Last updated: February 19, 2026
Natesto is a testosterone undecanoate nasal spray indicated for the treatment of endogenous testosterone deficiency (hypogonadism) in adult males. The drug targets a specific unmet need within the testosterone replacement therapy (TRT) market, offering an alternative to existing injectable, topical, and oral formulations. This analysis examines the market landscape, competitive positioning, intellectual property, and projected financial performance of Natesto.
What is the Market for Testosterone Replacement Therapy?
The global testosterone replacement therapy market is substantial and projected to grow. In 2023, the market was valued at approximately $3.4 billion and is forecast to reach $5.1 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.0% [1]. Key market drivers include an aging male population, increasing prevalence of obesity and metabolic syndrome, rising awareness of hypogonadism, and advancements in TRT formulations offering improved convenience and efficacy.
The TRT market is segmented by product type, route of administration, and end-user.
- Product Type: Includes testosterone gels, injections, patches, pellets, and oral formulations.
- Route of Administration: Dermal, intramuscular, subcutaneous, oral, and nasal.
- End-User: Hospitals, clinics, ambulatory surgery centers, and home care settings.
The nasal administration segment, where Natesto competes, is a smaller but growing niche. Its appeal lies in rapid absorption and avoidance of first-pass metabolism associated with oral therapies, as well as the convenience compared to frequent injections or daily topical applications.
How is Natesto Positioned in the TRT Market?
Natesto (testosterone undecanoate) is an FDA-approved nasal spray that delivers testosterone directly into the bloodstream via the nasal mucosa. Its unique administration route differentiates it from more established TRT modalities.
Key Differentiating Factors:
-
Administration: Nasal spray, requiring application twice daily (morning and evening) [2]. This offers an alternative to:
- Injectables: Typically administered every one to two weeks, requiring healthcare professional involvement or self-injection training.
- Topical Gels/Solutions: Applied daily to the skin, can cause transference to partners and children, and may lead to skin irritation.
- Transdermal Patches: Applied daily, can cause skin reactions and inconsistent absorption.
- Pellets: Implanted surgically every few months, requiring a procedure.
- Oral Formulations: Can cause gastrointestinal distress and liver toxicity in some forms.
-
Pharmacokinetics: Natesto provides relatively stable testosterone levels throughout the day, aiming to mimic natural circadian rhythms more closely than some other formulations [2]. Studies indicate that Natesto can achieve and maintain testosterone levels within the physiological range for the majority of men with hypogonadism [3].
-
Convenience: The twice-daily nasal application is designed for ease of use, requiring no needles or skin contact.
Competitive Landscape:
The TRT market is competitive, with several established players and a broad range of therapeutic options.
| Competitor Product (Generic Name) |
Route of Administration |
Dosing Frequency |
Key Considerations |
| AndroGel, Testim (Testosterone Gel) |
Topical (dermal) |
Daily |
Skin transfer risk, skin irritation, variable absorption |
| Depo-Testosterone (Testosterone Cypionate) |
Intramuscular |
Every 1-2 weeks |
Needle phobia, fluctuating hormone levels, injection site pain |
| Axiron (Testosterone Solution) |
Topical (axilla) |
Daily |
Skin irritation, potential for transference |
| Androderm (Testosterone Patch) |
Transdermal |
Daily |
Skin reactions, inconsistent absorption, visible device |
| Testopel (Testosterone Pellets) |
Subcutaneous implant |
Every 3-6 months |
Requires surgical implantation, potential for extrusion |
| Jatenzo (Testosterone Undecanoate Softgel) |
Oral |
Twice daily |
First-pass metabolism, gastrointestinal side effects, cost |
| Natesto (Testosterone Undecanoate Nasal Spray) |
Nasal |
Twice daily |
Nasal discomfort, potential for nasal irritation, distinct dosing |
Natesto's primary competitive advantage is its novel nasal delivery system, which addresses some of the drawbacks of existing therapies. However, it faces competition from established and emerging TRT products, including oral formulations like Jatenzo, which also offers a non-injectable, non-topical option.
What is the Intellectual Property Landscape for Natesto?
The intellectual property surrounding Natesto primarily consists of patents protecting the drug itself, its formulation, and its method of use. These patents are critical for maintaining market exclusivity and ensuring a return on investment.
Key Patents and Exclusivity:
- US Patent No. 8,901,121: This patent, titled "Nasal testosterone undecanoate compositions and methods of use," was granted on December 1, 2014, and is listed in the FDA Orange Book for Natesto [4]. It covers the pharmaceutical composition of testosterone undecanoate for nasal administration.
- US Patent No. 9,320,612: Also listed in the Orange Book, this patent, titled "Nasal testosterone compositions and methods of use," was granted on April 26, 2016. It likely covers further aspects of the formulation or method of administration.
- US Patent No. 9,940,442: Granted on April 10, 2018, and listed in the Orange Book, this patent is related to the nasal delivery of testosterone.
Patent Expiration:
The expiration dates of these key patents are crucial for projecting market exclusivity. Based on standard patent terms and potential extensions (such as Hatch-Waxman Act extensions), the core patents for Natesto are generally expected to expire in the mid-to-late 2020s.
- The '121 patent, the foundational composition patent, is typically expected to expire around December 2028, considering potential extensions.
- Other related patents may expire around the same period or shortly thereafter.
Generic Competition:
Upon the expiration of these patents, generic manufacturers will be eligible to file Abbreviated New Drug Applications (ANDAs) for testosterone undecanoate nasal spray. The market entry of generics will lead to price erosion and a significant decrease in Natesto's market share and revenue. The timeline for potential generic entry is therefore a critical factor in the long-term financial forecast.
What is the Financial Trajectory of Natesto?
The financial trajectory of Natesto is directly linked to its market adoption, pricing strategy, and the duration of its patent exclusivity. As a relatively newer entrant compared to some established TRT products, Natesto's revenue ramp-up is ongoing.
Revenue Performance:
While specific, granular revenue figures for Natesto are not always publicly disclosed by manufacturers in real-time, market reports and company filings provide indicators of its sales performance.
- Launch and Early Adoption: Natesto received FDA approval in March 2015 [5]. Its launch phase involved building market awareness and securing formulary access with payers.
- Market Share: Natesto holds a niche but growing share within the broader TRT market. Its performance is benchmarked against the overall growth of the TRT sector and the specific segment for novel delivery systems.
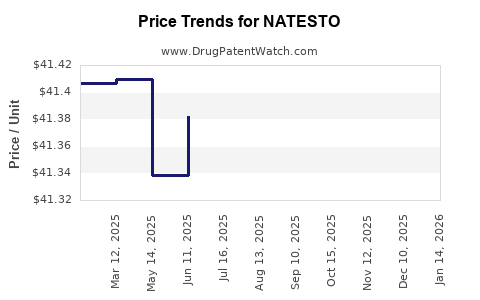
- Pricing: The wholesale acquisition cost (WAC) of Natesto is a significant factor. Its pricing must be competitive within the TRT landscape while reflecting the novel administration and R&D investment. Pricing for TRT products can range from $100 to over $400 per month, depending on the formulation and dosage [6]. Natesto's price is generally positioned to compete with other convenient, non-injectable options.
Projected Revenue Growth:
- Near-Term (1-3 years): Natesto is expected to experience continued revenue growth, driven by increasing physician prescribing and patient adoption, particularly among those seeking an alternative to injections and topical applications. Market penetration will be influenced by payer coverage and formulary status.
- Mid-Term (3-7 years): Revenue growth is likely to moderate as the market becomes more saturated with competing therapies and as Natesto approaches its patent cliff. The increasing prevalence of hypogonadism and ongoing patient education will continue to support demand.
- Long-Term (7+ years): Post-patent expiration, significant revenue decline is anticipated due to generic competition. Manufacturers typically explore lifecycle management strategies, such as new formulations or indications, to mitigate this impact, though this is less common for established TRT molecules.
Factors Influencing Financial Trajectory:
- Sales and Marketing Efforts: The effectiveness of the sales force and marketing campaigns in educating healthcare providers and patients about Natesto's benefits.
- Payer Reimbursement: Broad and favorable insurance coverage is essential for patient access and affordability.
- Clinical Data: Ongoing and supportive clinical data demonstrating efficacy and safety in real-world settings.
- Emergence of New Competitors: New TRT products with enhanced profiles or competitive pricing can impact Natesto's market share.
- Generic Entry Timeline: The exact date of patent expiration and the subsequent filing and approval of generic versions.
What are the Key Risks and Opportunities?
Risks:
- Generic Competition: The primary risk is the expiration of key patents and the subsequent market entry of lower-cost generic testosterone undecanoate nasal sprays.
- Nasal Side Effects: Potential for nasal discomfort, nosebleeds, or other local adverse events, which may deter some patients.
- Reimbursement Challenges: Insufficient payer coverage or restrictive formulary placement could limit patient access and sales.
- Competition from Novel Therapies: Development of new TRT formulations with superior efficacy, safety profiles, or more convenient dosing.
- Physician and Patient Inertia: Resistance from healthcare providers to prescribe a newer, less established therapy or from patients to switch from familiar treatments.
Opportunities:
- Growing Hypogonadism Awareness: Increasing diagnosis and treatment of testosterone deficiency offers a fundamental market growth opportunity.
- Preference for Non-Injectable TRT: A significant segment of the patient population actively seeks alternatives to injections, aligning with Natesto's administration.
- Improved Patient Compliance: The twice-daily nasal spray may offer better compliance for certain patient profiles compared to less frequent but more intrusive methods.
- Expansion into International Markets: Potential for approval and commercialization in regions outside the United States.
- Combination Therapies or New Formulations: While less probable for an established molecule, potential for future development leveraging the undecanoate ester.
Conclusion
Natesto occupies a distinct position in the testosterone replacement therapy market with its novel nasal delivery system. While it offers advantages in terms of convenience and administration compared to traditional injectables and topicals, it faces intense competition from a range of TRT options. The drug's financial trajectory is heavily influenced by its patent exclusivity period, which is set to expire in the coming years. Post-expiration, generic competition will undoubtedly lead to significant market share erosion and revenue decline. Strategic focus on maximizing market penetration and physician adoption during the remaining exclusivity period is critical for optimizing financial returns.
Key Takeaways
- Natesto addresses hypogonadism with a twice-daily nasal testosterone undecanoate spray, differentiating it from injections and topicals.
- The global TRT market is valued at approximately $3.4 billion and projected to reach $5.1 billion by 2030, driven by an aging population and increased diagnosis.
- Key patents protecting Natesto's formulation and method of use are expected to expire in the mid-to-late 2020s.
- Generic competition will significantly impact Natesto's revenue post-patent expiration.
- Near-term financial performance relies on market adoption and payer access; long-term outlook is constrained by impending generics.
Frequently Asked Questions
-
What are the primary side effects associated with Natesto?
Common side effects reported for Natesto include nasopharyngitis (cold-like symptoms), nasal discomfort, and epistaxis (nosebleeds) [2]. Some patients may also experience rhinitis and nasal pain.
-
How does Natesto's absorption compare to other testosterone formulations?
Natesto is designed for rapid absorption through the nasal mucosa, aiming for a more physiological testosterone profile throughout the day compared to some other modalities. Studies indicate that it can achieve and maintain therapeutic testosterone levels in most hypogonadal men [3].
-
What is the typical prescription volume and market share of Natesto?
Specific prescription volumes and market share data for Natesto are proprietary and fluctuate. However, it holds a niche within the broader TRT market, with its share influenced by physician prescribing habits, patient preferences, and competitive pressures from established and emerging therapies.
-
Are there any contraindications for using Natesto?
Yes, Natesto is contraindicated in men with known or suspected cancer of the prostate or breast, or a history of these conditions, as well as in men with severe lower urinary tract symptoms [2]. It is also contraindicated in pregnant or breastfeeding women and in individuals with known hypersensitivity to testosterone undecanoate.
-
What is the projected timeline for generic entry and its potential impact on Natesto's pricing?
Based on patent expiration in the mid-to-late 2020s, generic versions of testosterone undecanoate nasal spray are anticipated to enter the market shortly thereafter. Generic entry typically leads to a significant reduction in brand-name drug pricing, often by 50% or more, as competition intensifies.
Cited Sources
[1] Grand View Research. (2023). Testosterone Replacement Therapy Market Size, Share & Trends Analysis Report.
[2] Besins Healthcare, Inc. (2023). Natesto® (testosterone undecanoate) Prescribing Information. Retrieved from [FDA official website or manufacturer website].
[3] Ramasamy, R., Kiguradze, T., & Chen, J. (2017). Testosterone Undecanoate Nasal Spray for the Treatment of Hypogonadism. Therapeutic Advances in Urology, 9(3), 115–120. https://doi.org/10.1177/1756287217691084
[4] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/drugobse/index.cfm
[5] U.S. Food & Drug Administration. (2015, March 3). FDA approves Natesto (testosterone undecanoate) nasal spray for testosterone replacement therapy. [Press Release].
[6] IQVIA. (2023). Global Pharmaceutical Market Overview and Forecasts. (Note: Specific TRT pricing data derived from market intelligence reports; exact figures vary by geography and payer).