Part I: The $15 Billion Paradox — A Market That Grows With the Brake On
The pharmaceutical excipients market is projected to reach $15.43 billion by 2034, expanding at a CAGR of between 5% and 8.4% depending on the segment, according to multiple market analyses published between 2024 and 2025. That trajectory is real. The forces behind it, including the wave of post-patent-cliff generic launches, the structural shift toward biologics, and the explosion of patient-centric dosage forms, are not slowing down.
And yet, the market grows in spite of a near-total failure to bring truly novel excipients to patients at scale.
A 2019 USP survey of formulation scientists put hard numbers on this dysfunction: 84% reported that being limited to approved excipients had constrained their products, 64% experienced development delays of one to five years, and 28% stopped development entirely because the excipient toolkit could not solve their problem. Those figures have not meaningfully improved. The FDA’s Novel Excipient Review Pilot (PRIME), launched in 2021, exists precisely because the agency recognized this failure mode. Fewer than a dozen companies have sought entry to that program since its launch.
The root cause is a well-documented coordination failure. Drug developers will not stake an NDA on an excipient with no track record. An excipient with no drug approvals behind it has no track record. So the cycle holds. Industry insiders describe this as wanting to be ‘the first to be the second or third’ — a formulation equivalent of nobody wanting to go first.
The practical consequence is that the market grows on volume, not on innovation breadth. It grows because more generic tablets need standard binders, more biologic vials need polysorbate, and more pediatric liquids need the same preservatives that were already marginal forty years ago. The growth does not reflect a resolved innovation problem; it reflects an avoided one.
This is the paradox. And it is precisely where the opportunity lives.
A rigorous analysis of the FDA’s National Drug Code (NDC) directory — a public, continuously updated, and almost entirely underutilized strategic dataset — can dismantle the primary barrier to novel excipient R&D: the inability to prove the existence of a market need before the first dollar is committed to the lab. When an R&D team can demonstrate, using the FDA’s own product data, that 95% of approved high-concentration monoclonal antibodies rely on two surfactants with documented degradation pathways, the risk calculation changes. The need is no longer speculative. It is quantified. That distinction is worth more than most pilot studies.
Key Takeaways: The Paradox
The excipient market’s growth rate obscures a genuine innovation deficit. The economic opportunity for companies that can de-risk excipient R&D before entering the lab is substantial — not marginal. NDC data is the primary public instrument for that de-risking. Every month that data goes unmined, a competitor could be building the proprietary analytical infrastructure that shapes formulation strategy for the next decade.
Part II: Why ‘Inactive Ingredient’ Is a Dangerous Misnomer
The term ‘inactive ingredient’ is an FDA administrative designation, not a functional description. It means the component is not the labeled active therapeutic agent. It does not mean the component is pharmacologically inert, formulation-neutral, or interchangeable. Treating it that way has caused failed programs, delayed NDAs, and billions in avoidable R&D waste.
The reality, well-established in formulation science if not always in boardroom strategy, is that the excipient profile of a drug product directly governs its pharmacokinetic behavior. The dissolution rate of a BCS Class II compound in the gastrointestinal tract is not primarily a function of the API’s chemistry; it is a function of the polymer or surfactant excipient co-formulated with it. The aggregation behavior of a monoclonal antibody during storage and transit is not primarily a function of the protein sequence; it is a function of the stabilizer, buffer, and surfactant system surrounding it. The in vivo release rate of a depot injectable is controlled almost entirely by the PLGA polymer excipient, not by the peptide it encapsulates.
This reality has commercial consequences that compound across the drug lifecycle:
Excipients influence whether a compound can reach the clinic at all. For BCS Class IV compounds — low solubility, low permeability, which now represent roughly 40% of development pipelines — formulation strategy is the primary determinant of whether a molecule produces meaningful plasma exposure. No excipient innovation, no drug.
Excipients determine whether a patient takes the drug as prescribed. The pill burden in geriatric patients, the palatability of pediatric liquids, and the injection volume tolerance in self-administering biologic patients are all formulation problems. They are all excipient problems. Adherence is a formulation outcome.
Excipients create and defend IP positions. A novel stabilizer in a biologic formulation is patentable. A novel polymer matrix in a controlled-release tablet is patentable. A new co-processed excipient enabling direct compression of a combination therapy can anchor a formulation patent that extends commercial exclusivity years beyond the API’s basic patent expiry — a tactic the industry calls evergreening, and one that generates billions in protected revenue for companies that execute it well.
The analyst or R&D lead who understands excipients as enabling IP assets rather than commodity inputs has a fundamentally different product development conversation. The conversation shifts from cost-per-kilogram to value-per-approval, and the commercial relationships that follow are structured accordingly.
Part III: The Four Strategic Functions of Modern Excipients — With IP Implications
Function 1: API Enabling for Complex Modalities
The pharmaceutical pipeline’s center of gravity has shifted. Small molecules still dominate approvals by volume, but the high-value, high-complexity drugs now entering late-stage development include mRNA therapeutics, siRNA, antibody-drug conjugates (ADCs), cell therapies, and high-concentration biologics. Each of these modalities has formulation requirements that the standard excipient toolkit was not built to meet.
The clearest recent example is mRNA delivery. Without ionizable lipid nanoparticle (LNP) excipients capable of encapsulating and protecting single-stranded mRNA, endosomal escape, and cell membrane fusion, neither the Moderna nor the Pfizer-BioNTech COVID-19 vaccines would have been manufacturable. The Acuitas Therapeutics LNP formulation technology licensed by BioNTech, and the SNALP-derived LNP platform used by Moderna, were not incremental improvements on existing excipients. They were prerequisite technologies. The vaccines did not succeed despite their excipient platforms; they succeeded because of them.
The IP valuation implications are direct. Acuitas holds foundational patents on the ionizable lipid ALC-0315 and the LNP formulation architecture used in Comirnaty. That patent estate is not adjunct to the vaccine franchise; it is, from a freedom-to-operate standpoint, the franchise’s foundation. Any competitor building an mRNA product must either license these formulation patents, design around them, or challenge their validity — a process that has already generated litigation between Moderna and Alnylam, and between Moderna and Arbutus Biopharma, over LNP formulation IP.
For excipient developers, the strategic implication is clear: novel excipients that enable new modalities carry patent potential that is not captured by traditional excipient market sizing. The market for ionizable lipids is not limited to the excipient price per gram. It is bounded by the total revenue of all mRNA drugs requiring that delivery technology.
For ADCs, the comparable enabling technology is the linker-payload chemistry and the solubilizing excipient systems required to keep hydrophobic drug payloads stable in aqueous formulation. AstraZeneca and Daiichi Sankyo’s trastuzumab deruxtecan (Enhertu) is formulatd with a specifically engineered tetrapeptide-based cleavable linker; that linker technology is the basis of substantial IP protection alongside the mAb and payload. Excipient developers with novel solubilization systems for hydrophobic payloads are positioned at the center of the ADC formulation problem, which the market expects to grow to over $15 billion in drug revenue by 2028.
Function 2: Patient-Centric Formulation and Adherence Economics
Non-adherence costs the U.S. healthcare system an estimated $100 to $300 billion annually in preventable hospitalizations, according to the Annals of Internal Medicine. A meaningful fraction of that figure traces back to formulation failures: tablets too large to swallow, liquids too bitter to take twice daily, injections too frequent to maintain. These are excipient problems with excipient solutions.
For pediatric patients, taste is the primary barrier. The oral bioavailability of a compound is irrelevant if the child spits it out. Ion exchange resin taste-masking, in which the API is bound to a resin matrix that releases the drug only in the low-pH environment of the stomach rather than in the mouth, has been commercially deployed for decades in products like dextromethorphan suspensions. Microencapsulation with ethylcellulose or Eudragit E PO polymer coatings produces similar results by creating a physical barrier between the API and taste receptors on the tongue. The precision formulation problem — matching the masking technology to the API’s molecular charge, solubility, and release requirements — remains largely unsolved for large classes of compounds, particularly broad-spectrum antivirals and second-generation antibiotics.
For geriatric patients, the dominant formulation challenge is dysphagia. Orally disintegrating tablets (ODTs) and thin oral films solve the mechanical swallowing problem but introduce new excipient requirements. ODTs require excipients that provide rapid disintegration in saliva, often in under 30 seconds, while maintaining adequate mechanical strength to survive packaging and handling. The performance boundary between those two requirements is narrow, and navigating it typically demands either superdisintegrant excipients (crospovidone, modified croscarmellose) combined with highly water-soluble fillers like mannitol, or flash-melt technology using specialized sugar alcohol matrices. Neither solution works universally across API types, which is why a large portion of the oral geriatric market still relies on standard tablets and the associated adherence losses.
Controlled-release polymer systems for chronic disease management represent a third distinct domain. Hypromellose (HPMC) gel-matrix tablets have been standard for extended-release oral solids since the 1980s. The HPMC matrix controls drug release by forming a hydrated gel layer that slows drug diffusion as the tablet core erodes. The commercial limitation is that standard HPMC grades produce first-order release kinetics — the release rate declines as the tablet erodes — rather than zero-order kinetics, which would maintain constant plasma drug levels throughout the dosing interval. Achieving zero-order release typically requires more complex layered or osmotic pump architectures (the OROS system, originally developed by Alza, now part of Johnson & Johnson’s portfolio) or novel polymer combinations that have not yet achieved broad commercial adoption. The gap between what controlled-release formulators need (true zero-order kinetics from a simple matrix tablet) and what available HPMC grades provide is a documented, persistent, and quantifiable unmet need.
Function 3: Manufacturing Economics and Co-Processed Excipients
The economic case for advanced excipients in solid oral manufacturing is straightforward: wet granulation costs more than direct compression, and direct compression requires excipients with flow and compressibility properties that most APIs lack on their own.
Wet granulation involves blending the API with binders and other excipients, wetting the blend with a granulating solution, drying the resulting granules, milling to the target particle size distribution, and then blending again with lubricants before compression. Each step requires capital equipment, validated process controls, energy input, and time. For a generic manufacturer producing high-volume oral solids — metformin, lisinopril, atorvastatin — the difference between a wet granulation process and a direct compression process for the same product can represent a 15% to 25% reduction in manufacturing cost of goods.
Co-processed excipients are the enabling technology for that transition. A co-processed excipient is not a physical mixture of individual components; it is an engineered composite material produced by spray drying, wet granulation, or co-crystallization, in which the component materials are intimately associated at the particle level. The result is a material with emergent functional properties — flow, compressibility, dilution capacity — that exceed what a simple blend of the same ingredients would produce. Commercially available co-processed excipients include MicroceLac 100 (a spray-dried composite of microcrystalline cellulose and lactose monohydrate, from Meggle), Ludipress (polyvinylpyrrolidone and lactose, from BASF), and Prosolv SMCC (silicified microcrystalline cellulose, from JRS Pharma). Each of these has achieved commercial adoption because it solves a specific direct-compression processing problem for a class of APIs with known flow or compressibility limitations.
The IP architecture around co-processed excipients is instructive. JRS Pharma holds patents on the process for manufacturing Prosolv SMCC, specifically the method of co-processing MCC with colloidal silicon dioxide to produce a material with silicon dioxide distributed uniformly on the cellulose surface. Those process patents have created a durable competitive moat even though both component materials are long-off-patent commodities. The novel element is the manufacturing method and the resulting particle morphology, not the chemical identity of the components. This is the same patent strategy available to any excipient developer creating a new co-processed composite: process claims, particle morphology claims, and method-of-use claims in specific formulation contexts can all provide protection even when the individual ingredients are generic.
Function 4: Formulation Patents as Life-Cycle Management Tools
For branded pharmaceutical companies, the formulation patent is the primary instrument of post-API-expiry life-cycle management (LCM). The basic mechanics: file a patent claiming a novel formulation, dosage form, or delivery system; use that patent to list an Orange Book entry; require any generic filer to either design around the formulation patent or mount a Paragraph IV challenge asserting the patent is invalid or not infringed.
AstraZeneca’s strategy with quetiapine (Seroquel) is the canonical example. When Seroquel IR’s basic composition-of-matter patent expired, AstraZeneca launched Seroquel XR, protected by formulation patents on the extended-release matrix system. Seroquel XR generated several billion dollars in revenue before its own patents faced successful generic challenges. The revenue was not extracted by changing the API; it was extracted by changing the excipient architecture.
Novo Nordisk’s transition from semaglutide injection (Ozempic) to oral semaglutide (Rybelsus) is a more recent and technically sophisticated example. Oral delivery of a peptide drug like semaglutide was not commercially viable until Novo Nordisk developed its Absorption Enhancer Technology, based on the excipient sodium N-[8-(2-hydroxybenzoyl)aminocaprylate] (SNAC). SNAC transiently increases gastric permeability to allow semaglutide to be absorbed in the stomach before it reaches the proteolytic environment of the small intestine. The SNAC formulation is covered by multiple patents that Novo Nordisk has licensed defensively, creating a significant barrier for any competitor attempting to bring an oral GLP-1 receptor agonist to market using the same permeation-enhancement mechanism.
For excipient developers, the implication is that a novel excipient with a defined mechanism of action in a specific therapeutic context can anchor IP that is worth far more than the excipient’s standalone commercial value. SNAC’s value is not the price of the SNAC powder; it is the contribution to Rybelsus’ commercial exclusivity in a drug franchise that generates over $2 billion annually.
The NDC database is the instrument for identifying where the next Seroquel XR or Rybelsus-type opportunity exists, before a competitor files the patent.
Key Takeaways: Excipient Strategic Functions
Excipients operate simultaneously across four value dimensions: API enabling, patient adherence, manufacturing economics, and IP generation. R&D teams that evaluate excipient development opportunities against only one dimension will systematically underestimate the commercial case for investment. The IP dimension, in particular, is frequently underweighted in excipient R&D budgeting, even though formulation patents represent a multi-billion-dollar contribution to branded pharma revenues annually.
Part IV: Decoding the NDC — Structure, Segments, and Strategic Leverage
What the Three NDC Segments Actually Tell You
The NDC is a unique, three-segment product identifier assigned to every human drug product commercially distributed in the United States. The FDA issues the Labeler Code. The labeler assigns the Product Code and Package Code. The directory is updated daily and is publicly available as downloadable text files, Excel files, and via API.
The three segments carry distinct strategic intelligence:
The Labeler Code (four, five, or six digits) identifies the specific firm that manufactures, repacks, or distributes the product. For competitive analysis, this segment is the primary key. Filtering the entire NDC database by a single Labeler Code yields the complete commercial product portfolio of that company. Analyzing excipient patterns across that portfolio reveals the company’s formulation preferences, manufacturing technology constraints, preferred supplier relationships, and areas where formulation performance is likely suboptimal. A generic manufacturer whose entire oral solid portfolio relies on wet granulation — identifiable by the consistent co-occurrence of granulating binders like povidone K30 alongside colloidal silicon dioxide as a glidant — is a candidate for a direct compression excipient pitch grounded in data, not in general sales arguments.
The Product Code (three or four digits, assigned by the labeler) identifies the specific drug entity, including its dosage form, route of administration, and formulation type. Two products from the same labeler with the same API and strength will carry different Product Codes if one is an immediate-release tablet and the other is an extended-release capsule. This is the segment that enables formulation-level competitive benchmarking. Tracking how a company’s Product Codes for the same API evolve over time — from IR to ER to ODT, for example — provides real-time intelligence on their LCM strategy.
The Package Code (one or two digits) identifies pack size and type. By itself, this segment has limited formulation intelligence value. Cross-referenced with prescription data, however, it indicates whether a product is positioned for acute or chronic use, which informs both dosing frequency design and the likely excipient requirements for stability over long shelf lives.
The Critical Data Gap: Ingredients Are Not in the Core Product File
The main product.txt file in the FDA’s NDC directory contains the NDC, proprietary name, dosage form, route, and basic product metadata. It does not contain the ingredient list. The inactive ingredients are contained in the FDA’s inactive ingredient database (IID), in the Structured Product Labeling (SPL) XML files associated with each drug application, and in DAILYMED, the NIH’s database of FDA-approved drug labeling.
Building a complete formulation intelligence database requires linking these sources. The NDC product file provides the product catalog. The SPL files, parseable via the FDA’s open API or via DAILYMED’s bulk download, provide the ingredient lists. The FDA’s Unique Ingredient Identifier (UNII) code is the standardization key: every ingredient, regardless of how it is labeled in the SPL text (HPMC, hypromellose, hydroxypropyl methylcellulose, Methocel K4M), maps to a single UNII code, enabling consistent cross-product counting.
The data engineering workflow required is not trivial, but it is within the capability of a small team with Python and SQL expertise. The core steps are: download the NDC product file, download or API-pull the corresponding SPL files, parse the inactive ingredient XML nodes from each SPL, map ingredient names to UNII codes using the FDA’s substance registration system, and load the normalized data into a relational database structured for analytical query. A PostgreSQL schema with three primary tables — products (NDC as primary key), ingredients (UNII as primary key), and product_ingredients (junction table with quantity data where available) — supports the full range of gap analysis queries described in Part VI.
Once built and maintained, this database is a proprietary strategic asset. It provides an analytical capability that off-the-shelf market research reports do not offer: the ability to query, filter, and segment the actual formulation decisions of every commercially active pharmaceutical company in the U.S. market, in real time.
Key Takeaways: NDC Architecture
The NDC directory’s value as a formulation intelligence source depends entirely on the quality of the data engineering applied to it. The product file alone is insufficient; the SPL-derived ingredient data, normalized to UNII codes, is the operational layer. Teams that invest in building this database properly gain a competitive intelligence capability that takes months to replicate. Teams that rely on the product file alone miss the entire formulation dimension.
Part V: From Raw Directory to Analytical Goldmine — Data Engineering for Formulation Intelligence
Building the Analytical Sandbox
Raw, undifferentiated NDC data produces noise, not insight. The first structuring step is to tag every product record with multi-dimensional categorical variables that allow meaningful analytical segmentation. The relevant dimensions are:
Therapeutic area (TA), using standard coding like ICD-11 indication categories or ATC classification. This allows formulation trend analysis within specific disease contexts — cardiovascular, CNS, oncology, infectious disease — where different API chemistries and patient populations create different formulation requirements.
API class and mechanism of action (MoA), at a more granular level than TA. Within oncology, kinase inhibitors and antibody-drug conjugates have completely different formulation challenges. Tagging at this level allows the analysis to align with actual physicochemical problems rather than broad disease categories.
Dosage form and route of administration (RoA): oral solid, parenteral, topical, transdermal, inhalation, ophthalmic. This is the most operationally critical segmentation for excipient analysis. The excipient requirements and regulatory frameworks for an IV infusion are wholly different from those of an oral tablet. All subsequent analysis must be conducted within homogeneous RoA sandboxes to be interpretable.
Release profile, within oral solids: immediate release (IR), delayed release (DR/enteric coated), extended release (ER), orally disintegrating (ODT). Each profile type has a characteristic excipient signature — superdisintegrants for ODT, enteric polymers like HPMCP or Eudragit L100-55 for DR, hydrophilic matrix polymers for ER — and analyzing formulation complexity or diversity without this distinction generates misleading results.
Patient population flag (pediatric, geriatric, neonatal), where inferrable from product labeling, indication, or dosage form type. Pediatric liquids, for example, carry a distinct set of safety-critical excipient concerns that require a separate analytical lens.
Reverse-Engineering Competitor Formulation Philosophy
With this tagging structure in place, filtering the database by a single Labeler Code produces a cross-portfolio formulation map for that company. Patterns that emerge from this analysis are operationally significant.
A major generic manufacturer with 40% of its oral solid portfolio still relying on wet granulation processes — identifiable by the systematic co-occurrence of wet-granulation binders (povidone, copovidone, HPMC at high viscosity grades) with glidants and lubricants across products where dry binders would be technically feasible — has a structural manufacturing cost burden relative to competitors who have transitioned to direct compression. That burden is quantifiable, and an excipient developer with a solution can walk into a business development conversation with the data already on the table.
A branded biologics company whose entire mAb portfolio uses polysorbate 80 as the sole surfactant, without a single commercial product using polysorbate 20, the newer poloxamer-based stabilizers, or the recombinant human albumin alternatives, has a formulation uniformity that suggests either strong supplier preference or technical lock-in. Either represents a commercial opportunity for a developer with a technically superior alternative, provided the data is used to frame a specific, evidence-based problem statement rather than a generic pitch about surfactant performance.
Part VI: The Four-Step Gap Analysis Framework
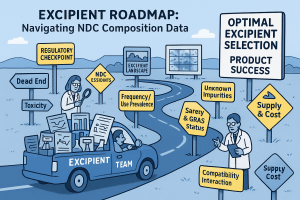
The gap analysis framework converts the structured NDC database from a descriptive catalog into a prescriptive R&D prioritization tool. The four steps are sequential and iterative.
Step 1: Segmentation and Baseline Mapping
Isolate the target analytical sandbox using the categorical tags described in Part V. Calculate the baseline formulation archetype for that sandbox: the modal combination of excipients, the median excipient count per formulation, the distribution of dosage forms, and the top-10 most frequently used ingredients by UNII code. This establishes the ‘normal’ formulation practice that subsequent analysis measures deviation from.
Visualization at this step is operationally useful. A co-occurrence heatmap — with UNII codes on both axes and cell color intensity representing how often two excipients appear together in the same formulation — immediately shows the dominant excipient clusters. A frequency-ranked bar chart of ingredient usage shows where the market has consolidated onto a small number of solutions. Both charts become reference assets for the hypothesis-generation step.
Step 2: Archetype Identification and Clustering
Apply unsupervised clustering (k-means or hierarchical clustering on the binary ingredient presence/absence matrix) to identify distinct formulation archetypes within the sandbox. Each cluster represents a coherent formulation strategy. For a typical immediate-release oral solid sandbox in a chronic disease TA, three to five clusters typically emerge: a wet-granulation archetype, a direct-compression archetype, a film-coated extended-release archetype, and one or two outlier clusters representing specialty platforms or older formulation approaches.
Characterize each archetype by its centroid ingredients, its prevalence in the market, and the types of companies that use it. This last point matters: if the wet-granulation archetype is used predominantly by companies with older manufacturing facilities, while the direct-compression archetype is concentrated among newer entrants, that pattern tells a story about where the industry is heading and what excipient transitions are already underway.
Step 3: Hypothesis-Driven Gap Identification
This is the analytically intensive step, and the one that produces differentiated insight. Three categories of gap are addressable:
Functional gaps appear as unusually high excipient counts in a formulation segment. A median of twelve excipients in a formulation segment where six would be standard for comparable APIs is a strong signal of compensatory formulation design: the formulator added multiple agents to overcome problems — poor flow, high compressibility force requirements, API-excipient incompatibilities — that a purpose-built co-processed or multifunctional excipient could address in a single component. The query for identifying this gap is straightforward: calculate the median and 90th percentile excipient count for each dosage form/TA combination, then rank sandboxes by deviation from the expected count for that RoA class.
Modality gaps appear as extreme concentration of ingredient usage within a drug class. If 95%+ of approved products in a growing therapeutic category use the same two surfactants, the same three sugars, and no novel stabilizers whatsoever, the toolkit is at a performance ceiling. The query: for each TA/RoA/dosage form combination, calculate the Gini coefficient or Shannon entropy of ingredient usage across the top functional categories (stabilizers, surfactants, buffers, tonicity agents). Low entropy signals low diversity, which signals high vulnerability to a superior new entrant.
Patient-centric gaps appear at the intersection of excipient usage data and known safety or palatability concerns. Cross-referencing the NDC ingredient data for pediatric and geriatric formulations against regulatory guidance on excipients of concern — ICH Q3C for residual solvents, FDA’s concerns around benzyl alcohol in neonates, EMA’s 2019 guideline on excipients in pediatric medicines — identifies formulations where the current excipient choice is not a preference but a constraint imposed by the absence of a better alternative.
White-space technology transfer gaps appear as proven formulation technologies that are densely adopted in one TA but absent in another where the physicochemical problem is comparable. Self-emulsifying drug delivery systems (SEDDS) have been commercially validated in HIV antiretroviral therapy (lopinavir/ritonavir, the original Kaletra formulation) and in antifungal therapy (cyclosporine soft-gel formulations). The underlying technology addresses BCS Class II/IV solubility problems. Yet SEDDS penetration in oral oncology kinase inhibitors — many of which are BCS Class II or IV and show highly variable oral bioavailability — remains limited. That gap is identifiable from the NDC data and represents a low-risk development opportunity built on an established and regulatorily precedented technology platform.
Step 4: External Validation and Time-Series Trending
Gap hypotheses generated from cross-sectional NDC analysis require external validation before R&D investment is committed. The validation stack includes: published formulation science literature confirming that the identified gap corresponds to a real technical barrier (not just a market preference for familiar excipients); patent database analysis confirming that the gap is not already being addressed by a competitor’s unpublished pipeline; and expert interviews with formulators at target customer companies confirming that the pain is active and unsolved.
The most powerful analytical extension is the time-series version of the gap analysis. Because the FDA updates the NDC directory daily and historical snapshots can be reconstructed, it is possible to plot excipient usage trends over time. A rising trend in formulation complexity for a specific drug class — the average excipient count per oral tablet in that class increasing by 15% over five years — is predictive. It says the problem is getting harder, not easier. That prediction justifies committing R&D resources to the solution before the need becomes acute and before competitors recognize the opportunity.
Key Takeaways: Gap Analysis
The four-step framework is repeatable, scalable, and directly connected to R&D investment decisions. Functional gap analysis produces leads for co-processed excipient development. Modality gap analysis produces leads for high-performance novel excipient R&D. Patient-centric gap analysis produces leads for reformulation and safety-improvement platforms. Technology transfer gap analysis produces leads for lower-risk new application development. Each gap type has a different risk-return profile and maps to a different regulatory pathway.
Part VII: High-Value R&D Targets — Three Case Studies With IP Valuation Models
Case Study 1: Co-Processed Multifunctional Excipient for High-Volume Oral Anti-Diabetics
The Gap
An NDC composition analysis of the top 50 NDCs in the oral anti-diabetic market — covering metformin HCl, empagliflozin (Jardiance), dapagliflozin (Farxiga), and combination tablets — reveals a median excipient count of 8.7 components per formulation. The ingredient co-occurrence data shows systematic separation of fill, bind, and disintegration functions across individual components: microcrystalline cellulose as filler, povidone K30 as granulating binder, croscarmellose sodium as disintegrant, colloidal silicon dioxide as glidant, and magnesium stearate as lubricant appearing together in over 65% of formulations. This pattern is the fingerprint of wet granulation as the dominant manufacturing process. The 5-component minimum plus the granulation process itself adds approximately 35% to the per-unit manufacturing cost of goods relative to a direct compression process.
The Opportunity
The target product profile: a co-processed composite of microcrystalline cellulose, pregelatinized starch, and crospovidone, spray-dried to produce a particle with embedded superdisintegrant activity and a bulk density and angle of repose compatible with direct compression tableting at API loads up to 60%. Development benchmarks would demonstrate: Carr’s index below 16 (good flow), ejection force below 200 N at 10 kN compression, disintegration time below 180 seconds in USP apparatus I, and dilution potential above 30% w/w with a model BCS Class III API.
IP Valuation
The co-processed composite is patentable via process claims (the spray-drying method and the slurry composition) and product claims (the particle morphology and the characteristic SEM/XRPD signatures of the co-processed structure). Comparable IP positions, such as JRS Pharma’s Prosolv SMCC patents, have defended premium pricing 3x to 5x above commodity MCC for over 15 years.
Market sizing: The top 50 oral anti-diabetic NDCs by volume represent approximately 8 to 12 billion tablets annually in the U.S. market alone. At an average excipient loading of 150 mg/tablet, the total addressable excipient mass is 1.2 to 1.8 million kg/year. A 15% share at $28/kg yields $5 to $7.5 million per year in U.S. revenue, scaling with generic volume growth as SGLT2 inhibitor patents expire between 2028 and 2032.
The manufacturing cost savings delivered to the customer — eliminated granulation equipment capex, reduced energy, faster batch turnaround — provide the basis for value-based pricing at a significant premium to commodity MCC while still generating a positive ROI for the adopting manufacturer.
Investment Strategy for Analysts
Excipient developers with existing MCC or starch production infrastructure are best positioned to enter this space with capital efficiency. Watch for patent filings on co-processed composites in the 2024-2026 window from JRS Pharma, Roquette, and Meggle. Any company filing process claims on a novel co-spray-drying method for multi-component excipient composites in this period is building the moat now.
Case Study 2: Novel Surfactant/Stabilizer System for High-Concentration mAb Subcutaneous Formulations
The Gap
Analysis of the parenteral biologics sandbox for approved monoclonal antibodies in immunology and oncology reveals that over 92% of commercial mAb formulations list either polysorbate 80 or polysorbate 20 as the sole surfactant, with the remaining 8% using recombinant human albumin (rHA) or poloxamer 188 as alternatives. Cross-referencing with published formulation science literature confirms the performance ceiling: polysorbates undergo auto-oxidation and hydrolysis under standard accelerated stability conditions, generating lyso-phospholipids and fatty acid peroxides that have been documented to accelerate protein aggregation in adalimumab, trastuzumab, and bevacizumab formulations. At concentrations above 150 mg/mL, neither polysorbate 80 nor polysorbate 20 adequately suppresses aggregation during the agitation stress typical of autoinjector operation.
Products affected by this limitation include the subcutaneous formulations of tocilizumab (Actemra SC), secukinumab (Cosentyx), and guselkumab (Tremfya), all of which have documented viscosity management challenges at their commercial concentrations and all of which rely on the standard polysorbate/amino acid/sugar stabilization platform.
The Opportunity
The target product profile: a novel amphiphilic block copolymer or glycolipid-based surfactant that demonstrates: critical micelle concentration below 0.01% w/v (comparable to polysorbate 80), viscosity reduction of at least 40% in a model 150 mg/mL IgG1 solution at pH 6.0 relative to polysorbate 80 control, sub-5% increase in aggregation by SEC-HPLC after 25 agitation cycles at 250 rpm, and peroxide generation below 0.5 nmol/mL after 6 months at 25°C/60% RH.
The mRNA vaccine LNP precedent is directly relevant here. Ionizable lipid excipients were not on any standard formulator’s toolkit in 2015. By 2021, they had enabled two $10+ billion drug franchises. A novel surfactant that solves the high-concentration mAb stabilization problem enters a market where the top 10 commercial mAbs by revenue collectively generate over $100 billion annually, and where the trend toward subcutaneous self-administration is structural and accelerating.
IP Valuation
Formulation patents covering the use of a novel surfactant in specific mAb product types (IgG1, bispecific, etc.) at defined concentration ranges can be structured as method-of-formulation patents or as composition patents (novel surfactant + mAb + buffer combination). Licensing this IP to branded biologics companies at a royalty rate tied to the commercial value enabled (subcutaneous versus IV formulation premium) rather than at a per-kilogram commodity price is the commercially rational approach.
A comparable IP licensing precedent: Halozyme’s ENHANZE drug delivery technology, based on the enzyme rHuPH20 that degrades subcutaneous hyaluronan to allow large-volume subcutaneous injection, has been licensed to Roche (Herceptin SC, Ocrevus SC), Janssen (Darzalex SC), AbbVie, and others at royalty rates that have generated over $1 billion in cumulative licensing revenue for Halozyme. A novel surfactant that solves the high-concentration formulation problem has comparable licensing potential if the IP position is structured correctly from the outset.
Investment Strategy for Analysts
Track IND-enabling submissions and early-phase formulation studies filed with the FDA’s PRIME Novel Excipient Review program. Any application to PRIME for a surfactant or stabilizing excipient in the biologic parenteral category is a signal that a company has generated sufficient safety and performance data to seek pre-competitive regulatory feedback — which means they are 18 to 36 months ahead of where the NDC data shows the market need. Halozyme (HALO), Evolva, and Evonik’s Health & Nutrition segment are publicly visible players in adjacent spaces whose patent filings and regulatory submissions are worth monitoring.
Case Study 3: Pediatric-Safe Preservation and Sweetening System for Oral Liquids
The Gap
NDC composition analysis of the top 20 most-prescribed oral liquid antibiotics and analgesics for children under 12 shows that sodium benzoate is present in 41% of formulations, parabens (methylparaben, propylparaben) appear in 38%, and sucrose concentrations above 40% w/v appear in 62%. Ethanol appears as a co-solvent in 18% of formulations, including some products indicated for use in children under 6 years.
These are not incidental choices. Sodium benzoate in the presence of ascorbic acid generates benzene, a documented carcinogen, as a degradation product. Parabens have documented endocrine disruption potential at repeated exposure levels relevant to chronic pediatric dosing. The WHO’s 2012 guidance on excipients in pediatric medicines lists all three as substances requiring risk-benefit assessment in children under 2 years, and the EMA’s 2019 guideline on excipients in the label identifies sodium benzoate and propylene glycol as requiring explicit risk-benefit justification in neonatal and infant formulations. Yet NDC data shows continued widespread commercial use across the pediatric liquid market, including in formulations approved post-2019.
The conclusion is not that manufacturers are acting improperly; most pediatric liquid formulations received approval before the current guidance frameworks existed, and reformulation under existing NDA is expensive. The conclusion is that formulators continue using suboptimal excipients because no commercially viable, regulatory-supported alternative with equivalent microbial efficacy and palatability has displaced them.
The Opportunity
A ‘pediatric-first’ preservation and sweetening platform would consist of: a non-cariogenic, pediatric-acceptable sweetener system (steviol glycosides at grade and particle size optimized for solution stability, or a defined xylitol/sorbitol blend with documented non-cariogenic status and acceptable laxative threshold in pediatric dosing); a broad-spectrum preservative with a well-characterized neonatal safety profile (potassium sorbate alone, or a combination of potassium sorbate with medium-chain fatty acid esters as potential alternatives to parabens); and a buffering system that maintains pH 4.5 to 6.0 stability without contributing to taste complaints. The platform would be co-developed with a Pediatric Research Equity Act (PREA)-driven drug development program to accumulate the in-use safety data that establishes regulatory precedent.
IP Valuation
The IP architecture for a pediatric excipient platform has two layers. First, the composition itself — the specific ratio, particle size, and grade of the sweetener and preservative combination — may be patentable as a novel pharmaceutical composition. Second, the method-of-use patents covering the use of the platform with specific drug classes (antibiotics, antipyretics, antiepileptics) in defined age groups create a licensing framework that is difficult for competitors to design around without generating a functionally different — and therefore separately regulatable — formulation.
The commercial prize is access to the pediatric liquid market across all indications, estimated at $8 to $12 billion in annual revenue globally. An excipient platform established as the go-to solution for pediatric liquid preservation and palatability earns its value through specification-in at the early formulation stage, at which point switching costs for the drug manufacturer are high.
Investment Strategy for Analysts
The regulatory driver here is the EU Pediatric Regulation and the FDA’s PREA requirements, both of which are generating new pediatric investigation plans (PIPs) and pediatric study plans (PSPs) that require age-appropriate formulations. Companies submitting PIPs with novel oral liquid formulations are the primary near-term customers for a pediatric excipient platform. EMA’s Committee for Medicinal Products for Human Use (CHMP) annual PIP database is a public, searchable resource for identifying the drug development programs where this platform will be needed in the next three to five years.
Key Takeaways: Case Studies
Each case study follows the same logic: the NDC data identifies the gap, formulation science literature validates the technical barrier, IP analysis defines the defensible position, and bottom-up market sizing quantifies the prize before a single preclinical experiment is run. The sequence is deliberate. R&D budgets committed before the gap is quantified are speculative; R&D budgets committed after this analysis are strategic.
Part VIII: FDA’s PRIME Program — Using NDC Evidence to Win Regulatory Acceptance
The FDA launched the Novel Excipient Review Pilot Program (PRIME) in 2021 specifically to address the regulatory barrier to excipient innovation. PRIME allows excipient manufacturers to submit quality and safety data for FDA review before the excipient appears in any NDA or ANDA. A positive PRIME review creates a reference data package that drug developers can cite in subsequent drug applications, reducing the per-application regulatory burden and making novel excipients commercially viable.
The key requirement for PRIME acceptance is a credible ‘Drug Development Need Statement’ — documentation that the novel excipient addresses a real, quantifiable unmet formulation need rather than a theoretical improvement. This is exactly where the NDC gap analysis framework becomes a regulatory tool.
Instead of a qualitative assertion that ‘better stabilizers are needed for biologics,’ a PRIME applicant who has completed the NDC analysis can present the FDA with a quantified evidence package: the specific number of approved mAb products relying exclusively on polysorbates, the documented degradation pathways of those polysorbates, the peer-reviewed data on their performance limitations at target concentration ranges, and the downstream consequences for patient access to subcutaneous self-administration formats. The FDA’s own data — the NDC directory — becomes the foundation of the case.
This approach does three things: it demonstrates rigor, it grounds the need statement in data the FDA already accepts as authoritative, and it pre-empts the most common reason for PRIME application rejection, which is an insufficiently specific demonstration that the need exists and is not already addressed by approved alternatives.
For the programs described in Case Studies 2 and 3, the NDC analysis provides the core quantitative argument. For Case Study 1 (co-processed excipient for direct compression), the PRIME pathway may be less relevant since the components are individually known — but the NDC data remains central to building the 505(b)(2) or ANDA-support strategy by demonstrating that the co-processed material will be used in formulations spanning multiple marketed drug categories.
Part IX: NDC + Patent Intelligence — The Dual-Lens Strategy
NDC composition data shows what is on the market. Patent data shows what companies are planning to put on the market. Neither dataset alone is sufficient for a complete competitive strategy. Together, they produce a forward-looking market map with a 3- to 5-year predictive horizon.
The operational integration works as follows. For each high-priority gap identified through NDC analysis, run a parallel patent search — using DrugPatentWatch, Derwent Innovation, or PatSnap — covering the last five years of formulation patent filings in the same therapeutic and formulation space. The search should capture: composition of matter claims covering novel excipient structures, method-of-formulation patents claiming the use of specific excipients with specific APIs, process patents covering new manufacturing methods for excipient composites, and Orange Book-listed formulation patents on existing drugs that signal active LCM investment.
The result of this overlay produces one of two strategic scenarios:
In Scenario A, the NDC gap is real and the patent landscape is sparse. No competitor has filed claims on the solution space, the innovator companies in the relevant TA have not filed formulation patents suggesting a proprietary approach is already in development, and the White Space is genuinely open. This scenario justifies accelerated R&D investment, because first-mover IP protection is available.
In Scenario B, the NDC gap is real and the patent landscape shows recent, concentrated filings. One or two players have already identified the gap and are building their position. This scenario does not close the commercial opportunity — it means the window for leading IP is closing — but it does change the strategy. The options are: enter the space as a second-mover with a differentiated technical approach that avoids the blocking claims, partner with the patent-holder to co-develop the commercial platform, or pivot to an adjacent gap with cleaner IP geography.
The SGLT2 inhibitor case is instructive here. The core empagliflozin and dapagliflozin composition patents began expiring in 2025-2026. Generic ANDA filings for both drugs were filed in 2021-2023, which is visible in the FDA’s paragraph IV certification database. An excipient developer who overlaid NDC composition data (showing the complex wet-granulation-dominated formulation landscape) with the paragraph IV filing timeline in 2020 could have structured a direct compression excipient development program timed precisely to be commercially available as the generic wave breaks, when dozens of generic manufacturers need a cost-competitive manufacturing solution simultaneously. That is not a coincidence-dependent business plan; it is a data-derived strategic sequence.
Key Takeaways: Dual-Lens Strategy
The NDC-patent overlay converts gap analysis from a snapshot into a timeline. It identifies not just where the need exists but when the commercial window will be largest. The predictive utility of this approach increases with the granularity of both datasets and with the discipline to run the analysis before making R&D commitments rather than as a post-hoc rationalization.
Part X: ROI Architecture — Building the Investment-Ready Business Case
Bottom-Up Market Sizing From NDC Data
The standard top-down approach to excipient market sizing — take the global pharmaceutical market, apply a percentage for excipient content, multiply by CAGR — produces estimates that are useful for industry overviews and useless for R&D investment decisions. They cannot tell you the size of the market for a co-processed excipient replacing wet granulation binders in SGLT2 inhibitor tablets. Bottom-up NDC-based sizing can.
The calculation: count the target NDCs (products where the gap excipient would be used), estimate annual unit volume per NDC (using IMS/IQVIA prescription data, or conservative reference estimates from similar drug categories), calculate the mass of excipient per dosage unit from typical formulation loadings, and sum. This produces a TAM in kilograms per year for the target excipient in the target application, before applying any assumptions about market penetration or pricing.
For the oral anti-diabetic direct compression excipient (Case Study 1), the calculation produces a U.S. TAM of approximately 1.2 to 1.8 million kg/year. At 15% market penetration after five years and a price of $28/kg (premium positioning relative to commodity MCC at $8-12/kg, justified by process economics delivered to the customer), the annual revenue projection is $5 to $7.5 million in the U.S. alone, with comparable opportunity in Europe and Asia as generic SGLT2 inhibitor volume grows.
Value-Based Pricing and the Full ROI Model
| Metric | Case Study 1: Oral Anti-Diabetic DC Excipient | Case Study 2: mAb Stabilizer | Case Study 3: Pediatric Liquid Platform |
|---|---|---|---|
| U.S. TAM (kg/year) | 1.5M | 85K | 210K |
| Estimated Market Penetration (Year 5) | 15% | 10% | 20% |
| Target Price ($/kg) | $28 | $2,200 | $380 |
| Projected U.S. Revenue (Year 5) | $6.3M | $18.7M | $15.96M |
| Estimated R&D + Regulatory Cost | $4.5M | $18M | $9M |
| Payback Period | ~6 years | ~5 years | ~4 years |
| Net Present Value (10%, 10-year) | $11M | $62M | $38M |
| Key IP Lever | Process patent on co-processing method | Composition + method-of-use patent | Composition + pediatric method-of-use |
The mAb stabilizer case commands 80x the per-kilogram price of the direct compression excipient because the value it delivers — enabling subcutaneous administration of a blockbuster biologic, potentially preserving or extending commercial exclusivity — is not denominated in manufacturing cost savings. It is denominated in the revenue delta between IV hospital administration and subcutaneous home administration, a difference that can be worth hundreds of millions in annual drug revenue for the branded company. Value-based pricing captures a fraction of that delta. Even a 0.1% royalty on the commercial mAb product enabled by the stabilizer technology outperforms per-kilogram commodity pricing by orders of magnitude.
Key Takeaways: ROI Architecture
The business case for novel excipient R&D requires a pricing model that reflects the value delivered, not the cost of goods. NDC-based bottom-up market sizing provides the volume estimates. Patent analysis provides the IP leverage. The combination allows an R&D investment case to be framed in terms that resonate with a CFO or investment committee: a quantified market, a defensible IP position, a credible market penetration trajectory, and a payback period grounded in actual formulation economics.
Part XI: Key Takeaways by Section
Part I (The Paradox): The excipient market’s CAGR obscures an innovation deficit. The opportunity lies in de-risking R&D before lab investment, using public FDA data.
Part II (Misnomer): Excipients govern pharmacokinetics, adherence, manufacturing cost, and IP position. The ‘inactive ingredient’ framing is a strategic liability.
Part III (Four Functions): Evaluate excipient development opportunities across all four value dimensions simultaneously. IP generation is systematically underweighted in excipient R&D budgeting.
Part IV (NDC Architecture): The product file alone is insufficient. SPL-derived ingredient data, normalized to UNII codes and loaded into a relational database, is the operational asset.
Part V (Data Engineering): Multi-dimensional segmentation (TA, API class, RoA, release profile, patient population) is required before any gap analysis is interpretable.
Part VI (Gap Analysis): Four distinct gap types — functional, modality, patient-centric, technology transfer — each map to a different risk-return profile and regulatory pathway.
Part VII (Case Studies): NDC data quantifies the gap; literature validates the technical barrier; patent analysis defines the IP opportunity; bottom-up sizing quantifies the prize. Run this sequence before any lab commitment.
Part VIII (PRIME): NDC evidence is the most credible foundation for an FDA Drug Development Need Statement. The regulatory barrier to novel excipients is real but addressable with the right evidence package.
Part IX (Dual-Lens): NDC data shows what is on the market; patent data shows what is coming. The overlay converts gap analysis from a snapshot to a strategic timeline with predictive horizon.
Part X (ROI): Value-based pricing, grounded in the economics of the drug product enabled rather than the cost of the excipient produced, is the correct framework for novel excipient commercial strategy.
Part XII: Investment Strategy for Analysts
Institutional investors and corporate development teams evaluating excipient companies or excipient-adjacent specialty chemicals businesses should apply the following analytical framework derived from this methodology:
Differentiated IP pipeline is the primary value driver in excipient companies, as in branded pharma. A company with a process-patent-protected co-processed excipient platform is worth a fundamentally different multiple than one selling commodity MCC or lactose. Assess the patent portfolio for depth (number of claims), breadth (number of drug applications covered), and durability (remaining patent life relative to the development pipeline of drugs that will require the excipient).
Formulation regulatory momentum is an emerging value signal. Watch the FDA’s PRIME program submissions and approvals. A company that has received positive PRIME feedback for a novel excipient has cleared a major de-risking milestone; the probability of commercial adoption by a drug developer increases materially because the regulatory unknown has been converted to a known. Monitor the Federal Register and FDA transparency reports for PRIME program activity.
Modality adjacency to high-growth drug categories is the most scalable growth driver. Excipient companies with formulation IP adjacent to mRNA therapeutics, ADCs, and high-concentration biologics are participating in markets that will grow at 20%+ CAGR through 2030, regardless of the overall pharmaceutical market trajectory. Evonik’s acquisition of CordenPharma’s lipid manufacturing capabilities, and Merck KGaA’s continued investment in its Biopharma Materials platform, reflect this thesis in corporate action.
Patent cliff timing creates excipient volume tailwinds that are predictable 3 to 5 years in advance. Use the paragraph IV filing database (publicly available via the FDA’s Orange Book) to identify the next major waves of generic entry. The generic manufacturers who win those markets will need cost-competitive manufacturing solutions. The excipient developer with a direct compression platform ready when a high-volume generic wave hits captures durable volume share. The companies to watch for 2026-2028 entry are those with development programs aligned to the empagliflozin, dapagliflozin, and apixaban generic waves, all of which are in active litigation and approaching resolution.
Pipeline concentration risk applies to excipient developers as it does to drug developers. A single-product excipient business whose revenue depends on a co-processing patent expiring in four years, with no pipeline behind it, should trade at a significant discount to a company with a staged portfolio of three to five excipient development programs at different stages of the NDC-to-PRIME-to-commercial pathway described in this report.
Part XIII: Frequently Asked Questions
What are the most common errors in NDC excipient data analysis, and how do you avoid them?
The two most consequential errors are name normalization failure and scope confusion. Name normalization failure occurs when the analysis counts ‘HPMC,’ ‘hypromellose,’ ‘hydroxypropyl methylcellulose,’ and various Methocel trade grades as separate ingredients. The result is severe undercounting of HPMC’s true market penetration. The solution is to map all ingredient names to their FDA UNII codes before any counting or co-occurrence analysis. The FDA’s Global Substance Registration System (GSRS) provides the definitive UNII lookup.
Scope confusion occurs when the analysis conflates ingredients listed in the inactive ingredient database (which reflects approved formulations historically) with ingredients actually present in currently marketed products (which requires filtering by active marketing status in the NDC product file). A formulation listed in the IID for a drug withdrawn from the market in 2008 is historical data, not current market intelligence. Always filter the NDC product file to ‘Active’ listing status before constructing the analytical sandbox.
Can this framework be used for biologics, where formulations are more proprietary than for small molecules?
Yes, with a modified emphasis. The NDC-derived ingredient data for biologics will show the types and categories of excipients (buffers, surfactants, amino acids, sugars, tonicity agents) even where the exact concentrations are proprietary. The value comes from diversity analysis, not from concentration analysis. Calculating the Shannon entropy of stabilizer choice across all approved mAbs, for example, quantifies the degree of toolkit constraint even without knowing formulation ratios. Layering in published stability studies from journals like the Journal of Pharmaceutical Sciences and AAPS PharmSciTech, and formulation-related patent claims from the associated Orange Book entries, builds a substantially complete picture of both what is used and why.
How does the NDC-based approach handle combination drug products, where multiple APIs complicate excipient attribution?
Combination products are a legitimate analytical challenge. The standard approach is to segment them as a separate sandbox from single-entity products for the same drug class. Excipient complexity in combination tablets systematically exceeds that in single-entity products because each API may require compatibility-protective excipients that would not be needed in isolation. Analyzing combination product formulations separately, and specifically flagging excipients that appear only in combination products (suggesting they are managing inter-API incompatibility rather than pure processing performance), produces higher-quality insight. These compatibility-management excipients are a distinct and often underserved R&D target: any novel excipient that reduces the complexity of co-formulating two APIs with incompatible physical or chemical properties has an immediate value proposition for the growing fixed-dose combination market.
What is the minimum credible investment to build the NDC analytical infrastructure described here?
A functional baseline database can be built by a single data scientist with pharmaceutical science domain knowledge in six to ten weeks of focused effort, using Python (pandas, xml.etree, sqlalchemy), a PostgreSQL instance (cloud-hosted for under $200/month at the required scale), and the FDA’s public bulk download files. The primary time investment is in building the SPL parser and the UNII normalization pipeline; these are well-documented engineering problems with publicly available reference implementations on GitHub. Annual maintenance, covering quarterly database updates and monitoring of new drug approvals, can be managed in approximately two to four hours per week.
A more capable system with real-time daily updates, automated trend monitoring, and integration with commercial patent databases (Derwent, PatSnap) is a three- to six-month project for a two-person team, with ongoing infrastructure costs of $1,500 to $3,000 per month. The ROI comparison is asymmetric: this infrastructure investment is recoverable from a single well-targeted excipient development program, and it fundamentally changes the quality of R&D prioritization decisions across the entire portfolio.
How does the methodology apply to the EU market, where the equivalent of the NDC database is structured differently?
The European Medicines Agency’s European Public Assessment Reports (EPARs) and the EMA’s product database contain formulation data, but the SPL-equivalent machine-readable labeling infrastructure is less uniformly available than in the U.S. The practical workaround is to use the EMA’s product information files (which include the Summary of Product Characteristics, or SmPC) as the data source for EU formulation analysis, supplementing with the EMA’s published list of approved excipients and the EDQM’s Handbook on Excipients qualification data. U.S. NDC analysis is typically conducted first because the data infrastructure is superior; EU analysis then serves as a validation and market-sizing complement, confirming that identified gaps exist in both regulatory jurisdictions and therefore justify a global development program rather than a U.S.-only one.
This analysis was produced using publicly available FDA data, peer-reviewed pharmaceutical science literature, patent databases, and market research. All financial projections are illustrative models, not investment advice. Consult a qualified financial advisor before making investment decisions based on market estimates.