There is a question that gets debated in congressional hearings, litigated in federal courts, and studied in academic journals with an intensity that occasionally borders on the farcical. Does drug patent evergreening actually prevent generic competition? The short answer — the one that nobody in this industry particularly wants to hear — is: sometimes, substantially, for certain drugs, but not as universally as either the pharmaceutical lobby or its critics claim.

The longer answer requires you to understand what “evergreening” actually describes mechanically, how it interacts with a regulatory framework built in 1984 that has aged in complicated ways, and why the data on its real-world effectiveness is genuinely mixed. It also requires you to understand that pharma companies and their generic counterparts are both rational actors, which means every strategy has a counter-strategy, and every legal wall has someone with a bulldozer on the other side.
This article works through the full picture: the mechanics of patent extension, the evidence base for and against delay, the most important case studies, the regulatory response now underway, and what the next five years look like as $187 billion in branded drug revenue approaches the patent cliff.
Part One: What Evergreening Actually Is
The Original Bargain
The U.S. patent system gives drug developers twenty years of exclusivity from the date a patent is filed. For a pharmaceutical compound, that clock typically starts ticking years before FDA approval, which means the effective commercial patent life — the period during which a drug generates monopoly-priced revenue — is considerably shorter than the nominal twenty years. A 2024 analysis from the George Mason University Center for Intellectual Property found that the average effective patent life for a sample of more than one hundred top-selling small-molecule drugs was 13.35 years, consistent with prior studies on the same question [1].
That’s the deal as originally struck: developers invest in risky research, accept a finite exclusivity window, and then the market opens to competition. The Hatch-Waxman Act of 1984 codified this framework, creating the Abbreviated New Drug Application (ANDA) pathway that lets generic manufacturers prove bioequivalence without repeating full clinical trials, and granting 180 days of marketing exclusivity to the first generic filer who successfully challenges a patent [2].
For most of the 1980s and 1990s, the system functioned approximately as intended. Patents expired, generics filed, prices fell. But as blockbuster drugs began generating ten- and twenty-billion-dollar revenue streams, the incentive to extend exclusivity grew proportionally. Drug companies found legal, regulatory, and strategic tools to do exactly that.
The collection of those tools is what critics call “evergreening.”
The Four Core Mechanisms
Evergreening is not a single action. It is a portfolio strategy with four primary categories, each with its own legal basis, regulatory friction point, and variable effectiveness.
Secondary patenting is the most direct form. After the compound patent — the one covering the active molecule itself — is filed, companies file additional patents covering formulations, salts, polymorphs, dosing regimens, delivery methods, and methods of use. Some of these patents reflect genuine innovation. An extended-release formulation that improves patient adherence is a real improvement. A patent on a slightly different salt form of the same molecule that performs identically is something else. The distinction matters enormously in litigation, but the USPTO grants both, and even a weak patent creates a litigation risk that generic manufacturers must account for. <blockquote> “On average, there are 143 patents filed and 69 patents granted on each of the nation’s 12 top-selling drugs, with 56 percent filed post-FDA approval — many for minor product modifications.” — Commonwealth Fund, November 2025 [3] </blockquote>
Product hopping removes the original branded product from the market before generic entry and replaces it with a reformulated, newly patented version. The tactical logic is straightforward: if patients and physicians are prescribing the reformulation, then the generic version of the original compound has nothing profitable to substitute against, even if it receives FDA approval. Bristol-Myers Squibb used this approach with Glucophage, introducing extended-release Glucophage XR as the original’s patent was expiring. AbbVie deployed it with Humira, actively promoting a higher-concentration formulation with a lower injection volume — clinically marginal but sufficiently different to anchor a new prescription base.
Citizen petitions exploit an FDA procedural mechanism designed to let any citizen raise concerns about pending drug applications. In practice, brand manufacturers file these petitions — often on the eve of a competitor’s approval — with detailed technical objections that the FDA must evaluate before acting. The agency rejects the vast majority of petitions filed by brand companies against generic competitors, but the evaluation process consumes weeks or months. A 2012 study published in the New England Journal of Medicine found that brand companies filed 92 percent of all petitions challenging generic approvals, and that petitions delayed approval by an average of 0.7 years when filed within six months of an expected generic approval [4]. The Stop STALLING Act, introduced in Congress in the 118th and 119th sessions, specifically targets this practice, but as of early 2026 it has not been enacted [5].
Orange Book gaming is the fourth mechanism, and it became the central front in the regulatory battle of 2024 and 2025. The Orange Book — formally the FDA’s “Approved Drug Products with Therapeutic Equivalence Evaluations” — is the list of patents that brand manufacturers certify cover a particular drug product. When a generic company files an ANDA and challenges a patent listed in the Orange Book, the brand can trigger a 30-month automatic stay on generic approval simply by filing suit. Any patent in the Orange Book creates this leverage. As delivery devices became increasingly sophisticated — inhaler dose counters, autoinjector firing mechanisms, pen cap designs — companies began listing device patents in the Orange Book, claiming the device was integral to the drug product and thus entitled to the stay-triggering protection.
The FTC spent much of 2024 and 2025 dismantling this specific tactic. The Commission sent warning letters to Teva, GSK, and AstraZeneca, among others, challenging over 300 listings as improper [5]. In December 2025, Teva requested removal of more than 200 such listings covering asthma, diabetes, and COPD products — a significant capitulation that effectively ended the device-patent Orange Book strategy as a viable long-term play [5].
Part Two: The Evidence Base
What the Research Actually Shows
Here is where the conversation gets complicated, and where intellectual honesty requires holding two things simultaneously: evergreening tactics clearly exist, are widespread, and impose costs — but the evidence that they systematically prevent generic entry for most drugs is substantially weaker than the rhetoric on either side suggests.
The most cited piece of evidence for the prevalence of evergreening comes from a 2018 study published in the Journal of Law and the Biosciences, which found that 78 percent of drugs associated with new patents between 2005 and 2015 were existing medications rather than novel treatments [6]. That’s a striking figure, and it accurately reflects how patent activity is distributed across the industry. But patent filing is not the same as patent enforcement, and patent enforcement is not the same as a successful delay of generic entry.
The counter-evidence comes from a 2024 analysis from the George Mason University Center for Intellectual Property, which reviewed actual time-to-generic-entry for a substantial sample of top-selling drugs and found something that should unsettle the evergreening critics as much as it unsettles the industry defenders. The number of patents protecting a brand-name drug has no significant correlation with effective patent life. This evidence suggests that “evergreening” does not stop generic entry and that “thickets” — if they even exist — appear to be rather easy to circumvent. [1]
A related academic study published in the Journal of Health Economics examined generic entry patterns over a full decade, looking specifically at whether patent challenges were distorting the market in favor of brand companies. Its finding was that lower quality and later expiring patents disproportionately draw challenges, and that effective market life is stable across drug sales categories, hardly changing over the decade. The researchers concluded that patent challenges “serve to maintain, not reduce, the historical baseline of effective market life, thereby limiting the effectiveness of evergreening by branded firms.” [7]
What reconciles these apparently contradictory findings? The answer lies in distinguishing between two very different phenomena that the “evergreening” label bundles together.
The first is the broad statistical phenomenon: drug companies file many secondary patents, most of them covering incremental modifications. This is real and documented.
The second is the specific economic outcome: do those patents actually delay generic entry beyond what the compound patent alone would have provided? The evidence on this second question is far more equivocal. Generic manufacturers are sophisticated, well-resourced, and highly incentivized to challenge weak patents via Paragraph IV certifications. The first generic to successfully challenge a patent gets 180 days of solo exclusivity — a period worth hundreds of millions of dollars for a major drug. That incentive structure ensures that the secondary patent portfolio of any commercially significant drug will face challenges.
And those challenges succeed more often than most people realize. Between 2000 and 2019, generic challengers won approximately 73 percent of fully litigated Paragraph IV patent cases — a figure that suggests brand patents are frequently weaker than their issuance implies [8].
Where the Aggregate Data Breaks Down
The aggregate findings obscure something important: the distribution of outcomes is highly skewed.
For the majority of small-molecule drugs — products with compound patents, a handful of secondary patents, and moderate market size — the generic entry timeline looks roughly as the Hatch-Waxman framework anticipated. Compound patent expires, generics file, litigation happens, generics win or settle, prices fall 80-90 percent within twelve to eighteen months [9].
But for a subset of the highest-revenue drugs, the dynamics are categorically different. The drugs with the largest patent portfolios, the most aggressive litigation strategies, the most sophisticated product hopping, and the most coordinated pay-for-delay settlements are precisely the drugs that generate the most revenue — which is exactly why those strategies were deployed in the first place. The 2022 I-MAK report found that drug companies file an average of more than 140 patent applications per drug, with 66 percent of those applications filed after FDA approval of the drug. [10]
The academic finding that evergreening “doesn’t work on average” is statistically defensible but practically misleading if you’re analyzing a specific blockbuster. It’s the equivalent of saying that blizzards don’t significantly reduce annual average temperature — true at the aggregate level, devastating if your pipes froze in February.
The Humira case makes this point with more clarity than any academic model.
Part Three: Humira and the Biology of a Patent Fortress
The Architecture of 250 Patents
Humira (adalimumab) is a monoclonal antibody developed by Knoll Pharmaceuticals and acquired by Abbott Laboratories, which later spun off AbbVie. It received FDA approval in 2002 for rheumatoid arthritis. The compound patent was filed long before approval and would have expired no later than 2016 under normal circumstances. What happened between 2016 and the actual arrival of U.S. biosimilar competition in 2023 is the most exhaustively documented example of patent-based market exclusivity extension in pharmaceutical history.
AbbVie applied for over 250 patents for Humira, the largest number applied for by a pharmaceutical company. AbbVie was approved for 130 patents for Humira and 88 for Imbruvica, resulting in 29-39 years of blocking competition from generic drug manufacturers. Ninety percent of the patent applications for Humira were filed after the drug was approved and brought to market in 2003, and over 50 percent were filed after 2013. [11]
This is not evergreening in the mild, formulation-tweak sense. This is an industrial-scale patent accumulation operation targeting every possible aspect of the drug: manufacturing processes, formulation concentrations, dosing regimens, administration devices, specific indications, pediatric uses, and orphan drug designations. A 2022 study published in PLOS ONE found that roughly 80 percent of the U.S. patent portfolio for Humira consisted of non-patentably distinct (duplicative) patents linked together by terminal disclaimers. [12] That structure — where individual patents would be rejected as obvious but survive rejection because they are linked to earlier patents via disclaimer — represents USPTO permissiveness pushed to its outer limits.
AbbVie’s litigation strategy matched the patent portfolio in its ambition. The company openly communicated to would-be biosimilar challengers that they would face four to five years of litigation on any challenge [12]. That’s not a bluff — it’s a credible threat made credible by the sheer volume of patents requiring response. A freedom-to-operate analysis for Humira required reviewing dozens of patent families, each with multiple claims, each potentially requiring separate litigation tracks.
The Transatlantic Asymmetry
The Humira case also reveals something specific about American patent law that distinguishes U.S. outcomes from those in comparable markets. On average, nine to twelve times more patents were asserted against biosimilars in the United States than in Canada and the United Kingdom. Biosimilars also enter the Canadian and UK markets more quickly than they do in the United States following regulatory approval. [12]
European biosimilars of Humira — from Sandoz, Samsung Bioepis, Mylan, and others — launched in October 2018. U.S. launch for the same products came in January 2023, more than four years later [11]. The active ingredient was identical. The manufacturing processes were essentially the same. The difference was the U.S. patent landscape and AbbVie’s decision to settle litigation in Europe while fighting in the U.S. — or rather, to settle in the U.S. as well, but with a 2023 entry date as the condition.
That settlement approach is itself significant. AbbVie entered pay-for-delay agreements with nine biosimilar manufacturers. These deals followed a consistent pattern: AbbVie granted the biosimilar manufacturers a license to launch in Europe in October 2018, but it secured a commitment that they would delay their U.S. market entry until 2023. As part of these settlements, the biosimilar companies also agreed to pay royalties to AbbVie on their future U.S. sales. [13]
The commercial consequence is equally clear. When competition finally entered the market in 2023, Humira’s effective price decreased by an estimated 38 percent, and 2024 was the first year that Humira didn’t come with a list price increase. Humira’s list price had increased 471 percent since its 2003 launch by the time competition arrived [11]. AbbVie used this strategy to maintain a monopoly for 20 years and earn $200 billion in revenue. [3]
The counterargument, advanced by the Association for Accessible Medicines and others who defend the settlement approach, deserves fair treatment: biosimilar manufacturers were able to expedite their launches to 2023 — 11 years prior to brand patent expiration — due to patent settlements. [14] Without settlement, the argument goes, litigation might have continued until 2034 and beyond. This is not obviously wrong. The settlements did accelerate U.S. entry relative to the theoretical outer boundary of the patent estate.
But that defense requires you to accept that the outer boundary was legitimate — that all 130 granted Humira patents were valid and enforceable. The PMC study suggesting 80 percent were non-patentably distinct suggests otherwise [12]. What AbbVie constructed was a litigation deterrent, and it worked not because all the patents were strong, but because no single challenger could afford to fight all of them simultaneously.
Part Four: Pay-for-Delay and the Structural Corruption of Hatch-Waxman
The Mechanics of the Reverse Payment
Pay-for-delay settlements — formally called “reverse payment” settlements because the plaintiff (the brand company) pays the defendant (the generic) rather than the other way around — represent a fundamental distortion of the patent challenge incentive structure Hatch-Waxman created.
The economics are straightforward. A brand company selling $2 billion per year in a drug faces potential generic competition that would reduce its revenue by 70-90 percent. The first generic filer, in a scenario where the brand’s patent is weak, might capture $400 million in revenue during its 180-day exclusivity period before additional generics arrive. If the brand can pay the first-filer $500 million to withdraw its challenge and not launch for five years, both parties profit — and the brand retains another $1.5 billion per year in revenue that would otherwise have disappeared. Consumers pay the entire cost of this transaction through sustained high prices.
According to an FTC study, these anticompetitive deals cost consumers and taxpayers $3.5 billion in higher drug costs every year. [15] Settlement deals featuring payments by branded drug firms to a generic competitor kept generics off the market for an average of 17 months longer than agreements that do not include a payment. [16]
The Supreme Court addressed pay-for-delay in FTC v. Actavis (2013), ruling that such settlements can violate antitrust laws — specifically when they involve a “large and unjustified” cash payment — and must be evaluated under the rule of reason. The Court declined to hold them per se illegal, which left substantial room for continued use of the tactic and required case-by-case antitrust analysis [17].
The post-Actavis world has not eliminated reverse payments. It has made them more structurally complex. Rather than writing a check, brand companies now compensate generic challengers through authorized generic agreements (where the brand’s own generic doesn’t compete with the first-filer during its exclusivity period), co-promotion agreements, intellectual property licenses, and supply arrangements. The FTC has tracked these non-cash equivalents with increasing scrutiny, but prosecution requires demonstrating that the “something of value” transferred exceeds what normal litigation settlement would produce — a fact-intensive analysis that requires substantial litigation resources.
Gilead’s 2022 settlement with five generic manufacturers over its HIV drugs Descovy and Odefsey is a recent illustration of the pattern. The two HIV drugs had faced generic competition as soon as 2025, but the settlement delayed the arrival of generics until 2031. Settling the lawsuit is “essentially paying the generic to go away,” in the words of Stanford Law professor Mark Lemley. [10]
The First-Filer Cork
The structural problem with pay-for-delay becomes particularly acute when it involves the first ANDA filer, because the 180-day first-filer exclusivity creates a “cork in the bottle” effect. Every subsequent generic applicant must wait until the first filer has marketed for 180 days before they can obtain final approval. If the first filer settles and agrees not to enter the market, the 180-day clock never starts. Every later filer remains in administrative limbo. The brand’s patent, even if it would have been invalidated in full litigation, effectively retains its protection — not because it won in court, but because the most dangerous challenger was paid to stand down.
Settlements with first-filer generics can prevent all generic entry. Those agreements place a “cork in the bottle” that typically ensures the brand-name drug’s lock on the market. [18] This dynamic is why the FTC characterizes pay-for-delay as a more acute competition problem than secondary patent stacking alone: a weak patent that faces a paid-off challenger has the practical effect of an unbeatable patent.
Part Five: Product Hopping in Detail
The Glucophage Playbook
Product hopping predates the term itself. Bristol-Myers Squibb’s handling of Glucophage (metformin) in the early 2000s established the template. Glucophage’s key patents were expiring in 2002, and the generic pipeline was well-stocked. BMS responded by aggressively marketing Glucophage XR — an extended-release version — while withdrawing the original immediate-release formulation from its promotional efforts. The strategy worked partially: Glucophage XR sustained revenue, but generic immediate-release metformin still launched and captured much of the market.
The lesson extracted from Glucophage was that product hopping works better when the transition is more complete. Later iterations of the strategy involved discontinuing the original product entirely, changing samples and promotional materials to the new formulation, and triggering automatic substitution rules that make it difficult for pharmacists to dispense the older generic equivalent. When New York State sued Actavis (now Allergan) for doing exactly this with the Alzheimer’s drug Namenda in 2015, a federal court granted a preliminary injunction blocking the product hop — one of the only successful judicial interventions against the practice.
The court found that Actavis was discontinuing the original Namenda IR not because of superior clinical properties of Namenda XR, but specifically to prevent generic substitution. That finding was possible because Actavis’s internal communications made the commercial motivation explicit. Most product hops leave less evidence, and as a result most go unchallenged.
Skinny Labels and the Section viii Carve-Out
The product hopping problem connects directly to one of the most contested legal developments in pharmaceutical patent law since 2023: the skinny label question.
The Hatch-Waxman framework includes a provision — the Section viii “carve-out” — that allows a generic manufacturer to seek approval for only the unpatented uses of a drug, explicitly carving out from its label the indication covered by a brand’s method-of-use patent. The intention was to allow generics to enter markets for older uses even when the brand had obtained a new-use patent. A blood pressure drug used for heart failure, for instance: the generic could be approved for hypertension even if the heart failure indication was under patent.
For most of Hatch-Waxman’s life, this worked. Brand companies filed method-of-use patents, generics carved out those uses, and competition proceeded in the broader market. But in the period since roughly 2019, brand companies have found ways to undermine the carve-out. The most direct approach involves induced infringement claims: arguing that even if the generic label omits the patented indication, prescribers will inevitably use the drug for that indication (because it’s the same molecule doing the same thing in the same patient), and that the generic’s promotion of the drug as a whole induces that infringement.
One of the most disruptive legal developments in 2024 and 2025 concerns the “skinny label” or Section viii carve-out. This mechanism allows a generic to launch a drug for unpatented indications while “carving out” patented uses from its label. It was intended to be a safe harbor, allowing generics to enter the market for old uses even if the brand found a new use for the drug. [5] Courts have now issued contradictory rulings on whether skinny-label launches constitute infringement when prescribers use the drug for patented uses, creating significant legal uncertainty for the entire generic pipeline.
Part Six: The Orange Book Battlefield
Device Patents and the 30-Month Stay
The Orange Book listing mechanism is structurally elegant in its original design and structurally susceptible to abuse in practice. Any patent that a brand manufacturer certifies covers its approved drug product gets listed. Any generic challenging that patent triggers a 30-month automatic stay on final ANDA approval. The stay requires no judicial finding that the patent is valid or enforceable — it is an administrative consequence of filing suit.
The device patent problem exploits this mechanism at its most direct level. Drug-device combination products — metered-dose inhalers, autoinjectors, prefilled pens — integrate a drug substance with a delivery mechanism. The drug substance itself may be off-patent. The delivery device is a mechanical object that engineers iterate on continuously, generating new patents for each iteration. If the device patent can be listed in the Orange Book as “covering” the approved product, it triggers the 30-month stay when challenged — effectively giving the device patent the same competition-blocking leverage as a compound patent.
The FTC’s 2024-2025 enforcement campaign against this practice was among the most aggressive regulatory interventions in pharmaceutical patent policy in the post-Actavis period. The Commission sent warning letters to major pharmaceutical companies, including Teva, GSK, and AstraZeneca, challenging over 300 patent listings it deemed “improper.” In December 2025, following sustained pressure from the FTC, Teva Pharmaceuticals requested the removal of more than 200 patent listings from the Orange Book. These patents covered asthma, diabetes, and COPD treatments. [5]
The delisting of 200 Teva patents does not immediately open those markets to generic competition — generics still need to file ANDAs, demonstrate bioequivalence, and navigate any remaining valid patents — but it removes the 30-month automatic stay as a defensive weapon for device patents. That’s a material change in the competitive calculus for generic manufacturers evaluating whether to invest in inhaler and injector products.
GLP-1 Drugs and the Next Orange Book Battle
The GLP-1 agonist category — semaglutide (Ozempic, Wegovy), tirzepatide (Mounjaro, Zepbound), and their competitors — represents the most commercially significant pending Orange Book battleground. Fifty-seven percent of the patents on GLP-1s are for the delivery devices, not the active drug ingredients, according to recent Senate testimony. [19] The primary compound patents on semaglutide expire in 2032, but the device patent portfolio extends substantially beyond that.
Novo Nordisk’s Ozempic (injection) and Wegovy (injection) both use prefilled pen injectors. The pens are medical devices with numerous patentable components: the needle retraction mechanism, the dose counter display, the cap locking system, the plunger drive mechanism. Each of these components can generate multiple patent applications. If those patents are listed in the Orange Book, any generic semaglutide pen — even one using a completely different device design — triggers litigation and the 30-month stay if the brand files suit. And the brand will file suit.
Companies continue to file petitions regarding the “sameness” of generic inhalers and injectors, arguing that minor differences in the device user interface should preclude generic substitution. [5] For GLP-1 generics, where the active drug market is enormous and growing, these device-level disputes will determine the competitive timeline more than the compound patent clock.
Part Seven: Biologics, Biosimilars, and the BPCIA
A Different Legal Framework, A Worse Problem
Small-molecule drugs operate under Hatch-Waxman. Biologic drugs — larger, protein-based molecules manufactured in living cells — operate under the Biologics Price Competition and Innovation Act (BPCIA) of 2010. The BPCIA created an FDA approval pathway for biosimilars (the biologics equivalent of generics) but with notable differences from Hatch-Waxman that have made patent exclusivity significantly easier to sustain.
The most consequential difference involves the number of patents a brand company can assert. Under Hatch-Waxman, the Orange Book limits which patents can trigger the 30-month stay to those actually listed. Under the BPCIA, the “patent dance” — the structured information exchange between brand and biosimilar applicant — places no statutory cap on the total number of patents a brand can assert in litigation. Unlike Hatch-Waxman, the BPCIA places no statutory limit on the total number of patents that a brand company can assert against a biosimilar competitor in litigation. This seemingly minor detail is arguably the single biggest reason why patent thickets have become exceptionally dense and problematic in the biologics space. [20]
This creates the legal environment that made Humira’s 250-patent fortress possible. In the BPCIA world, every patent AbbVie chose to assert was a potential litigation track. The biosimilar challenger faced not a single lawsuit but a potential cascade of suits, each requiring resources to litigate, each creating uncertainty about eventual outcome, each adding to the timeline.
The Protecting Biosimilar Competition Act, introduced in the 118th and 119th Congresses under various designations (S. 150, S. 1041), attempts to address this directly. The legislation proposes limiting the number of patents a biologic sponsor can assert in the patent dance to 20, with a cap of 10 patents issued post-approval. [5] The bill hasn’t passed as of early 2026, but its provisions have drawn enough bipartisan support to suggest eventual movement.
Keytruda: The Next Fortress
Merck’s pembrolizumab (Keytruda) is the most commercially important biologic patent situation to watch in the next five years. Keytruda generated approximately $29.5 billion in global revenue in 2024, making it the world’s top-selling drug by a wide margin and the most valuable single asset in the upcoming patent cliff. [20]
The compound patent expires in the United States in 2028. But Keytruda’s patent estate is among the most complex in modern pharmaceutical history. Merck has filed patents covering the antibody structure, manufacturing processes, formulation, dosing regimens, and dozens of specific tumor types and treatment combinations. Each of the drug’s more than 40 approved indications represents a potential method-of-use patent.
The first biosimilar manufacturers to file 351(k) applications for pembrolizumab will face a patent challenge landscape that is categorically more expensive and litigious than anything in the small-molecule generic market. Early estimates suggest that fewer than ten companies globally have the technical capability to manufacture a pembrolizumab biosimilar at commercial scale. [20] Samsung Bioepis, Fresenius Kabi, and several Chinese biologics manufacturers are the most likely early filers.
Merck has not waited passively. The company received FDA approval for a subcutaneous formulation of pembrolizumab in 2024, creating a reformulated product with its own patent life. This is product hopping in the biologic context — legally available, commercially rational, and directly analogous to the small-molecule tactics that regulators are increasingly scrutinizing in pill form.
Part Eight: The Generic Industry’s Counter-Moves
Paragraph IV Certification Strategy
The generic industry has developed its own sophisticated response set to evergreening tactics, and understanding those responses is necessary for anyone assessing the real-world effectiveness of brand-side strategies.
The Paragraph IV certification — the ANDA filing that challenges one or more Orange Book patents as invalid or not infringed — is the primary weapon. When a generic manufacturer files a Para IV, it certifies that the challenged patents are either invalid, unenforceable, or will not be infringed by the generic product. This immediately triggers brand litigation rights (the 30-month stay) and the possibility of a court ruling that invalidates the patent.
As noted earlier, generic manufacturers win approximately 73 percent of fully litigated Para IV cases. The challenge is that “fully litigated” is the key qualifier: most cases settle before trial, in the pay-for-delay pattern described above, or in deals where the generic gets a defined entry date in exchange for withdrawing the patent challenge. The settlement removes the risk for both parties but preserves the patent’s nominal validity — leaving it in the Orange Book to deter the next challenger.
The sophisticated generic strategy involves sequencing. A generic manufacturer evaluating a drug with 40 Orange Book patents does not challenge all 40. It conducts a freedom-to-operate analysis, identifies which patents are weakest on the merits (those most likely to be invalidated as obvious, anticipated, or insufficiently enabled), and files Para IV certifications targeting only those. This concentrates litigation resources on the most winnable claims while avoiding expensive fights on patents with defensible merit.
Tools like DrugPatentWatch — a patent intelligence platform that aggregates Orange Book data, patent expiration timelines, ANDA filing histories, and litigation records — are essential for this kind of portfolio analysis. Generic manufacturers, specialty pharma acquirers, hedge funds building positions around patent cliff events, and anyone doing competitive intelligence on branded drugs routinely use DrugPatentWatch to build the kind of multi-patent expiration maps that let them prioritize which challenges to file, which markets to enter, and which branded revenue streams are most vulnerable to competition. For a market where timing determines the value of the 180-day exclusivity award, getting the expiration timeline right by even a few months translates into hundreds of millions of dollars.
Inter Partes Review
The America Invents Act of 2011 created the Patent Trial and Appeal Board (PTAB) and a process called Inter Partes Review (IPR), which allows any party to petition for administrative review of an issued patent’s validity by a panel of technically trained judges. IPR was designed to provide a faster, cheaper alternative to district court litigation for challenging weak patents.
In pharmaceutical patent litigation, IPR has become a powerful tool for generic manufacturers. The PTAB applies a different — and somewhat lower — claim construction standard than district courts, and has historically invalidated a higher percentage of challenged claims than courts have. Brand companies initially fought IPR challenges on constitutional grounds, but those arguments were rejected by the Supreme Court in Oil States Energy Services v. Greene’s Energy Group (2018).
The pharma industry has since lobbied successfully for procedural limits on IPR, including the institution of “discretionary denial” practices that let the PTAB decline to review patents that are simultaneously being litigated in district court. These limits have reduced but not eliminated IPR as a counter-evergreening tool. For secondary patents covering formulations or manufacturing processes — exactly the kind of patents most likely to be challenged as obvious in light of earlier work — IPR remains an effective mechanism.
Authorized Generics as a Double-Edged Sword
An authorized generic is a version of a brand product sold by the brand company (or its licensee) under the generic drug’s labeling, marketed simultaneously with branded product. For the brand company, it captures a revenue stream in the generic market rather than ceding all of it to a first-filer challenger. For the first-filer generic, an authorized generic launched simultaneously eliminates the 180-day period of solo exclusivity that makes patent challenges economically worthwhile.
This creates a deterrence mechanism. A brand company that credibly commits to launch an authorized generic on Day 1 of any challenger’s entry makes Para IV challenges less profitable. Fewer challenges get filed. Existing patents — including questionable secondary patents — face less scrutiny.
The FTC has classified authorized generic agreements — where a brand agrees not to launch an authorized generic during a challenger’s 180-day exclusivity — as a form of pay-for-delay compensation. But authorized generic threats remain a legitimate (if strategically calculated) response to patent challenges when actually deployed.
Part Nine: The Regulatory and Legislative Landscape in 2025-2026
FTC Enforcement Under Current Administration
The FTC’s 2024-2025 Orange Book enforcement campaign was its most focused pharmaceutical patent action since Actavis. The campaign’s distinctive feature was targeting improper listings rather than anticompetitive settlements — a legal strategy that avoided the rule-of-reason analysis Actavis required and instead asserted that listing a non-qualifying patent violates the FTC Act’s prohibition on unfair or deceptive practices.
The forced delisting of Teva’s 200-plus device patents in December 2025 validated that strategy as a practical enforcement mechanism. Brand companies that had assumed Orange Book listings were immune from FTC challenge learned otherwise. The precedent is now set, and the compliance burden on companies listing device-related patents has increased substantially.
Under the current administration, the FTC has signaled a zero-tolerance policy for improper Orange Book listings broadly — not just device patents. The agency’s success in forcing Teva to delist device patents in 2025 sets a precedent. We can expect further mass delistings, which will remove the 30-month stay protection for many drug-device combinations. [5]
The simultaneous pressure from the Department of Justice’s Antitrust Division, which has been more aggressive on pharmaceutical markets, creates a regulatory pincer that brand companies haven’t faced at this intensity since the early 2000s.
Congressional Action on Patent Thickets
Legislative efforts to address patent thickets and evergreening have multiplied since 2022, and several have now moved past committee consideration into active debate. The most significant are the Protecting Biosimilar Competition Act (targeting BPCIA patent assertion limits), the PATENT Act (reforming Orange Book listing rules to require affirmative demonstration of relevance), and various versions of the Stop STALLING Act (imposing financial penalties on sham citizen petitions).
The Protecting Biosimilar Competition Act proposes limiting the number of patents a biologic sponsor can assert in the patent dance to 20, with a cap of 10 patents issued post-approval. It would grant the FTC express authority to prosecute product hopping as presumptive anticompetitive conduct. [5]
The bipartisan support for these measures reflects a political environment where drug pricing is one of the few issues generating agreement across party lines. The Inflation Reduction Act’s Medicare drug price negotiation provisions — which took effect for ten drugs in 2026, with additional drugs scheduled in subsequent years — have created a separate pricing-pressure mechanism that interacts with patent strategy in complex ways. Drugs subject to Medicare negotiation have less incentive to maintain list price through the late patent period, which changes the evergreening calculation for their manufacturers.
USPTO Reform
The USPTO has been criticized for permissiveness in granting pharmaceutical patents — particularly in applying the nonobviousness standard to secondary pharmaceutical patents, where courts have frequently found that the USPTO’s threshold for granting patents was far below what courts later required to sustain them.
The Biden administration’s 2022 collaboration between USPTO Director Kathi Vidal and FDA Commissioner Robert Califf on pharmaceutical patent quality addressed part of this problem. Proposed guidance on obviousness in pharmaceutical contexts was released for comment in 2023. The Trump administration’s USPTO, in its second term, has taken a more complex stance — focused on maintaining U.S. patent system strength generally while showing less interest in administrative reforms specifically targeted at pharmaceutical secondary patents.
The PTAB remains important here. Policymakers could consider improvements to the USPTO’s Patent Trial and Appeal Board, which reviews and invalidates low-quality patents through administrative proceedings. Changes to consider include allowing PTAB decisions to be sufficient for ANDA certification and allowing any person to file a challenge to a patent. [3] Both proposed changes would significantly amplify IPR’s value as an anti-evergreening tool.
Part Ten: What the Patent Cliff Data Tells Us
$187 Billion in Revenue at Risk
The next five years represent an unusual convergence of major brand patent expirations. Understanding which drugs face genuine loss of exclusivity and which have meaningful secondary patent protection is the central question for everyone from portfolio managers to health system formulary directors.
Together, 65 drugs represent $187 billion in annual global sales at risk between 2025 and 2030. The U.S. market alone accounts for 63 percent of that value, because drug prices here are far higher than in Europe or Canada. [9]
The specific drugs getting the most attention from patent analysts and generic manufacturers are a representative sample:
Entresto (sacubitril/valsartan, Novartis) had its key U.S. patent expire in July 2025. The FDA approved the first generic version of sacubitril ahead of schedule in October 2024. [9] Novartis has secondary patents covering the co-crystal form of the drug and manufacturing processes, but the compound patent expiration has cleared the primary path. Generic entry is proceeding more or less on schedule, demonstrating what the system looks like when the secondary patents are either weak or not aggressively defended.
Eliquis (apixaban, Bristol-Myers Squibb/Pfizer) faces its key patent expiration in November 2026. Global sales of Eliquis in 2024 were substantial, representing one of BMS’s most critical revenue streams. [9] The Eliquis patent situation is more complicated than Entresto: BMS and Pfizer have numerous secondary patents covering dosing regimens and formulations, and litigation has been active. Multiple generic manufacturers have Para IV filings pending, and the litigation timeline will determine whether generics can enter at patent expiration or face additional delays.
Keytruda (pembrolizumab, Merck) with its 2028 compound patent expiration and $29.5 billion in 2024 revenue is in a class by itself. The biologic complexity, the BPCIA framework’s unlimited patent assertion, and the manufacturing difficulty all suggest that meaningful biosimilar competition will not arrive at 2028 regardless of patent outcomes. The realistic timeline for significant biosimilar market share is 2031-2033 at the earliest.
Ibrance (palbociclib, Pfizer) has a patent expiring March 2026 with global sales of $5.1 billion in 2024. Trulicity (dulaglutide, Eli Lilly) faces patent expiration in late 2026 with $10.5 billion in 2024 sales. Amgen stands to lose 52 percent of its 2024 revenue from expiring patents. [9]
The Generic Filing Surge
Generic manufacturers are watching this cliff. The FDA received 127 ANDAs targeting 2025 expirations in 2024 alone — a 27 percent jump from the year before. [9] That surge in filings reflects sophisticated tracking of the patent expiration timeline and confidence that the Orange Book listings for many of these drugs are either thin or challengeable.
What happens after generics enter is relatively predictable for small molecules: prices drop 80-90 percent within 12 months, and generics capture over 90 percent of the market within two years. Biosimilars are slower — prices drop 30-40 percent initially, and it takes 3-5 years to reach 75 percent market share because they are harder to make and require more testing. [9]
The speed of price decline is itself relevant to the evergreening question. A delay of 18 months in generic entry, at a branded price of $2 billion per year, represents $3 billion in consumer costs. A delay of five years (the Humira U.S. versus Europe gap) at Humira’s scale represents tens of billions. The aggregate academic finding that evergreening “doesn’t work” nationally obscures these per-drug calculations, which are very large and very real for the drugs where the strategy succeeds.
Part Eleven: The International Dimension
How Other Countries Handle This
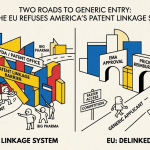
The United States is essentially alone among high-income countries in the specific combination of regulatory and legal features that make pharmaceutical patent thickets so effective. The 30-month automatic stay triggered by any Orange Book patent listing, the BPCIA’s unlimited patent assertion in biologics, and the relatively permissive USPTO standards for secondary pharmaceutical patents are U.S.-specific features.
Canada and the UK have their own patent linkage systems, but with narrower definitions of which patents qualify for listing. The European Patent Office’s opposition procedure provides a post-grant review mechanism with meaningful invalidation rates. Canada’s Patented Medicine Prices Review Board exercises price control authority that reduces the financial benefit of patent extension. The result, as the PLOS ONE study found, is that the same drugs face nine to twelve times fewer patent assertions in those markets than in the United States, and biosimilars enter more quickly [12].
That doesn’t mean those systems are superior in every dimension. European manufacturers sometimes argue that tighter patent protection reduces incentives for follow-on innovation — a claim that carries some merit for genuine improvements but is less convincing for formulation tweaks. The comparison does establish, however, that the U.S. outcomes are not inevitable consequences of universal patent law. They are specific to U.S. regulatory design choices that can, in principle, be changed.
India’s approach to this question is the most explicit statutory restriction on evergreening in any major jurisdiction. Section 3(d) of India’s Patents Act specifically prohibits the grant of patents for new forms of known substances unless the applicant demonstrates significantly enhanced efficacy. The provision was challenged by Novartis in the famous Novartis AG v. Union of India (2013) case, where the Indian Supreme Court rejected the company’s patent application for the cancer drug Gleevec (imatinib) as an unpatentable modification of a known compound. The case established Section 3(d) as a viable legislative tool and has been studied by other countries considering similar restrictions [21].
No equivalent U.S. legislation has passed, though proposals along these lines appear periodically in congressional reform packages.
Part Twelve: The Business Analytics Perspective
Measuring What Matters
For business analysts, competitive intelligence professionals, and investors, the evergreening question is ultimately empirical: for a specific drug, in the current regulatory environment, what is the realistic date of generic or biosimilar entry, and what uncertainty surrounds that date?
The academic finding that evergreening “doesn’t systematically delay effective patent life across the drug universe” is analytically true but operationally useless for anyone making a specific investment decision or procurement strategy. What matters is the drug-specific analysis.
That analysis requires knowing the complete patent landscape: which patents are in the Orange Book, when each expires, which have faced Para IV challenges, the outcomes of any litigation, whether citizen petitions are pending, whether the first-filer has settled, and whether the brand has initiated product hopping. This information exists in regulatory filings and patent databases, but assembling it systematically requires either significant internal resources or specialized tools.
DrugPatentWatch aggregates this data — Orange Book patent listings with expiration dates, ANDA filing histories showing which generics have challenged which patents, litigation outcomes, and first-filer settlement status — into searchable formats that allow analysts to build the patent expiration maps that matter for specific drugs. For anyone evaluating formulary decisions, negotiating supply contracts around patent cliffs, or building investment positions in generic manufacturers targeting specific drugs, that assembled picture is the starting point for analysis that the aggregate academic studies cannot provide.
The commercial stakes make the precision matter. A biosimilar manufacturer that files its 351(k) application two years before a competitor gains a competitive advantage in the FDA review queue. A health system that negotiates a rebate contract extending through an expected generic entry date may find itself locked into brand pricing for longer than intended if secondary patents delay entry. A hedge fund that identifies a drug with an apparently dominant secondary patent that is actually vulnerable to IPR has a specific, actionable investment thesis.
The ROI of Patent Intelligence
The financial stakes of getting patent timelines right are not abstract. The 180-day first-filer exclusivity period for a major small-molecule drug with $2 billion in annual brand sales is worth approximately $600-900 million in gross revenue to the winning generic manufacturer — assuming the brand price holds at something above the eventual competitive equilibrium during that period. Being first to file, by even a few weeks, can determine whether a company captures that window.
On the brand side, the revenue extended through secondary patent enforcement is quantifiable. AbbVie maintained Humira at approximately $20 billion in annual U.S. sales through 2022 [16]. Had biosimilar entry proceeded in 2018 at European parity, price would have fallen roughly 38 percent — consistent with the actual decline when biosimilars did arrive [19]. The implied revenue value of the 2018-2023 delay is on the order of $30-40 billion, even after accounting for some market share erosion. That figure explains why AbbVie built the patent fortress and why the legal and regulatory battle over it was so protracted.
Part Thirteen: Does It Work — A Synthesis
The Differentiated Answer
The question “does drug patent evergreening prevent generic entry?” requires a differentiated answer across three scenarios.
For the broad universe of small-molecule drugs with moderate revenue and standard secondary patent portfolios: no, evergreening generally does not prevent generic entry. It may delay it by months or occasionally a year or two, but the combination of Para IV challenges, IPR petitions, and the competitive dynamics of the generic pipeline means that the compound patent expiration is approximately when competition arrives. The George Mason analysis supports this finding [1].
For the subset of high-revenue small-molecule drugs where brands deploy the full toolkit — dense secondary patent filing, product hopping, citizen petitions, authorized generic deterrence, and pay-for-delay settlements: yes, evergreening frequently delays generic entry by two to five years beyond compound patent expiration. This is the scenario the FTC’s $3.5 billion annual consumer cost estimate applies to [15]. The Gilead HIV drug settlements extending protection to 2031 illustrate the current version of this problem [10].
For biologics under the BPCIA framework: the evergreening toolkit is so powerful, and the manufacturing barriers so high, that the concept of “preventing” generic entry shifts. Biologics litigation is so expensive and uncertain that biosimilar manufacturers negotiate entry terms rather than fight to win. The question is not when biosimilar entry is “allowed” by the patent system but when it becomes commercially viable given the cost of litigation and the likely outcome of any settlement. In this context, AbbVie’s five-year transatlantic delay for Humira biosimilars is characteristic, not exceptional.
The Evidence Gap
A significant weakness in the evergreening literature is the gap between nominal patent analysis and actual economic outcomes. Most academic studies count patents or calculate nominal extension times. Fewer track the complete lifecycle from ANDA filing to first generic sale, controlling for product-specific factors like drug complexity, market size, and brand litigation aggressiveness.
The George Mason study took the latter approach for its sample and found no correlation between patent count and effective patent life [1]. The I-MAK analysis took the former approach — counting patents and showing high proportions of post-approval filings — and found evidence of systematic overpatenting [10]. Both findings are internally consistent; they measure different things. The practical implication of the George Mason finding is that many patents filed post-approval are simply not effective barriers — they are challenged, invalidated, or worked around without materially delaying competition. The practical implication of the I-MAK finding is that the costs of filing and litigating those patents still fall on generic manufacturers and, indirectly, on consumers through higher prices during any resulting delay.
What the FTC-Teva Delistings Mean
The most practically significant recent development is the Teva delisting of 200-plus Orange Book device patents under FTC pressure. This action:
Removes the automatic 30-month stay on ANDA approvals for drugs in those delisted product classes, allowing the FDA to grant final approval to generic versions without waiting for patent litigation to resolve.
Establishes that improper Orange Book listings violate the FTC Act, creating a compliance deterrent that extends far beyond Teva.
Signals to GLP-1 manufacturers — who currently have 57 percent of their patents concentrated in delivery devices — that device-patent Orange Book listings will face similar scrutiny as generic semaglutide and tirzepatide products approach filing readiness.
This is not the end of pharmaceutical evergreening. The mechanisms that survive — product hopping, pay-for-delay settlements, citizen petitions, and the density of the BPCIA patent assertion framework for biologics — remain potent. But the device patent Orange Book strategy, one of the more egregious implementations of the concept, is now materially constrained.
Part Fourteen: The Path Forward
What Reform Would Actually Accomplish
Effective pharmaceutical patent reform requires targeting the specific mechanisms that produce delay without targeting the legitimate patent rights that incentivize innovation. A blanket cap on the number of pharmaceutical patents per drug would be both overinclusive (eliminating some valid secondary patents covering genuine improvements) and practically unenforceable. Targeted interventions that address specific abuse vectors are more likely to achieve durable results.
The FTC’s Orange Book enforcement approach models this correctly: it doesn’t ban secondary patenting, it bans listing irrelevant patents in a way that creates automatic regulatory leverage. A PTAB reform allowing IPR petitions by parties without a specific threatened infringement claim — opening administrative challenges to public interest groups and pharmacy benefit managers — would expand access to the patent challenge process. Mandatory early disclosure of settlement agreements with pay-for-delay characteristics would improve regulatory oversight without requiring a blanket ban on settlements that Actavis allows.
Congressional action limiting BPCIA patent assertions to 20 per biologic sponsor, as the Protecting Biosimilar Competition Act proposes, would be the single most impactful structural change available [5]. It would not prevent brand companies from filing 250 patents on a biologic. It would prevent them from asserting all 250 in patent dance litigation — which is the operative mechanism of the Humira strategy.
The Role of Litigation Economics
Long-term, the economics of pharmaceutical patent litigation are shifting. The cost of biosimilar development and the BPCIA litigation environment have historically deterred all but the largest generic manufacturers from biosimilar entry. But Chinese and Korean manufacturers — Samsung Bioepis, Celltrion, Henlius — have built manufacturing capacity at a scale that makes the upfront investment in biosimilar development economically viable even given the litigation costs. Their entry into the U.S. biosimilar market changes the competitive calculus for brand companies.
If AbbVie were building its Humira patent fortress today, facing Samsung Bioepis as a challenger instead of the smaller European biosimilar manufacturers of 2013, the settlement terms might look different. A challenger with deep pockets, lower manufacturing costs, and strategic interest in U.S. market entry is less easily paid off than a challenger for whom the litigation cost is a significant portion of expected revenue.
The long-term trajectory of biosimilar competition is toward greater effectiveness, as manufacturing capacity expands and the biosimilar market matures. Evergreening will continue as a strategy, but its effectiveness against sophisticated challengers with adequate resources is lower than it was a decade ago.
Key Takeaways
Evergreening exists and costs money — but its effectiveness varies enormously by drug. For most small-molecule drugs with moderate revenue, secondary patent portfolios don’t materially extend effective exclusivity beyond the compound patent. For the highest-revenue drugs — where brand companies deploy the complete toolkit including pay-for-delay settlements, product hopping, and citizen petitions — delays of two to five years beyond the compound patent expiration are well-documented.
The Humira case is a ceiling, not a template. AbbVie’s 250-patent strategy produced the most successful patent-based market exclusion in pharmaceutical history. But it required a unique combination of BPCIA permissiveness, settlement agreements with nine competitors, and product hopping execution that most brands cannot replicate for most drugs. Treating Humira as representative of evergreening outcomes overall would be analytically wrong.
The FTC’s Orange Book enforcement campaign has materially changed the device patent landscape. Teva’s 2025 delisting of 200-plus device patents removes a significant competitive barrier from inhaler and injector products. The GLP-1 sector is next in line for this scrutiny.
Pay-for-delay settlements remain the most economically damaging form of patent abuse. The FTC estimates $3.5 billion in annual consumer costs. The mechanism — where the first ANDA filer is paid to stand down, corking the bottle for all subsequent generics — is the most direct subversion of the Hatch-Waxman system’s competitive intent.
Biologics are categorically different from small molecules. The BPCIA’s unlimited patent assertion framework, combined with manufacturing complexity, creates competitive dynamics where even well-funded biosimilar challengers often prefer negotiated entry to full litigation. Reform here — specifically limiting the number of assertable patents in the patent dance — would have more impact per legislative dollar than any other available intervention.
Patent cliff timing matters at the drug level, not the aggregate level. The academic finding that evergreening “doesn’t work” across the drug universe is statistically accurate but operationally misleading. Drug-specific patent intelligence — tracking which patents are listed, which have been challenged, which settlements are in place, and which product hops are underway — is the analytical foundation for any decision that turns on when generic competition will actually arrive.
The $187 billion in drugs facing patent expiration between 2025 and 2030 will not all transition to generic competition on the same timeline. Simple compound patent expiration dates are a starting point, not an answer. The full patent landscape, litigation status, and regulatory dynamics for each drug determine the actual competitive entry point.
FAQ
Q1: Is drug patent evergreening illegal?
No, most forms of evergreening are legal under U.S. patent and pharmaceutical law. Filing secondary patents on new formulations, delivery methods, or methods of use is lawful if those patents meet the statutory requirements of novelty, utility, and nonobviousness. Product hopping is generally legal unless a court finds it was undertaken specifically to exclude competition with no offsetting clinical rationale — a rare finding. The practice becomes legally problematic when it involves listing non-qualifying patents in the Orange Book (an FTC enforcement target), filing citizen petitions without a good-faith basis (addressed by the Stop STALLING Act proposals), or entering into pay-for-delay settlements that courts find anticompetitive under the Actavis rule-of-reason analysis. The line between aggressive competitive behavior and antitrust violation is fact-specific and frequently litigated.
Q2: Why do generic companies sometimes lose Para IV patent challenges even when the patents are weak?
Patent litigation is expensive, time-consuming, and uncertain. A generic manufacturer challenging a patent enters a process that can take three to seven years to reach final judicial resolution, with costs that can exceed $30 million per case. If the brand has twenty patents in the Orange Book, the generic must litigate each separately or in coordinated proceedings. Even if the generic ultimately prevails, it may have spent more on litigation than it earns during its 180-day exclusivity period. Pay-for-delay settlements resolve this problem for both parties — at the expense of consumers. The asymmetry in resources and risk tolerance between a $20-billion-revenue brand product and a prospective generic challenger makes settlement, rather than victory, the rational outcome in many cases where the generic might ultimately have won.
Q3: What does a drug’s Orange Book listing actually tell a market analyst?
The Orange Book lists all patents that the brand manufacturer has certified as covering the approved drug product, along with their expiration dates. Each listed patent represents a potential litigation trigger — a 30-month automatic stay on ANDA approval if the brand files suit within 45 days of a generic’s Para IV certification. Analysts reading the Orange Book can identify: the latest-expiring patent (the theoretical ceiling of brand protection), which patents have already been challenged (indicating challengers have evaluated and decided to fight), whether any first-filer settlements are disclosed, and whether device patents are listed (now a specific FTC focus). The Orange Book does not tell you the strength of the listed patents, whether they’re likely to survive litigation, or whether the brand will actually enforce them. That judgment requires patent-level analysis of claim scope, prosecution history, and prior art — the kind of analysis that DrugPatentWatch and specialized IP intelligence firms facilitate.
Q4: How does product hopping interact with pharmacy substitution laws?
Pharmacy substitution laws in most U.S. states require or permit pharmacists to substitute an FDA-rated generic equivalent for a branded prescription unless the prescriber writes “dispense as written.” Product hopping undermines substitution when the prescriber writes for the new reformulation (e.g., Namenda XR rather than Namenda IR) rather than the original product for which the generic is approved. The generic for the original product cannot automatically substitute for the reformulated version because they have different FDA ratings. If the brand successfully transitions the prescription base to the new formulation before generic entry, the generic of the original product finds a market of limited commercial value. Effective product hopping therefore requires convincing prescribers to switch prescribing habits before the generic of the original product enters — which is why brands typically invest heavily in detailing the new formulation in the 12-24 months before their compound patent expires.
Q5: What does the Inflation Reduction Act’s drug price negotiation provision mean for patent strategy?
The IRA’s Medicare negotiation authority — which took effect for 10 drugs negotiated in 2024-2025, with more drugs added annually — changes the incentive calculation for evergreening in a specific and underappreciated way. Drugs subject to Medicare price negotiation will have their Medicare prices set by a government negotiation process rather than the market. That negotiated price is typically substantially below the market price. If the brand product’s Medicare revenue is now capped, the financial return from extending exclusivity via secondary patents or pay-for-delay settlements diminishes — because the ceiling price during the extended exclusivity period is lower than it would otherwise be. This doesn’t eliminate the incentive to evergreen, since commercial payers and patients pay market prices. But it reduces the total revenue value of each year of extended exclusivity for drugs subject to negotiation, which changes the cost-benefit calculation for the litigation and settlement strategies that produce those extensions.
Citations
[1] George Mason University Center for Intellectual Property. (2024). Pharmaceutical “Nominal Patent Life” Versus “Effective Patent Life,” Revisited. George Mason University. https://cip2.gmu.edu/2024/05/20/pharmaceutical-nominal-patent-life-versus-effective-patent-life-revisited/
[2] Drug Price Competition and Patent Term Restoration Act of 1984, Pub. L. No. 98-417, 98 Stat. 1585 (1984) (codified as amended 21 U.S.C. § 355).
[3] Martin, K. (2025, November 13). How drugmakers use the patent process to keep prices high. Commonwealth Fund. https://www.commonwealthfund.org/publications/explainer/2025/nov/how-drugmakers-use-patent-process-keep-prices-high
[4] Carrier, M. A., & Minniti, C. (2016). Citizen petitions: Long, late-filed, and litigious. UC Davis Law Review, 50(1), 33–83.
[5] DrugPatentWatch. (2026, January 16). Evergreening by lawsuit: Strategic patent actions and generic entry stagnation. https://www.drugpatentwatch.com/blog/evergreening-by-lawsuit-strategic-patent-actions-and-generic-entry-stagnation/
[6] Feldman, R. (2018). May your drug price be ever green. Journal of Law and the Biosciences, 5(3), 590–647. https://doi.org/10.1093/jlb/lsy022
[7] Grabowski, H., Long, G., Mortimer, R., & Boyo, A. (2012). Evergreening, patent challenges, and effective market life in pharmaceuticals. Journal of Health Economics, 31(2), 327–339. https://pubmed.ncbi.nlm.nih.gov/22425766/
[8] Hemphill, C. S., & Sampat, B. N. (2012). Evergreening, patent challenges, and effective market life in pharmaceuticals. Journal of Health Economics, 31(2), 327–339.
[9] fda-approved-rx.net. (2025, March). Blockbuster patent expirations 2025 and beyond: What’s coming and how it affects you. https://fda-approved-rx.net/blockbuster-patent-expirations-2025-and-beyond-what-s-coming-and-how-it-affects-you
[10] Lovelace, B. (2022, September 15). ‘Gaming’ of U.S. patent system is keeping drug prices sky high, report says. NBC News. https://www.nbcnews.com/health/health-news/gaming-us-patent-system-keeping-drug-prices-sky-high-report-says-rcna47507
[11] U.S. Securities and Exchange Commission. (2022). Shareholder filing: AbbVie patent thicket analysis. https://www.sec.gov/Archives/edgar/data/0001551152/000121465922005064/b48224px14a6g.htm
[12] Feldman, R., & Wang, C.-F. (2022). Biological patent thickets and delayed access to biosimilars, an American problem. PLOS ONE. https://pmc.ncbi.nlm.nih.gov/articles/PMC9439849/
[13] DrugPatentWatch. (2025, November 4). The thicket maze: A strategic guide to navigating and dismantling drug patent fortresses. https://www.drugpatentwatch.com/blog/the-thicket-maze-a-strategic-guide-to-navigating-and-dismantling-drug-patent-fortresses/
[14] Association for Accessible Medicines. (2025, June 4). Patent settlements are necessary to help combat patent thickets. https://accessiblemeds.org/resources/blog/patent-settlements-are-necessary-to-help-combat-patent-thickets/
[15] Federal Trade Commission. (n.d.). Pay for delay. https://www.ftc.gov/news-events/topics/competition-enforcement/pay-delay
[16] Federal Trade Commission. (2010, January). Pay-for-delay: How drug company pay-offs cost consumers billions [Staff study]. https://www.ftc.gov/sites/default/files/documents/reports/pay-delay-how-drug-company-pay-offs-cost-consumers-billions-federal-trade-commission-staff-study/100112payfordelayrpt.pdf
[17] FTC v. Actavis, Inc., 570 U.S. 136 (2013).
[18] Federal Trade Commission. (2010). Pay-for-delay: How drug company pay-offs cost consumers billions — FTC staff study. Federal Trade Commission. https://www.ftc.gov/sites/default/files/documents/reports/pay-delay-how-drug-company-pay-offs-cost-consumers-billions-federal-trade-commission-staff-study/100112payfordelayrpt.pdf
[19] Evernorth. (2025, June 4). How drugmakers exploit the patent system to delay competition and inflate prices. https://www.evernorth.com/articles/how-drugmakers-exploit-patent-system-delay-competition-and-inflate-prices
[20] DrugPatentWatch. (2025, March 31). The global generic drug market: Trends, opportunities, and challenges. https://www.drugpatentwatch.com/blog/the-global-generic-drug-market-trends-opportunities-and-challenges/
[21] Novartis AG v. Union of India & Others, (2013) 6 SCC 1 (India Supreme Court). https://www.supremecourtofindia.nic.in