Biosimilar-to-Biosimilar Switching: The Complete IP, Clinical, and Market Playbook
For pharma IP teams, portfolio managers, R&D leads, and institutional investors navigating the cross-switching era.
I. What Biosimilar-to-Biosimilar Switching Actually Is — and Why It Matters Now
Defining Cross-Switching with Precision
Biosimilar-to-biosimilar switching — also called cross-switching — is the practice of transitioning a patient from one approved biosimilar to a different biosimilar referencing the same originator biologic. It is not originator-to-biosimilar switching, which is the scenario that most clinical trial data, regulatory guidance, and published literature address. The distinction matters enormously for IP teams and clinicians alike, because the regulatory frameworks governing the two scenarios are categorically different, and the evidentiary requirements for each diverge sharply.
Cross-switching occurs for at least four distinct reasons. Payers mandate it to extract lower net prices through formulary exclusions or preferred-product contracting. Clinicians initiate it when a patient experiences tolerability issues — injection site reactions, citrate-related burning — that are excipient-specific rather than molecule-specific. Patients request it for device reasons, including needle gauge, auto-injector ergonomics, or the availability of latex-free presentations. Health systems execute it en masse when a new tender cycle changes the preferred supplier without changing the therapeutic molecule. Each driver implies a different risk profile, a different communication burden, and a different IP exposure for the product losing share.
As of late 2025, the global biosimilar market carries projected CAGRs between 17.3% and 25.5% through the late 2020s. That growth rate compresses into one implication: the number of cross-switching decisions executed annually will rise faster than the clinical guidance infrastructure designed to manage them. Clinicians, payers, and manufacturers are all operating ahead of the formal evidence base. That is the central tension this document addresses.
Why the Regulatory Gap Is Structurally Permanent
Neither the FDA nor the EMA requires — or anticipates requiring — dedicated randomized controlled trials for biosimilar-to-biosimilar comparisons. This is not an oversight. The regulatory logic is explicit: if Product A is biosimilar to Reference R, and Product B is biosimilar to Reference R, then the transitivity of biosimilarity makes a direct A-vs.-B RCT scientifically redundant. The two products are each anchored to the same molecular reference point through extensive analytical, pharmacokinetic, and comparative immunogenicity characterization.
The consequence is that cross-switching will always lack the canonical Phase III switching study that physicians historically used as clinical permission. Real-world evidence is therefore not a supplement to trial data for this indication — it is the primary evidentiary category, and it will remain so indefinitely. Portfolio managers and R&D leads should treat RWE generation programs not as post-marketing niceties but as core commercial assets, comparable in strategic value to a Phase IIIb extension study for a novel biologic.
Key Takeaways — Section I
The absence of RCT data for biosimilar-to-biosimilar switching is a regulatory design feature, not a data gap. The market is moving faster than formal guidelines. Cross-switching volumes will accelerate with biosimilar market expansion, and the stakeholders who build robust RWE infrastructure earliest will hold durable competitive and formulary advantages.
II. The Regulatory Framework: FDA, EMA, and the Interchangeability Overhaul
FDA: From Switching Studies to ‘Totality of Evidence’
The FDA’s historical position required biosimilar sponsors seeking an ‘interchangeable’ designation to conduct dedicated switching studies — typically three-period crossover designs demonstrating that alternating between the biosimilar and the reference product produced no greater immunogenicity or loss of efficacy than continued reference product use. That requirement governed the first generation of U.S. interchangeability designations.
In June 2024, the FDA issued draft guidance proposing to eliminate the dedicated switching study requirement. The agency’s rationale cited accumulated data showing ‘effectively insignificant’ incremental risk from switching, as well as the maturation of analytical characterization techniques that can now establish biosimilarity at a level of molecular resolution that was not achievable a decade ago. Under the proposed framework, a sponsor seeking interchangeability can submit either historical switching study data or a comprehensive analytical-and-clinical comparative package demonstrating that the switching standard is met without new trial execution.
The FDA’s FY2025 budget proposal went further, requesting authority to amend the Public Health Service Act to deem all approved biosimilars interchangeable with their reference products by statute, without any additional evidentiary submission. As of Q2 2026, that legislative proposal has not been enacted, but it signals the directional intent of U.S. biosimilar policy. If enacted, it removes the interchangeability designation as a market differentiator entirely — every approved biosimilar becomes substitutable at the pharmacy counter, subject to state law.
As of November 2023, the FDA had approved 44 biosimilars across 14 reference products, with 13 carrying interchangeable designation. Nine of those 13 interchangeability approvals occurred without switching study data, already applying a ‘totality of evidence’ standard before the June 2024 draft guidance formalized the approach.
EMA: The Mature Position the FDA Is Converging Toward
The EMA, in its September 2022 joint statement with the Heads of Medicines Agencies, stated that EU-authorized biosimilars are interchangeable with their reference products and with each other, and that there is ‘no limit’ on the number of times a patient can be switched among biosimilars of the same reference product. This position rests on two pillars. The first is the rigorous comparability exercise required for initial EU authorization. The second is the accumulated real-world experience across EU member states — over one million patient-treatment years of safety data, across multiple therapeutic areas and reference products, without systematic evidence of harm attributable to switching.
The EMA position is also explicit that molecular complexity does not modify the interchangeability principle. Monoclonal antibodies, fusion proteins, and other complex biologics are all covered by the same framework. Individual EU member states retain authority over pharmacy-level substitution policy, meaning the EMA statement governs the science while national formulary and substitution rules govern the commercial execution.
State-Level Fragmentation in the U.S. — A Persistent Operational Complexity
Even with the FDA’s proposed statutory interchangeability expansion, pharmacy-level substitution in the U.S. remains a state-by-state issue. State substitution laws vary on notification requirements, patient consent, prescriber override rights, and documentation obligations. For biosimilar manufacturers, this fragmentation means that FDA interchangeability designation does not translate into uniform pharmacy substitution access — commercial teams must map state-specific substitution law as part of launch planning, and those maps must be updated as state legislatures revise their biologic substitution statutes.
For cross-switching specifically, the state law framework matters less, because cross-switches are typically payer-driven or prescriber-initiated rather than pharmacist-substituted. But if the FDA’s proposed statutory change passes, it would convert most biosimilar-to-biosimilar transitions from prescriber-managed events into pharmacist-executable substitutions in states with permissive laws, fundamentally changing the commercial and clinical management model.
Regulatory Technology Roadmap: What Comes Next
The trajectory of biosimilar regulatory science points in a consistent direction. Advanced analytical characterization — including high-resolution mass spectrometry for glycoform profiling, hydrogen-deuterium exchange mass spectrometry for higher-order structure assessment, and single-molecule imaging — is replacing clinical trial data as the primary basis for biosimilarity determinations. The FDA’s Critical Quality Attributes (CQA) framework, which links analytical fingerprinting to functional activity, will likely expand in scope, further compressing the clinical development burden for biosimilars.
For cross-switching, this means the regulatory evidence base will increasingly be analytical and computational rather than clinical. Sponsors that invest in advanced characterization platforms now are building regulatory capital that will accelerate future biosimilar approvals and interchangeability designations. From an IP standpoint, novel analytical methods may themselves be patentable, creating a secondary layer of proprietary advantage within the biosimilar development process.
Key Takeaways — Section II
The FDA is converging toward the EMA’s science-based, trial-free interchangeability standard. The proposed statutory change to deem all biosimilars interchangeable would eliminate interchangeability as a commercial differentiator and shift competitive dynamics entirely toward price, formulation, device, and support programs. U.S. state law fragmentation remains the practical constraint on pharmacy-level substitution regardless of federal regulatory posture.
Investment Strategy — Section II
Sponsors holding interchangeable biosimilar designations as a moat should model scenarios where that moat disappears. If the PHS Act amendment passes, the value of interchangeability-specific IP (e.g., switching study data packages, interchangeability-designated BLAs) declines sharply. Reallocation toward formulation IP, device design patents, and patient support infrastructure becomes the logical hedge.
III. IP Landscape and Patent Thicket Exposure by Reference Product
The Patent Thicket Problem: Mechanism and Scale
A patent thicket is a dense cluster of overlapping patents — covering the molecule, manufacturing process, formulation, delivery device, dosing regimen, and individual indications — that a reference biologic sponsor constructs around a core asset to delay biosimilar entry beyond primary patent expiration. AbbVie’s adalimumab (Humira) is the most extensively documented case. AbbVie built a portfolio of over 130 U.S. patents around adalimumab, covering citrate-free formulations, high-concentration subcutaneous delivery, and specific auto-injector designs. This portfolio delayed U.S. biosimilar entry until 2023, despite European adalimumab biosimilars entering market in 2018. The resulting price differential — European adalimumab biosimilars achieving 85% discounts by 2019 through competitive tendering, while U.S. net prices remained elevated through the litigation period — represents one of the most quantifiable examples of IP strategy suppressing biosimilar cross-switching opportunity.
For biosimilar-to-biosimilar switching specifically, the thicket problem has a secondary dimension. A biosimilar entrant that differentiates itself through a citrate-free formulation — a clinically meaningful attribute driving switch demand — may itself hold formulation patents that restrict competitors from replicating that specific tolerability advantage. Cross-switching among adalimumab biosimilars in the U.S. is not just a clinical or payer decision; it is shaped by which formulation patents each entrant holds and whether citrate-free status is exclusively associated with a particular product’s IP position.
The ‘Patent Dance’ Under the BPCIA: Strategic Leverage Points
The Biologics Price Competition and Innovation Act (BPCIA) established the ‘patent dance’ — a structured information exchange between reference product sponsors and biosimilar applicants governing which patents are litigated and in what sequence. The dance has two phases. In the first, the biosimilar applicant shares its Biologics License Application with the reference sponsor, triggering a 60-day window in which the reference sponsor identifies patents it believes would be infringed. The parties negotiate which of those patents to litigate immediately. In the second phase, the reference sponsor can assert additional patents 30 days before commercial launch.
The strategic implications for cross-switching are specific. A biosimilar entrant that navigates the patent dance successfully and launches commercially becomes a potential cross-switch target. Its launch price, device characteristics, and formulary positioning all influence whether payers and prescribers direct cross-switches toward or away from it. Entrants that resolve the patent dance through settlement — rather than litigation to judgment — may obtain launch rights while paying royalties that compress their pricing flexibility, directly affecting their competitiveness in cross-switch scenarios.
IP Valuation Framework for Biosimilar Assets
For M&A and portfolio management purposes, a biosimilar asset’s IP value is a composite of at least five components. The first is freedom-to-operate: the degree to which the biosimilar’s manufacturing process, formulation, and device avoid active reference product patents. The second is proprietary manufacturing process IP: the extent to which the biosimilar’s cell line selection, bioreactor conditions, purification sequences, and quality control methods are themselves patented, creating barriers to generic biosimilar competition. The third is formulation IP: patents covering excipient composition, buffer systems, concentration, or pH that affect stability, tolerability, or shelf life. The fourth is device IP: patents on auto-injector design, prefilled syringe specifications, or novel delivery mechanisms. The fifth is data exclusivity: the 12-year reference product exclusivity period under the BPCIA, and analogous periods under EMA law, that restricts when biosimilar regulatory submissions can be made.
Each of these IP layers affects cross-switching economics differently. Formulation and device IP most directly influence which biosimilar a payer selects when executing a formulary cross-switch, because they govern the product attributes — citrate-free status, injector ergonomics — that distinguish otherwise biosimilar-equivalent options.
Adalimumab IP Valuation Case Study
Adalimumab is the highest-revenue biologic in history, with global peak sales exceeding $20 billion annually. Its IP architecture is instructive for understanding cross-switching market structure. AbbVie’s core composition-of-matter patent expired in 2016 in Europe and 2023 in the U.S. following settlement agreements with multiple biosimilar entrants. As of 2025, the U.S. adalimumab market has seven or more approved biosimilars, several carrying citrate-free formulations. AbbVie’s Humira High Concentration citrate-free formulation was itself developed as an evergreening strategy, patented separately from the original molecule.
In the cross-switching context, Coherus BioSciences’ Yusimry, Organon’s Hadlima (citrate-free version), Boehringer Ingelheim’s Cyltezo (the first FDA-interchangeable adalimumab biosimilar), and Samsung Bioepis’s Hadlima SD and Hyrimoz by Sandoz all occupy different positions in the patent landscape. Cyltezo’s interchangeability designation — achieved with switching study data — gives it pharmacy substitution access in permissive states. Formulation distinctions among the citrate-free products create the primary clinical rationale for biosimilar-to-biosimilar cross-switching within this reference product class. Understanding which entrants hold formulation patents versus which are operating under license or in freedom-to-operate positions is essential for predicting cross-switch dynamics over the next formulary cycle.
Infliximab IP Valuation Case Study
Infliximab (Remicade, Janssen/J&J) entered biosimilar competition earlier in Europe, with Celltrion’s Remsima and Pfizer’s Inflectra launching in 2015. The U.S. market followed, with CT-P13 SC (Remsima SC) adding a subcutaneous route of administration that the originator IV formulation does not offer — a formulation and delivery strategy generating new patent coverage around a molecule whose primary IP has substantially expired. This is a textbook evergreening-adjacent maneuver: the biosimilar entrant acquires IP for a formulation innovation that the reference product sponsor has not deployed, creating switching rationale between the IV-administered originator biosimilars and the SC-administered CT-P13 SC.
For cross-switching among infliximab biosimilars, the route-of-administration distinction is the primary clinical driver. A patient stable on IV infliximab biosimilar who transitions to SC CT-P13 represents a cross-switch with distinct pharmacokinetic and immunogenicity implications. Clinical data on this specific transition, while limited, support the safety of the switch; the NOR-SWITCH trial, though examining the originator-to-biosimilar transition, established the foundational principle of non-inferiority in the infliximab class. Real-world data from Scandinavian registries have extended this confidence to multiple switches within the class.
Key Takeaways — Section III
Patent thickets delay biosimilar entry and compress cross-switching opportunity within a product class. Formulation and device IP — not the core molecule — is the primary IP layer governing cross-switch competitive dynamics. Adalimumab’s citrate-free landscape and infliximab’s IV-vs.-SC formulation distinction are the two most commercially significant current examples. IP valuation for biosimilar assets must account for all five IP component layers; composition-of-matter expiry alone does not define competitive exposure.
Investment Strategy — Section III
Biosimilar acquirers should conduct full IP audits at the formulation and device layer, not just at the molecule level. Assets with proprietary citrate-free or buffer-free formulation patents in high-prevalence autoimmune reference product classes carry durable competitive moats independent of molecule patent status. Conversely, biosimilar programs launching ‘me-too’ formulations against already-differentiated competitors in the same class face compressed pricing power and limited cross-switch appeal.
IV. Clinical Evidence: What the Data Actually Show
Foundational Biosimilarity Evidence and Its Transitivity
Every approved biosimilar has cleared a rigorous comparability exercise against its reference product: analytical characterization of primary structure, higher-order structure, glycosylation, charge variants, and oxidation profiles; pharmacokinetic and pharmacodynamic studies demonstrating equivalent exposure and target engagement; and head-to-head immunogenicity assessment with anti-drug antibody (ADA) profiling. This package, which the FDA and EMA both require, establishes that the biosimilar and reference product are highly similar with no clinically meaningful differences in safety, purity, or potency.
The transitivity argument for cross-switching follows directly: if A ≈ R and B ≈ R, then A ≈ B, where the equivalence is defined by the shared reference point. This is not a loose assumption — it is grounded in the same analytical characterization data that earned each biosimilar its approval. The practical question for clinical teams and payers is not whether the science supports cross-switching, but how to operationalize it with acceptable residual uncertainty about patient-level immunogenicity trajectories and device-specific tolerability.
Meta-Analytic Evidence: What 31 Studies and 44 Switch Periods Show
A FDA-reviewed meta-analysis covering 44 switch treatment periods from 31 studies across eight reference products found no statistically significant differences in mortality, serious adverse events, or treatment discontinuation rates between patients who switched between a biosimilar and its reference product and those who maintained continuous therapy. Incidence of ADAs and neutralizing antibodies was comparable between groups. These findings held irrespective of reference product class, the direction of the switch, and whether the switch was performed once or multiple times.
This is the bedrock evidentiary foundation for cross-switching. If single and multiple originator-biosimilar switches produce no measurable harm at the population level across eight reference product classes, the scientific basis for anticipating harm from biosimilar-to-biosimilar switches — where both products are equally similar to the same reference — is thin. The FDA cited this evidence base explicitly in its June 2024 draft guidance as justification for removing the dedicated switching study requirement.
Cross-Switching-Specific Data: The Systematic Review Finding
A systematic review focused specifically on biosimilar-to-biosimilar switching, published in a peer-reviewed journal, analyzed data across infliximab, adalimumab, etanercept, and rituximab biosimilars. The authors concluded that available data indicate cross-switching is safe and effective, with no evidence of loss of effectiveness or increased adverse events compared to maintaining continuous biosimilar therapy. The review acknowledged that the evidence base is thinner than for originator-biosimilar switching, that most studies are observational rather than randomized, and that immunogenicity reporting is inconsistent across studies. These limitations do not reverse the directional conclusion — they set the research agenda for the next five years of RWE collection.
Specific efficacy data from cross-switch cohorts: infliximab biosimilar cross-switches showed clinical remission maintenance at approximately 75% at six months post-switch; adalimumab biosimilar cross-switches maintained remission at approximately 82% at six months. These rates are consistent with remission maintenance rates reported for originator-to-biosimilar transitions in the same therapeutic areas, providing additional support for the transitivity argument.
The Derikx Double-Switch Dataset
The Derikx et al. (2021) retrospective cohort study examined a clinically rare but commercially important scenario: patients with inflammatory bowel disease who underwent a double biosimilar switch — from adalimumab originator to biosimilar SB5 (Imraldi, Samsung Bioepis), and subsequently to ABP 501 (Amgevita, Amgen). The second switch was clinically driven in most cases by injection site tolerability issues with SB5, typically attributable to its citrate-containing formulation.
The study found stable adalimumab trough levels across both switches, no new detectable ADA emergence after the second switch, and zero adalimumab discontinuations following the second transition. This dataset is particularly valuable for IP and market access teams because it describes the exact mechanism driving citrate-based cross-switching: a formulation attribute creates tolerability differentiation within a biosimilar class, generates a clinically justified cross-switch, and the receiving product (citrate-free ABP 501 in this case) captures durable patient retention with no pharmacological loss. The business case for citrate-free formulation investment in adalimumab biosimilars is directly visible in this data.
Key Takeaways — Section IV
Meta-analytic evidence across 44 switch periods and eight reference product classes shows no statistically meaningful harm from switching. Cross-switch-specific systematic review data supports the same conclusion for infliximab, adalimumab, etanercept, and rituximab. The Derikx double-switch dataset is the most granular available evidence for the commercial and clinical dynamics of formulation-driven cross-switching. Remission maintenance rates post-cross-switch (75-82% at six months) are consistent with originator-to-biosimilar switch outcomes.
V. Immunogenicity: The Mechanism-Level Risk Analysts Must Model
ADA Cascade Mechanics in Cross-Switching
Anti-drug antibody (ADA) formation is the primary immunogenicity concern in any biologic switching scenario. When a patient transitions between two biosimilars of the same reference product, the immune system encounters a molecular entity that is highly similar — but not identical — to the previous product. Differences may exist in glycosylation patterns, charge variant distribution, host cell protein impurity profiles, and formulation excipients. Each of these attributes can theoretically influence immunogenicity.
The practical risk depends on whether the patient has already developed ADAs against the first biosimilar. Pre-existing ADAs directed against epitopes shared between the two biosimilars could theoretically neutralize the second product immediately. Pre-existing ADAs specific to an epitope unique to the first biosimilar might not cross-react with the second, potentially resulting in ADA clearance and immunological ‘re-naivety’ that actually reduces immunogenicity risk post-switch. The Derikx study’s observation of no new detectable ADAs after a double switch is consistent with this second scenario in at least a subset of patients.
Critically, current biosimilar cross-switch studies do not consistently report ADA prevalence at baseline (before the switch) or characterize whether post-switch ADAs are new versus pre-existing. This is the most significant methodological gap in the cross-switching evidence base. For high-stakes cross-switch programs — particularly those involving immunosuppressed oncology patients or patients on concurrent immunomodulators — baseline ADA status should be assessed before executing a payer-mandated transition.
Monitoring Protocols That Close the Evidence Gap
A rigorous cross-switch immunogenicity monitoring protocol includes at minimum: baseline ADA titer and antibody isotype characterization before the switch; ADA reassessment at four, 12, and 24 weeks post-switch; trough level monitoring at the same intervals; and correlation of ADA status with clinical outcomes scores (e.g., DAS28, Harvey-Bradshaw Index, or disease-specific metrics). For infliximab cross-switches, this protocol is feasible within existing therapeutic drug monitoring (TDM) infrastructure used by most academic gastroenterology and rheumatology centers. For adalimumab, TDM implementation is less uniform, creating an evidence collection opportunity for health systems with established monitoring programs.
Manufacturers whose cross-switch programs generate this level of immunogenicity characterization data have a direct path to differentiating their product in formulary negotiations: a documented safety profile for cross-switch patients, with ADA prevalence rates and trough level stability data, is a more credible formulary submission than a general claim of biosimilarity.
Key Takeaways — Section V
ADA formation is the primary immunogenicity mechanism in cross-switching. Pre-existing ADA status at the time of switch is the most important variable for individual patient risk stratification, and it is systematically under-assessed in current RWE. Manufacturers and health systems that build structured TDM-linked immunogenicity monitoring into cross-switch programs generate commercially valuable safety data. The clinical guidance gap — no formal guidelines for cross-switch monitoring — is an opportunity for specialty society publication that can directly influence formulary access.
VI. Formulation and Device IP as Competitive Differentiators
Buffer-Free and Citrate-Free Formulation Technology
Citrate is a commonly used buffer in biologics formulations, providing pH stability. It is also associated with injection site pain — a ‘burning sensation’ complaint that is well-documented in the adalimumab biosimilar class and was the primary tolerability driver in the Derikx double-switch cohort. The development of citrate-free, high-concentration subcutaneous formulations is therefore simultaneously a tolerability improvement, a cross-switch commercial magnet, and an IP asset.
Buffer-free formulation technology — including phosphate-free and histidine-buffered systems — allows biologics to maintain stability without the irritating excipients that drive patient-reported injection site reactions. From a regulatory standpoint, a buffer-free biosimilar must demonstrate analytical comparability to its reference despite the formulation change; from a patent standpoint, the specific buffer-free excipient composition, the concentration range, and the pH window can each support independent patent claims. Sandoz’s Hyrimoz citrate-free formulation and Organon’s Hadlima citrate-free version each occupy distinct positions in the adalimumab IP landscape for this reason.
The commercial logic is clear in the data. Patients who cross-switch away from a citrate-containing adalimumab biosimilar for tolerability reasons and land on a citrate-free alternative show high retention rates — the Derikx cohort showed zero adalimumab discontinuations post-second-switch. Formularies that position citrate-free biosimilars as the preferred alternative to citrate-containing options can reduce overall adalimumab discontinuation rates, reducing the downstream costs of biologic failure and transition to alternative mechanism therapies.
Auto-Injector and Device Patent Strategy
Device patents are a second-order IP layer that directly affects cross-switching. Auto-injector design — needle gauge, injection depth, force required to actuate, visual confirmation of dose completion, needle concealment for needle-phobic patients — varies meaningfully across biosimilar manufacturers. Patients who have adapted to a specific device design may experience a learning curve or tolerability regression when cross-switched to a product with a different device, even when the molecule is identical.
Manufacturers have addressed this by patenting specific device design features. The needle shield configuration, cap removal torque specification, dose indicator window design, and needle retraction mechanism are each patentable device elements. A biosimilar that holds device patents on features associated with patient preference — low actuation force, hidden needle, audible completion click — has a product characteristic that payer formulary decisions can legitimately consider when selecting a preferred cross-switch target.
For IP teams, the device patent landscape should be assessed alongside the formulation and manufacturing process patent landscape. Device patents typically have 20-year terms from filing, often filed years before product launch, and may outlast formulation patents if the formulation patents are filed earlier in the development timeline. Device IP can therefore provide a longer competitive runway than molecule-adjacent formulation IP in a maturing biosimilar market.
Technology Roadmap: Next-Generation Biosimilar Formulation Strategy
The formulation innovation trajectory for biosimilars over the next five to seven years includes several specific directions. High-concentration, low-volume subcutaneous formulations reduce injection burden for patients on frequent dosing schedules and can support at-home administration, shifting site-of-care economics in favor of self-injection over infusion center delivery. Co-formulation approaches — combining a biologic with a hyaluronidase enzyme (as Halozyme’s ENHANZE platform does for established products) to enable rapid subcutaneous delivery — are a potential route for biosimilar manufacturers to achieve device and formulation differentiation. Extended half-life engineering for Fc-fusion proteins and antibodies is more complex for biosimilars but is appearing in the pipeline for next-generation bio-betters that overlap with the biosimilar market.
For cross-switching specifically, the formulation trajectory implies that the driver of future patient transitions within a biosimilar class will increasingly be convenience and tolerability rather than price alone. A biosimilar with lower injection frequency, better tolerability profile, or a more patient-preferred device will attract formulary-driven cross-switches from less-convenient competitors, even at comparable net prices. Manufacturers investing in these attributes now are building cross-switch pull at the payer and prescriber level simultaneously.
Key Takeaways — Section VI
Citrate-free formulation and device design are the two most commercially actionable IP-linked cross-switching differentiators in the current market. Buffer-free formulation patents covering excipient composition, concentration, and pH window generate IP moats independent of molecule patents. Device patents are a long-duration secondary IP layer that directly influences patient preference in cross-switch scenarios. The formulation technology roadmap points toward high-concentration, low-volume subcutaneous delivery and convenience optimization as the next competitive axes.
Investment Strategy — Section VI
Biosimilar developers with proprietary citrate-free or buffer-free formulation technology in the adalimumab class, or SC delivery technology in the infliximab class, carry cross-switch commercial advantages that are quantifiable through discontinuation rate reduction and patient retention data. These assets should be valued with a cross-switch market share premium relative to formulation-equivalent competitors. Acquirers should assess device patent portfolios alongside formulation IP; a biosimilar with both a citrate-free formulation patent and a preferred auto-injector design patent has the most durable cross-switch competitive position.
VII. Economic Drivers, Pricing Architecture, and Legislative Landmines
The Cost-Saving Arithmetic of Cross-Switching
The economic case for biosimilar-to-biosimilar switching is straightforward at the system level and complicated at the product level. At the health system level, switching between biosimilars of the same reference product can generate savings relative to a higher-priced competitor within the class. In Norway, competitive tendering among adalimumab biosimilars has produced an 85% discount versus the originator’s list price, with a single preferred biosimilar capturing 100% market share in tender cycles. BC’s provincial biosimilar initiative, if extrapolated nationally across Canada, was projected to generate C$452.2 million in annual savings. A single health system switching infliximab patients to a lower-cost biosimilar alternative has saved an estimated $500,000 per transition initiative.
Cumulative biosimilar savings in the U.S. since the first biosimilar launch in 2015 total approximately $24 billion. Cross-switching within the biosimilar class will deepen those savings as competition intensifies and net prices fall further below originator levels. The marginal savings from a cross-switch are smaller than from an originator-to-biosimilar switch, but they are operationally simpler to execute (no physician persuasion campaign against biosimilars as a category) and are likely to become the dominant form of managed biosimilar spending as originator market share approaches zero in competitive categories.
Pricing Dynamics: List Price, Net Price, and Rebate Architecture
Biosimilar pricing does not follow a simple ‘lower than originator’ pattern. Pricing strategy depends on the benefit channel (pharmacy benefit vs. medical benefit), the payer type, and the market structure of the specific therapeutic class.
In the pharmacy benefit channel, Pharmacy Benefit Managers (PBMs) favor high list prices with large rebates, because rebate revenue is a core component of their business model. A biosimilar with a low list price and no rebate may generate lower net cost to the payer than a biosimilar with a high list price and a 40% rebate — or it may not, depending on whether the rebate is passed through to the plan or retained by the PBM. This opacity is a structural barrier to straightforward cross-switching economics. Biosimilar manufacturers playing in the pharmacy benefit space often price with dual strategies: a high list price product for rebate-driven formulary positioning, and a low-cost alternative for value-based or transparent-pricing contracts.
In the medical benefit channel, the ASP-plus-add-on reimbursement model governs. The IRA increased biosimilar ASP add-ons from 6% to 8% for five years — a meaningful difference for high-cost infused biologics where the add-on payment represents absolute dollar revenue to the administering provider. This creates an economic incentive for infusion centers and hospital outpatient departments to prefer biosimilars over reference products when the add-on payment calculation favors the biosimilar.
The IRA’s Two-Year Window Problem
The IRA’s biosimilar incentive increase comes packaged with a structural risk that has not received sufficient attention in the commercial literature. When the CMS selects a reference biologic for price negotiation under the IRA’s drug pricing provisions, a two-year window opens around that selection date. If a biosimilar cannot launch commercially within that window, it will enter a market where the reference product’s price has already been negotiated downward. The biosimilar must then price below the negotiated reference product price — a price that was already reduced by Medicare negotiation — to compete on cost. Depending on the negotiated price reduction, this may render the biosimilar commercially unviable.
The practical implication is that biosimilar manufacturers must track the IRA negotiation selection calendar with the same precision they track patent expiry dates. A biosimilar delayed by patent litigation or manufacturing scale-up issues may miss its commercial window entirely if the reference product enters IRA negotiation during that delay. IP teams and business development functions need to coordinate IRA timeline modeling with litigation strategy and manufacturing readiness planning.
340B Program Misalignment
340B-covered entities — primarily safety-net hospitals, federally qualified health centers, and specific other provider types — purchase covered outpatient drugs at deeply discounted 340B prices and bill payers at the higher standard rate, retaining the spread as margin. For reference biologics, this spread can be substantial: 340B hospitals have been documented charging between 3.2 and 11.3 times acquisition cost for reference drugs.
The perverse incentive this creates for biosimilar adoption is well-documented. If a 340B hospital switches a patient from a reference biologic to a biosimilar, the biosimilar’s 340B acquisition price is lower (consistent with its lower commercial price), but so is its billed rate — meaning the dollar margin from the 340B spread may actually be smaller for the biosimilar than for the reference product. Historically, this dynamic has suppressed biosimilar uptake in 340B institutions. The IRA’s ASP+8% add-on for biosimilars partially mitigates this misalignment, but does not eliminate it. 340B hospitals with high biologic prescribing volumes remain a distinct market segment requiring separate formulary strategy.
Key Takeaways — Section VII
Cross-switching generates meaningful but incremental system-level savings relative to the primary originator-to-biosimilar transition. PBM rebate architecture creates opacity in net price economics that complicates cross-switch formulary decisions. The IRA’s two-year window is a specific commercial risk for biosimilar programs with delayed launches. 340B institutions require separate market access strategies because of the systematic misalignment between biosimilar cost savings and 340B spread economics.
Investment Strategy — Section VII
Model IRA negotiation timelines for target reference products as a core component of biosimilar commercial planning. Programs targeting reference biologics with anticipated IRA selection dates within three to five years need accelerated launch timelines or explicit regulatory risk mitigation. 340B market penetration is structurally distinct from the non-340B market; commercial projections should segment the two populations with different uptake curves and net price assumptions.
VIII. Stakeholder Dynamics: Who Blocks Adoption and Why
Physician Hesitancy: The Gatekeeper Problem
Physicians are the primary determinants of biologic and biosimilar selection for the overwhelming majority of commercially insured patients. Despite measurable improvement in physician familiarity with biosimilar science over the past five years, a substantial proportion of U.S. prescribers remain reluctant to initiate cross-switches in patients who are clinically stable on an existing biosimilar. The concerns physicians express consistently are not random — they cluster around four specific themes.
The first is loss of therapeutic response in a stable patient. Physicians who spent months titrating a patient to adequate disease control are rationally reluctant to introduce a new variable that might disrupt that equilibrium, even when the probability of disruption is low. The second is immunogenicity uncertainty. Many prescribers retain an intuitive — and scientifically outdated — concern that each switch event accumulates immunogenicity risk independently. The third is lack of formal guidelines. Physicians trained in evidence-based medicine are uncomfortable executing a clinical action for which no specialty society guideline exists, even when the available evidence supports that action. The fourth is loss of prescriptive control, particularly in pharmacist-led substitution models. Physicians report that automatic substitution removes their ability to individualize therapy based on patient-specific factors, including device preference, comorbidities, and insurance dynamics.
More than 80% of U.S. physicians surveyed in 2019 expressed concern about non-medical switching. The specific concern was not biosimilar safety as a category — most surveyed physicians accepted the general safety equivalence of biosimilars — but the execution of switches for economic rather than clinical reasons, which they perceived as removing clinical judgment from the process.
Patient Resistance and the Nocebo Effect: Quantified
The nocebo effect — negative health outcomes generated by negative expectations rather than pharmacological activity — is documented at substantial rates in biosimilar switch populations. Published analysis attributes up to 84% of biosimilar discontinuations in some cohorts to nocebo responses rather than genuine loss of efficacy or adverse pharmacology. This is a staggering proportion. It means that in programs with poor patient communication, most observed ‘treatment failures’ after a cross-switch are psychologically mediated, not pharmacologically real.
Patients who undergo a switch without advance notification, without explanation of the clinical rationale, and without understanding of the equivalence basis for the switch are more likely to attribute any subsequent symptom change — including symptom changes that are within normal disease variability — to the switch. This attribution error drives unnecessary discontinuations, which then generate false signals of biosimilar inferiority in uncontrolled real-world datasets.
The implication for cross-switch program design is specific: notification lead time, communication channel, information content, and the framing of the switch (medical recommendation vs. insurance mandate) are each controllable variables that predict discontinuation rates more accurately than the pharmacology of the products involved. Programs that notify patients at least 30 days in advance, provide written and verbal explanation of the equivalence basis, offer helpline access, and frame the switch as a clinical recommendation rather than a cost-cutting measure consistently outperform programs that do not.
Payer Incentive Structures
Payers drive a large proportion of cross-switch decisions. Their motivations are transparent: lower net cost per biologic-treated patient, achieved either by negotiating preferred biosimilar contracts with steeper discounts or by moving patients off a higher-cost formulary tier. The sophistication of payer cross-switch programs varies significantly. Navitus Health Solutions’ adalimumab transition program — which achieved a 94% transition rate from Humira to biosimilars, with 91% of transitioned patients remaining on a biosimilar for at least three months — is an example of a rigorously designed program that coordinated provider outreach, patient communication, and claims monitoring to achieve and sustain the transition.
Payers face their own structural barrier: the rebate model. If a reference product manufacturer is offering rebates that make the reference product cheaper on a net basis than the biosimilar, a payer that switches patients to the biosimilar may actually increase its drug spending. This scenario has occurred repeatedly in the U.S. market, creating what is publicly documented as ‘perverse rebating’ dynamics. PBM contracts that pass through rebates to plan sponsors align incentives toward net price optimization. PBM contracts that retain rebates as PBM revenue create incentives that may favor high-list, high-rebate reference products over low-list biosimilars even when the biosimilar has lower net cost.
Key Takeaways — Section VIII
Physician hesitancy is rational and evidence-responsive — it responds to clinical data, formal guidelines, and transparent incentive structures. Patient nocebo effects are the dominant driver of cross-switch discontinuations and are directly addressable through communication program design. Payer rebate architecture can systemically misalign formulary decisions against biosimilars even when biosimilars have lower net prices; this requires plan sponsor vigilance at the PBM contract level.
IX. Real-World Program Dissection: BC Initiative, NOR-SWITCH, Navitus
BC Biosimilars Initiative: Mandatory Switch at Scale
British Columbia’s biosimilar initiative, launched in 2019, mandated that patients on government drug plan coverage receiving reference biologics for non-rare disease indications transition to approved biosimilars within a specified period or lose coverage. By March 2020, 78% of targeted patients had successfully transitioned. The initiative covered etanercept and infliximab in the first phase, with adalimumab added subsequently.
The BC program’s success factors are instructive for cross-switch program designers. Patient support infrastructure — dedicated transition nurses, pharmacist counseling, helpline access — was built into the program before launch. Physicians received targeted education on the biosimilar evidence base. The mandate was presented as a policy decision with a clear evidence rationale, not as a cost-cutting exercise, which reduced prescriber antagonism. Projected national savings from a pan-Canadian version of the program exceeded C$450 million annually.
The BC program was an originator-to-biosimilar switch, not a biosimilar-to-biosimilar cross-switch. Its relevance to cross-switching is the process model: mandatory switch programs can achieve high transition rates when patient support, prescriber education, and clear communication are deployed systematically. The same model translates to cross-switch programs within a biosimilar class, with the added advantage that prescribers who already accept biosimilar use are more receptive to cross-switch mandates than they were to the initial originator-to-biosimilar transition.
NOR-SWITCH: The Foundational RCT
The NOR-SWITCH trial randomized 482 Norwegian patients stable on reference infliximab to either continue on originator or switch to biosimilar CT-P13 (Remsima) for 52 weeks. The primary endpoint was disease worsening, assessed using disease-specific criteria across six indications. The trial demonstrated non-inferiority of CT-P13 relative to continued originator therapy on the primary endpoint, with disease worsening in 26.2% of switchers vs. 29.6% of continuers — a difference within the pre-specified non-inferiority margin.
NOR-SWITCH does not directly address biosimilar-to-biosimilar cross-switching — it is an originator-to-biosimilar design. Its relevance is foundational: it established the methodological and evidentiary framework for assessing switching in the context of a large biologic class (TNF inhibitors), demonstrated that a well-designed mandatory switch program can collect interpretable clinical data, and provided the reference point against which all subsequent infliximab switching studies are benchmarked.
For cross-switching specifically, NOR-SWITCH’s most important contribution is the principle that 26% disease worsening in a biologic-stable population is the expected background rate even without switching — meaning that some proportion of patients in any switch cohort will worsen for reasons unrelated to the switch. Distinguishing switch-attributable worsening from background disease fluctuation requires a control arm, which is absent from most cross-switch observational studies.
Navitus Adalimumab Transition: Commercial Program Design at Its Best
The Navitus Health Solutions transition program moved insured patients from reference adalimumab (Humira) to biosimilar alternatives through a coordinated commercial program. The 94% overall transition rate and 91% sustained biosimilar use at three months are the headline numbers. The program’s operational design is more informative than its outcomes.
Navitus coordinated outreach to prescribers before patient communication, ensuring that physicians were aware of the formulary change and had the clinical information needed to counsel patients before patients received their first communication. Patient notification was delivered through multiple channels — written letter, patient portal message, and pharmacist outreach — with an average lead time exceeding 30 days. The communication explicitly identified the specific biosimilar to which the patient would be transitioned, described whether it was citrate-free, explained the device differences if any, and quantified the patient’s out-of-pocket cost change.
Within the transitioned cohort, 6% of patients subsequently cross-switched to a different adalimumab biosimilar. This is the specific cross-switch rate within a rigorously managed transition program. The 6% figure represents the residual cross-switch demand after device and formulation preferences were addressed upfront — meaning it likely reflects genuine tolerability-driven cross-switching rather than communication failures. This number is a useful baseline for modeling cross-switch volume in payer programs that adopt similar communication standards.
Key Takeaways — Section IX
The BC initiative demonstrates that mandatory cross-switch programs achieve high transition rates when patient support infrastructure is pre-built. NOR-SWITCH establishes the background disease worsening rate that must be controlled for in cross-switch outcome analysis. Navitus demonstrates that a 6% residual cross-switch rate within a well-managed transition program represents genuine tolerability-driven switching demand. These three programs together define the outer bounds of achievable transition rates and the operational requirements to reach them.
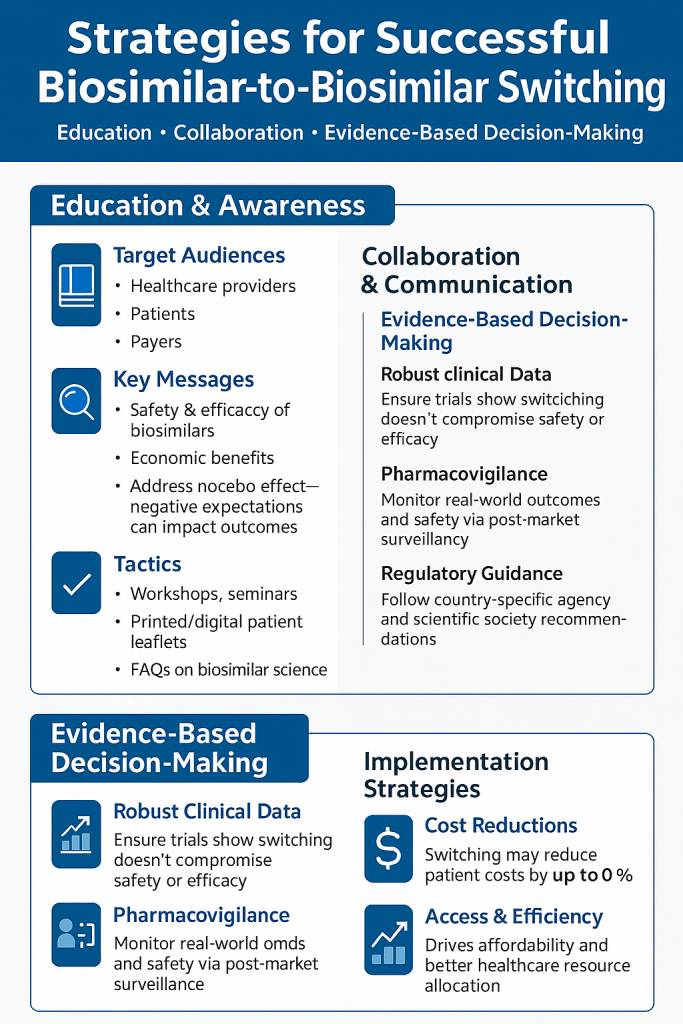
X. Implementation Playbook for Health Systems and Payers
Formulary Architecture for Biosimilar-to-Biosimilar Switching
Health system pharmacy and therapeutics (P&T) committees executing cross-switch formulary decisions should evaluate biosimilar alternatives along at least five dimensions: net acquisition price after all applicable discounts and rebates; formulation characteristics specifically citrate-free status and excipient profile; device design and patient preference data; available immunogenicity and trough level monitoring infrastructure for the specific molecule; and manufacturer patient support program depth.
Bundling all biosimilars of the same reference product into a single P&T review cycle — rather than reviewing each new biosimilar entrant independently — reduces administrative burden and provides a comparative framework that allows formulary decisions to reflect genuine clinical distinctions rather than sequential approvals. Health systems with established formulary review infrastructure should add formulary slots for new biosimilar entrants 30 to 60 days before their projected commercial availability date, enabling pre-negotiated contracts to be in place at launch.
Prior Authorization Streamlining
Prior authorization remains one of the most operationally burdensome elements of biosimilar transitions. For cross-switches, prior authorization requirements may require re-initiation of a PA process even for patients who were already approved for a biosimilar of the same reference product, because the product code changes when the specific biosimilar changes. Health systems should work with their payer partners to establish ‘biosimilar class’ PA designations that cover all approved biosimilars of a given reference product under a single authorization, eliminating re-PA requirements for intra-class cross-switches.
TDM Integration for High-Risk Cross-Switch Patients
Therapeutic drug monitoring integration is the clinical quality standard for cross-switch programs in gastroenterology and rheumatology. Trough level assessment before the switch establishes individual patient pharmacokinetic baseline. Post-switch trough levels at 4, 12, and 24 weeks detect early loss of exposure that may predict loss of clinical response before clinical deterioration is apparent. ADA baseline assessment before the switch allows risk stratification: patients with pre-existing ADAs, particularly high-titer or neutralizing antibodies, require more intensive post-switch monitoring and may be candidates for prescriber-directed continuation of their current biosimilar rather than a mandatory cross-switch.
Patient Communication Protocol: Operational Specifics
A cross-switch communication program that achieves the Navitus-equivalent transition rates requires specific operational commitments. Prescriber notification must precede patient communication by at least one to two weeks. Patient notification must identify the specific receiving product by name and trade name, not just as ‘an adalimumab biosimilar.’ The communication must address the question of citrate-free status explicitly, because this is the most common patient-reported concern for adalimumab cross-switches. Cost impact must be quantified in specific dollar terms, not general statements about savings. Post-transition follow-up outreach at 30 days, from either the manufacturer’s patient support program or the health plan’s care management team, reduces nocebo-driven discontinuation by providing an explicit opportunity for patients to report concerns before they present at their next scheduled physician visit.
Key Takeaways — Section X
Formulary bundling by reference product class reduces P&T administrative burden and enables comparative evaluation of biosimilar alternatives on clinically meaningful dimensions. PA streamlining through biosimilar class designations eliminates re-authorization requirements for cross-switches. TDM integration provides the clinical risk stratification infrastructure needed to identify patients for whom a mandatory cross-switch is contraindicated. Patient communication protocols with prescriber-first sequencing, product-specific information, and post-transition follow-up achieve materially higher transition and retention rates than generic formulary change notices.
XI. Investment Strategy: Portfolio Positioning in a Cross-Switching Market
What Cross-Switching Does to Market Share Dynamics
In a mature biosimilar class — defined as one where multiple biosimilars of the same reference product have achieved commercial launch and where originator market share has declined below 20% — the primary commercial competition shifts from originator vs. biosimilar to biosimilar vs. biosimilar. This is the cross-switching market. The reference product’s IP protection is largely exhausted or settled, manufacturing scale is established across multiple suppliers, and the incremental price reduction from each new biosimilar entrant is smaller than in the initial competition phase.
In this environment, cross-switch market share is determined by net price competitiveness, formulary positioning, formulation attributes, device design, patient support program quality, and TDM data availability. Manufacturers who competed on ‘we are biosimilar to reference product X’ now compete on ‘our citrate-free formulation retains patients who cross-switch from citrate-containing competitors.’ This is a fundamentally different commercial proposition requiring different IP, different manufacturing investment, and different medical affairs strategy.
Portfolio Construction for Institutional Investors
Institutional investors evaluating biosimilar manufacturer portfolios should weight several factors differently in a cross-switching market than in an early-entry biosimilar market.
Formulation IP breadth matters more than number of approved biosimilars. A manufacturer with two biosimilars, each carrying citrate-free formulation patents and proprietary auto-injector design patents, has a more durable cross-switch competitive position than a manufacturer with five biosimilars that are formulation and device equivalents of each other.
Manufacturing cost structure determines floor price in tender competition. In ‘winner-takes-all’ national tenders like Norway’s, the lowest sustainable net price wins 100% of the tendered market. Manufacturers with the lowest cost-of-goods through process intensification, continuous manufacturing adoption, or single-use bioreactor efficiency have structurally superior tender competitiveness.
Geographic diversification of biosimilar portfolios across FDA, EMA, and PMDA (Japan) jurisdictions provides cross-switch opportunity diversification. European biosimilar markets are further along the adoption curve; U.S. markets are accelerating. Manufacturers with established European cross-switch data can use that evidence in FDA submissions and U.S. formulary negotiations.
Pipeline Assessment Criteria
For biosimilar pipeline assets specifically targeting cross-switch market positioning, the due diligence criteria should include: confirmed citrate-free or buffer-free formulation status; proprietary device design with at least one granted device patent; availability of SC formulation option where the reference product class has IV-dominant originator usage (a SC conversion strategy); institutional payer access strategy specifically addressing the 340B and IRA reimbursement dynamics; and post-market RWE commitment sufficient to generate cross-switch-specific immunogenicity and trough level data within the first three years of launch.
Key Takeaways — Section XI
Mature biosimilar classes compete on cross-switch dynamics rather than originator displacement. Formulation IP and device design are the primary differentiators in cross-switch market share competition. Portfolio construction for institutional investors should weight formulation IP breadth over sheer number of biosimilar approvals. Manufacturing cost structure is the determinant of tender competitiveness. Pipeline assets should be evaluated against a cross-switch-specific due diligence checklist that includes formulation IP, device patents, SC conversion strategy, and RWE commitment.
XII. Key Takeaways by Segment
For Biosimilar IP Teams
Formulation and device IP are the most commercially relevant patent layers in a mature biosimilar class. Citrate-free formulation patents in adalimumab and SC delivery patents in infliximab are the current high-value IP assets for cross-switch market positioning. Composition-of-matter expiry does not define the competitive IP horizon — assess all five IP component layers for each reference product class in your portfolio. Track the IRA negotiation calendar as carefully as patent expiry dates; a missed two-year window can eliminate commercial viability for a biosimilar program.
For Portfolio Managers and Institutional Investors
The cross-switching market is now the primary competition landscape for adalimumab and infliximab, and will be for etanercept and rituximab within the next two to three years. Manufacturers with citrate-free formulation IP and proprietary device design patents in these classes hold the most durable cross-switch competitive moats. Evaluate portfolio companies on their formulation IP breadth, manufacturing cost structure, and 340B market access strategy, not just on number of biosimilar approvals.
For R&D Leads
The next formulation frontier for biosimilar cross-switch positioning is high-concentration, low-volume SC delivery combined with buffer-free excipient systems. Invest in analytical characterization platforms — including high-resolution glycoprofiling and higher-order structure assessment — that accelerate regulatory submissions and generate proprietary data packages for interchangeability applications without dedicated switching studies. Build post-launch TDM-linked immunogenicity monitoring into clinical development plans as a commercial data asset, not as a pharmacovigilance cost center.
For Payers and Health System Decision-Makers
Mandatory cross-switch programs achieve high transition rates when prescriber-first communication, 30-plus day patient notification lead times, product-specific information (including citrate-free status), and post-transition follow-up are operationalized systematically. Nocebo-driven discontinuations are the primary driver of cross-switch failure; communication program design has a larger effect on retention rates than pharmacology. Establish biosimilar class PA designations to eliminate re-authorization burden. Integrate TDM monitoring for infliximab and adalimumab cross-switch programs to identify the small proportion of patients for whom a mandatory switch is clinically contraindicated.
For Medical Affairs and Clinical Teams
The evidence base for biosimilar-to-biosimilar cross-switching is positive and directionally consistent across systematic review, meta-analysis, and specific real-world cohorts. The critical gap is baseline ADA characterization before cross-switches — this is the most important unmeasured variable in existing datasets and the most impactful addition to future cross-switch studies. Advocate for specialty society guidelines on cross-switch monitoring protocols; the gap between clinical practice and formal guideline availability is now wider than the evidence gap justifies.
Reference Compounds and Regulatory Milestones Summary Table
| Reference Product | Primary Cross-Switch Driver | Key IP Layer | U.S. Cross-Switch Status | Formulation Differentiator |
|---|---|---|---|---|
| Adalimumab (Humira) | Payer formulary, citrate tolerability | Citrate-free formulation patents | Active, 7+ biosimilars | Citrate-free vs. citrate-containing |
| Infliximab (Remicade) | IV-to-SC conversion, price | SC delivery patents (CT-P13 SC) | Active, 4+ biosimilars | IV vs. SC route |
| Etanercept (Enbrel) | Price, payer mandate | Device design patents | Emerging in U.S. | Device ergonomics |
| Rituximab (Rituxan) | Price, oncology formulary | Manufacturing process IP | Active, 3+ biosimilars | Concentration, infusion time |
| Bevacizumab (Avastin) | Oncology formulary, 340B | Manufacturing process IP | Active, 5+ biosimilars | Infusion concentration |
| Trastuzumab (Herceptin) | Oncology formulary, combination | Formulation/device IP | Active, 5+ biosimilars | Subcutaneous co-formulation |
Patent landscapes, FDA regulatory positions, and commercial market shares are subject to rapid change. Patent intelligence platforms including DrugPatentWatch provide real-time monitoring of the patent and regulatory developments that determine cross-switch commercial timing and IP exposure.