1. What Excipients Actually Do (and Why the ‘Inactive’ Label Is Costing You Sales)

The phrase ‘inactive ingredient’ is a regulatory classification, not a commercial one. It tells you something about therapeutic intent. It tells you nothing about formulation criticality, supply chain leverage, or pricing power.
Excipients can constitute up to 90% of a finished dosage form by mass. In a 500 mg tablet, the API might occupy 50 mg. Everything else, including the binder holding the matrix together, the disintegrant that ruptures it in the duodenum, the coating that delays that rupture until the correct gastrointestinal segment, and the glidant ensuring uniform powder flow through the tablet press, is excipient. Strip any one of those components out and you do not have a cheaper drug. You have no drug.
That functional density translates into commercial leverage that most excipient sales organizations systematically under-monetize. The reasons are partly structural: excipient businesses typically sell into procurement departments that benchmark on raw material price per kilogram, not on formulation-enabling value. Reframing that conversation requires fluency in the IP and regulatory mechanisms that actually drive pharmaceutical R&D decision-making.
This article is built for that reframing. It is written for excipient commercial teams, IP strategists, portfolio managers at pharma companies evaluating formulation partnerships, and institutional investors assessing the competitive durability of excipient manufacturers.
Key Takeaways: Section 1
Excipients are decision-determining components in drug performance, regulatory approval, and patent lifecycle strategy. The ‘inactive’ designation is a toxicity classification, not a commercial diminishment. Excipient businesses that sell on price per kilogram rather than formulation-enabling value are leaving substantial margin on the table.
2. The $14 Billion Excipient Market: Segment-Level Revenue Anatomy
Overall Market Size and Growth Trajectory
Three major research firms have produced consistent projections across different base years. MarketsandMarkets estimated the global pharmaceutical excipients market at $10.5 billion in 2024, projecting a CAGR of 6.1% to $14.86 billion by 2030. Grand View Research puts the 2023 baseline at $9.88 billion, reaching $13.05 billion by 2030 at a 4.1% CAGR. Precedence Research projects growth from $8.37 billion in 2023 to $14.72 billion by 2033 at a 5.81% CAGR. The variance in base estimates reflects differing scope definitions, particularly around whether biopharmaceutical and parenteral excipients are included in the core figure.
The directional consensus is firm: the market grows above general pharma sector rates, driven by formulation complexity rather than simple volume expansion.
High-Growth Sub-Segments
The biopharmaceutical excipients sub-segment, valued at $2.45 billion in 2022, grows at approximately 5.5% annually to reach an estimated $3.76 billion by 2030. That growth is concentrated in stabilizers and tonicity agents for parenteral biologics, particularly high-purity carbohydrates like trehalose and sucrose, which protect protein structure during lyophilization cycles. The instability challenges unique to monoclonal antibodies (mAbs), fusion proteins, and ADCs create a technical barrier that commodity excipient suppliers cannot easily clear.
The sustained-release excipients sub-segment grows faster still, at 8.4% CAGR from a $1.26 billion base in 2022 to a projected $2.36 billion by 2030. This segment’s growth has a direct structural driver: patent-holding branded manufacturers routinely file secondary patents on sustained-release formulations as their base API patents near expiry, creating consecutive waves of formulation demand.
Product Category Breakdown
Polymers, primarily microcrystalline cellulose (MCC) and pregelatinized starch, account for over 45% of revenue and dominate sustained-release oral applications. Lactose-based excipients hold the highest overall market share by excipient type at approximately 41%. By function, binders and fillers at 49.72% of 2023 revenues make up the largest single functional category.
Alcohols show the highest projected growth rate among product categories, driven primarily by their role as solubilizers and co-solvents in parenteral and semi-solid formulations. Coating agents, disintegrants, and flavoring agents round out the high-revenue functional tiers.
Regional Demand Distribution
North America held approximately 38% of 2023 revenues, anchored by FDA-regulated NDA/ANDA filing volumes and the density of pharmaceutical manufacturing in the US. Europe’s market share reflects a high concentration of branded drug manufacturing and biosimilar development. Asia Pacific projects the highest CAGR at 6.9%, driven by ANDA filing activity in India, generic manufacturing expansion in China, and growing domestic drug consumption in Southeast Asia.
The asymmetry matters strategically: the US and EU generate the most technically demanding excipient requirements, particularly for controlled release and biologics. Asia Pacific generates the highest volume growth but competes primarily on cost efficiency.
Key Takeaways: Section 2
The global excipient market is not a single growth story. Biopharmaceutical and sustained-release sub-segments grow at twice the rate of the overall market and carry materially higher margins. Excipient companies that have not explicitly segmented their commercial strategy by these sub-segments are allocating resources against a blended average that obscures the actual value creation opportunity.
3. The Patent Cliff Is Not a Threat to You, It’s a Pipeline
Scale of the Opportunity
Between 2025 and 2030, approximately $300 billion in annualized pharmaceutical revenue faces loss of exclusivity (LOE). That figure comes from a wave of blockbuster expirations including Keytruda ($25 billion in 2023 revenues), Eliquis ($12 billion), Stelara ($10 billion), and Opdivo ($9 billion), along with roughly 186 additional products spanning oral solids, injectables, and biologics.
For excipient manufacturers, this is a demand scheduling problem, not a threat. When a patent expires and generic manufacturers file ANDAs or biosimilar 351(k) applications, they need formulation-grade excipients. They need them fast, in quantity, with clean regulatory support documentation, and often with technical application support for bioequivalence studies. Companies that have tracked these expirations and pre-positioned supply and application data will capture those contracts. Companies that respond reactively will not.
The Generic Entry Mechanics
Generic market share transfer is not gradual. Within 12 months of LOE for a typical small-molecule oral solid, the reference listed drug often loses 70% to 80% of unit volume to generic entrants. For a drug generating $5 billion annually, that represents roughly $4 billion in revenue shifting to manufacturers who need cost-efficient, BE-validated formulations on an accelerated timeline.
The first generic filer who receives 180-day exclusivity under Paragraph IV certification has a particularly acute need: they must launch quickly to capture the exclusivity window before additional generics enter. Speed-to-formulation directly determines the commercial value of that 180-day window, and excipient quality, consistency, and availability are a rate-limiting factor in the ANDA review process.
Beyond Oral Solids: The Biosimilar Opportunity
The biologic LOE wave, anchored by Keytruda’s 2028 expiry and Enbrel’s staggered US/EU expirations, generates a parallel but technically distinct demand signal. Biosimilar development requires high-purity biopharmaceutical excipients with documented lot-to-lot consistency, low endotoxin specifications, and in many cases, excipient-specific comparability data to support the biosimilar’s analytical similarity package.
The concentration of stabilizers, tonicity agents, and surfactants in a biologic formulation is not a commodity procurement decision. It is a regulatory document. Excipient manufacturers who have invested in the quality infrastructure to provide Type IV Drug Master File (DMF) support, lot release documentation, and excipient characterization data for biologics applications will be preferred suppliers as the biosimilar pipeline expands.
Key Blockbuster LOE Schedule, 2025-2030
| Drug | API Class | US LOE | 2023 Revenue (est.) | Primary Dosage Form |
|---|---|---|---|---|
| Stelara (ustekinumab) | Biologic mAb | 2025 | $10B | SC injection |
| Revlimid (lenalidomide) | Oral IMiD | 2025 | $9B | Oral capsule |
| Ozempic (semaglutide) | GLP-1 agonist | 2025 | $3.6B | SC pen injection |
| Eylea (aflibercept) | Biologic fusion | 2025-2026 | $5.9B | Intravitreal |
| Eliquis (apixaban) | Oral anticoagulant | 2026 | $12B | Oral tablet |
| Xarelto (rivaroxaban) | Oral anticoagulant | 2026 | $4.5B | Oral tablet |
| Ibrance (palbociclib) | CDK4/6 inhibitor | 2027 | $5.39B | Oral capsule |
| Trulicity (dulaglutide) | GLP-1 agonist | 2027 | $7B | SC pen injection |
| Keytruda (pembrolizumab) | Biologic mAb | 2028 | $25B | IV infusion |
| Opdivo (nivolumab) | Biologic mAb | 2028 | $9B | IV infusion |
| Enbrel (etanercept) | Biologic fusion | 2028-2029 | $3B | SC injection |
Key Takeaways: Section 3
The $300 billion LOE wave is an excipient demand pipeline with a published schedule. Each drug on that list represents a formulation project that will require specific excipient packages. Excipient companies that build product-level intelligence on these drugs, including their current formulation compositions and the technical requirements for BE or biosimilar equivalence, will have a material commercial advantage over those entering the market reactively.
4. Drug Patent Architecture: A Technical Decoder for Formulation Teams
The Patent Stack: From Base Compound to Tertiary Device Integration
A drug on the market at any given time typically has not one patent but a layered portfolio of patents with staggered expiration dates. Understanding that stack is the foundation of any serious excipient intelligence operation.
The base patent covers the active moiety, either the chemical compound itself or, for biologics, the protein sequence or DNA construct. This is the primary IP asset established during drug discovery and typically filed well before NDA submission. Because the 20-year statutory patent term runs from filing date, not approval date, and because the average time from IND filing to NDA approval spans 7 to 12 years, the effective market life of the base patent at launch is often 8 to 12 years, sometimes less.
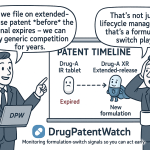
Secondary patents build the protective structure that matters most to excipient businesses. These patents cover crystalline polymorphs and substantially pure forms of the compound, specific enantiomers, novel formulations, methods of use, new dosage strengths, new routes of administration, and combination products. Secondary patents are typically filed closer to NDA submission or post-approval, meaning their 20-year term extends further from the base patent’s expiry date. This is the structural mechanism behind evergreening: a brand can maintain effective exclusivity well past its base patent’s expiration by converting its generic-entry timeline from the base expiry date to the expiry of a later-filed formulation or method-of-use patent.
Tertiary patents integrate medical devices with an off-patent active ingredient. A pre-filled autoinjector for a biologic, a dry powder inhaler for an established respiratory compound, or a digital sensor-embedded patch for a controlled-release API each create fresh IP even when the molecule has been off-patent for years.
Formulation Patents: The Direct Commercial Interface with Excipients
Formulation patents are, by definition, patents about how an API is combined with other ingredients. They protect the composition, the ratio, the physical form, the release mechanism, and the method of administration. Excipients determine all of those attributes. A formulation patent that claims a novel controlled-release matrix exists because a specific combination of polymer excipients, their concentration ranges, and their physical interactions with the API produces a release profile that is patentable.
This creates a two-sided commercial dynamic for excipient manufacturers. On one side, formulation patents filed by branded companies signal specific functional requirements for excipients. The claims in those patents describe what the excipient must do. On the other side, the expiration of formulation patents opens the door for generic manufacturers to replicate those formulations, again requiring the same excipient package. Tracking formulation patents gives excipient companies a forward-looking demand signal for both sides of that market.
Paragraph IV Certifications as a Timing Signal
When a generic company files an ANDA and certifies under Paragraph IV that a listed patent is invalid or will not be infringed by their product, that filing represents an explicit commercial bet on a specific drug’s patent vulnerability. The certification triggers a 45-day window during which the patent holder can file an infringement suit, imposing a 30-month stay on FDA approval. Paragraph IV filings are public through the FDA’s Orange Book and litigation records.
For excipient manufacturers, active Paragraph IV litigation on a drug product tells you that a well-capitalized generic company has already decided to enter that market and is willing to litigate to do so. That is an earlier and more reliable demand signal than waiting for LOE to occur.
Patent Term Extension and Pediatric Exclusivity
Patent Term Extension (PTE) under the Hatch-Waxman Act allows the FDA to restore patent life lost during regulatory review. The calculation adds all of the NDA review period plus half of the IND-to-NDA clinical development time, capped at a total extension of five years, with the remaining patent term after extension not exceeding 14 years from approval. Only one patent per drug product qualifies for PTE.
Pediatric exclusivity, a separate 6-month protection attached to existing patents and exclusivities, delays LOE when a company conducts FDA-requested pediatric studies. Unlike NCE or new clinical investigation exclusivity, pediatric exclusivity attaches to any patent or exclusivity listed in the Orange Book for that drug. A drug with 3 months of base patent remaining plus 3-year new clinical investigation exclusivity plus 6 months of pediatric exclusivity has an effective LOE date that may be 3.5 years later than the base patent expiry suggests. Excipient companies tracking only patent expiry dates without accounting for these layered exclusivity mechanisms will consistently misforecast the timing of generic entry.
Key Takeaways: Section 4
The effective generic entry date for any drug is determined by the full patent and exclusivity stack, not the base patent alone. Excipient intelligence programs that track only API patent expirations miss the formulation patents, method-of-use patents, PTEs, and exclusivity periods that set the actual LOE timeline. Paragraph IV certifications are a real-time commercial signal indicating generic companies have committed resources to entering a specific market.
5. IP Valuation Frameworks for Excipient Portfolio Decisions
Excipient-Specific IP as a Balance Sheet Asset
Excipient manufacturers with proprietary formulation-enabling technologies have IP portfolios that are materially undervalued on most internal assessments. The reason is methodological: standard IP valuation approaches in the pharmaceutical sector, typically income-based NPV models or market transaction comparables, calibrate against API or drug product patents. Excipient patents, which protect compositions, manufacturing processes, and applications rather than therapeutic agents, do not map cleanly onto those templates.
A more accurate framework treats excipient IP in two distinct categories. Defensive IP consists of patents that prevent competitors from replicating a proprietary excipient technology. These patents protect market position rather than generate direct licensing revenue. Their value is calculated as the margin premium the proprietary excipient commands over commodity alternatives, multiplied by addressable volume over the remaining patent term, discounted at an appropriate rate.
Offensive or licensing IP consists of patents that the excipient manufacturer can license to pharmaceutical companies who need access to the technology for their own formulations. A patented co-processed excipient that enables direct compression of otherwise poorly compressible APIs, or a novel lipid-based delivery system that substantially improves bioavailability for BCS Class II molecules, has licensing value that can be structured as upfront fees plus per-unit royalties tied to the drug product’s commercial success.
Case Study: Captisol (Ligand Pharmaceuticals)
Captisol, chemically sulfobutylether beta-cyclodextrin (SBECD), is a modified cyclodextrin excipient developed to solubilize poorly water-soluble drugs for parenteral administration. Ligand Pharmaceuticals holds the Captisol IP and has licensed it to pharmaceutical manufacturers across multiple approved drug products, including Pfizer’s Vfend IV (voriconazole) and Acrotech’s Evomela (melphalan for injection).
As of Ligand’s most recent public filings, Captisol-related royalties contribute a recurring, high-margin revenue stream tied directly to the commercial performance of drugs that could not be formulated without it. Ligand’s market capitalization reflects substantial value attributable to the Captisol IP portfolio, demonstrating that excipient IP is a fundable, monetizable asset class when structured correctly.
This model, a proprietary excipient IP platform generating royalties on downstream drug product sales, is replicable. Excipient companies with novel solubilization technologies, stabilization systems for high-concentration biologics, or controlled-release polymer systems that enable new patent claims for brand manufacturers should be evaluating whether their IP is structured for licensing rather than product sale only.
Valuing Formulation-Enabling Technologies in the Context of Drug LOE
The commercial value of a formulation-enabling excipient technology scales with the revenue at risk in the drugs it addresses. A polymer system that enables a sustained-release formulation of a $5 billion drug generates a licensing value ceiling proportional to the brand’s incentive to pay for that extension. At typical LOE revenue erosion rates of 70-80% in year one, a brand manufacturer holding a $5 billion drug facing patent expiry in 24 months has an NPV-positive case for paying substantial licensing fees for a formulation patent that extends exclusivity by 3 to 5 years.
Excipient IP teams should build a drug-by-drug analysis mapping their proprietary technologies against approaching LOE dates. The output is a prioritized licensing pipeline, ranked by the revenue value the brand manufacturer stands to protect, the probability that the excipient technology supports a viable formulation patent, and the regulatory timeline for establishing the new formulation as a distinct drug product.
Investment Strategy: Excipient IP as a Pharma Portfolio Hedge
For institutional investors with pharmaceutical sector exposure, excipient companies with proprietary IP portfolios provide a structural hedge against LOE risk in branded drug positions. When blockbuster drugs lose exclusivity, excipient suppliers to the resulting generic market benefit from volume. When branded companies execute lifecycle management strategies using novel formulations, excipient IP licensors benefit from royalty streams tied to those new formulations.
Publicly traded excipient or specialty chemical companies with pharmaceutical application IP, including companies like Ashland Global Holdings (Klucel, Benecel polymers), Evonik Industries (EUDRAGIT polymer line), and Croda International (pharmaceutical lipids and surfactants), have revenue streams that are partially counter-cyclical to patent cliff dynamics. Pure-play excipient IP platforms like Ligand are more directly sensitive to drug product commercial performance.
Key Takeaways: Section 5
Excipient IP is a fundable asset class. The Captisol/Ligand model demonstrates that royalty streams on formulation-enabling excipient technologies can generate durable, high-margin revenues independent of direct product sales volume. Excipient companies that have not evaluated whether their novel technologies are structured for licensing are undermonetizing their IP portfolios. For investors, excipient IP platforms offer pharmaceutical sector exposure with structural hedging against LOE volatility.
6. How Patent Expiry Creates Specific Excipient Demand, Drug by Drug
Reading Formulation Composition from Orange Book and NDC Data
Each NDA contains a qualitative and quantitative formulation composition. That information is partially accessible through the FDA’s National Drug Code (NDC) directory, inactive ingredient database, and published labeling, and in greater detail through commercial patent intelligence platforms. For excipient commercial teams, this data is not background context, it is prospecting intelligence.
A drug with a complex modified-release formulation, for example Venlafaxine ER (Effexor XR), contains a specific package of polymer coatings, spheronization aids, and sustained-release rate-controlling polymers that will be replicated in exact or near-exact form by each ANDA applicant. The ANDA must demonstrate bioequivalence to the reference listed drug (RLD), and significant deviation from the RLD’s qualitative excipient composition, particularly for modified-release products, requires additional justification and potentially additional BE studies. This creates a strong gravitational pull toward the same excipient suppliers who served the innovator formulation.
Drug-by-Drug Excipient Demand Profiles
Semaglutide (Ozempic/Rybelsus) — LOE 2025-2026
The injectable semaglutide formulation is a subcutaneous solution containing the GLP-1 peptide with a buffer system (sodium phosphate dibasic), a tonicity agent (propylene glycol), a preservative (phenol), and water for injection. Biosimilar and generic semaglutide developers will need injectable-grade versions of these excipients with endotoxin specifications consistent with parenteral use. Oral semaglutide (Rybelsus) is more technically complex: it contains the absorption enhancer sodium N-(8-[2-hydroxybenzoyl]amino) caprylate (SNAC) at high concentrations (300 mg per tablet) to facilitate GI absorption of the peptide. SNAC itself is patented by Emisphere Technologies, now owned by Novo Nordisk, creating a patent thicket around the oral formulation that will delay generic replication independent of the API’s LOE date.
Apixaban (Eliquis) — LOE 2026
Eliquis tablets are immediate-release oral solids with a formulation comprising lactose monohydrate, microcrystalline cellulose (MCC), croscarmellose sodium, sodium lauryl sulfate (SLS), and magnesium stearate. This is a conventional compressed tablet composition. The ANDA opportunity for excipient suppliers is high-volume and relatively low-technical-complexity. The critical variable is BE: apixaban’s aqueous solubility is low (BCS Class II), meaning particle size control in both the API and filler excipients can influence dissolution and BE outcome. Suppliers of milled lactose and MCC with documented particle size distribution data will be preferred partners for ANDA developers targeting the Eliquis generic market.
Pembrolizumab (Keytruda) — LOE 2028
Keytruda is a lyophilized powder for IV infusion containing pembrolizumab, sucrose as a cryoprotectant, polysorbate 80 as a surfactant, histidine and histidine hydrochloride monohydrate as a buffer system, and water for injection. Biosimilar pembrolizumab development requires lot-to-lot consistency data for all excipients, particularly the sucrose (tonicity and cryoprotection), polysorbate 80 (interfacial stabilization), and histidine buffer. Regulatory expectations for biosimilar analytical similarity packages require excipient characterization data at a level of detail that commodity sucrose or polysorbate 80 suppliers cannot typically provide. Pharmaceutical-grade, compendial-compliant, well-documented suppliers with Type IV DMF support are the only viable options. Given Keytruda’s $25 billion revenue base, this is the single largest excipient demand event projected through the decade.
Key Takeaways: Section 6
The LOE schedule translates directly into a drug-by-drug excipient demand forecast when cross-referenced against formulation compositions. Different drug classes, oral solids, modified-release tablets, SC injectables, IV biologics, require fundamentally different excipient packages and documentation standards. Excipient commercial teams that have built product-level LOE intelligence will enter RFP processes ahead of competitors who have not.
7. Bioequivalence Mechanics: Where Excipient Properties Determine Generic Approval
What the 80-125% Rule Actually Measures
The most common misstatement about bioequivalence is that the generic must contain 80% to 125% of the active ingredient found in the reference drug. That is wrong. The 80-125% range applies to the 90% confidence interval of the ratio of the generic product’s pharmacokinetic parameters, specifically Cmax (peak plasma concentration) and AUC (area under the plasma concentration-time curve), to the same parameters measured for the RLD. For the 90% CI to fall within that range, the point estimate of the mean PK ratio must be very close to 1.0 in practice, often within 5-10%.
This distinction matters for excipient suppliers because it clarifies the mechanism by which excipient selection affects BE outcomes. Excipients do not change the quantity of API in the tablet. They change how the API dissolves, how quickly it dissolves, and how available it is at the intestinal epithelium. Those are the factors driving Cmax and AUC. A change in MCC grade that alters tablet porosity changes dissolution rate. A change in disintegrant concentration changes the time before drug release begins. A change in lubricant concentration can coat drug particles and retard dissolution. Any of these shifts can move the PK ratio outside the acceptance window, generating a BE failure that delays or prevents ANDA approval.
Particle Size Distribution and Dissolution
For BCS Class II drugs (low solubility, high permeability), dissolution is the rate-limiting step for absorption. The solubility of a drug particle increases as its surface area increases, which increases as particle size decreases. Micronization, reducing particles to the 1-1000 micrometer range, increases dissolution rate without affecting equilibrium solubility. Nanonization, reducing particles below 1 micrometer, can increase both dissolution rate and apparent solubility.
Excipients play a direct role in both processes. Polymers like PVPK-25 act as precipitation inhibitors in nanosuspension formulations, maintaining drug particles in a nanosized state during dissolution. For milled excipients used as fillers or diluents, the particle size distribution of the excipient itself affects blend homogeneity, compressibility, and the uniformity of drug distribution within the tablet matrix. Suppliers of milled lactose, MCC, or dicalcium phosphate for BCS Class II generic formulations should be providing particle size characterization data as a standard component of technical support packages.
Lipid-Based Drug Delivery Systems and Excipient Selection
BCS Class II and Class IV molecules with high lipophilicity are increasingly formulated as lipid-based drug delivery systems (LBDDS), including self-emulsifying drug delivery systems (SEDDS) and self-microemulsifying drug delivery systems (SMEDDS). In these systems, the drug is dissolved in a mixture of oils, surfactants, and co-solvents that spontaneously emulsify upon contact with gastrointestinal fluids.
Excipient selection is the formulation. The identity and HLB value of the surfactant system determines emulsion droplet size. The oil phase composition determines the drug’s solubility within the vehicle. The co-solvent selection affects both drug loading capacity and the viscosity of the fill for liquid-filled hard capsules or softgels. Critical pharmaceutical lipids for LBDDS include Cremophor EL (polyoxyl 35 castor oil), Labrasol (caprylocaproyl polyoxylglycerides), propylene glycol monocaprylate (Capmul PG-8), and medium-chain triglycerides. Each of these requires pharmaceutical-grade purity specifications, consistent fatty acid profiles across lots, and clean regulatory support to be viable in an ANDA or NDA formulation.
Binder Concentration Effects on Tablet Performance
Binders used in wet granulation (PVP, HPMC, HPC) and dry granulation (directly compressible MCC, Pearlitol) affect tablet hardness, porosity, and disintegration time through related but distinct mechanisms. Higher binder concentrations generally produce harder tablets with longer disintegration times. PVP K30 and HPMC E5 form dense matrices that can delay API release in immediate-release formulations if used at concentrations optimized for structural integrity but not dissolution. This creates a BE risk that is not apparent until dissolution testing against the RLD.
The practical consequence for excipient manufacturers is that technical data packages for binders should include concentration-response data on disintegration time and dissolution rate, ideally using a model BCS Class II drug to illustrate dissolution sensitivity. ANDA development teams evaluating binder options need this data before committing to a formulation, and suppliers who provide it differentiate on technical value rather than price.
Superdisintegrants: Mechanism and Selection Criteria
Crospovidone (crosslinked PVP, e.g., Kollidon CL), sodium starch glycolate (SSG), and croscarmellose sodium (Ac-Di-Sol) are the dominant superdisintegrants in contemporary tablet formulations. They outperform starch and MCC in disintegration rate because their disintegration mechanisms, swelling and wicking for SSG, wicking and swelling for crospovidone, and swelling and capillary action for croscarmellose sodium, operate at low concentration levels (typically 2-5% w/w).
The selection between these three affects formulation performance in ways that are not interchangeable. Crospovidone’s wicking mechanism makes it effective under high-compression conditions where SSG’s swelling mechanism may be partially suppressed. SSG is sensitive to high concentrations of soluble excipients that can suppress its water uptake. Croscarmellose sodium performs well across compaction pressures but is sensitive to moisture. For a generic developer targeting an immediate-release tablet with a tightly controlled dissolution specification, the choice of superdisintegrant is a formulation variable with direct BE implications, not a commodity substitution decision.
Key Takeaways: Section 7
Excipient selection directly determines whether a generic ANDA achieves BE on first attempt or requires costly reformulation and additional clinical studies. Suppliers who provide formulation-specific technical data, dissolution characterization, particle size documentation, and lipid system performance profiling give ANDA developers information that reduces their development risk. That risk reduction has a commercial value that justifies supplier differentiation beyond price.
8. Formulation Patent Strategy: How Branded Companies Use Your Products to Evergreen Their IP
The Mechanics of Evergreening
Evergreening refers to the strategy of filing sequential secondary patents on a drug product to extend the period during which no generic can enter the market without risk of patent infringement litigation. The structure of secondary patents makes this possible: while a base compound patent has a fixed expiration, a new formulation patent filed 10 years after the compound patent starts its own 20-year term. If the new formulation is listed in the Orange Book and becomes the reference listed drug, generic applicants must certify against it.
The pharmaceutical industry’s use of evergreening is well-documented. A Commonwealth Fund analysis identified several strategic mechanisms: authorized generics (the brand manufactures the generic itself), settlement agreements following Paragraph IV litigation, and compound patent filings on new polymorphs or enantiomers. Each of these involves formulation decisions that depend on specific excipient technologies.
Technology Roadmap: How Branded Companies Convert LOE Risk into Secondary Patent Filings
The lifecycle management strategy for a branded oral solid facing base patent expiry typically follows a recognizable sequence. In the 3-7 years before the base patent expires, the company’s formulation development team evaluates the following options, roughly in order of regulatory and IP complexity:
A controlled-release or extended-release reformulation is the most common first move. It requires a new NDA (typically a 505(b)(2) using the IR formulation as reference), a new clinical study demonstrating pharmacokinetic equivalence or superiority over the IR form, and new formulation patents covering the rate-controlling excipient system. Hydrophilic matrix systems using HPMC (hydroxypropyl methylcellulose) are standard for this approach. Ethocel (ethylcellulose) or Eudragit membrane coatings are used for pellet-based extended-release systems. Both are patentable composition claims when combined with specific concentration ranges and manufacturing parameters.
A fixed-dose combination (FDC) adds a complementary API to the formulation, often in a single tablet or bilayer configuration. This requires new formulation development, a new NDA, and generates fresh compound and formulation patents if the combination itself is novel. Excipient selection for bilayer tablets is technically constrained: the two layers must not interact chemically, the compression parameters must account for different layer hardness requirements, and the release profiles for both APIs must meet their respective pharmacopeial or clinical specifications simultaneously.
A new delivery route, for example converting an oral product to a transdermal patch or a subcutaneous injectable, represents the most technically complex lifecycle management approach. Transdermal formulations require pressure-sensitive adhesives, permeation enhancers, and backing membranes that are themselves patentable components. Injectable reformulations of previously oral drugs often require solubilizing excipients (cyclodextrins, lipid vehicles, polysorbates) and buffer systems specifically adapted to the new route.
Each step generates formulation patents, and each formulation patent requires a specific excipient package. Excipient manufacturers who track lifecycle management filings are, in effect, reading the branded pharmaceutical industry’s R&D pipeline.
Orange Book Patent Listing as a Demand Signal
When a pharmaceutical company lists a new patent in the Orange Book, specifically a formulation or method-of-use patent filed post-approval, it is signaling a lifecycle management move. That listing tells you what patent the company is betting its exclusivity extension on. The patent claims, which are public, describe the formulation. Those claims describe what excipients do, if not always what they are by name. A claim covering ‘a rate-controlling matrix comprising a water-soluble cellulose ether at 15-40% w/w’ is a demand specification for HPMC in a particular concentration range.
Excipient commercial teams should be monitoring new Orange Book listings for their key pharmaceutical customers and potential customers. New formulation patent listings are early indicators of active product development projects that need excipient support.
Key Takeaways: Section 8
Evergreening strategies by branded pharmaceutical companies generate the specific formulation patent filings that represent excipient demand at the high-value end of the market. Excipient manufacturers who track Orange Book patent listings and analyze formulation patent claims can identify branded company development priorities before an RFP is issued. This intelligence shortens the sales cycle and allows technical pre-positioning that competitors without patent intelligence cannot replicate.
9. The Regulatory Gauntlet: FDA, EMA, ICH, and the PRIME Program
The FDA’s Indirect Approval Architecture for Excipients
The FDA does not approve excipients through a standalone regulatory pathway. An excipient receives de facto regulatory acceptance only through its inclusion in an approved drug product. This architecture has two significant implications. First, excipients already listed in the FDA’s Inactive Ingredient Database (IID) carry a lower regulatory burden for new drug applications. The IID entry shows the excipient has been reviewed and accepted in at least one approved product, in a specific route of administration, at a documented concentration range. Drug developers use IID listings to minimize regulatory uncertainty in ANDA and NDA submissions.
Second, novel excipients, defined by the FDA as excipients with no prior use in approved drug products and no established history in food use, require a comprehensive safety evaluation embedded within the drug application. There is no standalone pathway for excipient approval. The entire NDA can be held up by unresolved questions about a novel excipient’s safety profile, making drug developers strongly averse to using excipients not already in the IID.
This structural bias toward established excipients creates a significant market protection for incumbents and a substantial market entry barrier for novel excipient technologies. It also creates a ceiling on innovation: drug developers who might benefit from a superior novel excipient often default to an established one because the regulatory cost of the novel option is internalized into their drug application timeline.
The PRIME Program: A Partial Solution with Real Limitations
In September 2021, the FDA launched the Pilot Program for the Review of Innovation and Modernization of Excipients (PRIME). The program’s explicit goal was to create a pre-drug-application review mechanism for novel excipients, allowing excipient manufacturers to submit toxicology and quality data to the FDA before a pharmaceutical company incorporates the excipient into a drug formulation.
Submissions to PRIME must describe the proposed use of the excipient, the specific drug development need it addresses, and the public health benefit it provides. Qualifying public health rationales include enabling a new route of administration, substantially enhancing bioavailability for a drug that currently cannot be adequately formulated, or supporting a new technology for treatment of a serious disease.
The program’s real limitation is structural: PRIME review does not produce an approvable excipient. It produces feedback on the data package required for FDA to evaluate the excipient within a future drug application. The excipient remains unapproved in any standalone sense. Drug developers still face the risk that the entire application could be delayed by excipient-related questions, meaning PRIME reduces but does not eliminate the regulatory risk premium that makes developers prefer IID-listed excipients.
For excipient manufacturers with truly novel technologies, the practical value of PRIME participation is two-fold. First, pre-submission FDA feedback on the data package can prevent a costly and timeline-damaging deficiency letter later in the drug application process. Second, PRIME participation signals to potential pharmaceutical development partners that the excipient manufacturer has engaged proactively with the regulatory process, reducing the partner’s perception of adoption risk.
Type IV DMFs: The Practical Regulatory Currency
A Type IV Drug Master File (DMF) is a submission to the FDA of confidential technical information about an excipient’s composition, manufacturing process, quality controls, and safety profile. The DMF is not reviewed or approved in isolation. It is referenced by a drug application (NDA or ANDA) when that application uses the excipient, and the FDA reviews the DMF in the context of the drug review.
The commercial value of a Type IV DMF is that it allows excipient manufacturers to protect proprietary manufacturing information while making it available to the FDA through the drug application review. Without a DMF, pharmaceutical companies using a proprietary excipient must either include the full manufacturing detail in their own application (disclosing it to their competitors) or rely entirely on a letter of authorization from the excipient supplier. The DMF pathway protects both parties’ interests.
For excipient manufacturers, maintaining current, well-documented Type IV DMFs for all commercially relevant excipients is a market access requirement, not a competitive differentiator. Companies without DMFs are excluded from consideration for regulated pharmaceutical formulations regardless of product quality.
ICH Q3C and Q3D: Elemental Impurity and Residual Solvent Obligations
ICH Q3C (Impurities: Residual Solvents) classifies organic volatile solvents into three classes based on toxicity. Class 1 solvents, including benzene, carbon tetrachloride, and 1,2-dichloroethane, are carcinogenic or have severe toxic potential and must be avoided in excipient manufacturing unless technically unavoidable. Class 2 solvents carry organ toxicity risks and are controlled to Permitted Daily Exposure (PDE) limits. Class 3 solvents, including ethanol, acetone, and ethyl acetate, have low toxic potential and are acceptable with limits.
Excipient manufacturers are obligated to characterize residual solvent content in their products, provide that data to pharmaceutical customers, and document that cumulative solvent levels in the finished drug product remain within PDE limits. Customers formulating multiple excipients into a single dosage form must aggregate solvent contributions across all components, meaning a supplier who provides incomplete solvent characterization data creates a downstream compliance gap.
ICH Q3D (Elemental Impurities) requires a risk-based assessment of elemental impurity levels from all sources in a drug product, including excipients, manufacturing equipment, and container closure systems. Class 1 elements, specifically arsenic, cadmium, mercury, and lead, require evaluation regardless of source or likelihood. This is particularly relevant for excipients derived from natural sources such as talc, stearic acid, or plant-derived polymers, where metal uptake from soil or processing equipment can introduce Class 1 impurities at concentrations that require documentation.
EMA Requirements and EU Market Access
The EMA’s marketing authorization dossier requirements for excipients align closely with FDA expectations but have distinct features relevant to EU market access. The EMA publishes an excipient labeling annex that specifies which excipients with known pharmacological activity must be disclosed in the medicine’s summary of product characteristics (SmPC) and patient information leaflet. This list includes lactose, sorbitol, aspartame, benzyl alcohol, tartrazine (E102), propylene glycol, and several others, with specific warnings required for patient populations at risk.
For excipient manufacturers targeting EU market access, the practical implication is that formulations incorporating these flagged excipients require additional labeling consideration during drug development. This creates a mild commercial incentive for pharmaceutical developers to avoid flagged excipients where functionally equivalent alternatives exist, which is relevant to excipient business development positioning.
Key Takeaways: Section 9
Regulatory infrastructure, specifically the IID, DMF system, PRIME program, and ICH quality guidelines, defines the barrier structure of the excipient market. Incumbents with well-documented DMFs and IID listings have structural market access advantages that new entrants cannot overcome quickly. Novel excipient developers should engage with the PRIME program as a risk-reduction tool, not as an approval pathway. ICH Q3C and Q3D compliance documentation is a mandatory commercial deliverable for any excipient supplied into regulated pharmaceutical formulations.
10. Building a Patent Intelligence Operation: Tools, Workflows, and Signal Prioritization
Data Sources and Their Relative Utility
A functional pharmaceutical patent intelligence operation uses several data sources in combination. No single source provides complete coverage of the signals that matter to excipient businesses.
The FDA Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations) is the primary source for patent expiration dates for approved US drug products, associated exclusivity periods, and ANDA applicant information. The Orange Book is updated daily and is the authoritative source for 30-month stay calculations and first-filer Paragraph IV eligibility determinations. Its limitation is that it covers only FDA-approved products and only patents that have been formally listed by the NDA holder.
The USPTO Patent Full-Text Database provides access to patent claims, specifications, and prosecution history for all US-issued patents. Patent claims are the legally operative text that defines the scope of protection. For formulation patents, the claims describe the excipient composition in functional or compositional terms. Systematic claim analysis of formulation patents covering drugs approaching LOE is the most reliable method for identifying specific excipient requirements.
Commercial patent intelligence platforms, including DrugPatentWatch, Cortellis Drug Intelligence, and Evaluate Pharma, integrate Orange Book data, USPTO filings, ANDA filing information, and market data into searchable databases with alert functionality. These platforms reduce the manual effort of patent monitoring and can generate automated alerts when new patents are listed for drugs of commercial interest.
The FDA ANDA filing database, accessible through FDA’s Drugs@FDA portal, shows which generic companies have filed applications for specific reference listed drugs. This data, combined with Paragraph IV certification records (accessible through FDA’s 180-day exclusivity database), identifies which drugs are actively in generic development, who the likely first filers are, and what the timeline to approval might be.
Signal Prioritization Framework
Not all patent expirations are equal commercial opportunities. An excipient company with limited business development capacity needs a prioritization framework that ranks opportunities by revenue potential, technical fit, and competitive positioning.
Revenue potential is a function of the drug’s current annual sales multiplied by the estimated excipient intensity of its formulation. A $10 billion injectable biologic with a complex stabilizer system represents a higher-value excipient opportunity than a $500 million oral solid with a conventional immediate-release formulation, even though the oral solid’s generic entry may be simpler to support.
Technical fit scores how well the company’s existing excipient product portfolio aligns with the formulation requirements of the target drug. A polymer supplier specializing in HPMC matrix systems should prioritize controlled-release drug expirations. A lipid excipient supplier should prioritize drugs formulated as LBDDS or soft gelatin capsules. A biopharmaceutical excipient manufacturer should weight biologic LOEs.
Competitive positioning assesses how many other excipient suppliers can provide the required excipient to the same specification. For commodity excipients like lactose monohydrate or MCC, the opportunity is high volume but low margin, and competitive intensity is high. For specialized excipients with limited qualified suppliers, the margin opportunity is substantially higher.
Freedom-to-Operate Analysis for Novel Excipient Development
Before committing R&D resources to a novel excipient technology, a Freedom-to-Operate (FTO) analysis is mandatory. FTO analysis maps the claims of issued patents in relevant technology spaces against the proposed composition, manufacturing process, and intended use of the new excipient. The goal is to identify patents that could be infringed by the development program before significant capital has been deployed.
Advanced FTO tools provide element-by-element claim comparison against proposed compositions and processes, identifying specific claim elements that represent potential infringement risk. Where FTO analysis identifies blocking patents, the response options include designing around the blocking claims, licensing the relevant patent, challenging the patent’s validity through inter partes review (IPR), or selecting an alternative technology path. FTO analysis conducted at program initiation rather than near commercial launch is materially cheaper and less disruptive to execute.
Competitive Intelligence: Reading Competitor Patent Portfolios
Patent portfolios are public documents. A systematic review of patent filings by competitor excipient manufacturers, organized by technology area and filing date trend, reveals R&D investment priorities that are not disclosed in earnings calls or product catalogs. An excipient competitor filing 15 patents per year on lipid-based delivery systems over 24 months is signaling a major strategic investment in that space. That signal is relevant for competitive positioning and for anticipating where that competitor’s new product launches will occur.
Cross-referencing competitor patent filings with pharmaceutical company ANDA pipelines and drug LOE timelines shows which competitor is positioning for which drug’s generic market. This is not theoretical analysis; it is pipeline mapping. It identifies where competitive pressure will be highest at specific future points in time and allows commercial resource allocation to avoid head-to-head competition on low-margin opportunities in favor of higher-value positions where the competitor has not yet established a technical presence.
Key Takeaways: Section 10
Effective patent intelligence for excipient companies requires integrating Orange Book data, USPTO patent claims analysis, ANDA filing records, and commercial intelligence platforms into a single workflow with prioritization criteria based on revenue potential, technical fit, and competitive positioning. FTO analysis is a mandatory gate before any novel excipient R&D commitment. Competitor patent portfolio analysis is a prospecting tool that identifies where market pressure will intensify before it appears in pricing or customer RFPs.
11. Investment Strategy for Excipient Portfolio Managers and Institutional Analysts
The Excipient Sector as a Pharmaceutical Infrastructure Play
The excipient industry does not carry clinical development risk. Excipient companies do not run Phase II trials. They do not face FDA Complete Response Letters on safety or efficacy grounds. Their revenue visibility, particularly for suppliers to generic manufacturers against a published LOE schedule, is higher than for drug developers or specialty pharma companies.
This combination of above-market growth rates (6-8% for high-specialty sub-segments), recurring demand driven by a multi-year LOE wave, and structurally lower binary risk than API or drug product companies positions the excipient sector as a pharmaceutical infrastructure investment with characteristics closer to contract research organizations (CROs) or CDMOs than to drug developers.
Public Market Exposure
Pure-play pharmaceutical excipient exposure is limited in public equity markets. Most excipient businesses are either private companies or subsidiaries of large specialty chemical conglomerates. Public market access points include:
Ashland Global Holdings (NYSE: ASH) generates approximately 40% of revenues from pharmaceutical excipients, including HPMC (Benecel), HPC (Klucel), and related polymers. The pharmaceutical segment has higher margins than Ashland’s other business lines and direct exposure to the controlled-release and tablet binding polymer markets.
Evonik Industries (XETRA: EVK) markets the EUDRAGIT polymer line for pharmaceutical coating and matrix systems, Aerosil (colloidal silicon dioxide) for flow and stability, and a range of lipid excipients through its Health Care division. Evonik’s pharmaceutical excipient business is a segment within a larger specialty chemicals company.
Croda International (LSE: CRDA) has a dedicated Life Sciences segment that includes pharmaceutical excipients, primarily lipid-based systems and polysorbates used in injectable and oral formulations. Croda’s excipient portfolio is highly relevant to biologic formulation.
Ligand Pharmaceuticals (NASDAQ: LGND) is the closest available pure-play excipient IP company, with royalty revenues tied directly to Captisol-containing approved drugs. Ligand’s revenue model is structurally different from product sale excipient companies: it collects royalties on downstream drug product revenues, making it a direct proxy for the commercial performance of the drugs in its licensee portfolio.
Private Market Considerations
Several significant excipient manufacturers operate as private companies or as divisions of large private chemical groups. IMCD Group (AMS: IMCD), a publicly traded specialty chemical distributor with substantial pharmaceutical excipient distribution revenues, provides an indirect exposure point. JRS Pharma (Julius Hoesch group), a major supplier of specialty cellulose-based excipients, is private.
For private equity, the excipient sector offers platform acquisition opportunities in the co-processed excipient space, where novel functionality is created by combining established excipients into proprietary co-processed materials with enhanced performance characteristics. Co-processed excipients command price premiums over their constituent individual excipients, carry defensible IP, and address the pharmaceutical industry’s preference for minimizing the number of excipient components while maximizing functional performance.
Demand Drivers to Monitor
The GLP-1 receptor agonist class, specifically semaglutide and related peptides, represents an emerging structural demand driver for injectable-grade excipients. Commercial GLP-1 production is scaling globally at extraordinary rates. The subcutaneous pen injection format for semaglutide, tirzepatide, and related peptides requires pharmaceutical-grade phenol, propylene glycol, and phosphate buffer systems at scale. Oral peptide delivery, if it advances commercially, would create sustained demand for absorption-enhancing excipients like SNAC.
The ADC (antibody-drug conjugate) pipeline creates demand for specialized buffer systems, surfactants, and stabilizers capable of maintaining the stability of a bispecific or complex biologic payload. ADC formulation is technically demanding and requires high-purity, well-characterized excipients with endotoxin specifications consistent with parenteral oncology applications.
Gene therapy and RNA therapeutic formulations, specifically lipid nanoparticle (LNP) delivery systems for siRNA, mRNA, and gene editing payloads, represent the highest-growth technical frontier for excipient demand. LNP formulations require ionizable lipids, helper lipids (DSPC), PEGylated lipids (PEG2000-DMG), and cholesterol, along with precise ratio and particle size control. These are specialty chemicals with pharmaceutical application requirements rather than traditional pharmaceutical excipients, but the demand signal is excipient-relevant.
Key Takeaways: Section 11
The excipient sector provides pharmaceutical revenue exposure with structurally lower clinical development risk than drug companies. Public market access through Ashland, Evonik, Croda, and Ligand offers distinct risk-return profiles ranging from specialty chemical conglomerate exposure (Ashland, Evonik) to pure pharmaceutical application growth (Croda Health) to excipient IP royalty streams (Ligand). The GLP-1 scale-up, ADC pipeline expansion, and LNP technology adoption are the three forward demand drivers most relevant to excipient investor positioning over the 2025-2030 period.
12. Key Takeaways by Section: Executive Summary
The excipient market is a $14 billion sector growing at 6-8% in its highest-value sub-segments. Biopharmaceutical and sustained-release excipients grow faster than the overall market and carry materially higher margins. Companies that have not segmented their strategy by these sub-segments are competing on a blended average that obscures actual value creation opportunity.
The $300 billion LOE wave through 2030 is an excipient demand pipeline with a published schedule. Keytruda, Eliquis, Stelara, Ozempic, and roughly 186 other products will lose exclusivity within this period. Each product represents a specific excipient requirement that can be identified from formulation composition data available in public regulatory documents.
Drug patent architecture has multiple layers, and only tracking base patents misses most of the commercial signal. Formulation patents, method-of-use patents, PTEs, and pediatric exclusivity all affect the actual LOE date. Paragraph IV certification filings identify drugs where generic companies have already committed development resources.
Excipient IP is a fundable asset class. The Captisol/Ligand model demonstrates royalty revenue generation from formulation-enabling excipient technologies. Excipient companies with novel technologies have IP that can be structured for licensing rather than product sale alone.
Bioequivalence mechanics give excipient properties direct regulatory consequence. Particle size distribution, binder concentration, disintegrant selection, and lipid system composition are not formulation aesthetics. They determine whether an ANDA achieves BE on first attempt. Technical data packages that address these parameters differentiate suppliers in ANDA development procurement.
Evergreening generates the formulation patent filings that represent the highest-value excipient demand. Orange Book new patent listings by branded companies are readable demand signals that precede formal RFP processes by 12 to 36 months.
Regulatory infrastructure is a market structure determinant. IID listings and Type IV DMF support are market access prerequisites. The PRIME program reduces risk for novel excipient adoption but does not create an independent approval pathway.
Effective patent intelligence integrates Orange Book data, USPTO claims analysis, ANDA filing records, and competitive portfolio review. Prioritization by revenue potential, technical fit, and competitive positioning is the resource allocation mechanism that converts large-scale patent data into actionable commercial targeting.
For investors, the excipient sector is a pharmaceutical infrastructure play with above-market growth, structurally lower clinical development risk, and exposure to the GLP-1 scale-up, ADC pipeline, and LNP technology adoption, the three highest-growth technical demand frontiers of the current pharmaceutical cycle.