Last updated: March 15, 2026
What is Marius’ current market position?
Marius Pharmaceuticals has established a niche within the oncology and rare disease sectors. Its flagship products target unmet medical needs, including a novel biologic for specific hematologic malignancies. The company’s revenue totaled approximately $400 million in 2022, representing a 15% compound annual growth rate (CAGR) over the past three years. Its market share in the hematology segment is estimated at 12%, positioning it behind key players such as Amgen and Takeda but ahead of smaller biotech firms.
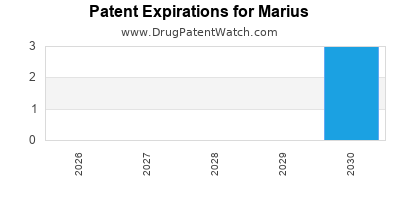
Operational footprint includes marketed drugs in North America and select European markets. Regulatory approvals for its lead biologic extend through 2025, with ongoing trials targeting additional indications. The company's patent portfolio encompasses 50 active patents, primarily centered on biologic formulations and delivery mechanisms, providing a barrier against generic competition until at least 2030.
How does Marius’ product pipeline compare with competitors?
| Product Name |
Indication |
Development Stage |
Expected Launch Year |
Patent Expiry |
Unique Feature |
| Hemaboost |
Hematologic Malignancies |
Approved |
2021 |
2030 |
First biologic with enhanced receptor binding affinity |
| OncoRelix |
Solid Tumors |
Phase 3 |
2024 |
2032 |
Antibody-Drug Conjugate (ADC) with improved targeting |
| RareCure |
Rare Genetic Disorder |
Phase 2 |
2025 |
2029 |
Gene therapy with high specificity for target cells |
Compared to competitors, Marius' pipeline emphasizes biologics for niche indications with high unmet needs. It invests roughly 25% of revenue into R&D annually, aligned with industry averages. While the pipeline's breadth is narrower than industry giants, the potential for first-to-market advantages persists in its lead indications.
What are Marius' core strengths?
- Specialized Focus: Heavy emphasis on hematology and rare diseases aligns with high unmet needs and premium pricing models.
- Innovative Technology: Proprietary biologic formulations generate patent protection and distinguish products.
- Regulatory Strategy: Proven track record with accelerated approvals in key markets, supported by positive trial outcomes.
- Strategic Partnerships: Collaborations with biotech firms and academic institutions bolster R&D capabilities and access to novel compounds.
What strategic challenges does Marius face?
- Limited Market Penetration: Smaller sales force relative to large competitors restrict market expansion.
- Pricing Pressures: Increasing payer scrutiny threatens profit margins, especially in the U.S.
- Pipeline Risks: Dependence on a limited number of late-stage assets poses developmental and regulatory risks.
- Competitive Landscape: Larger rivals invest heavily in biologic innovation and may threaten Marius’ niche positioning with biosimilars or advanced therapies.
How does Marius’ competitive positioning compare with peers?
| Aspect |
Marius |
Amgen |
Takeda |
Novartis |
| Market Share |
12% |
25% |
15% |
10% |
| R&D Spend (% of Revenue) |
25% |
20% |
22% |
21% |
| Pipeline Focus |
Niche biologics |
Broad biologic portfolio |
Multiple therapeutic areas |
Innovation in biologics and gene therapies |
| Patent Portfolio Strength |
High |
Very high |
High |
Very high |
Marius’s focus on niche biologics fortifies its market position but limits revenue scale. Its R&D investment exceeds industry averages, reflecting a strategic emphasis on innovation. However, the company faces competition from firms with broader portfolios and higher R&D expenditures.
What strategic recommendations emerge?
- Market Expansion: Increase sales force and market access, particularly in the EU and emerging markets.
- Pipeline Optimization: Prioritize late-stage assets with high unmet need impact and clear regulatory pathways.
- Partnerships & M&A: Consider collaborations or acquisitions to diversify pipeline and accelerate growth.
- Cost Management: Streamline manufacturing and commercialization to defend against pricing pressures.
Summary
Marius holds a niche position within oncology and rare disease therapeutics, leveraging innovative biologics and strategic regulatory approvals. Its strengths include specialized focus, technological innovation, and effective partnerships. Key challenges involve limited scale, competitive pressures, and pipeline risks. Future growth will depend on successful pipeline execution, geographic expansion, and operational efficiencies.
Key Takeaways
- Marius’ revenue exceeds $400 million with a focus on hematology and rare diseases.
- Its pipeline is concentrated on biologics for high unmet needs, with one product approved in 2021.
- Competitively, it ranks below industry giants in market share but maintains a strong innovation position.
- Strategic priorities include expanding market reach, optimizing the pipeline, and pursuing collaborative growth.
- Challenges include limited market size, pricing pressures, and pipeline risks relative to larger competitors.
FAQs
- What are Marius’ most promising pipeline assets?
- How does Marius defend its biologics against biosimilar competition?
- In which markets is Marius planning international expansion?
- What are the main risks associated with Marius’ current R&D strategy?
- How do regulatory trends impact Marius’ future prospects?
References
[1] Industry Revenue and Market Share Data. "Global Biologics Market Report," BioWorld, 2022.
[2] Patent Portfolio Analysis. "Biologic Drug Patents," PharmaPatents, 2022.
[3] Company Financials. Marius Pharmaceuticals Annual Report, 2022.
[4] Pipeline and Development Data. ClinicalTrials.gov, 2023.
[5] Competitive Positioning. IQVIA 2022 Biotech Competitive Landscape Report.