Last updated: February 19, 2026
This analysis examines Pfizer's current market standing, core competencies, and strategic directions within the pharmaceutical industry, focusing on patent expirations, pipeline developments, and recent M&A activity. Pfizer's portfolio, while facing patent cliffs for key products, is supported by a robust pipeline and strategic acquisitions aimed at future growth.
What is Pfizer's Current Market Position?
Pfizer is a leading global pharmaceutical company with a diversified portfolio across multiple therapeutic areas. Its market position is characterized by the significant revenue generated by its blockbuster drugs, alongside the ongoing challenge of managing patent expirations and the need to replenish its product offerings.
Key Revenue Drivers and Patent Expirations
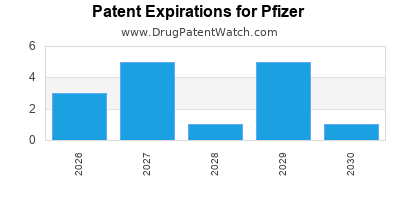
Pfizer's top revenue-generating products, such as Comirnaty (COVID-19 vaccine) and Paxlovid (COVID-19 antiviral), have seen substantial sales but are subject to market fluctuations and evolving public health needs. The company is also navigating patent expirations for other significant drugs.
| Product Name |
Therapeutic Area |
Estimated 2023 Revenue (USD Billion) |
Key Patent Expiration Year (US) |
| Comirnaty |
Infectious Disease |
~20-25 (Varies significantly by quarter) |
Ongoing regulatory approvals and market demand |
| Paxlovid |
Infectious Disease |
~10-15 (Varies significantly by quarter) |
Ongoing regulatory approvals and market demand |
| Eliquis (co-developed with Bristol Myers Squibb) |
Cardiovascular |
~6.4 |
2031 |
| Prevnar 13/Pneumococcal 13-valent Conjugate Vaccine |
Vaccine |
~3.9 |
2026 |
| Xeljanz |
Immunology |
~2.5 |
2027 |
Source: Pfizer Investor Relations, internal estimates based on market trends.
The expiration of patents for major drugs exposes these products to generic competition, leading to significant revenue erosion. Pfizer's strategy involves a proactive approach to portfolio management, including the development of next-generation products and strategic acquisitions to mitigate these impacts.
Geographic Reach and Market Share
Pfizer operates in over 120 countries. Its market share varies by therapeutic area and region, with strong positions in oncology, inflammation and immunology, vaccines, and internal medicine. Emerging markets represent a growth opportunity, though penetration is influenced by pricing, regulatory environments, and healthcare infrastructure.
What are Pfizer's Core Strengths?
Pfizer's strengths lie in its extensive research and development capabilities, global commercial infrastructure, and a proven track record in bringing novel therapies to market.
Research and Development Prowess
Pfizer invests heavily in R&D, maintaining a broad pipeline across various therapeutic areas. The company employs a multi-pronged R&D strategy that includes internal discovery, external collaborations, and strategic acquisitions of innovative technologies and early-stage assets.
- Pipeline Breadth: Pfizer's pipeline includes over 90 investigational medicines across 7 therapeutic areas. This diverse pipeline aims to address unmet medical needs and capitalize on emerging scientific advancements.
- Advanced Modalities: The company is actively developing therapies using novel modalities such as mRNA, gene therapy, and antibody-drug conjugates (ADCs), positioning it for future market leadership in innovative treatments.
- Clinical Trial Expertise: Pfizer has extensive experience in conducting large-scale global clinical trials, enabling efficient development and regulatory approval processes.
Global Commercial Infrastructure
The company possesses a formidable global commercial infrastructure, which is critical for the successful launch and widespread adoption of new medicines.
- Sales and Marketing Network: A vast network of sales representatives and marketing teams in key markets ensures effective product promotion and physician engagement.
- Supply Chain and Distribution: Pfizer's robust supply chain and distribution capabilities enable the timely and reliable delivery of its products worldwide, a critical factor in market access and patient care.
- Market Access and Reimbursement: Extensive experience in navigating complex market access and reimbursement landscapes in different countries facilitates patient access to its medicines.
Strategic Acquisitions and Partnerships
Pfizer has a history of leveraging acquisitions and partnerships to enhance its portfolio and pipeline.
- Seagen Acquisition: The acquisition of Seagen Inc. for approximately $43 billion (announced March 2023) significantly bolsters Pfizer's oncology pipeline with Seagen's leading antibody-drug conjugate (ADC) technology. This move is central to Pfizer's strategy to establish leadership in oncology.
- BioNTech Partnership: The long-standing collaboration with BioNTech SE for the development and commercialization of the COVID-19 vaccine has been a major success and serves as a model for future partnerships.
- Other Strategic Deals: Pfizer has engaged in numerous smaller collaborations and licensing agreements to access novel drug candidates and technologies.
What are Pfizer's Strategic Imperatives?
Pfizer's strategic focus is on achieving sustainable growth by capitalizing on its R&D investments, expanding its oncology business, and navigating the evolving healthcare landscape.
Oncology as a Growth Engine
Oncology is a key strategic priority for Pfizer, driven by the high unmet need and the potential for significant market growth. The acquisition of Seagen is instrumental in this strategy.
- ADC Expansion: Pfizer aims to become a leader in ADCs, a class of drugs that targets cancer cells with high precision, minimizing damage to healthy tissues. Seagen's portfolio includes approved ADCs and a promising pipeline.
- Portfolio Diversification: Beyond ADCs, Pfizer is investing in other oncology modalities, including small molecules, biologics, and immunotherapy combinations, to create a comprehensive oncology offering.
- Targeted Therapies: The company is focused on developing targeted therapies that address specific genetic mutations or biomarkers in cancer, offering personalized treatment approaches.
Portfolio Renewal and Pipeline Acceleration
Pfizer is committed to accelerating the development and commercialization of its R&D pipeline to offset revenue losses from patent expirations.
- Focus on High-Unmet-Need Areas: R&D efforts are concentrated on therapeutic areas with significant unmet medical needs, such as rare diseases, autoimmune disorders, and neurodegenerative diseases, alongside oncology and infectious diseases.
- Speed to Market: Pfizer emphasizes efficiency in clinical development and regulatory submissions to ensure timely access to new therapies for patients.
- Biologics and Advanced Therapies: Continued investment in the development of complex biologics, including gene therapies and cell therapies, is a cornerstone of its long-term innovation strategy.
Navigating the Evolving Healthcare Landscape
Pfizer is adapting its strategies to address challenges and opportunities presented by changes in healthcare policy, market access, and the competitive environment.
- Value-Based Pricing and Outcomes: The company is increasingly exploring value-based pricing models and demonstrating the real-world value of its medicines to payers and healthcare systems.
- Digital Health Integration: Pfizer is investing in digital health solutions to enhance patient engagement, improve treatment adherence, and gather real-world evidence for its products.
- Geographic Expansion: Strategic efforts are in place to expand its presence and market access in key emerging markets where significant patient populations and growth potential exist.
What are the Key Risks and Challenges?
Pfizer faces several risks and challenges, including intensifying competition, regulatory hurdles, and the inherent uncertainties of drug development.
Patent Expirations and Generic Competition
The expiration of patents for major revenue-generating drugs remains a significant threat.
- Revenue Erosion: Generic entry typically leads to a rapid and substantial decline in sales for originator drugs.
- Market Share Loss: Generic competitors often offer lower prices, making it difficult for originator products to maintain market share.
- Mitigation Strategies: Pfizer's strategy relies on launching "next-generation" products or biosimil versions before patent expiry and leveraging its pipeline to fill the revenue gap.
Clinical Trial Success and Regulatory Approval
The success of drug development is not guaranteed, with a high rate of attrition in clinical trials.
- High Failure Rates: Many drug candidates fail to demonstrate efficacy or safety in clinical trials, leading to significant R&D investment losses.
- Regulatory Scrutiny: Regulatory agencies worldwide are increasingly stringent in their review of new drug applications, requiring robust data and evidence.
- Post-Market Surveillance: Even after approval, drugs can face challenges related to unexpected side effects or safety concerns identified in post-market surveillance.
Competitive Landscape and Pricing Pressures
The pharmaceutical market is highly competitive, with significant pricing pressures from governments and payers.
- Rival Companies: Major pharmaceutical companies and emerging biotechs are actively developing therapies for the same diseases, intensifying competition.
- Government Regulations: Healthcare systems globally are implementing measures to control drug spending, leading to price negotiations and rebate systems.
- Public Scrutiny: Pharmaceutical pricing practices are often subject to public and political scrutiny, which can influence pricing strategies and market access.
Key Takeaways
Pfizer is navigating a dynamic market characterized by significant opportunities and challenges. Its strengths in R&D and global commercialization provide a solid foundation. The strategic acquisition of Seagen signals a strong commitment to oncology leadership. However, managing patent expirations and ensuring pipeline productivity remain critical for sustained long-term growth. The company's ability to innovate, adapt to evolving healthcare policies, and execute its strategic initiatives will determine its future market position.
Frequently Asked Questions
-
What is Pfizer's strategy for addressing upcoming patent cliffs on its key products?
Pfizer aims to mitigate the impact of patent expirations through the development and launch of "next-generation" therapies, biosimil products, and by strategically acquiring companies with promising pipelines, as exemplified by the Seagen acquisition in oncology.
-
How diversified is Pfizer's current R&D pipeline?
Pfizer's R&D pipeline is diversified across more than seven therapeutic areas, including oncology, immunology, internal medicine, vaccines, and rare diseases, with a significant focus on advanced modalities like mRNA and antibody-drug conjugates.
-
What are the primary therapeutic areas targeted by Pfizer's recent major acquisitions?
Pfizer's most significant recent acquisition, Seagen, is focused on bolstering its oncology portfolio, specifically in the area of antibody-drug conjugates.
-
What impact is the COVID-19 pandemic and its associated products having on Pfizer's long-term strategy?
While Comirnaty and Paxlovid provided significant revenue and accelerated the company's mRNA capabilities, Pfizer's long-term strategy is focused on diversifying beyond COVID-19 products, leveraging its R&D expertise in other therapeutic areas and advanced platforms for sustainable growth.
-
How is Pfizer addressing global pricing pressures and market access challenges?
Pfizer is adapting by focusing on value-based pricing models, demonstrating the clinical and economic benefits of its therapies, and engaging with payers and policymakers to navigate complex reimbursement landscapes in different global markets.
Citations
[1] Pfizer Investor Relations. (n.d.). Investor presentations and financial reports. Retrieved from [Official Pfizer Investor Relations Website]
[2] Bloomberg Terminal. (various dates). Company financial data and market analysis for Pfizer Inc.
[3] SEC Filings. (various dates). Pfizer Inc. Form 10-K and 10-Q filings. U.S. Securities and Exchange Commission.
[4] Press Releases. (various dates). Pfizer Inc. Official Press Releases.