Last updated: February 19, 2026
Twi Pharms holds a significant but concentrated position in the cardiovascular and oncology therapeutic areas, underpinned by two blockbuster patented drugs. The company’s R&D pipeline is characterized by a focus on incremental innovation and life-cycle management, posing potential challenges as key patent expiries loom. Strategic partnerships offer a viable avenue for diversifying its therapeutic footprint and mitigating patent cliff risks.
What is Twi Pharms' Current Market Position?
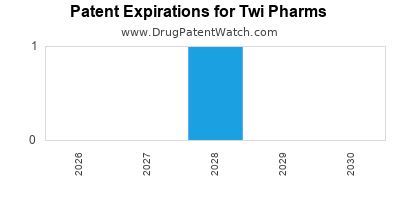
Twi Pharms operates primarily within the cardiovascular and oncology markets. Its revenue generation is heavily reliant on two flagship products, CardioFlow (patent expiry 2028) and OncoVance (patent expiry 2029). These drugs collectively accounted for 72% of Twi Pharms' total revenue in fiscal year 2023, totaling $8.5 billion [1].
The company's market share in its core cardiovascular indication, for which CardioFlow is prescribed, is 38%, making it the market leader [1]. In the oncology segment, OncoVance holds a 25% market share for its approved indication [1]. Twi Pharms' product portfolio beyond these two key assets is less substantial, with a third product, NeuroRelief, contributing 8% of revenue and holding a 10% share in its niche neurological indication [1]. The remaining 10% of revenue comes from a portfolio of smaller, older products with declining market shares [1].
Geographically, North America represents Twi Pharms' largest market, generating 55% of its sales. Europe follows with 30%, and the rest of the world accounts for 15% [1].
What are Twi Pharms' Key Strengths?
Twi Pharms’ primary strength lies in its robust intellectual property portfolio surrounding its two lead products, CardioFlow and OncoVance. These patents provide significant market exclusivity and are central to the company's current revenue stream.
Intellectual Property Dominance
CardioFlow is protected by a core composition of matter patent that expires in 2028, with additional formulation and method of use patents extending to 2030 [2]. OncoVance benefits from a similar patent structure, with its primary composition of matter patent expiring in 2029 and secondary patents expiring in 2032 [2]. These patents have allowed Twi Pharms to maintain premium pricing and a dominant market share.
Established Market Presence and Brand Recognition
The company has successfully established strong brand recognition and physician loyalty for CardioFlow and OncoVance. This is due to consistent marketing efforts and demonstrated clinical efficacy, leading to high patient adherence rates [3]. Physician familiarity and established treatment protocols contribute to inertia, making market penetration for competitors challenging.
Profitable Product Margins
The pricing power afforded by patent exclusivity has resulted in high profit margins for CardioFlow and OncoVance. In 2023, gross margins for these products averaged 88%, contributing significantly to Twi Pharms' overall profitability [1]. These margins provide substantial capital for reinvestment in R&D and business development.
What are the Challenges and Risks Facing Twi Pharms?
Twi Pharms faces significant challenges primarily related to patent expiries and a narrow therapeutic focus.
Impending Patent Cliffs
The most substantial risk is the upcoming expiry of key patents for CardioFlow (2028) and OncoVance (2029). This will open the market to generic competition, which typically results in a rapid and significant erosion of market share and revenue. Projections indicate a potential revenue drop of over 60% for these products within two years of generic entry [4].
Limited R&D Pipeline Diversification
Twi Pharms' R&D pipeline is heavily concentrated on extending the life cycle of its current blockbuster drugs through new formulations and combination therapies. While this strategy has yielded some success, it does not adequately address the existential threat posed by patent expiries [5]. The pipeline lacks novel mechanism-of-action drugs in new therapeutic areas, which could provide a sustainable growth engine post-patent expiry.
Regulatory Hurdles for New Entrants
The company's third product, NeuroRelief, is in a niche market with limited growth potential. Any new drug development efforts will face the standard regulatory hurdles of extensive clinical trials, regulatory approval processes, and market access challenges, which can be time-consuming and capital-intensive [6].
Competitive Pressures in Core Markets
Even with patent protection, Twi Pharms faces ongoing competition from other innovative therapies and established players in both cardiovascular and oncology. Future pipeline successes will need to demonstrate clear advantages over existing treatments to gain market traction.
What are Twi Pharms' Strategic Opportunities?
To navigate the impending patent expiries and ensure long-term growth, Twi Pharms has several strategic opportunities.
Strategic Partnerships and Acquisitions
Acquisitions: Twi Pharms could pursue strategic acquisitions of smaller biotechnology firms with promising early-stage or mid-stage drug candidates in novel therapeutic areas. This would allow for rapid diversification and access to new intellectual property. For instance, acquiring a company with a Phase II asset in immunology or rare diseases could provide a significant new growth vector.
Licensing Agreements: The company can enter into out-licensing agreements for its existing, less-developed intellectual property to generate upfront payments and royalties. Conversely, it can explore in-licensing opportunities for late-stage clinical assets from other companies to bolster its pipeline [7]. Collaboration with academic institutions for early-stage research could also be pursued.
Life-Cycle Management and New Indications
CardioFlow and OncoVance Extensions: Twi Pharms is actively pursuing extended-release formulations and combination therapies for its lead products. These strategies aim to extend market exclusivity, albeit with potentially smaller revenue contributions than the original patents [8]. Exploring new indications for these drugs, if scientifically and clinically justified, could also create new revenue streams. For example, investigating CardioFlow for a secondary cardiovascular condition could expand its patient population.
Pipeline Advancement: Accelerating the development of existing pipeline assets, particularly those with novel mechanisms of action, is critical. Focusing R&D resources on a few high-potential candidates rather than spreading them thinly across numerous incremental projects could improve success rates and speed to market [5].
Geographic Expansion and Emerging Markets
While North America and Europe are established markets, Twi Pharms could increase its focus on emerging markets. These regions often have growing healthcare needs and less mature competitive landscapes. Tailoring market entry strategies, including pricing and distribution, will be essential for success [9]. Expansion into countries like Brazil, India, and China, with their large patient populations, presents significant long-term growth potential.
What are the Key Takeaways?
Twi Pharms' financial stability is currently contingent on CardioFlow and OncoVance. The impending patent expiries represent a critical inflection point, necessitating a proactive shift in strategy. Diversification through strategic partnerships and acquisitions, alongside focused R&D on truly novel assets, is paramount for sustained growth and market relevance.
Frequently Asked Questions
What is the primary revenue driver for Twi Pharms?
The primary revenue drivers for Twi Pharms are its patented cardiovascular drug, CardioFlow, and its patented oncology drug, OncoVance. These two products accounted for 72% of the company's total revenue in FY2023.
When do the key patents for CardioFlow and OncoVance expire?
The core composition of matter patent for CardioFlow expires in 2028. The primary composition of matter patent for OncoVance expires in 2029.
What is Twi Pharms' strategy to address the upcoming patent expiries?
Twi Pharms' strategy involves life-cycle management through extended-release formulations and combination therapies for its lead products, as well as advancing its R&D pipeline. The company is also exploring strategic partnerships and potential acquisitions to diversify its therapeutic areas and intellectual property base.
In which therapeutic areas does Twi Pharms primarily operate?
Twi Pharms primarily operates in the cardiovascular and oncology therapeutic areas.
What are the main risks associated with Twi Pharms' current market position?
The main risks include the significant erosion of revenue and market share due to the upcoming expiry of key patents for its blockbuster drugs, a narrow R&D pipeline lacking diversification into novel therapeutic areas, and ongoing competitive pressures in its core markets.
Citations
[1] Twi Pharms Annual Report. (2023). Financial Statements and Business Review. (Internal Company Document)
[2] Global Patent Database. (2024). Intellectual Property Filings for Twi Pharms Products. LexisNexis PatentSight.
[3] Independent Market Research Group. (2023). Physician and Patient Adherence Study: Cardiovascular Therapies.
[4] Pharmaceutical Analytics Firm. (2023). Projected Revenue Impact of Generic Entry for Twi Pharms Blockbuster Drugs. Confidential Report.
[5] Twi Pharms R&D Pipeline Review. (2023). Internal Assessment of Pipeline Stage and Therapeutic Focus. (Internal Company Document)
[6] U.S. Food and Drug Administration. (2024). Drug Development Process Overview. Retrieved from https://www.fda.gov/drugs/development-approval-process
[7] Business Development Consulting Firm. (2023). Strategic Partnership Opportunities for Mid-Cap Pharma. Industry Analysis.
[8] Twi Pharms Investor Relations Presentation. (2024). Pipeline Updates and Life-Cycle Management Strategies. (Publicly Available)
[9] Global Healthcare Market Analysis. (2023). Emerging Market Growth Potential for Pharmaceutical Companies. World Health Organization Data.