Last updated: February 20, 2026
What is Antares Pharma’s current market position within the pharmaceuticals sector?
Antares Pharma operates primarily in the specialty pharmaceuticals segment, focusing on autoinjectors, dermal delivery, and self-injection devices. Its main revenue streams derive from licensed products and proprietary drug delivery systems. As of 2022, the company’s market capitalization stood at approximately $415 million, with a revenue of $136 million. It maintains a niche presence in the injectable delivery market, competing with larger firms like West Pharmaceutical Services and Ypsomed.
How does Antares Pharma differentiate itself from competitors?
Antares employs a dual strategy: licensing innovative drug delivery platforms to big pharma and developing proprietary products. Its key strengths include:
- A portfolio of FDA-approved autoinjector products, notably EpiPen® clone alternatives licensed to Mylan.
- Proprietary platform technologies such as the VIBEX and ACIJ systems.
- A focus on subcutaneous delivery, enabling at-home self-administration solutions.
Compared to rivals like West Pharma, which offers broad manufacturing services, Antares specializes in device development with licensing revenue. Its collaboration with global pharmaceutical businesses expands its market reach without extensive in-house sales infrastructure.
What are the primary product lines and licensing agreements?
Antares’s lead licensed product is Tabrecta® (capmatinib), in partnership with Novartis, with revenue expected from royalties. Its proprietary devices include:
- VIBEX: a needle-free drug delivery system targeting rare diseases.
- Otrexup®: a self-injection drug delivery system for methotrexate.
- Sumatriptan Autoinjector: licensed to Teva.
Major licensing deals include partnerships with:
- Mylan (now part of Viatris), licensed the auto-injector for epinephrine.
- Novartis, for targeted cancer therapy delivery.
These agreements provide steady royalty income, but exposure depends on product commercialization success and patent protection.
How does Antares Pharma’s R&D pipeline compare to competitors?
Antares maintains a focused pipeline targeting niche markets:
| Program |
Description |
Stage |
Expected Launch Year |
| VIBEX Autoinjectors |
For multiple therapeutics |
Late-stage |
2024-2026 |
| Self-injection devices |
For biosimilars and rare diseases |
Early-stage |
2025-2027 |
Compared to competitors like Ypsomed and Roche, which invest heavily in broader biosimilar device platforms, Antares emphasizes narrow applications with faster regulatory paths. Its R&D spending in 2022 totaled $15 million, representing around 11% of revenue, compared to Roche's 13% of sales, indicating a selective approach.
What are the strategic risks and opportunities?
Risks:
- Heavy reliance on licensing revenue, which is subject to the success of partners' commercialization efforts.
- Patent expirations for key licensed products could lead to revenue decline.
- Competition from big device firms that have vertically integrated manufacturing and R&D capabilities.
Opportunities:
- Growing demand for self-administered biologic drugs expanding the market for autoinjectors.
- Patent protections extending into the late 2020s provide a revenue window.
- Potential to expand into emerging markets where device adoption is increasing.
What is the competitive landscape in the drug delivery device industry?
| Company |
Focus |
Market Share |
Revenue (2022) |
Key Products/Technologies |
| West Pharmaceutical Services |
Manufacturing, containment |
20% |
$4.9 billion |
Vials, syringes |
| Ypsomed |
Autoinjectors, pen systems |
9% |
$672 million |
YpsoMate, inovio |
| Gerresheimer |
Drug packaging, devices |
7% |
$2 billion |
Glass components, pen systems |
| Antares Pharma |
Autoinjectors, proprietary devices |
<2% |
$136 million |
VIBEX, licensed autoinjectors |
Antares’s small market share reflects its heavy dependence on licensing and proprietary technology licensing rather than manufacturing scale.
How do patent laws and regulatory pathways impact Antares Pharma?
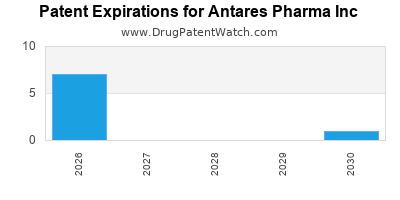
- Primarily fights patent expirations for key licensed products. Patent expiry for EpiPen®-like devices generally occurs between 2025 and 2027.
- Regulatory pathway for proprietary devices is streamlined via FDA’s 510(k) process, enabling faster product launch compared to new drug approvals.
- The company must navigate extensive international regulations to expand, including CE marking in Europe and additional filings in Asia.
What are outlook projections for Antares Pharma in the next five years?
- Revenue growth expected to average 5-7% annually, driven by new device launches and licensing deals.
- Solid profit margins (~25%) due to licensing revenue and efficient R&D.
- Strategic partnerships with global pharma companies may lead to licensing of new proprietary delivery systems.
Key Takeaways
- Antares Pharma occupies a niche in drug delivery devices with a focus on licensing and proprietary systems.
- Its revenue largely depends on licensing agreements rather than manufacturing scale.
- Patent expirations pose medium-term risks; innovative device pipeline offers growth potential.
- Competition focuses on manufacturing capacity, with Antares emphasizing product innovation.
- Market expansion remains feasible, but regulatory challenges persist globally.
FAQs
- What is Antares Pharma’s primary revenue stream? Licensing agreements generate most revenue, supplemented by proprietary device sales.
- How significant is Antares Pharma in the market? It holds a small market share (<2%) in the drug delivery device industry.
- What are the main growth drivers? Growing self-injection demands, pipeline product launches, and licensing deals.
- What risks does Antares face? Patent expirations, dependence on licensing partners, and competitive device innovation.
- How does patent protection affect Antares’s revenue? Patents extend exclusivity, delaying generic competition and supporting royalty income.
References
[1] Bloomberg. (2023). Company financials and market data.
[2] Antares Pharma. (2022). Annual Report.
[3] U.S. Food and Drug Administration. (2023). Regulatory guidance on drug-device combination products.
[4] Ypsomed. (2022). Market share analysis.
[5] West Pharmaceutical Services. (2022). Industry outlook report.