 CFR 314.55 or the Pediatric Research Equity Act. This exclusivity attaches to and extends all existing Orange Book patents and exclusivities by 6 months, rather than being a standalone exclusivity period. For a blockbuster drug with multiple listed patents, a single pediatric study can be worth a billion dollars in extended revenue with relatively low incremental investment, making Written Requests one of the highest-ROI lifecycle management tools available.
GAIN Act Exclusivity
Under the Generating Antibiotic Incentives Now Act (GAIN, enacted as part of FDASIA 2012), antibiotics and antifungals that receive Qualified Infectious Disease Product (QIDP) designation earn 5 additional years added to any applicable NCE, ODE, or 3-year new clinical study exclusivity. A novel antibiotic that would otherwise receive 5 years of NCE exclusivity gets 10 under GAIN. Given the commercial challenges of antibiotic development, GAIN exclusivity is often the primary financial justification for investment.
180-Day First-Filer Exclusivity
The first ANDA applicant to submit a substantially complete application with a Paragraph IV certification earns 180 days of generic market exclusivity after the first commercial marketing of the generic, or the court’s decision that the Orange Book patent is invalid or not infringed, whichever comes first. ‘Substantially complete’ is a term with technical meaning: the ANDA must be ready for substantive review, not merely a shell filing. Multiple ANDA filers can share 180-day exclusivity if they are all first filers (filed on the same day with the same Paragraph IV certifications), a scenario that has become common for high-value drugs where multiple generic companies race to file simultaneously.
180-Day Competitive Generic Therapy (CGT) Exclusivity
CGT exclusivity, created by the FDA Reauthorization Act of 2017, provides 180 days of generic exclusivity for drug products with inadequate generic competition (typically, three or fewer approved generics). It is designed to incentivize generic entry into markets that have not attracted sufficient competition, addressing the phenomenon of single-source generic price spikes. Unlike the Paragraph IV first-filer exclusivity, CGT does not require a patent challenge; it attaches to the first ANDA approved for a CGT-designated drug.
Investment Strategy: Reading the Orange Book Like a Sell-Side Analyst
The most actionable Orange Book intelligence for institutional investors is not the patent expiration date in isolation; it is the gap between the last listed patent expiration and the last listed exclusivity expiration, combined with the Paragraph IV litigation timeline. That gap, measured in months, represents the zone of maximum revenue risk for brand holders and maximum opportunity for generic entrants.
For a long position in a branded drug company, the critical questions are: How many active Paragraph IV certifications exist against the most commercially significant patent? What is the probability of surviving litigation given the claim scope and prosecution history? Is there a 30-month stay currently in effect, and what is its expiration date? Are there any authorized generic agreements that would cushion the revenue decline even if a generic wins? Is there a lifecycle extension product with its own exclusivity and patent stack that could absorb revenue migrating off the original formulation?
For a short position or a competitive intelligence exercise, the questions invert: Is the latest patent expiration the same as the compound’s only real defensive layer, or are there multiple overlapping exclusivities? Are there multiple ANDA filers, reducing the likelihood of the first generic being able to sustain a premium? Has the brand company been filing new patents with expiration dates 3 to 5 years out, indicating an active evergreening program, or is the IP wall stagnant?
The Orange Book’s downloadable data files allow for systematic screening across the entire portfolio of branded drugs. Analysts who build custom databases from these files can identify all drugs whose last patent or exclusivity expires within a rolling 24-month window, cross-reference those against ANDA filing activity, and generate a ranked list of imminent patent cliff exposures with revenue estimates from public IMS/IQVIA data.
Key Takeaways: Orange Book
The Orange Book is both a legal disclosure mechanism and a strategic intelligence source. The FDA’s ministerial role in listing means that brand companies control the database, and NDA holders have strong incentives to list early and broadly. Paragraph IV certifications and the 30-month stay are the primary litigation levers. Patent use codes and the section viii carve-out are the primary entry levers for generics on method-of-use patents. The interaction between Orange Book patents and regulatory exclusivities creates a stacked protection system that the most sophisticated lifecycle management programs exploit through formulation patents, new indication exclusivities, and pediatric study timing. No Orange Book analysis is complete without checking the active Paragraph IV litigation docket maintained by the FDA, as the litigation outcome is the variable that most frequently forces revision of revenue models.
4. Decoding the FDA Purple Book: Biologics, Reference Product Exclusivity, and the Patent Dance
Scope, Access, and the BPCIA Framework
The Purple Book lists all biological products licensed under the Public Health Service (PHS) Act, whether regulated by the Center for Drug Evaluation and Research (CDER) or the Center for Biologics Evaluation and Research (CBER). Its primary informational outputs are the date of first licensure for each reference biological product (which anchors the 12-year exclusivity calculation), the reference product exclusivity expiration date, the biosimilarity and interchangeability status of licensed biosimilars, and, since 2021, the patent lists exchanged during the BPCIA patent dance.
The Purple Book is an online-only database, updated monthly. There is no print edition. Monthly reports containing all database records are available for download, which is the format preferred for systematic analysis. The database’s search functionality allows filtering by applicant, BLA number, proprietary name, proper name, and biosimilar-to-reference relationship.
Reference Product Exclusivity: The 12-Year Clock and Its Exceptions
Under BPCIA Section 351(k)(7), a reference biological product receives 12 years of exclusivity from the date of its first licensure. During this period, no aBLA for a biosimilar or interchangeable product referencing that biologic can receive effective FDA approval. The 4-year mark is also significant: no aBLA can even be submitted until 4 years have elapsed from first licensure of the reference product.
The ‘first licensure’ date is not always obvious. If a biological product was previously licensed under a different regulatory pathway (e.g., as a biologics license application converted from an NDA during the transition of protein products to biologics regulation), the first licensure date may significantly predate the current BLA holder’s commercial launch. That discrepancy matters: a biosimilar applicant tracking only the current BLA approval date may miscalculate the exclusivity expiration by years.
Pediatric exclusivity adds 6 months to the 12-year exclusivity clock when the reference product sponsor completes pediatric studies in response to a Written Request, extending it to 12.5 years. Orphan drug exclusivity does not attach to reference product exclusivity in the Purple Book (in contrast to the Orange Book system), which is a critical asymmetry that biosimilar developers must account for when planning aBLA timing.
The practical effect of 12-year exclusivity on biosimilar development economics is substantial. A biosimilar program typically requires $100-300 million in development costs over 7-9 years. That means developers must initiate programs well before exclusivity expiration to be ready for launch, and they must finance those programs against the risk that the reference product sponsor extends exclusivity through lifecycle management, litigation, or new indication approvals that restart certain protections. The biosimilar programs with the highest NPV are those targeting reference products where 12-year exclusivity is the only meaningful barrier, where the market is large, and where manufacturing complexity is manageable.
The Patent Dance: Mechanics, Obligations, and Strategic Implications
The BPCIA patent dance is a sequential information exchange governed by strict timelines. When a biosimilar applicant submits its aBLA, it must provide the reference product sponsor with a copy of the application and manufacturing information within 20 days of FDA acceptance. This disclosure is confidential and is the biosimilar’s most sensitive trade secret: the sponsor now knows how the biosimilar is made.
Within 60 days of receiving the aBLA disclosure, the reference product sponsor must provide a list of patents it believes could be asserted if the biosimilar were marketed. The biosimilar applicant then has 60 days to provide its positions on non-infringement or invalidity of each listed patent, along with any additional patents it believes the sponsor should have included. The sponsor then has 60 days to respond. The final output of this exchange is a ‘list of patents for immediate litigation,’ which governs which patents can be litigated in the first wave of BPCIA litigation. Subsequent patents can be litigated later but only in specified circumstances.
The dance is optional in a technical but practically limited sense. The Supreme Court’s Amgen v. Sandoz decision (2017) held that the aBLA disclosure requirement is not enforceable by injunction but that reference product sponsors can file a declaratory judgment action on non-dance patents if the applicant declines to participate. In practice, most biosimilar applicants participate in the dance because declining exposes them to a broader set of potential patent challenges without the structured limitation the dance provides.
The strategic calculus for reference product sponsors is to list as many patents as possible in the 60-day window, knowing that the FDA’s role is ministerial and that listing does not require the sponsor to defend the patent’s validity or enforceability. That incentive structure has produced Purple Book listings that include patents of wildly variable quality, from core biologic composition claims to peripheral formulation and manufacturing patents of questionable relevance to the biosimilar’s specific process.
For biosimilar developers, the patent dance creates a structured disclosure that is simultaneously a legal obligation and an intelligence opportunity. Receiving the reference product sponsor’s patent list reveals which patents the sponsor considers most critical to defending market share, which patents the sponsor intends to litigate first, and which aspects of the biologic’s IP portfolio are considered strong enough to assert in a formal legal proceeding.
IP Valuation: How Purple Book Listings Affect Biologic Asset Pricing
Biologic products command the highest per-unit prices in the pharmaceutical market, and their patent and exclusivity portfolios are correspondingly the most complex to value. The key inputs for a Purple Book-anchored IP valuation model include the first licensure date, the reference product exclusivity expiration, the composition-of-matter patent expiration on the biologic molecule, the formulation and manufacturing process patent expiration dates, the number of aBLA filers and their anticipated timelines, and the interchangeability designation status of any approved biosimilar.
For reference products, the revenue model hinges on the assumption of full-price period from launch through the earlier of reference product exclusivity expiration or biosimilar launch date, followed by a rapid price erosion curve once an interchangeable biosimilar enters. The erosion curve for biologics without interchangeable competitors has historically been much shallower than the small-molecule generic erosion curve (30-50% price erosion versus 70-90%), because physician inertia, formulary dynamics, and payer contracting have kept reference products competitive. The entry of interchangeable biosimilars changes that calculus materially, since interchangeable products can be substituted at the pharmacy level without physician intervention, replicating the small-molecule generic substitution dynamic.
Humira (adalimumab) is the definitive case study. AbbVie’s biologic generated approximately $21.2 billion in U.S. revenue in 2022. The biosimilar entry wave, which began in January 2023, was anticipated for years in advance and built into consensus AbbVie earnings models. Yet the actual market share erosion in the first year of biosimilar competition was slower than analysts projected, primarily because AbbVie’s aggressive rebate contracting secured continued formulary position for the reference product. The interchangeable biosimilars (Hadlima from Samsung Bioepis, Cyltezo from Boehringer Ingelheim) have an easier path to formulary parity than non-interchangeable biosimilars but still face the structural advantage of AbbVie’s existing rebate relationships with pharmacy benefit managers.
For acquirers evaluating a biologic asset, the Purple Book patent listing provides a floor estimate of the IP defense period, while the patent dance exchanges provide a ceiling on what the sponsor is willing to actively litigate. The gap between those two figures represents the zone of negotiated settlements and authorized biosimilar agreements, which are increasingly common as sponsors prefer royalty revenue to uncontrolled market share erosion.
Recent Legislative Changes: Purple Book Continuity Act in Practice
Before the Purple Book Continuity Act took effect in March 2021, the Purple Book contained no patent information at all. Biosimilar developers had no centralized, public source of the patents their reference product sponsor intended to assert, meaning that competitive intelligence required litigation docket analysis, Freedom of Information Act requests, and private research. The 2021 changes require reference product sponsors to submit patent dance lists to the FDA within 30 days of providing them to a biosimilar applicant. The FDA then publishes these lists in the Purple Book database.
The practical limitation of this system is that the publication is reactive and applicant-specific. A patent list appears in the Purple Book only after a biosimilar applicant has submitted an aBLA and the patent dance has begun. If no one has filed a biosimilar application against a given reference product, the Purple Book contains no patent information for that product beyond the exclusivity dates. This means that for drugs with no current biosimilar filers, the Purple Book is far less informative than the Orange Book, which requires proactive patent disclosure from NDA holders regardless of whether any ANDA has been filed.
There is also no ongoing update obligation. Once a patent list is published from a specific patent dance, updates to that list require another triggering event. Reference product sponsors who obtain new patents after the initial dance exchange are not required to proactively add them to the Purple Book database.
Biosimilar Interchangeability: The Designation That Changes Pharmacy-Level Competition
A biosimilar that receives an interchangeability designation from the FDA has cleared a higher evidentiary bar than standard biosimilarity, demonstrating that it can be expected to produce the same clinical result as the reference product in any given patient and that switching between the two carries no additional safety or efficacy risk. The interchangeability designation is the Purple Book’s most commercially significant data point for competitive analysis.
Under the BPCIA, the first biosimilar to receive interchangeability designation for a reference product earns one year of exclusivity against other interchangeability designations, assuming the first interchangeable product is commercially marketed and no court has found a relevant patent invalid or not infringed. This one-year interchangeability exclusivity, which runs from commercial marketing or court decision (whichever comes first), is a distinct exclusivity type from the reference product’s 12-year period.
State pharmacy substitution laws govern whether an interchangeable biosimilar can be dispensed in place of the reference product without a physician’s specific authorization. As of 2025, all 50 states have enacted biosimilar substitution laws, but they vary in notification requirements. The practical effect of interchangeability is that it moves the biosimilar into the same competitive dynamic as a small-molecule generic: pharmacy dispensing decisions drive substitution rather than physician prescribing decisions alone. For high-volume self-injectable products like insulin analogs and adalimumab, interchangeability is the difference between 10% and 40% market share within 24 months of launch.
Technology Roadmap: Biosimilar Development from aBLA Filing to Market
Biosimilar development timelines are substantially longer than generic drug timelines, and the Purple Book exclusivity dates set hard boundaries around the earliest points in that timeline.
Phase 1: Analytical Characterization (Years 1-3)
The first stage is reverse-engineering the reference product’s structural and functional profile. This requires sourcing multiple lots of the reference product (both U.S. and ex-U.S. supply if the approval strategy includes EU data) and conducting extensive analytical characterization: primary sequence confirmation, glycosylation profiling, higher-order structure analysis (circular dichroism, hydrogen-deuterium exchange mass spectrometry), Fc receptor binding studies, and in vitro bioassays. For monoclonal antibodies, the glycan profile is particularly critical because glycosylation affects Fc effector function and, for some molecules, receptor binding. A biosimilar developer cannot submit its aBLA without demonstrating analytical similarity to the reference product’s lot-to-lot variability range.
Phase 2: Cell Line Development and Process Development (Years 2-4)
Simultaneously with analytical work, the developer’s manufacturing team constructs the expression system, typically a CHO (Chinese hamster ovary) cell line for monoclonal antibodies, and iterates upstream and downstream process parameters to hit the analytical target profile. This phase is where manufacturing patent risk is highest: the reference product sponsor’s process patents, which may not be listed in the Purple Book but are discoverable through independent patent searching, can constrain the biosimilar developer’s process choices. Using a significantly different purification train or cell culture medium formulation reduces process patent risk but may introduce product quality differences that complicate analytical similarity.
Phase 3: Clinical Program (Years 4-6)
The FDA expects biosimilar aBLAs to include at minimum a Phase 1 pharmacokinetic/pharmacodynamic study in healthy volunteers (or patients if the product is too toxic for healthy volunteers) and, in most cases, a comparative efficacy study in the most sensitive indication. ‘Most sensitive’ means the indication where the biological mechanism is most directly tied to the molecule’s properties, and where measurable differences in clinical outcome would most likely emerge if there were meaningful differences between the biosimilar and reference product. The comparative efficacy study is the most expensive component, often accounting for 50-60% of total development cost. Regulatory-only programs that rely heavily on analytical similarity data without comparative efficacy studies are possible for some products under the FDA’s evolving totality-of-the-evidence standard but carry higher approval risk.
Phase 4: Regulatory Submission and Review (Years 6-8)
The aBLA can be submitted no earlier than 4 years after the reference product’s first licensure date. FDA’s review clock for biosimilar aBLAs is 12 months standard, 6 months priority (priority review for biosimilars is rare). During this period, the patent dance commences within 20 days of aBLA acceptance. The litigation phase can run concurrently with FDA review; BPCIA patent litigation is filed in federal district court and proceeds on a separate track.
Phase 5: Commercial Launch (Year 8 onward, subject to exclusivity)
Final approval cannot become effective before reference product exclusivity expires (12 years from first licensure, plus any pediatric extension). If the developer has received a complete response letter or an FDA request for additional information, the effective approval date shifts accordingly. Commercial launch planning must account for the interchangeability designation timeline if that designation is part of the strategy, as well as payer contracting timelines (typically 12-18 months ahead of launch for formulary placement), manufacturing scale-up, and device or combination product approvals for auto-injectors or prefilled syringes.
Investment Strategy: Biologic Patent Cliffs and Biosimilar Entry Windows
For investors with exposure to biologic franchises, the Purple Book provides a structured way to map competitive risk over a 5-10 year horizon. The primary tool is the reference product exclusivity expiration date combined with the number of aBLA filers currently listed in the Purple Book or disclosed in FDA approval actions.
The Humira biosimilar experience established a market precedent: the entry of seven or more biosimilars simultaneously does not produce the same rapid price erosion that occurs in small-molecule markets. The formulary and rebate contracting dynamics of the U.S. biologic market, combined with the lack of universal interchangeability designations at launch, allow reference product sponsors to defend significant volume share for 18-36 months after biosimilar entry. Investors who modeled Humira’s revenue decline using small-molecule erosion curves significantly over-projected the speed of AbbVie’s revenue loss.
The better model for biologic IP cliff analysis distinguishes between interchangeable and non-interchangeable biosimilar entry, between therapeutic areas where physician inertia is high (oncology, immunology) and those where pharmacy substitution is more readily accepted (insulin analogs), and between markets with large commercial payer exposure (where PBM contracting is decisive) and those with high Medicare Part B exposure (where ASP-based reimbursement makes biosimilars structurally advantaged).
From the reference product sponsor’s perspective, the most effective defense is not litigation but authorized biosimilar strategy: licensing a biosimilar partner to produce a version of the molecule under the sponsor’s own brand or at a deep discount, capturing the biosimilar market while preventing uncontrolled price erosion. Pfizer’s authorized biosimilar strategy for certain oncology biologics, and AbbVie’s own authorized biosimilar program for Humira (Hyrimoz), demonstrate that the reference product sponsor with a sophisticated biosimilar division can be both the incumbent and the biosimilar entrant.
Key Takeaways: Purple Book
The Purple Book’s 12-year reference product exclusivity dates are the hard constraint for biosimilar development investment decisions. The patent dance patent lists, published since 2021, provide structured intelligence on which patents the reference product sponsor considers most defensible, but those lists are reactive, applicant-specific, and not updated proactively after the initial exchange. The interchangeability designation is the single most commercially significant binary in the Purple Book: a biosimilar with interchangeability in a high-volume self-injectable market operates in a fundamentally different competitive environment than one without it. For investors, biologic IP cliff modeling must account for formulary contracting dynamics and the authorized biosimilar strategy, not just the patent and exclusivity expiration dates.
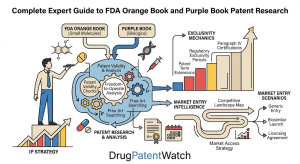
5. Orange Book vs. Purple Book: A Side-by-Side Technical Analysis
| Feature | FDA Orange Book | FDA Purple Book |
|---|---|---|
| Drug type covered | Small-molecule drugs (chemically synthesized) | Biological products (derived from living organisms) |
| Legal framework | Hatch-Waxman Act (1984) / FD&C Act | BPCIA (2010) / PHS Act |
| Patent listing trigger | Mandatory at NDA submission; proactive obligation | Reactive; triggered only by biosimilar aBLA and patent dance |
| Patent types listed | Drug substance, drug product, method of use | Patents disclosed in patent dance (including, potentially, manufacturing) |
| Patent use codes | Yes, ‘U-‘ format with specific indication descriptions | No |
| Manufacturing patent exclusion | Generally excluded | May be included if raised during patent dance |
| Exclusivity types | NCE (5yr), ODE (7yr), NCS (3yr), PED (6 mo add-on), GAIN (5yr add-on), 180-day first-filer, CGT (180-day) | Reference product (12yr), interchangeable biosimilar (1yr), pediatric (6 mo add-on) |
| Orphan drug exclusivity | Yes (ODE, 7yr) | Not listed in Purple Book |
| Paragraph IV / patent challenge mechanism | Structured Paragraph IV certification; 30-month stay; 180-day first-filer exclusivity | Patent dance exchange; BPCIA litigation; no automatic stay equivalent |
| Update frequency | Daily (ANDA approvals), monthly supplements, annual edition | Monthly |
| Access format | Online database, downloadable data files, PDF | Online database, monthly downloadable reports |
| Patent data completeness | High for listed types; excludes manufacturing | Lower; limited to dance participants and their exchange |
| Post-approval patent update obligation | Yes (within 30 days of patent issuance) | No ongoing obligation beyond dance exchange |
| Therapeutic equivalence codes | Yes (A/B codes for substitutability) | Biosimilarity/interchangeability designations |
| FDA listability adjudication | Ministerial (FDA does not evaluate patent claims) | Ministerial (FDA does not evaluate patent claims) |
| Delisting mechanism | Orange Book Transparency Act; patent dispute process | No equivalent delisting mechanism currently |
| Key limitation | Excludes manufacturing and certain polymorph patents | Dance-reactive; no proactive disclosure; no use codes |
The structural asymmetry between the two books is not accidental. It reflects the different competitive dynamics of small molecules and biologics. Small molecules are easy to copy if the patents are cleared; the Paragraph IV system is designed to force the patent question into court promptly. Biologics are difficult to copy regardless of IP status; the 12-year exclusivity is designed to ensure the innovator recoups its investment before facing any competition at all, with the patent dance as a secondary layer of protection for the most commercially valuable claims.
6. Advanced Research Workflows: Integrating Both Books with External Data Sources
Cross-Referencing Paragraph IV Dockets with Orange Book Listings
The FDA maintains a public list of ANDA filers who have made Paragraph IV certifications, organized by the brand drug and the patent being challenged. Cross-referencing this list with the Orange Book patent listings reveals not just which patents are being challenged but which claims within those patents the generic filer believes are invalid or not infringed. Court dockets from the resulting 30-month stay litigation (available via PACER) provide the specific legal theories: obviousness, anticipation, enablement failures, or non-infringement arguments based on claim construction.
The most useful analytical workflow combines Orange Book patent expiration dates with Paragraph IV litigation outcomes in a searchable database. A patent that has survived three Paragraph IV challenges without invalidation is more commercially durable than one that has never been challenged, and its contribution to the brand drug’s IP valuation should be weighted accordingly. Conversely, a patent that has been challenged in every ANDA filer’s application is likely to be litigated to resolution in the next round, and the probability of invalidation increases with each additional challenger bringing potentially new prior art.
Mining Exclusivity Stacks for Lifecycle Management Insight
A drug with multiple overlapping exclusivities has a longer effective protection period than any single exclusivity alone would suggest. The practical tool for mapping exclusivity stacks is the Orange Book’s exclusivity table, downloadable as a data file. For each drug product (identified by application number and product number), the table lists all active exclusivity codes and their expiration dates.
A useful analysis sorts branded drugs by the gap between the last patent expiration and the last exclusivity expiration. A large gap means the exclusivity system, not the patent system, is the primary protection mechanism. Drugs in this category are particularly vulnerable to 505(b)(2) generic applications that use a different formulation but reference the same active moiety, because such applications only need to clear the specific exclusivity type covering the change, not the full NCE period. Brand companies that structure their lifecycle extensions to earn 3-year NCS exclusivity on each new dosage form can build a rolling exclusivity ladder that keeps any single formulation protected even as the base compound’s patents fall.
Using Downloadable Data Files for Quantitative Patent Analysis
The FDA provides Orange Book data files in pipe-delimited format, updated monthly. The key files are products.txt (all approved NDA and ANDA products), patent.txt (all listed patents with application number, product number, patent number, and expiration date), exclusivity.txt (all active exclusivities), and applications.txt (NDA holder information). These files can be imported into any relational database or analytical software and joined to create a complete view of the patent and exclusivity landscape for any branded drug.
For the Purple Book, the monthly downloadable report contains all licensed biologics with their BLA numbers, proper names, reference product relationships, first licensure dates, exclusivity expiration dates, and any listed patent information. Joining this file to aBLA approval action lists (available from FDA’s Drugs@FDA database) and BPCIA litigation dockets produces a comprehensive biosimilar competitive landscape model.
Firms that build and maintain these integrated databases hold a substantial competitive intelligence advantage over those that rely on ad hoc Orange and Purple Book lookups. The ability to screen the entire branded drug market for drugs entering a 36-month patent cliff window, identify the number of active ANDA filers against each, and estimate 180-day exclusivity value is a capability that fundamentally changes generic pipeline prioritization decisions.
7. Evergreening Strategies: A Full Technology Roadmap
Evergreening is the structured extension of commercial exclusivity through IP mechanisms layered onto an approved drug product. It is legal, widely practiced, contested by payers and generic manufacturers, and closely tracked by the FTC (whose 2002 and 2023 reports on Orange Book patent practices documented specific abuses). The following roadmap traces the full lifecycle of a hypothetical blockbuster small molecule through its evergreening program.
Stage 1: Composition of Matter Patent (Years 0-20 from filing)
The COM patent covers the active pharmaceutical ingredient itself, its salts, and often its major polymorphs. Filed during preclinical development, the COM patent typically expires 20 years from its filing date, before Patent Term Extension (PTE) under Hatch-Waxman. PTE can restore up to 5 years of patent life lost to FDA review, capped at 14 years of post-approval protection. This is the foundational IP layer; once it falls, the molecule itself is in the public domain.
Stage 2: Formulation and Delivery System Patents (Years 5-15 from NDA)
As the COM patent ages, the NDA holder commissions formulation research to develop new delivery mechanisms: extended-release beads, osmotic pump technology (OROS), abuse-deterrent matrices, nanoparticle formulations, or co-crystal forms that alter solubility or absorption. Each new formulation, if it produces a clinically meaningful benefit, can support an sNDA with new clinical studies, a new product listing, and new Orange Book patents with expiration dates 10-15 years beyond the NDA date. The formulation patents are listed in the Orange Book; ANDA filers targeting the reformulated product must now clear both the COM patent (if still active) and the formulation patents, or challenge them via Paragraph IV.
Stage 3: Method-of-Use Patent Expansion (Years 5-20 from NDA)
Post-approval clinical research frequently generates new uses for approved drugs, either as single agents in new indications or as components of combination regimens. Each new approved indication can be covered by a new method-of-use patent and listed in the Orange Book with a corresponding patent use code. A generic filer who wants to launch before the method-of-use patent expires must either carve out the new indication or file a Paragraph IV challenge. Carving out a high-revenue indication is commercially unattractive; a Paragraph IV challenge requires litigation investment. Either option benefits the brand company.
Stage 4: Dosage Form and Strength Expansion (Years 8-18 from NDA)
sNDAs for new dosage forms (e.g., tablet to capsule, immediate-release to extended-release, oral to patch), new strengths, or new routes of administration each earn 3-year NCS exclusivity if supported by new clinical investigations. The NDA holder can use these exclusivities to migrate the patient population to the newer formulation before generic entry on the original product. If the migration is successful, the market for the original formulation shrinks to the point where generic entry is commercially uninteresting, even after all exclusivities expire. This was the core of AstraZeneca’s Prilosec-to-Nexium strategy: launch the enantiomer ahead of omeprazole generic entry, migrate patients through aggressive managed care contracting, and let the generic Prilosec market develop in a category the brand has effectively abandoned.
Stage 5: Combination Product and Fixed-Dose Combination Patents (Years 10-20 from NDA)
Combining the original molecule with a complementary agent under a single NDA creates a new product entry, new Orange Book listings, and potentially NCE exclusivity for the combination partner if the partner is itself a new chemical entity. For molecules in chronic disease management (cardiovascular, metabolic, CNS), fixed-dose combinations increase adherence, create formulary differentiation, and reset the patent clock. The combination product’s Orange Book entries protect against combination generics; the original monocomponent remains separately exposed to ANDA competition once its own exclusivities fall.
Stage 6: Pediatric Exclusivity (Anytime Pre-Exclusivity Expiration)
A Written Request from the FDA for pediatric studies can be sought at any point in the product lifecycle where there is a remaining exclusivity or patent to extend. The 6-month extension applies to all existing listed patents and exclusivities simultaneously, not just the one that would otherwise expire soonest. For a drug with NCE exclusivity expiring in year 5 and a formulation patent expiring in year 12, both periods extend by 6 months upon completion of the pediatric study. The cost of the pediatric study, often $20-50 million for a single Phase 2 study, is trivially small relative to the revenue protected.
Stage 7: Authorized Generic and Patent Settlement Agreements (Years 10-25)
The final stage of the lifecycle management program is not strictly IP but directly interacts with Orange Book patent mechanics. An authorized generic agreement, in which the NDA holder licenses a generic company to sell a version of the drug under the NDA at a discount, captures a portion of the generic market without the reference product’s price collapsing. Authorized generics can launch immediately upon brand patent expiration; they do not need their own ANDA approval. They also compete with Paragraph IV first filers during the 180-day exclusivity window, reducing the first filer’s revenue and thereby lowering the economic incentive for future Paragraph IV challenges on similar molecules.
Patent settlement agreements (sometimes called ‘pay-for-delay’ agreements, now subject to antitrust scrutiny under FTC v. Actavis, 2013) are structured agreements between an NDA holder and a Paragraph IV filer in which the filer accepts a later launch date in exchange for compensation, often including an authorized generic license. The FTC monitors these agreements and publishes annual reports on their prevalence. The Actavis standard requires antitrust analysis under a ‘rule of reason’ framework; large unexplained payments from brand to generic companies are a red flag.
8. Common Research Errors and How to Avoid Them
Conflating patent expiration with loss of exclusivity. These are distinct events and rarely coincide. A drug’s last Orange Book patent may expire in year 12 while NCE or ODE exclusivity continues through year 15. ANDA final approval cannot issue until both have cleared.
Using only the Orange Book for freedom-to-operate analysis. The Orange Book excludes manufacturing process patents, which can be commercially significant barriers to generic entry. A freedom-to-operate analysis must extend to the USPTO’s patent database, European Patent Office records (for molecules with global development history), and the patent prosecution file wrappers that reveal claim scope narrowing during examination.
Assuming Purple Book patent lists are comprehensive. Patent lists in the Purple Book reflect only what the reference product sponsor chose to include in a specific patent dance exchange with a specific biosimilar applicant. The sponsor may have additional patents, may list fewer patents in future exchanges, and has no obligation to update the published list as new patents issue.
Treating the first-filer 180-day exclusivity as guaranteed. The 180-day exclusivity can be forfeited if the first filer fails to market commercially within a specified window after the FDA makes its application tentatively approvable, fails to comply with FDA requests, or is found to have engaged in patent settlement conduct that triggers forfeiture provisions under the MMA (2003). Losing the exclusivity period destroys the primary financial rationale for a Paragraph IV challenge on high-value drugs.
Ignoring the difference between ‘filed’ and ‘accepted’ in aBLA tracking. The FDA’s acceptance of an aBLA for filing triggers the patent dance clock; mere submission does not. A developer tracking competitor progress via Purple Book updates may miss the filing date if they are monitoring only submission records.
Failing to verify patent expiration dates against USPTO records. Orange Book expiration dates are submitted by NDA holders and are not independently verified by the FDA. Maintenance fee payment status, inter partes review outcomes, and any terminal disclaimers that cap a patent’s term relative to an earlier patent can all cause the true patent expiration to differ from the Orange Book listing. The USPTO’s patent term calculator and the Patent Center database are the authoritative sources.
9. Master Reference Tables
Orange Book Exclusivity Code Reference
| Code | Full Name | Duration | Blocking Effect |
|---|---|---|---|
| NCE | New Chemical Entity | 5 years from NDA approval | Blocks ANDA/505(b)(2) submission; Paragraph IV allowed at year 4 |
| ODE | Orphan Drug Exclusivity | 7 years from approval | Blocks same-drug same-indication applications |
| NCS | New Clinical Study | 3 years from sNDA approval | Blocks final ANDA/505(b)(2) approval for the same product/use |
| PED | Pediatric Exclusivity | 6 months added to existing patents and exclusivities | Extends existing protection periods |
| GAIN | Qualified Infectious Disease Product | 5 years added to NCE/ODE/NCS | Stacks onto qualifying exclusivity |
| PC | First-Filer (180-day) | 180 days from first commercial marketing or court decision | Blocks other generics from final approval during period |
| CGT | Competitive Generic Therapy | 180 days from approval | Blocks other ANDA approvals for same product |
| NP | New Product | 3 years (new formulation/indication) | Same as NCS in practice |
Purple Book Exclusivity Reference
| Exclusivity Type | Duration | Trigger | Notes |
|---|---|---|---|
| Reference Product Exclusivity | 12 years from first licensure | Automatic upon BLA approval | 4-year bar on aBLA submission also applies |
| Pediatric Exclusivity (Biologic) | 6 months added to 12-year period | Completion of pediatric studies per Written Request | Extends to 12.5 years total |
| Interchangeable Biosimilar Exclusivity | 1 year from first commercial marketing or court decision | First interchangeable designation for a given reference product | Blocks subsequent interchangeability designations only |
Hatch-Waxman vs. BPCIA: Key Procedural Comparison
| Element | Hatch-Waxman (Small Molecule) | BPCIA (Biologic) |
|---|---|---|
| Application type | ANDA | aBLA (351(k)) |
| Patent disclosure mechanism | Proactive Orange Book listing by NDA holder | Reactive patent dance exchange |
| Patent challenge vehicle | Paragraph IV certification | Patent dance + BPCIA litigation |
| Automatic approval stay | 30-month stay upon brand company lawsuit | No automatic stay; injunction must be sought |
| First-mover exclusivity | 180-day first-filer exclusivity | 1-year interchangeable biosimilar exclusivity (different mechanism) |
| Exclusivity period | 5-year NCE maximum; others shorter | 12-year reference product exclusivity |
| FDA delisting obligation | Yes (under Orange Book Transparency Act) | No equivalent |
10. Consolidated Key Takeaways for IP Teams and Investors
The Orange Book is a proactive, NDA-holder-driven disclosure system designed around the Paragraph IV litigation framework. Its primary utility for generic developers is patent challenge planning and exclusivity stack analysis. Its primary utility for brand companies is lifecycle management: each new sNDA-supported product listing creates a fresh patent and exclusivity layer that resets the ANDA timeline for that formulation. The Orange Book’s power as an IP intelligence tool scales directly with the analyst’s ability to connect its listings to litigation dockets, prosecution file histories, and product-level revenue data.
The Purple Book is a reactive, aBLA-triggered disclosure system operating within the BPCIA framework. Its most reliable data points are the first licensure date and reference product exclusivity expiration. Its patent information is meaningful but incomplete: it tells you what the reference product sponsor was willing to assert against a specific biosimilar applicant at a specific point in time, not what patents the sponsor has in reserve. Biosimilar developers who rely on the Purple Book alone for IP clearance will be surprised in litigation.
The interplay between the two books matters for companies with both small-molecule and biologic assets. The most sophisticated pharmaceutical lifecycles involve a succession of products, an oral small molecule followed by a subcutaneous biologic targeting the same pathway, each with its own patent and exclusivity stack, each designed to migrate patient revenue forward as the earlier product’s protection erodes. Reading the Orange and Purple Books together, as chapters of the same commercial story, is how the most experienced IP analysts and institutional investors approach drug portfolio valuation.
For analysts and investors, the practical workflow is this: start with the Purple Book first licensure date and 12-year exclusivity expiration for biologic assets; cross-reference aBLA filer activity from FDA approval action letters; build the biosimilar entry scenario incorporating interchangeability designation status and state substitution law. For small molecules, start with the Orange Book’s last patent expiration, stack exclusivities, identify active Paragraph IV certifications, model litigation outcome probabilities, and price in the authorized generic and 180-day exclusivity dynamics. Then integrate both views into a single product-level revenue model that accounts for the transition from brand to generic or biosimilar competition with mechanism-specific erosion curves.
The Orange and Purple Books are public records, maintained by a federal agency, and accessible to anyone with an internet connection. The competitive advantage is not access; it is the analytical framework applied to the data, and the speed with which that analysis can be acted upon when patent expiration or litigation resolution creates a market entry window.
All patent and exclusivity dates should be verified against current FDA Orange Book and Purple Book database records and USPTO patent term data before making commercial or investment decisions. This document does not constitute legal advice.