Last updated: June 3, 2026
Executive summary: Testosterone products sit in a mature, highly competitive market shaped by (1) patent/market-exclusivity fade cycles across multiple dosage forms, (2) recurring reimbursement pressure and payer utilization management, (3) manufacturing and supply constraints that can swing wholesaler inventories and short-cycle pricing, and (4) ongoing generic competition that shifts value from branded launches to net price erosion. Financial trajectory is dominated by the extent of brand-to-generic conversion by formulation (injectables, transdermal gels/solutions, patches, pellets) and by payer mix in the US and Western Europe. The market also remains sensitive to safety label perceptions and competitive “substitute” switching between delivery systems.
How big is the testosterone drug market and what drives demand?
Featured snippet answer: Testosterone demand is driven by hypogonadism prevalence, clinician prescribing patterns, payer coverage rules, and product accessibility (availability and formulary placement). Net sales track how quickly brand formulations convert to generics and how strongly prescribers can switch among alternative delivery systems.
Primary therapeutic and prescribing drivers
- Indication focus: Testosterone replacement therapy for primary and secondary hypogonadism in adult males (typical labeling frameworks in the US and EU). Off-label use varies by jurisdiction and has been a historical driver of volatility in prescribing, but payer controls increasingly target documented diagnosis.
- Diagnosis dynamics: Rates of testosterone deficiency diagnosis rise and fall with screening practices, endocrine/urology referral patterns, and changes in clinical guideline messaging.
- Patient adherence: Delivery system drives real-world persistence. Gels and solutions often capture adherence and flexible titration. Patches and pellets have adherence profiles that affect switching behavior.
Key demand headwinds
- Utilization management: Prior authorization, step therapy, and documentation requirements compress volumes unless the prescriber aligns with payer evidence standards.
- Safety and labeling perception: Boxed warnings and cardiovascular risk discussions shape prescriber comfort and payer risk scoring.
- Competitor switching: When a product becomes non-preferred, prescribers move to a covered formulation even if the clinical difference is limited.
What is the testosterone pricing and reimbursement landscape in the US?
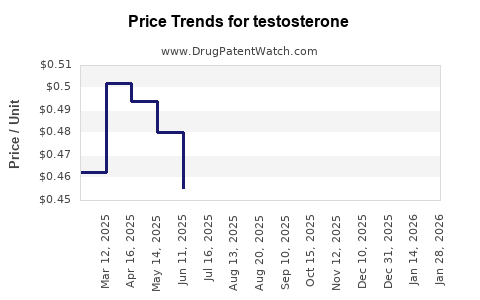
Featured snippet answer: US net pricing is pressured by generic substitution and payer formulary design. Loss of exclusivity for branded testosterone formulations typically triggers rapid erosion in average selling price unless the brand retains protected differentiation through formulation patents, device IP, or lifecycle “authorized” improvements.
Reimbursement and payer mechanics
- Commercial plans: Formulary exclusions and preferred-tier placement decide whether a branded product keeps volume. Generic-first rules often reduce share within months of generic entry.
- Medicaid and Medicare: Coverage is less about brand loyalty and more about lowest net cost and formulary status, which accelerates conversion.
- 340B and specialty channels: Certain buyer segments may influence near-term channel pricing and inventory behavior but do not typically prevent long-run net price declines after exclusivity breaks.
Net price sensitivity by delivery system
- Gels/solutions: High unit volume and strong generic penetration make pricing sensitive to discounting and wholesaler buying behavior.
- Injectables: Often face different competitive patterns depending on product-specific history, supply continuity, and whether payers perceive injection schedules as clinically standardized.
- Patches/pellets: Smaller market share and different competitive sets can sustain higher margins longer, but ultimately face generic pressure as manufacturing capacity scales.
How do patent expiration cycles and generic entry reshape the testosterone financial outlook?
Featured snippet answer: Testosterone sales generally show a pattern of brand peak followed by stepwise declines around exclusivity and patent expirations by formulation. The faster the conversion from branded to generic, the steeper the net revenue drawdown, with the timing controlled by FDA approvals, labeling carve-outs, and manufacturing ramp for generics.
What typically determines the timing of value erosion
- Exclusivity type and scope: Each product’s regulatory exclusivity (and the strength of formulation/device/method-of-use patents where applicable) determines how long “skin in the game” branded pricing lasts after first generic filings.
- Paragraph IV litigation cadence: If a brand’s relevant Orange Book-listed patents are challenged, settlement or court outcomes can delay generic launch, moving the erosion date out by months to years.
- ANDA approval and launch ramp: Even with legal clearance, generic uptake depends on packaging equivalence, supply assurance, wholesaler confidence, and prescriber switching.
Delivery-form cross-impact
- A generic entry in one delivery class can shift patient mix to covered options, accelerating overall therapeutic category erosion even if some products keep protection longer.
When does testosterone lose exclusivity by product and dosage form?
Featured snippet answer: Exclusivity loss is not a single date for “testosterone” as a molecule; it is formulation- and product-specific. The market’s financial trajectory follows the staggered end dates of branded products by gel/solution/injectable/patch/pellet category, with generic conversions typically clustering in waves.
What to map for each testosterone brand
- Active ingredient and salt: Testosterone (base) products may have different patent estates based on specific formulation and delivery system.
- Route and dosage form: Topical gels/solutions have distinct formulation and device patents relative to injectables or pellets.
- Strength and packaging: Patents can be strength-specific (or tied to multi-dose packaging), affecting how quickly generic SKUs replace branded revenue.
- Orange Book coverage: Patent listing patterns drive what generics can launch “at risk” and what they must carve out.
Which companies dominate testosterone sales and how is market share shifting?
Featured snippet answer: The category is split between legacy brand holders and a growing set of generic manufacturers that have scaled production and distribution. Market share shifts follow exclusivity timelines, formulary inclusion decisions, and availability.
Competitive landscape structure
- Brand segment: Typically includes one or more legacy branded testosterone formulations where patents or lifecycle protections slow generic conversion.
- Generic segment: Multiple ANDA entrants for gels and injectables plus additional competitors for patches/pellets, producing heavy price competition.
- Channel dynamics: Wholesalers and pharmacy benefit managers increasingly treat testosterone as a cost-driven substitution category once formulary rules switch.
How share erosion usually plays out
- Early phase: Brand keeps share through non-price factors (prescriber familiarity, patient stability).
- Middle phase: When generics launch, brand share compresses quickly, then stabilizes at a residual level due to switching frictions.
- Late phase: After multiple entrants and stronger PBM preference, brand share drops further, leaving mostly net price discounting to hold the remaining base.
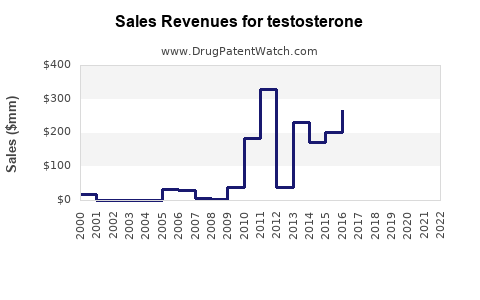
What financial trajectory should investors expect for testosterone brands?
Featured snippet answer: The expected trajectory is a gradual-to-sharp revenue decline after exclusivity loss, followed by stabilization at lower net sales driven by residual branded share or continued superiority in specific patient segments.
Revenue model by phase
- Pre-loss of exclusivity: Higher net pricing, stable volume, and premium position on formulary.
- Post first generic launch: Rapid net price erosion; revenue declines accelerate. Gross margin often compresses through discounting and customer mix changes.
- Post multi-generic normalization: Prices approach category floor; profitability depends on manufacturing cost efficiency, contract pricing discipline, and any remaining IP differentiation.
- Cycle-to-cycle replacement: Some companies offset declines via new strengths, delivery device improvements, or geographic expansion.
Profitability drivers
- Manufacturing scale and yield
- Channel rebates and PBM negotiations
- Inventory management around supply disruptions
- Legal and exclusivity spend
How does testosterone supply and manufacturing risk impact quarterly sales?
Featured snippet answer: Supply constraints can temporarily raise net pricing and maintain volume for constrained products, but the underlying trend after exclusivity loss still points to long-run erosion once supply normalizes and generic options remain available.
Mechanisms
- Allocation during shortages: Forced allocation can drive sales concentration but may also trigger payer substitution.
- Inventory swings: Wholesaler ordering behavior before anticipated shortages or price drops can distort near-term revenue.
- Regulatory inspections and manufacturing downtime: Quality events can disrupt continuity and trigger short-cycle reimbursement or switching.
What is the Orange Book status of testosterone products and why does it matter financially?
Featured snippet answer: Orange Book-listed patents define the “last mile” before generics can launch. For testosterone brands, the strongest financial lever is not the earliest patent expiration on the molecule but the final combination of formulation/device/method-of-use patents that blocks the most commercially relevant dosage forms.
How Orange Book coverage maps to risk
- If patents remain unchallenged or survive: Generic entry is delayed, protecting net sales.
- If patents are challenged and resolved early: Timelines compress and revenue erosion accelerates.
- If carve-outs exist: Generics can launch partial SKUs, creating fragmented revenue loss rather than a clean cliff.
What patent litigation and settlement patterns drive testosterone generic entry?
Featured snippet answer: In testosterone, litigation often determines whether generics launch “on” or “off” the scheduled exclusivity/patent dates. Settlement agreements are the key driver of delayed launches, while adverse court outcomes trigger earlier-than-planned erosion.
Litigation outcomes that matter
- Automatic stay triggered by first-filing ANDA: Can extend branded exclusivity exposure through the litigation window.
- Settlement: Can shift generic launch by the settlement-term or convert litigation risk into a timed entry date.
- Partial invalidation: May allow generic entry for some strengths or presentations.
How does testosterone compare with other hormone therapies on market maturity and price erosion?
Featured snippet answer: Testosterone shares market-maturity features with other commoditizing endocrine drugs: once generics secure broad formulary acceptance, net price erosion accelerates. Differences arise from (1) delivery system complexity and (2) patient adherence and switching frictions that can prolong branded share in localized segments.
Comparative dynamics
- Other androgen derivatives: May compete for similar prescribing pathways but are often differentiated by safety perceptions and availability.
- Alternative endocrine therapies: Where diagnosis pathways differ, payer rules can keep testosterone more price-sensitive due to substitution ease within class.
What generic entry risks exist for testosterone brands?
Featured snippet answer: Generic entry risk is highest for popular gel/solution strengths with broad coverage and for brands with weaker late-life formulation/device patent coverage. Risk decreases if the brand’s remaining patents protect a broad set of SKUs, or if settlement litigation agreements delay launch.
Risk factors
- Number of ANDA approvals already on file
- Breadth of Orange Book patent coverage
- Probability of “at-risk” launch after losing in court
- Manufacturing capacity of generic entrants
How do formulation changes and lifecycle management affect testosterone sales?
Featured snippet answer: Lifecycle changes that meaningfully impact dosing convenience, skin absorption profiles, or device usability can slow price erosion by giving payers and prescribers a reason to keep a brand. But lifecycle strategy cannot fully offset category-wide generic substitution once formulary rules turn.
Common lifecycle levers
- Delivery improvements: devices that reduce application variability or improve patient usability.
- Strength/packaging additions: which can segment the competitive set and buy time on specific SKUs.
- Lifecycle label updates: which can affect payer prior authorization behavior.
Key regulatory milestones that influence testosterone commercial timing
Featured snippet answer: FDA approval timing affects when generics can reach the market, which then shifts sales within quarters. For testosterone, the commercial impact is driven by ANDA approval and labeling alignment with Orange Book carve-outs.
Milestones that move sales
- ANDA approvals and labeling negotiations
- Patent expiry and exclusivity start-to-end dates
- Court outcomes and settlement terms
- Launch readiness: packaging, distribution agreements, and supply continuity
Key takeaways
- Testosterone is a mature, competition-driven category where financial trajectory follows formulation-specific patent and exclusivity timelines rather than a single molecule end date.
- Net price erosion is typically fast after generic entry, with residual branded share stabilizing only if formulation differentiation, formulary dynamics, or supply constraints persist.
- The biggest quarter-to-quarter sales swing factors are generic launch timing, payer preference changes, and supply availability.
- Orange Book patent coverage and patent litigation outcomes determine the calendar for revenue protection or accelerated category conversion.
- Investors and licensing counterparties should model value capture by dosage form (gels/solutions vs injectables vs patches/pellets) and SKU breadth, not by “testosterone” as a single monolith.
FAQs
1) Why do testosterone gels lose market share faster than injectables?
Because topical gels are high-volume and widely substituted at the pharmacy level, and generic packs usually reach formulary coverage with minimal prescriber friction.
2) What matters more for generic timing: patent expiry or FDA approval date?
FDA approval date governs launch readiness; patent expiry governs whether the generic can lawfully market when approval issues. Both determine the actual revenue impact calendar.
3) Can a testosterone brand keep pricing power after exclusivity loss?
Only if it retains meaningful SKU-level differentiation (device/formulation/patient-fit) that preserves formulary preference or if generic coverage is incomplete at first launch.
4) How do PBM formularies affect testosterone revenue?
PBM formulary placement and rebate design often determine whether prescribers keep using a brand or switch to a cheaper substitute once generics are available.
5) Does supply disruption protect brand sales long-term?
It can stabilize near-term sales by preventing switching during shortages, but it rarely prevents long-term erosion once generics are legally and operationally established.
References
- U.S. Food and Drug Administration. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA.
- U.S. Food and Drug Administration. Drug Approval Reports and related ANDA/505(b)(2) approval information. FDA.
- U.S. Patent and Trademark Office. Patent assignment and publication records for relevant testosterone formulation and method patents. USPTO.