I. Executive Summary

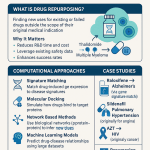
Drug repurposing — identifying new clinical indications for approved or investigational compounds — has moved from a cottage industry of accidental findings into a structured, capital-intensive discipline with a clearly quantifiable return profile. The global drug repurposing market was USD 34.98 billion in 2024. Precedence Research projects it reaches USD 59.30 billion by 2034, a CAGR of 5.42%. North America held 47% of 2024 revenue, driven by high chronic disease prevalence and active regulatory incentive programs.
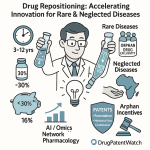
Those numbers are compelling on their own, but the deeper story is structural. Traditional new chemical entity (NCE) development costs north of USD 2.5 billion per approved drug, consumes 10 to 15 years, and fails more than 90% of the time at trial entry. Repurposing cuts average spend to roughly USD 300 million, compresses timelines to 3 to 12 years, and, for candidates that clear Phase I, delivers an approval rate approaching 30%. That math reshapes the risk-adjusted NPV of a dormant asset considerably.
Profitability, though, is not automatic. It depends on a layered strategy that combines method-of-use patent prosecution, regulatory exclusivity stacking, disciplined pricing anchored to clinical value, and a commercialization infrastructure built for the specific payer environment of the new indication. This guide walks through each layer in detail, with specific reference to the regulatory mechanisms, patent prosecution doctrines, and financial levers that determine whether a repurposed compound becomes a franchise asset or a low-margin generic play.
Key Takeaways for Executive Readers
Repurposing cuts NCE development cost by 50 to 60% and timeline by 5 to 7 years. The 505(b)(2) NDA pathway in the US and EMA Article 48 in the EU are the two most consequential regulatory levers available right now. Secondary patents on formulation, dosing regimen, or delivery system can restore 10 to 15 years of effective market exclusivity on a molecule whose composition-of-matter patent has expired. Biologics repurposing carries distinct pathway economics that separate it entirely from small-molecule strategy. And approximately 30% of drugs newly marketed in the US currently originate from computational repurposing approaches, a share that will grow as AI tooling matures.
II. What Drug Repurposing Actually Is
Definitions: Repositioning, Reprofiling, and Retasking
Drug repurposing, repositioning, and reprofiling are used interchangeably in the literature, though practitioners sometimes draw fine distinctions. For the purposes of this guide, all three refer to the same core activity: identifying a new clinical indication for a compound that already has human safety data, whether from prior approval or from a completed Phase I or Phase II program in a different indication.
The scope is broader than most people assume. Repurposing candidates include approved drugs prescribed off-label, investigational compounds that failed efficacy endpoints in their original indication but retain a clean safety profile, withdrawn drugs whose market removal was commercially rather than safety-driven, and nutraceuticals or live biotherapeutic products for which a new pharmacological mechanism has been characterized. Each of these starting points carries a different IP situation, a different regulatory ask, and a different commercial baseline — which is why a one-size-fits-all repurposing framework does not hold.
The distinction between “soft repurposing” and “hard repurposing” matters here. Soft repurposing extends a drug into a closely related indication — say, expanding a first-line oncology drug into a maintenance setting or a pediatric population — where the mechanistic hypothesis is straightforward and the regulatory bridge is short. Hard repurposing moves a compound into a biologically distant indication, such as an approved cardiovascular agent being developed for a neurological disease. Hard repurposing carries a richer IP opportunity (the prior art for the new indication is thinner) but requires a more substantial clinical program.
What Repurposing Is Not
Repurposing is not the same as off-label prescribing, though off-label use patterns often identify the repurposing hypothesis. Off-label prescribing happens at the level of the individual clinician and does not create regulatory approval, labeled prescribing information, or payer coverage certainty. Repurposing, as a commercial strategy, ends with an approved label change — a new indication in the package insert — which is the mechanism that converts clinical evidence into reimbursable revenue.
Repurposing is also not generic drug development. A generic ANDA requires demonstrating bioequivalence to an approved reference listed drug and offers no independent IP. A 505(b)(2) NDA for a repurposed use, by contrast, can generate new regulatory exclusivity and new patent rights even when the underlying molecule is off-patent. The legal and commercial structures are fundamentally different.
Key Takeaways for this Section
The starting point of a repurposing program — approved drug, failed IND, withdrawn compound — determines the IP situation, regulatory pathway, and clinical burden before a single trial is designed. Soft repurposing offers speed; hard repurposing offers richer IP optionality. Off-label use is a hypothesis generator, not a commercial endpoint.
III. The Cost and Timeline Math
Head-to-Head: NCE vs. Repurposed Drug Development
The cost advantage of repurposing comes from skipping preclinical compound discovery, lead optimization, and, in many cases, Phase I dose-escalation studies. The table below sets the comparison precisely.
| Metric | New Chemical Entity | Repurposed Drug |
|---|---|---|
| Total R&D cost | >USD 2.5 billion | ~USD 300 million (range USD 50M to USD 800M) |
| Time to first approval | 10 to 15 years | 3 to 12 years (average ~6 years) |
| Phase I requirement | Always required | Often waived if dose is equivalent or lower |
| Preclinical burden | Full toxicology package | Bridging studies only (if any) |
| Phase I to approval rate | <10% | ~30% |
| Composition-of-matter patent | Available | Usually expired or unavailable |
| Method-of-use patent | Available | Available; key IP mechanism |
| Regulatory exclusivity (US) | 5-year NCE exclusivity | 3-year new clinical investigation exclusivity |
| Orphan Drug designation | Available | Available; often more accessible for rare disease repurposing |
The cost figure deserves a calibration note. The USD 300 million average masks a wide range. A repurposing program that needs only a Phase II confirmatory study and a label extension for an existing NDA holder costs far less than a program where a new sponsor must file an independent 505(b)(2) NDA with a full CMC package, bridging PK studies, and a Phase III program in an immunology indication. The lower bound — roughly USD 50 million — applies to programs where a prior IND holder licenses the compound and builds on an existing clinical dataset. The upper bound — USD 800 million or more — applies to hard repurposing in large indications where payers will demand Phase III evidence before reimbursing.
Risk-Adjusted NPV: The Analyst’s Lens
For portfolio managers evaluating repurposing assets, the risk-adjusted net present value (rNPV) calculation differs from a standard drug development model in two key respects. First, the probability of technical success (PTS) is higher at each clinical stage: published meta-analyses put Phase II success rates for repurposed drugs 15 to 25 percentage points above the industry average for NCEs in the same indication class. Second, the cost to failure is lower, so the expected loss in failure scenarios is smaller.
The discount rate applied to the cash flow model, however, should reflect the IP risk specific to repurposed compounds. If a method-of-use patent has a weak prior-art position — because, for example, the off-label use was widely published before the priority date — that patent may not survive an inter partes review (IPR) petition, and the exclusivity supporting peak sales assumptions collapses. IP-adjusted discount rates for repurposed drugs in high prior-art-risk situations should carry a 3 to 5 percentage point premium over a composition-of-matter-protected NCE, all else equal.
Key Takeaways for this Section
The USD 300 million average cost is a median, not a ceiling. PTS is meaningfully higher than for NCEs, but IP risk must be priced into the discount rate. Programs where method-of-use patent validity is questionable require a separate exclusivity scenario analysis before committing to late-stage investment.
Investment Strategy Note
At the early-stage deal level, acquirers and licensors should weight payment structures toward milestones tied to patent prosecution outcomes (grant of first method-of-use claim, survival of any IPR challenge) rather than front-loaded upfront payments. This structure aligns compensation with the IP events that actually drive commercial value.
IV. Candidate Identification: From Serendipity to Systematic Search
The Three Strategic Approaches
Candidate identification in drug repurposing follows one of three strategic orientations. A drug-centric approach starts with a compound — usually one with known safety in humans — and asks what other diseases its mechanism of action, off-target effects, or pharmacokinetic properties might address. A disease-centric approach starts with an indication that lacks effective treatment and asks which approved or failed compounds could be redirected toward its molecular drivers. A target-centric approach maps a known molecular target implicated in a new disease and then screens for existing drugs that modulate that target, even if the original therapeutic context was entirely different.
Each orientation carries different IP implications. Drug-centric programs, particularly those built on widely-used generics, often face thin composition-of-matter protection from the outset, pushing all IP value into method-of-use or formulation claims. Disease-centric programs in rare disease, where prior art on the specific indication is sparse, offer the strongest method-of-use novelty arguments. Target-centric programs sit in between: the target itself may be well-characterized in a different therapeutic context, but the specific compound-target-indication combination may support a patentable claim.
Computational and Data-Driven Methodologies
The shift from serendipity to systematic identification has been driven by four converging methodological advances: multi-omics data integration, network pharmacology, AI and machine learning, and real-world data analysis. Each deserves specific treatment.
Multi-Omics Integration combines genomics, transcriptomics, proteomics, and metabolomics datasets to characterize the molecular architecture of a disease and identify where known drugs intersect it. Gene expression profiling — comparing the transcriptome of a disease state against the pharmacodynamic signature of a drug — can identify repurposing matches computationally before any wet-lab work. The Connectivity Map (CMap) database at the Broad Institute, now part of the LINCS program, has made this analysis accessible at scale. Practically, a multi-omics screen generates a ranked list of candidate compounds, not a development-ready hypothesis; the output requires biological validation and clinical plausibility assessment before an IND strategy is designed.
Network Pharmacology and Knowledge Graphs organize known biological interactions — protein-protein interactions, gene-disease associations, drug-target relationships — into queryable network structures. Researchers identify candidate repurposing pairs by looking for shortest-path connections between a drug’s known targets and the disease-relevant nodes in the network. Knowledge graph platforms, including those built on the National Drug File Reference Terminology and the Comparative Toxicogenomics Database, can surface hypotheses that conventional literature review would miss. The Exscalate4Cov project used supercomputing-scale in silico docking across 400,000 approved compounds to identify raloxifene as a candidate COVID-19 therapeutic — a result subsequently tested in clinical trials — illustrating both the power and the speed of this approach.
AI and Machine Learning accelerate all steps of the above processes and introduce new capabilities. Deep learning models trained on bioactivity data can predict drug-target binding affinities for targets outside a compound’s characterized profile. Graph neural networks (GNNs) learn from the structure of biological networks directly, without requiring manually curated features. Large language models trained on scientific literature can extract drug-disease associations from unstructured text at a scale that human curation cannot match. Baricitinib’s identification as a potential COVID-19 therapeutic was facilitated by a deep learning model that predicted its kinase and JAK inhibitor profile could suppress the cytokine storm underlying severe disease — a prediction later validated in the ACTT-2 trial.
The limitation of AI-driven screens is worth stating clearly: they generate hypotheses, not clinical evidence. The false positive rate in virtual screening remains high. Programs that commit capital to a repurposing hypothesis generated entirely in silico, without biological validation in relevant cellular or animal models, carry a disproportionate risk of late-stage failure for mechanism-related rather than safety-related reasons.
Real-World Data Analysis uses electronic health records (EHRs), claims databases, and pharmacovigilance data to detect drug-disease associations in actual patient populations. Pharmacoepidemiology studies comparing outcomes between patients who happen to have received a drug for one indication versus those who did not — a natural experiment structure — can surface protective associations that no prospective trial would have been designed to test. The FDA Sentinel system and European Network of Centres for Pharmacoepidemiology and Pharmacovigilance (ENCePP) provide infrastructure for this kind of analysis. For commercial strategy, RWD analysis also clarifies the size of the off-label prescribing opportunity that an on-label approval would capture, helping revenue models at an early stage.
Prioritization Criteria Before Entering Development
Not every computational hit should proceed to development. A structured prioritization framework evaluates candidates across four dimensions: biological plausibility (is there a coherent mechanistic rationale supported by multiple data streams?), clinical translatability (is there a validated biomarker or patient enrichment strategy?), regulatory feasibility (what clinical evidence will FDA or EMA require, and what is available?), and IP opportunity (is there a defensible patent position or regulatory exclusivity path?). Programs that score below a threshold on any single dimension warrant a pause before Phase II investment, regardless of how strong the other scores are.
Key Takeaways for this Section
AI and computational tools generate hypotheses; wet-lab validation and clinical plausibility review convert those hypotheses into development candidates. RWD analysis is underused as a commercial intelligence tool, not just a scientific one. The choice of identification methodology (drug-centric, disease-centric, or target-centric) shapes the entire downstream IP and regulatory strategy, so that choice belongs at the portfolio level, not the project level.
V. IP Architecture for Repurposed Drugs
The IP Challenge: Why Composition-of-Matter Is Usually Off the Table
The fundamental IP challenge in drug repurposing is that the most powerful form of pharmaceutical patent protection — a composition-of-matter (CoM) claim covering the active pharmaceutical ingredient — is generally unavailable. By definition, repurposing targets compounds that already exist in the literature or in commercial use. CoM claims require novelty; a known molecule fails that test regardless of how innovative the new clinical use may be. Patent offices in the US, EU, and Japan uniformly require a novel composition for a CoM claim, and prior publication or commercial sale of the compound defeats novelty.
What remains available is a spectrum of secondary claims, each with its own strength, duration, prosecution complexity, and enforceability profile. Understanding which secondary claim type fits a specific program — and how to construct a layered portfolio of such claims — is the central IP task in repurposing.
Method-of-Use Patents
A method-of-use patent covers the specific process of treating a defined condition with a known compound. Under 35 U.S.C. Section 101 and its foreign equivalents, a new method of treatment can be patentable even when the compound itself is not. The requirement is novelty and non-obviousness of the method, not the compound.
The novelty analysis in method-of-use prosecution centers on prior art specific to the indication. If a drug’s package insert, prior publications, or conference presentations already describe the clinical use being claimed, the claim will fail on anticipation. If the new use is mechanistically obvious from the drug’s known pharmacology — for example, adding a second indication that shares the same molecular pathway as the approved indication — the claim faces an obviousness rejection under 35 U.S.C. Section 103. The strongest method-of-use patents cover uses that are mechanistically non-obvious, often because the repurposing hypothesis was derived from off-target activity rather than the drug’s primary mechanism.
Enforcement is a persistent limitation. Method-of-use patents are enforceable against prescribers and manufacturers who induce the patented method, but they cannot directly block a generic drug manufacturer from selling the compound for unrelated approved uses. The “skinny label” issue — where a generic manufacturer omits a patented indication from its label to avoid inducement liability — has been extensively litigated under the Hatch-Waxman Act. The Federal Circuit’s GlaxoSmithKline v. Teva decision held that circumstantial evidence of induced infringement can support liability even when a generic uses a skinny label, but the legal landscape remains unsettled, and enforcement risk should be factored into any IP valuation for a method-of-use-only portfolio.
Formulation and Delivery System Patents
New formulation patents cover novel pharmaceutical compositions — different salt forms, polymorphic forms, prodrugs, nanoparticle encapsulation, liposomal delivery, extended-release matrix systems, or fixed-dose combinations — that were not previously described for the compound. These claims offer several advantages over pure method-of-use protection: they are easier to enforce (a generic manufacturer cannot avoid the claim by using a skinny label because the patented composition is physically the product), and they can be listed in the FDA Orange Book if they meet the requirements of 21 CFR Part 314.53.
Orange Book listing is consequential. When a generic applicant files an ANDA referencing a branded drug with Orange Book-listed formulation patents, it must certify to each listed patent. A Paragraph IV certification — asserting that the patent is invalid or will not be infringed — triggers a 30-month regulatory stay on the ANDA approval, giving the innovator time to litigate. That 30-month stay is effectively a free injunction pending trial, and it materially protects market exclusivity even when the outcome of litigation is uncertain.
The IP value of a formulation patent depends directly on whether the formulation change delivers a genuine clinical benefit. Formulation changes that improve bioavailability, reduce dosing frequency, shift administration from IV to subcutaneous, or enable outpatient use in a previously hospital-administered drug are defensible from both a patent prosecution and a pricing standpoint. Reformulations that offer no discernible clinical benefit are vulnerable to obviousness challenges and generate limited pricing power, limiting their commercial contribution.
Dosing Regimen and Patient Population Patents
Specific dosing regimen patents protect a novel schedule of administration — a particular dose, interval, or titration protocol — that is clinically non-obvious. These claims are narrow in scope but can block the exact dosing regimen referenced in clinical trial data, making it difficult for generic prescribers to replicate the studied regimen without technically practicing the claimed method.
Patient population patents cover the use of an existing drug in a novel, previously unrecognized patient subgroup. Genomic and biomarker characterization has made this approach increasingly viable: a companion diagnostic that identifies a responsive subpopulation can support both a narrow patent claim and a companion diagnostic approval, creating a bundled IP and regulatory exclusivity structure that is difficult for generics to replicate. Oncology has generated the most examples — KRAS G12C inhibitors repurposed across tumor types with the same genomic driver, or PARP inhibitors extended from BRCA-mutant ovarian cancer to BRCA-mutant prostate and breast cancer — but the approach is applicable across therapeutic areas wherever genomic stratification of responders is feasible.
Combination Therapy Patents
Fixed-dose combinations (FDCs) of a repurposed drug with a standard-of-care companion therapy can generate composition-of-matter claims on the combination even when neither component is individually patentable. The novelty requirement is met by the combination itself, provided the specific pairing was not previously disclosed. Non-obviousness is met when the combination produces an unexpected pharmacological effect — synergy, reduced toxicity, improved pharmacokinetics — that would not have been predicted from each agent’s individual profile. Combinations that produce only additive effects without a documented unexpected benefit are vulnerable to obviousness rejection.
FDC patents also open a formulation development pathway that supports higher pricing: a co-formulated tablet or capsule commands a price premium over two separate generic pills, even when the individual components are off-patent.
Key Takeaways for this Section
CoM protection is almost never available in repurposing. The IP portfolio must be built from method-of-use, formulation, dosing regimen, patient population, and combination claims in a layered structure where each claim type protects a different dimension of the product. Orange Book listing of formulation claims triggers the 30-month Hatch-Waxman stay, the most commercially valuable procedural benefit available to a repurposed drug’s IP holder.
Investment Strategy Note
When valuing a repurposing asset, map the IP portfolio against three scenarios: (1) all secondary claims survive challenge, (2) method-of-use claims are invalidated but formulation claims survive, and (3) all secondary claims fail. Scenario 3 reduces the asset to a data exclusivity play — 3-year new clinical investigation exclusivity in the US — with a market exclusivity window too short to justify most Phase III programs without an orphan drug designation or other supplemental protection. Assets where the IP portfolio is limited to a single method-of-use claim with thin prior art differentiation should be discounted aggressively relative to assets with layered formulation and combination patent coverage.
VI. Evergreening and the Secondary Patent Roadmap
What Evergreening Is and What It Is Not
Evergreening describes the strategic practice of filing successive secondary patents — on formulations, delivery systems, dosing regimens, metabolites, or new indications — to extend the period of effective market exclusivity beyond the expiration of an original composition-of-matter patent. The term carries a pejorative connotation in policy debates, but from an IP management perspective, it describes a legitimate and well-established patent portfolio strategy that is no different in principle from the continuation and continuation-in-part practice used in any other technology industry.
The legal and commercial question is not whether secondary patents are appropriate — they are — but whether each successive patent covers a meaningful innovation. Patent offices apply standard patentability criteria to secondary pharmaceutical claims regardless of the commercial context. Formulation patents on clinically meaningless modifications, such as a polymorph with identical bioavailability to the original, are routinely rejected or invalidated on obviousness grounds. Formulation patents covering a delivery system change that reduces dosing frequency from twice-daily to once-weekly, or that enables home administration of a previously hospital-only product, typically survive challenge because the clinical benefit supports a non-obviousness argument.
The Evergreening Technology Roadmap for Small Molecules
A systematic evergreening strategy for a small molecule drug follows a staged development plan tied to the patent lifecycle of the original compound. The roadmap below represents a generalized template applicable to most small molecule franchises.
Stage 1: Composition-of-matter patent filing (Year 0) The original CoM patent is filed at the time of compound discovery or synthesis. Patent term runs 20 years from the priority date. For a drug that takes 10 years to reach approval, the effective post-approval life of the CoM patent is roughly 10 years, extended by Patent Term Extension (PTE) under 35 U.S.C. Section 156 by up to 5 years (capped at 14 years of effective post-approval exclusivity). European Supplementary Protection Certificates (SPCs) provide an analogous extension, up to 5 years post-patent expiry, subject to the date of first EU marketing authorization.
Stage 2: Formulation development and filing (Years 5 to 8) While the original product is in Phase III or early post-launch, the development team pursues a next-generation formulation: extended-release, coformulation with a bioavailability enhancer, nanoparticle encapsulation, or route-of-administration shift. Patents on the new formulation are filed with a priority date 10 to 13 years after the original CoM priority date, giving them a 20-year term that extends well beyond CoM expiry. If the formulation patent generates Orange Book-eligible claims, it is listed and provides Hatch-Waxman protection for the new formulation product.
Stage 3: New indication development (Years 8 to 12) Systematic repurposing investigation — drawing on post-market pharmacovigilance data, RWE from EHR analysis, or academic collaboration — identifies one or more new indications. Method-of-use patents covering these indications are filed. If an indication qualifies for Orphan Drug Designation, the 7-year US market exclusivity or 10-year EU market exclusivity (reducible to 6 years if the designation criteria are no longer met) is secured in parallel with patent prosecution. Regulatory exclusivity and patent protection can run concurrently or sequentially, depending on the timing of approval.
Stage 4: Combination therapy and next-generation dosing (Years 10 to 15) FDC development with a complementary standard-of-care agent creates a combination product protected by novel composition-of-matter claims on the combination. Separately, a new dosing regimen optimized for the new indication — perhaps a lower dose, a less frequent schedule, or a weight-based or biomarker-stratified protocol — generates additional method-of-use claims with a later priority date.
Stage 5: Pediatric exclusivity (Year 12 to launch plus 6 months) Filing a Pediatric Study Plan (PSP) with FDA and conducting required pediatric studies under the Pediatric Research Equity Act (PREA) or the Best Pharmaceuticals for Children Act (BPCA) generates an additional 6-month pediatric exclusivity applicable across all Orange Book-listed patents and regulatory exclusivity periods. This is effectively a 6-month extension to every other protection period at minimal marginal cost, assuming the pediatric studies were already planned or required by FDA.
The cumulative effect of stages 1 through 5, properly executed, can maintain meaningful market exclusivity — either through patent protection or through regulatory exclusivity periods — for 25 to 30 years from the original approval date, against a CoM patent baseline of 14 years of effective post-approval protection.
Evergreening Roadmap for Biologics
The evergreening framework for biologics differs substantially from the small molecule roadmap. Biologic patent portfolios are structurally more complex: a typical monoclonal antibody is covered by 40 to 70 patents addressing the molecule itself, manufacturing processes, formulations, dosing regimens, and specific indications. Patent cliffs for biologics are rarely as sharp as for small molecules because no single patent dominates the way a CoM patent does for a small molecule.
The key mechanisms for biologic evergreening include: (1) continuation and continuation-in-part applications extending composition claims on post-translational modifications, glycoforms, or purification processes; (2) formulation patents covering subcutaneous (SC) co-formulation with hyaluronidase, enabling self-administration versus the original IV infusion; (3) biosimilar interchangeability blocking, where patent claims on the specific device used to administer a self-injectable biologic (autoinjector, prefilled syringe) create a product differentiation that biosimilar developers cannot easily replicate; (4) indication expansion patents covering new diseases, often in areas where the biologic’s mechanism of action is newly characterized; and (5) combination claims with small molecule adjuncts for specific patient subpopulations.
The biosimilar interchangeability question deserves particular attention. Under the Biologics Price Competition and Innovation Act (BPCIA), a biosimilar can be designated as interchangeable with the reference product if it meets additional switching study requirements. An interchangeable designation allows pharmacist-level substitution without prescriber intervention, analogous to automatic generic substitution for small molecules. For the originator, blocking interchangeability — through device patents or formulation differentiation that biosimilar developers cannot replicate without infringing — maintains a pharmacist-substitution barrier that significantly slows market share erosion even after the reference product’s 12-year regulatory exclusivity expires.
Key Takeaways for this Section
Evergreening is a standard IP portfolio management practice, not an anomaly. Its value depends entirely on whether each successive patent covers a genuine innovation. The 5-stage small molecule roadmap generates 25 to 30 years of layered exclusivity from a 14-year CoM baseline. For biologics, device patents and SC formulation patents are among the most commercially valuable secondary claims because they directly address the interchangeability substitution risk.
VII. IP Valuation as a Core Asset: Frameworks and Case Studies
Why IP Valuation Matters in Repurposing Transactions
Every licensing negotiation, acquisition, and partnering deal in drug repurposing assigns an implicit or explicit value to the IP portfolio of the asset being transacted. When the IP is strong — a granted method-of-use patent with a clean prosecution history, Orange Book-listed formulation claims, and an unexpired orphan drug exclusivity — the valuation premium over a program with no IP is substantial. When the IP is weak — a pending method-of-use application with obvious prior art, no formulation claims, and no regulatory exclusivity — the asset is essentially a data package, and its value depends almost entirely on the strength of the clinical results rather than on any exclusivity protection.
Formal IP valuation in pharma uses three methodological frameworks: the income approach, the market approach, and the cost approach.
The income approach discounts the future cash flows attributable to the IP-protected period of the drug’s commercial life. For a repurposed drug, this requires modeling: (1) peak sales in the new indication, (2) the duration of the exclusivity period supported by the IP portfolio, (3) the probability that key patents survive any challenge, and (4) the discount rate reflecting both time value and IP risk. The exclusivity period is not simply the remaining patent term; it is the term of the most commercially significant defensible claim, adjusted for the probability of invalidation and for the likelihood of an ANDA filer pursuing a Paragraph IV challenge. In high-value therapeutic areas, a Paragraph IV challenge should be assumed as near-certain when the market size exceeds USD 500 million annually.
The market approach benchmarks the IP-protected program against precedent transactions for comparable assets: similar indication, similar exclusivity position, similar clinical development stage. Drug repurposing transaction databases and licensing analytics platforms track deal economics and provide the comparable set. The challenge in repurposing specifically is that comparables are sparse in some indication categories; the method works best in oncology, CNS, and immunology, where deal volume is high.
The cost approach estimates the cost to recreate equivalent IP protection through independent development. This is most relevant when assessing freedom-to-operate and design-around cost, rather than the asset value itself. Knowing that it would cost USD 80 million and six years for a competitor to independently develop a clean IP position for the same indication informs both licensing terms and litigation strategy.
Case Study: GLP-1 Receptor Agonists — Repurposing IP as a USD Multi-Billion Asset
Semaglutide (Ozempic, Wegovy) offers the clearest recent example of how repurposing IP — specifically, method-of-use and formulation patents for a new indication — transforms a mature diabetes franchise into a new multi-billion-dollar commercial platform. Semaglutide received its first US approval for Type 2 diabetes in 2017. Novo Nordisk subsequently pursued a separate NDA for a higher dose formulation (2.4 mg vs. 1.0 mg) specifically for chronic weight management, approved as Wegovy in 2021.
The IP structure supporting Wegovy’s market exclusivity includes method-of-use patents covering the weight management indication at the higher dose, formulation patents covering the specific once-weekly injectable formulation at the 2.4 mg dose level, and device patents on the FlexTouch autoinjector system. These secondary patents extend commercially meaningful exclusivity well beyond the expiration of semaglutide’s original composition-of-matter protection. Novo Nordisk has listed multiple patents covering the weight management indication and formulation in the Orange Book for Wegovy, requiring any generic applicant to certify against each.
The IP valuation of the Wegovy franchise — distinct from the underlying semaglutide molecule — reflects the market size of the weight management indication (estimated peak annual sales exceeding USD 10 billion for Wegovy alone), the duration of the applicable exclusivity periods, and the probability that Orange Book-listed patents survive challenge. At a rough income approach calculation, the NPV of the Wegovy-specific IP position at the time of approval was in the range of USD 15 to 20 billion, assuming a 10-year protected commercial life and a 70% probability of key patents surviving challenge. This represents the value created by a targeted repurposing program — a new indication, a new dose, a new formulation — layered onto a known molecule.
The GLP-1 class is now being actively investigated for cardiovascular disease reduction (the SELECT trial confirmed cardiovascular event reduction for semaglutide), non-alcoholic steatohepatitis (NASH), sleep apnea, and addiction. Each new indication is a distinct repurposing program with its own method-of-use patent opportunity, its own regulatory exclusivity potential (including potential orphan designation in some rare subtypes), and its own commercial buildout.
Case Study: Thalidomide — From Notorious to Orphan Drug IP Architecture
Thalidomide was withdrawn from most markets in the early 1960s following its teratogenicity discovery. Celgene reintroduced it in 1998 for erythema nodosum leprosum, and subsequently for multiple myeloma, building a patent portfolio around specific dosing regimens, the Risk Evaluation and Mitigation Strategy (REMS) program — which itself functions as a de facto barrier to generic entry — and the related compound lenalidomide (Revlimid), an analog with superior efficacy and a cleaner safety profile. The REMS-based barrier is not a patent right, but it operates as an effective market protection mechanism in practice: any ANDA filer must meet the REMS requirements for thalidomide-class compounds, which adds regulatory complexity and liability exposure that deters generic development.
The IP architecture Celgene constructed around thalidomide and lenalidomide — layering composition-of-matter patents on the analogs, method-of-use patents on specific multiple myeloma dosing protocols, and REMS-related barriers — generated a combined franchise that Celgene sold to Bristol-Myers Squibb in 2019 for USD 74 billion. The bulk of that valuation reflected the lenalidomide franchise (USD 12 billion annual sales at peak), but thalidomide’s repurposing success was the proof of concept that launched the platform.
Key Takeaways for this Section
IP valuation in repurposing requires explicit modeling of exclusivity duration, patent challenge probability, and the income approach against a clearly defined commercial scenario. Semaglutide/Wegovy demonstrates that a well-constructed secondary patent portfolio on a repurposed indication can create USD 15 to 20 billion in IP-attributable value. REMS programs, while not IP instruments, can function as commercial barriers when they impose compliance infrastructure that deters generic development.
Investment Strategy Note
In due diligence for a repurposing asset acquisition, request a complete prosecution history for all secondary patents, a freedom-to-operate opinion covering the new indication, a list of all Orange Book-listed patents, and a legal assessment of IPR vulnerability for each claim family. Commission an independent IP valuation using the income approach with explicit scenario trees for patent challenge outcomes. Do not rely on the seller’s in-house IP assessment as the primary input.
VIII. Regulatory Pathways: US and EU
The 505(b)(2) NDA: The US Repurposing Highway
The 505(b)(2) New Drug Application pathway under the Federal Food, Drug, and Cosmetic Act is the primary US regulatory mechanism for repurposed drugs. It allows an applicant to rely, in part, on published literature or on FDA’s prior findings of safety and effectiveness for a previously approved product that the applicant did not develop. This reliance eliminates the need to independently generate all the data that a full 505(b)(1) NDA would require, particularly early preclinical toxicology and Phase I safety data when the dose in the new indication is comparable to or lower than the original approved dose.
The practical value of the 505(b)(2) pathway is in data bridging. An applicant developing a repurposed formulation of an approved drug can reference the reference listed drug’s (RLD’s) safety database, pharmacokinetic characterization, and CMC specifications, then add only the studies required to support the specific differences — new indication, new dose, new formulation, new patient population. Pre-IND meetings with the FDA’s Office of New Drugs are essential before an IND is filed, because the acceptable scope of data reliance is not codified in a single document and varies by the nature of the differences between the RLD and the proposed product. Sponsors who assume they can rely on the RLD’s full safety database without conducting bridging PK studies routinely receive clinical holds or complete response letters that set development back 12 to 18 months.
Expedited review designations available to 505(b)(2) applicants include: Fast Track designation (for drugs addressing an unmet medical need in a serious condition, with rolling review benefits); Breakthrough Therapy designation (for drugs showing preliminary clinical evidence of substantial improvement over existing therapies on a clinically meaningful endpoint, with intensive FDA guidance); Accelerated Approval (conditional approval on a surrogate endpoint reasonably likely to predict clinical benefit, with confirmatory trial requirement); and Priority Review (6-month PDUFA review timeline vs. standard 12 months). None of these designations are exclusive to repurposed drugs, but they are disproportionately accessible to repurposing programs because the existing safety data often allows earlier demonstration of preliminary efficacy, which is the threshold for Breakthrough Therapy designation.
Orphan Drug Designation (ODD) under the Orphan Drug Act applies to drugs intended to treat, diagnose, or prevent conditions affecting fewer than 200,000 US patients annually. ODD carries 7 years of market exclusivity from the date of approval (separate from and potentially stacking with patent protection), a 50% tax credit on qualified clinical trial expenses (critical for programs funded by smaller biotechs without large tax bases), and eligibility for orphan drug grants. For repurposing programs targeting rare diseases — a disproportionately large fraction of repurposing activity given the high unmet need and the favorable regulatory economics — ODD is the single most valuable regulatory tool available, and securing it should be a first-order priority before Phase II investment.
The timing of ODD filing matters. The FDA requires that the drug has not received prior approval for the orphan indication, but ODD can be granted before any clinical data exists. Filing ODD before Phase II, based on published prevalence data and a scientific rationale document, costs relatively little and protects the exclusivity option. Waiting until after Phase III results are in hand does not create additional exclusivity or other benefits, but creates a window during which a competitor could file ODD first for the same indication.
EU Regulatory Framework: EMA Article 48 and the Not-For-Profit Initiative
The European Medicines Agency provides several mechanisms relevant to drug repurposing, but the most consequential development in EU regulatory policy is the proposed revision to Article 48 of EU pharmaceutical legislation. Under the current framework, only the holder of a marketing authorization (MA) can submit clinical evidence to extend the MA to cover a new indication. For off-patent drugs where the original MA holder has no commercial incentive to fund new indication development, this creates a market failure: clinical evidence generated by academic or not-for-profit researchers cannot be incorporated into the official prescribing information, preventing payer coverage and systematic prescriber adoption.
Article 48, as proposed in the 2023 EU pharmaceutical legislation revision, would create a mechanism for not-for-profit entities — academic medical centers, disease foundations, national health systems — to submit clinical evidence for new indications of established medicines directly to the EMA. If the evidence supports a favorable benefit-risk assessment, the original MA holder would be required to update the product information to reflect the new indication. This is a structural shift: it removes the commercial gatekeeper from the repurposing regulatory pathway and creates a publicly funded route to label extension.
The practical implications for commercial players are mixed. On one hand, Article 48 creates a free-rider problem: if academic studies supported by public funding generate label extensions for off-patent drugs, commercial competitors can prescribe and sell those drugs for the newly labeled indication without having funded the development. On the other hand, if a commercial player has a reformulated or otherwise differentiated version of the same compound, the academic label extension for the new indication can validate the market opportunity and reduce prescriber education costs, to the advantage of the branded reformulation.
The EMA’s 2022 pilot program supporting not-for-profit repurposing submissions is the operational precursor to Article 48. The pilot accepted scientific advice requests from academic institutions and patient advocacy organizations for repurposing programs in oncology and rare disease, providing EMA regulatory guidance without the MA holder’s involvement. Early results from the pilot have been published but remain limited, and the programs are primarily early-stage. Article 48’s full implementation will take additional years through the EU legislative process.
Separately, the EMA’s adaptive pathways pilot — now incorporated into the standard Scientific Advice process as iterative development — allows interim data packages to inform regulatory requirements as a program evolves, reducing the risk of designing a Phase III study that generates data insufficient for approval. For repurposed drugs where Phase II data already exist (often from academic or investigator-initiated trials), adaptive pathway engagement can substantially reduce the Phase III program size and duration.
Cross-Jurisdictional Regulatory Comparison
| Mechanism | US | EU |
|---|---|---|
| Primary repurposing pathway | 505(b)(2) NDA | Article 10 hybrid application; Article 48 (proposed) |
| Orphan exclusivity | 7 years | 10 years (reducible to 6 years) |
| Pediatric exclusivity extension | 6 months | 2-year SPC extension (for new pediatric studies) |
| Expedited review | Fast Track, Breakthrough Therapy, Priority Review | PRIME scheme (priority medicines) |
| Patent term extension | Up to 5 years (PTE) | Up to 5 years (SPC), capped per member state |
| Regulatory data exclusivity (base) | 5 years (NCE) or 3 years (new clinical study) | 8+2+1 years (data exclusivity + market exclusivity + new indication extension) |
| Not-for-profit repurposing pathway | No formal analog | Article 48 (proposed 2023) |
Key Takeaways for this Section
Pre-IND engagement with FDA is not optional in 505(b)(2) programs; the scope of permissible data reliance is negotiated, not assumed. ODD should be filed before Phase II investment, not after. Article 48 represents a structural shift in EU repurposing regulation that commercial players with differentiated reformulations should monitor closely, as it may validate indication opportunities at public expense.
Investment Strategy Note
For US programs in potential orphan indications, model two regulatory scenarios: one with ODD (7-year exclusivity post-approval, 50% tax credit, potential grant funding) and one without. The NPV differential between these scenarios often exceeds the cost of the ODD application by an order of magnitude. In EU programs, track the Article 48 legislative timeline and identify whether academic studies in the target indication are progressing, as an academic label extension could pre-validate the market or, depending on your asset position, reduce the pricing premium available for a branded formulation.
IX. Biologics-Specific Repurposing: A Technology Roadmap
Why Biologics Repurposing Is Structurally Different
Small molecule repurposing and biologic repurposing share a conceptual framework but diverge at nearly every operational level. Biologics are large, complex molecules — monoclonal antibodies, fusion proteins, cytokines, enzymes — whose pharmacological activity often derives from highly specific target binding rather than broad off-target effects. This specificity limits the drug-centric repurposing hypothesis: it is less likely that an antibody against one target will have meaningful off-target activity at a clinically relevant second target. The more productive repurposing hypothesis for biologics is usually disease-centric or target-centric: identify a new disease driven by the same target the biologic modulates, rather than looking for off-target effects.
The regulatory framework for biologics repurposing in the US runs under the Public Health Service Act (PHS Act) Section 351, not the FDCA Section 505 framework governing small molecules. A new indication for an approved biologic can be pursued as a Biologics License Application (BLA) supplement under the 351(a) pathway. There is no analog to the 505(b)(2) NDA for biologics, meaning an applicant cannot formally rely on another sponsor’s BLA data through a 505(b)(2)-style bridging mechanism. An originator repurposing its own biologic relies on its own existing BLA data; an independent developer pursuing repurposing of a biologic manufactured by someone else must either license the original BLA data or generate independent safety and pharmacokinetic data.
The 351(k) Biosimilar Pathway and Repurposing Interactions
The 351(k) pathway governs biosimilar applications. A biosimilar approved under 351(k) is licensed for the same indications as the reference product unless the reference product’s indication is covered by orphan drug exclusivity. This creates an interaction between repurposing strategy and biosimilar defense strategy. If an originator’s repurposed indication carries orphan exclusivity, the biosimilar is blocked from that indication — but not from the original indications. The originator’s commercial strategy must therefore consider which indication generates the majority of revenue and whether orphan exclusivity for the repurposed indication materially protects total franchise value.
The biosimilar interchangeability designation, discussed earlier in the context of evergreening, is directly relevant here. A biosimilar that achieves interchangeable status can be substituted at the pharmacy level without prescriber authorization. The studies required for interchangeability — typically switching studies demonstrating that alternating between the reference product and the biosimilar does not produce increased immunogenicity or reduced efficacy — add 2 to 4 years of development time for the biosimilar developer. Device patents on the autoinjector and formulation patents on the SC co-formulation are the originator’s primary tools for preventing or delaying interchangeability, because the biosimilar developer must use the same device and formulation to demonstrate interchangeability with a specific reference product presentation.
Indication Sequencing for Biologics: A Technology Roadmap
Biologic indication expansion follows a logic distinct from small molecule repurposing because the cost of the pivotal trial program — typically a randomized Phase III study in an immunological or oncological indication — ranges from USD 200 million to USD 800 million per indication. The indication sequencing decision is therefore a portfolio capital allocation problem, not just a scientific one.
The roadmap for a biologic franchise typically sequences indications by the following criteria in order: regulatory exclusivity duration remaining on the first indication, strength of the mechanistic hypothesis in the second indication (a second indication driven by the same molecular pathway as the first requires less de-risking investment than a mechanistically distinct indication), size of the addressable patient population, payer willingness to pay in the new indication, and competitive density.
A representative roadmap for a monoclonal antibody targeting an inflammatory pathway might sequence: (1) approval in rheumatoid arthritis (primary indication), (2) label expansion to psoriatic arthritis (shared mechanistic basis, reduced Phase III burden through reference to existing pharmacology data), (3) expansion to inflammatory bowel disease (new Phase III but mechanistically related, and IBD market commands premium pricing), (4) expansion to a pediatric population (triggers pediatric exclusivity extension), and (5) investigation in a rare inflammatory disease qualifying for ODD (orphan exclusivity for 7 years in the new indication).
Each successive indication adds a new method-of-use patent with a later priority date, extends the portfolio’s commercial life, and adds distinct pricing scenarios in different payer environments. The cumulative exclusivity structure, when ODD is available for one indication, can protect the franchise through a period that effectively outlasts the original 12-year biologic regulatory exclusivity.
Key Takeaways for this Section
Biologics repurposing is primarily disease-centric or target-centric, not drug-centric. There is no 505(b)(2) analog for biologics; independent developers must generate independent safety data or license originator data. Interchangeability blocking via device and SC formulation patents is the most commercially significant secondary patent strategy for biologic originators facing biosimilar competition. Indication sequencing is a capital allocation decision requiring explicit modeling of exclusivity duration, trial cost, and payer pricing by indication.
X. Market Sizing and Commercial Dynamics
Market Size and Segmentation
The global drug repurposing market reached USD 34.98 billion in 2024 and is projected at USD 59.30 billion by 2034, a CAGR of 5.42%. North America’s 47% revenue share reflects the density of Phase II and Phase III repurposing programs in oncology and CNS, the availability of US orphan drug incentives, and the depth of the US market for premium-priced specialty drugs. The Asia-Pacific region is growing faster than North America in absolute terms, driven by expanding clinical trial infrastructure in China and India and government-supported repurposing programs in Japan.
By approach type, disease-centric programs held 43% of market share in 2024. This dominance reflects the concentration of repurposing activity in therapeutic areas — oncology, rare disease, neurodegeneration — where the disease mechanism is well-characterized but treatment options are limited. The disease-centric approach’s dominance also reflects the regulatory incentive structure: orphan drug designations, which provide the strongest commercial incentives, are by definition tied to a specific disease, pulling development activity toward disease-centric hypothesis generation.
By molecule type, biologics held 62% of market share in 2024. This is a reversal from 10 years ago, when small molecule repurposing dominated. The shift reflects two structural changes: the growing use of monoclonal antibodies across multiple indication categories (where a single approved antibody can be investigated in a dozen indications), and the computational advances in multi-omics analysis that make it easier to identify new diseases driven by an antibody’s known target. Biologic repurposing programs are also more likely to generate new clinical data packages that support premium pricing, which drives revenue share even when program count is lower.
Geographic Pricing and Market Access Variation
Revenue projections must account for significant pricing variation across geographies. The US market supports premium pricing for repurposed drugs in specialty indications, particularly when orphan designation, small patient populations, or no alternative therapy supports a value-based pricing argument. European markets operate under health technology assessment (HTA) frameworks — NICE in the UK, HAS in France, G-BA in Germany, AIFA in Italy — that require comparative effectiveness evidence against the standard of care, not merely placebo. A repurposed drug that has only placebo-controlled data is likely to receive a minor benefit rating in the German AMNOG process, which caps the negotiated price below the manufacturer’s preferred level.
Japan’s National Health Insurance (NHI) price list, which governs all drug pricing in Japan, uses a formula-based premium system for drugs classified as innovative. Repurposed drugs with a clearly novel mechanism of action (even if the compound is known) may qualify for the innovation premium, materially improving the NHI price versus a generic equivalent.
Key Takeaways for this Section
Biologics now hold 62% of repurposing market revenue, driven by multi-indication antibody platforms and multi-omics-driven indication identification. Disease-centric approach programs dominate because the regulatory incentive structure — orphan designation, HTA evidence requirements — is organized around diseases, not compounds. European market access requires head-to-head comparative data against the standard of care; US payers require real-world evidence post-approval in most cases.
XI. Pricing Strategy
Value-Based Pricing: The Mechanics
Value-based pricing for a repurposed drug links the price to the clinical benefit the drug delivers in the new indication, expressed in terms that payers can quantify: quality-adjusted life years (QALYs) gained, hospitalizations avoided, surgery prevented, or productivity restored. The standard ICER (incremental cost-effectiveness ratio) framework in the US, used by the Institute for Clinical and Economic Review (ICER) for non-binding reference pricing, expresses value as the cost per QALY gained versus the standard of care. At a willingness-to-pay threshold of USD 100,000 to USD 150,000 per QALY, a drug that adds 1 QALY in a disease where the alternative is best supportive care can theoretically justify an annual cost of USD 100,000 to USD 150,000, regardless of its manufacturing cost.
For repurposed drugs, the value-based case is often complicated by the reference point. If the repurposed drug is a formerly off-patent compound available as a generic, the payer’s implicit cost baseline is the generic price. Demonstrating that the reformulated, newly indicated version provides enough incremental clinical benefit to justify a 50x or 100x price premium over the generic requires robust clinical evidence, a compelling QALY model, and a clear narrative for why the generic version is not an adequate substitute. The administration route shift argument — IV to SC, hospital to home — is often the most defensible payer narrative, because it directly reduces healthcare system costs (infusion center costs, nursing time, hospital bed utilization) in a way that is easy to quantify.
Published analysis of marketed repurposed drugs shows that a change in administration route to a hospital or outpatient infusion center setting can produce a price increase of 150% to 300% over the oral generic equivalent, without any change to the underlying molecule. A shift from IV to self-injectable SC can produce a further 20% to 40% premium over the IV product, primarily because of patient preference and reduced healthcare utilization. These data points provide a practical pricing anchor for programs developing SC formulations of repurposed biologics.
Medicaid Best-Price Rule: The Structuring Problem
For US programs that include Medicaid in the payer mix — which is nearly all programs, given Medicaid’s coverage of large numbers of patients with chronic disease — the Medicaid best-price rule creates a structuring constraint. Manufacturers must report to CMS the lowest price at which they sell a drug to any customer in the US (with limited exceptions). This price becomes the Medicaid rebate base; if the manufacturer offers a deep discount or a value-based contract with a large commercial payer, that price could become the Medicaid best price, triggering commensurate rebate obligations across all Medicaid units. This dynamic has historically prevented manufacturers from offering steep outcome-based rebates to commercial payers, because the rebate would propagate through the Medicaid system.
The Center for Medicaid and Medicare Innovation (CMMI) has experimented with best-price exemptions for certain value-based arrangements, but the framework remains in flux. For a repurposing program entering a high-Medicaid indication — rare pediatric disease, severe mental illness, substance use disorder — structuring value-based contracts with commercial payers requires careful modeling of Medicaid best-price exposure and, in some cases, dedicated consultation with CMS before contracting.
Pricing for Orphan Drug Indications
Orphan indications support the highest price points in the pharmaceutical market, reflecting small patient populations (which require high per-patient revenue to generate acceptable total returns), typically high unmet need (which supports strong value-based pricing arguments), and 7-year market exclusivity (which limits payer leverage by preventing generic entry). Published data on approved orphan drugs show median annual treatment costs of USD 150,000 to USD 500,000 in most rare disease categories, with some ultraorphan therapies (fewer than 1,000 US patients) priced above USD 1 million per patient per year.
For a repurposing program in an orphan indication, the pricing ceiling is primarily constrained by payer willingness to pay — for rare disease, this is driven by the severity of the disease and the clinical magnitude of the benefit — rather than by the drug’s cost basis. A generic compound that costs USD 5 per dose to manufacture can legitimately be priced at USD 50,000 per year in a severe orphan indication if the clinical evidence supports it and the 7-year exclusivity holds. The price is set by the value in the new indication, not by the history of the compound.
Key Takeaways for this Section
Value-based pricing for repurposed drugs requires clinical evidence sufficient to support a QALY model against the standard of care. Administration route changes are the most easily quantified source of incremental value in payer negotiations. Medicaid best-price rule exposure must be modeled before commercial contracting structures are finalized in high-Medicaid indications. Orphan indications support price points driven by clinical benefit and small patient populations, fully independent of the compound’s original or generic price.
Investment Strategy Note
For portfolio managers evaluating repurposing assets in rare disease, the orphan pricing scenario deserves an explicit model: map annual treatment cost to patient population size, model Medicaid and commercial payer coverage separately, and apply the 7-year exclusivity duration to peak sales timing. In most well-constructed orphan repurposing programs, this model supports a valuation that makes the clinical development economics highly attractive relative to novel drug development in the same indication.
XII. Market Acceptance and Reimbursement
The Off-Label Use Problem
Off-label prescribing — using an approved drug in an unapproved indication, dose, or patient population — is legal in the US and common in clinical practice. Surveys suggest that 15% to 20% of all drug prescriptions in the US are written off-label, with rates exceeding 50% in certain specialty categories such as oncology and psychiatry. This off-label prescribing represents a market that exists before formal repurposing approval but that creates a commercial paradox: it validates the clinical hypothesis but creates the incentive problem that limits investment in formal approval.
Generic manufacturers cannot promote their products for off-label uses under FDA rules, and their incentive to pursue a new NDA for an indication where they would receive only 3 years of data exclusivity (not 7-year orphan exclusivity and not CoM patent protection) is limited. This is the core market failure identified in the FAS report on generic drug repurposing: the expected revenue from a new indication is shared across all generic manufacturers without any exclusivity mechanism to reward the first mover. The Incentivizing Generic Drug Repurposing report from the University of Chicago’s Market Shaping Accelerator (2025) specifically proposed an advance market commitment (AMC) structure — where a government or payer pre-commits to purchasing a defined quantity of a repurposed generic at a predetermined price — as a mechanism to restore investment incentives. This proposal has not yet been enacted in US law but has attracted bipartisan interest.
The practical implication for commercial repurposing strategy is: wherever possible, structure the program to generate exclusivity (orphan designation, formulation patent, new clinical investigation exclusivity) that creates a competitive moat even if competitors sell a generic version of the same compound for a different indication. The branded reformulation plus the formal label for the new indication plus the payer coverage certainty that comes with on-label status are collectively worth more than the off-label market share, even in therapeutic areas where off-label prescribing rates are high.
HTA and Comparative Effectiveness: The EU Access Hurdle
In EU markets governed by HTA, reimbursement for a repurposed drug depends on demonstrating comparative effectiveness against the standard of care, not merely efficacy versus placebo. The challenge for many repurposing programs is that their pivotal clinical evidence comes from placebo-controlled studies — designed when the repurposing hypothesis was being confirmed — rather than from head-to-head active-controlled trials. NICE in the UK routinely requires network meta-analyses (NMAs) to indirectly compare a new drug’s efficacy against standard-of-care treatments when direct head-to-head data are unavailable. NMAs are sensitive to methodology assumptions and are often contested by both manufacturers and HTA bodies.
The EU Joint Clinical Assessment (JCA) framework, which began rolling out in 2025, creates a single EU-level HTA assessment for certain oncology and advanced therapy medicinal products, with national HTA bodies making reimbursement and pricing decisions based on the EU-level clinical assessment. Repurposing programs in scope for JCA will need to submit a single clinical evidence dossier covering comparative effectiveness across EU member states, rather than separate HTA submissions in each country. This reduces administrative cost but also removes the flexibility to tailor evidence packages to individual country HTA requirements.
Patient Advocacy and Crowdfunding as Market Development Tools
For repurposed drugs targeting rare or neglected diseases, patient advocacy organizations (PAOs) operate as de facto market development partners. PAOs fund early-stage academic research, recruit patients into clinical trials, pressure payers for coverage of promising off-label uses, and generate demand for formal regulatory approval. For diseases where the patient community is organized and active — Duchenne muscular dystrophy, amyotrophic lateral sclerosis, certain rare metabolic disorders — PAO involvement can reduce patient recruitment timelines by 30% to 50% and can materially influence FDA priority review decisions through accelerated approval pathways tied to patient-centered outcome measures. Commercial sponsors who co-develop relationships with PAOs early — through research grants, trial infrastructure support, and transparent communication about development plans — gain access to recruitment networks and regulatory influence that cannot be replicated through standard clinical operations.
Crowdfunding platforms, including LifeArc’s Mission fund and the crowdsourced clinical trial model used by some rare disease advocates, have funded early-phase repurposing studies for compounds where commercial sponsors have not yet committed. These crowdfunded studies generate clinical data that can de-risk a subsequent commercial development program, effectively providing free Phase II data to any sponsor willing to pick up the program afterward.
Key Takeaways for this Section
The off-label use market validates clinical hypotheses but does not solve the commercialization problem; formal approval plus exclusivity protection is the required structure for sustainable revenue. EU HTA bodies require head-to-head comparative evidence; programs designed only with placebo controls face access barriers in major EU markets. PAO engagement in rare disease repurposing is a material commercial advantage when structured early.
XIII. The Collaborative Ecosystem
What Each Stakeholder Actually Brings
Drug repurposing programs that reach the market typically involve at least three distinct organizational types working in sequence or in parallel. Understanding what each stakeholder genuinely contributes — and what gaps remain — determines partnership structure, IP ownership splits, and milestone payment design.
Pharmaceutical companies bring capital, regulatory expertise, manufacturing infrastructure, commercial distribution, and global market access. They are most valuable in the late-stage development and commercialization phases. Their repurposing strategy is predominantly driven by lifecycle management — extending franchise value for compounds whose original CoM patents are approaching expiry — and by portfolio gap filling in therapeutic areas where internal R&D has not generated a sufficient pipeline. Companies with large existing sales forces in a specific therapeutic area can commercialize a repurposed drug in that area at near-zero incremental fixed cost, dramatically improving the commercial economics.
Biotechnology companies provide platform technologies — AI-powered target identification, multi-omics screening, proprietary biomarker characterization — and the organizational agility to run early-stage repurposing programs faster than large pharma. Recursion Pharmaceuticals, for example, uses a cellular imaging-based phenotypic screening platform that processes approved compounds across thousands of disease models at a throughput that traditional biochemical screening cannot match. Recursion’s collaborations with Bayer for fibrotic disease and with Roche/Genentech for oncology use this platform to generate repurposing hypotheses that the large pharma partner then develops and commercializes through its own infrastructure.
Academic institutions generate the scientific discoveries that underlie most repurposing hypotheses. Published analyses estimate that academic and public-sector institutions have contributed to nearly 90% of new indications developed for previously approved drugs. This contribution is upstream: basic research identifying a new disease mechanism, a new drug-target interaction, or a new patient subgroup. The challenge is translation — moving from a published finding to an IND-ready development candidate requires CMC work, regulatory strategy, and trial design expertise that most academic centers lack. The “valley of death” between academic discovery and clinical development is where most repurposing hypotheses are lost, and bridging it requires either a dedicated academic translational infrastructure (found at a small number of research-intensive universities) or a commercial partnership arranged early enough in the process that IP ownership is clearly established before publication or public disclosure.
Contract Research Organizations (CROs) provide execution infrastructure: clinical trial management, patient recruitment, regulatory submission preparation, pharmacovigilance, and biostatistics. In repurposing programs, where the development team is often leaner than in a standard NCE program, CRO selection and scope definition are critical early decisions. CROs with specific indication expertise — an oncology CRO with established networks at major cancer centers, or a rare disease CRO with experience running trials in small patient populations with geographically dispersed enrollment — can reduce trial timelines by 20% to 30% relative to general-purpose CROs working in an unfamiliar therapeutic area.
IP Ownership in Multi-Partner Collaborations
The IP ownership structure of a multi-partner repurposing collaboration deserves more attention than it typically receives at the partnership formation stage. Academic partners often operate under technology transfer frameworks that give the institution background IP ownership over discoveries made in academic laboratories, with commercial licenses negotiated separately. If an academic institution’s published finding provides the repurposing hypothesis, and a commercial sponsor then funds clinical development, the question of who owns the method-of-use patent that covers the new indication — the academic institution (as the source of the hypothesis) or the commercial sponsor (as the funder of the clinical evidence) — must be resolved in writing before any publication or clinical trial begins. Disputes over this question have derailed commercially promising repurposing programs when they arise after clinical data has been generated.
Standard contractual frameworks, including the Intellectual Property Management guidelines from the National Institutes of Health (NIH) and the AUTM standard license terms, provide starting points. In practice, commercial sponsors typically negotiate exclusive licenses to academic background IP relevant to the repurposing indication, coupled with assignments of foreground IP generated during the funded development program, in exchange for upfront fees, milestone payments, and tiered royalties. The royalty rate on repurposed drugs sourced from academic discoveries typically ranges from 2% to 6% of net sales, reflecting the significant commercial investment required to reach market.
Key Takeaways for this Section
Academic institutions generate most repurposing hypotheses but lack the commercial infrastructure to develop them. IP ownership must be contractually settled before publication or trial initiation. CRO selection by indication-specific expertise, rather than by price alone, reduces trial timelines meaningfully.
XIV. Future Outlook: AI, RWD, and Precision Medicine
AI in Repurposing: What the Technology Can and Cannot Do
AI and machine learning tools applied to repurposing fall into four functional categories: target prediction (predicting which molecular targets an existing drug interacts with, beyond its characterized pharmacology), indication prediction (predicting which disease phenotypes a drug’s known activity profile would address), patient stratification (predicting which patient subpopulations within a target disease would respond), and clinical trial optimization (predicting trial design parameters — sample size, endpoint selection, enrollment criteria — that maximize the probability of demonstrating a statistically significant effect).
Deep learning models for target prediction, particularly graph-based models trained on protein structure data from AlphaFold and experimental binding databases, have substantially improved the accuracy of off-target activity prediction. This makes the drug-centric repurposing hypothesis more tractable: rather than relying on serendipitous off-target observations, researchers can computationally predict them with meaningful accuracy. The Exscalate4Cov project’s identification of raloxifene as a COVID-19 candidate via supercomputing-scale docking was an early proof of principle; subsequent platforms have improved in speed and accuracy.
What AI cannot currently do is reliably predict clinical efficacy in humans from preclinical or computational data alone. The gap between target engagement and therapeutic benefit remains large, and no machine learning model trained on preclinical data has demonstrated reliable clinical outcome prediction across diverse indication categories. AI narrows the candidate list from thousands to dozens; it does not replace clinical trials, and programs that proceed to Phase II on the strength of computational evidence alone, without robust biological validation in relevant preclinical models, continue to fail at rates similar to those of conventional drug development.
Real-World Data as a Continuous Signal
Post-approval, repurposed drugs generate real-world evidence in the new indication that can be used in several commercially valuable ways. Pharmacovigilance data from the FDA’s Adverse Event Reporting System (FAERS) and the EMA’s EudraVigilance database may reveal safety signals specific to the new patient population that were not detectable in the pivotal trials. Equally, RWD can reveal unexpected efficacy signals — subgroups of patients who respond better than the trial average, clinical settings where the drug outperforms standard of care by a larger margin — that support future label expansion or patient enrichment strategies.
RWD analysis using claims databases and EHRs is increasingly accepted by FDA as supportive evidence in label expansion discussions, particularly for supplemental indications where a full randomized trial would be ethically or logistically impractical. The FDA’s Real-World Evidence framework, published in 2023, specifies the data quality standards and study design requirements that make RWD acceptable for regulatory purposes. For repurposing programs in rare disease, where randomized controlled trials may be impossible to power adequately given small patient populations, a well-designed RWD study meeting FDA’s framework criteria can constitute a primary evidence basis for a supplemental NDA.
Precision Medicine and Biomarker-Stratified Repurposing
The convergence of repurposing strategy with precision medicine — using genomic, proteomic, or imaging biomarkers to identify responsive patient subpopulations — is the most commercially promising direction for the next decade. Biomarker-stratified repurposing programs address two simultaneous problems: the problem of heterogeneous patient populations that dilute clinical signal in broad trials, and the problem of thin prior art for a method-of-use patent on a broad indication.
A repurposed drug developed for a genomically defined subpopulation of a common disease — say, a kinase inhibitor repurposed for a non-cancer disease driven by the same kinase mutation — can simultaneously achieve: (1) a cleaner Phase II signal in a more homogeneous patient population; (2) a method-of-use patent claim defined by the biomarker, which is narrower (more defensible against prior art challenges) and easier to enforce against generic prescribers (because the biomarker-defined patient population is tracked through companion diagnostic testing); and (3) a companion diagnostic revenue stream for the biomarker test, which creates an additional IP asset entirely separate from the drug itself.
The regulatory pathway for a biomarker-stratified repurposing program includes parallel review of the drug and the companion diagnostic under FDA’s in vitro diagnostic (IVD) approval process, and potentially a co-development and co-marketing agreement between the drug sponsor and a diagnostic company. These arrangements add structural complexity to the partnership architecture but are well-precedented in oncology (KRAS companion diagnostics, BRAF/MEK co-development programs) and increasingly practiced in other therapeutic areas.
Key Takeaways for this Section
AI tools are hypothesis generators, not clinical outcome predictors; biological validation remains mandatory. RWD as a regulatory evidence basis for supplemental indications is increasingly accepted under the FDA’s 2023 framework, particularly in rare disease where randomized trials are impractical. Biomarker-stratified repurposing programs generate cleaner clinical signals, stronger method-of-use IP, and companion diagnostic revenue — three simultaneous advantages that make this approach the direction most likely to dominate repurposing deal flow in the next 5 to 10 years.
Investment Strategy Note
Programs combining a repurposed compound with a validated companion diagnostic and a biomarker-defined patient population represent the highest IP density of any repurposing structure: the drug carries method-of-use claims defined by the biomarker, the diagnostic carries a separate IP position, and the co-development agreement creates a preferential distribution relationship with a diagnostic partner. For portfolio managers, the correct valuation framework for these programs models the drug and diagnostic IP separately and applies deal-level transaction comps from the oncology companion diagnostic market, not from the general drug repurposing deal database.
XV. Master Investment Strategy Checklist
The following checklist consolidates the actionable decision points from each section of this guide. It is intended for IP teams, portfolio managers, business development leads, and R&D decision-makers evaluating a repurposing asset at any stage from early discovery to pre-commercialization.
Candidate Assessment
Confirm the mechanism of action hypothesis is supported by at least two independent data streams (computational, in vitro, in vivo, or clinical RWE). Verify that prior art on the new indication is thin — meaning no published study has demonstrated the same compound in the same indication before the proposed patent priority date. Assess whether the program qualifies as soft repurposing (related indication, short regulatory bridge) or hard repurposing (distant indication, substantial clinical program required), and size the development budget accordingly. Determine whether biomarker stratification is available and whether a companion diagnostic can be co-developed.
IP Audit
Map all secondary patent claims: method-of-use, formulation, dosing regimen, patient population, combination. Assess each claim for prior art vulnerability and model IPR challenge probability. Confirm Orange Book listing eligibility for formulation claims. Calculate the cumulative effective exclusivity duration from all IP sources (patents plus regulatory exclusivity, plus any pediatric extension). Run scenario analysis for three IP strength levels: all claims valid, formulation claims only valid, no claims valid (data exclusivity only).
Regulatory Planning
File ODD before Phase II investment if the new indication qualifies. Schedule Pre-IND meeting with FDA to align on the 505(b)(2) data reliance scope before designing any bridging studies. Assess eligibility for Fast Track, Breakthrough Therapy, or Priority Review designation. In EU programs, map the PRIME scheme eligibility and the Article 48 legislative timeline for any indication involving off-patent compounds where academic studies are in progress.
Commercial and Pricing Strategy
Build an income approach IP valuation with explicit patient population sizing, exclusivity duration scenarios, and a clinical evidence strength-adjusted price assumption. Construct a payer-specific access model: separate Medicaid (best-price exposure), commercial, VA/DoD, and Medicare coverage scenarios. In EU programs, build a comparative effectiveness evidence gap analysis: what head-to-head data will NICE, G-BA, and HAS require, and what does the current evidence package provide versus that requirement? Model value-based pricing anchored to QALY calculations for the new indication, with sensitivity analysis to different willingness-to-pay thresholds.
Partnership Structure
In multi-partner collaborations, execute IP ownership agreements before any publication or trial initiation. Structure milestone payments around patent prosecution events, not just clinical milestones. In academic partnerships, negotiate exclusive licenses to background IP and assignments of foreground IP before funding any clinical development work. Select CROs based on indication-specific trial execution experience, not on headline price.
Portfolio-Level Governance
Assign a dedicated cross-functional team — IP, regulatory, commercial, medical affairs — to each repurposing program from the point of IND filing. Establish quarterly portfolio reviews that track IP prosecution status, regulatory exclusivity timeline, RWD evidence generation, and commercial payer access development in parallel, not sequentially. Treat IP events (patent grant, ODD approval, first Paragraph IV certification received) as portfolio-level milestones with defined commercial and legal response protocols, not as legal department notifications to be processed after the fact.
This guide reflects regulatory frameworks, market data, and IP prosecution doctrine current as of March 2026. Regulatory frameworks — particularly the EU Article 48 legislative process and FDA’s evolving RWE guidance — are subject to change. Confirm current status of cited provisions before relying on them in regulatory or legal strategy.