GILEAD SCIENCES INC Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GILEAD SCIENCES INC, and when can generic versions of GILEAD SCIENCES INC drugs launch?
GILEAD SCIENCES INC has twenty-three approved drugs.
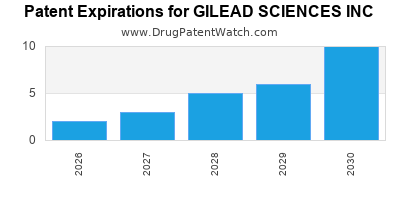
There are eighty-two US patents protecting GILEAD SCIENCES INC drugs.
There are two thousand two hundred and twenty-eight patent family members on GILEAD SCIENCES INC drugs in sixty-six countries and three hundred and seventy supplementary protection certificates in nineteen countries.
Summary for GILEAD SCIENCES INC
| International Patents: | 2228 |
| US Patents: | 82 |
| Tradenames: | 18 |
| Ingredients: | 18 |
| NDAs: | 23 |
| Drug Master File Entries: | 7 |
| Patent Litigation for GILEAD SCIENCES INC: | See patent lawsuits for GILEAD SCIENCES INC |
Drugs and US Patents for GILEAD SCIENCES INC
Expired US Patents for GILEAD SCIENCES INC
Paragraph IV (Patent) Challenges for GILEAD SCIENCES INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 150 mg, 200 mg, and 250 mg | ➤ Subscribe | 2012-05-17 |
| ➤ Subscribe | Tablets | 150 mg, 150 mg, 200 mg, 300 mg | ➤ Subscribe | 2018-10-04 |
| ➤ Subscribe | Tablets | 300 mg | ➤ Subscribe | 2010-01-26 |
| ➤ Subscribe | Tablets | 200 mg/25 mg/300 mg | ➤ Subscribe | 2015-05-20 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2015-12-09 |
International Patents for GILEAD SCIENCES INC Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2014040471 | ⤷ Try a Trial |
| Norway | 20071745 | ⤷ Try a Trial |
| Philippines | 12016502499 | ⤷ Try a Trial |
| Iceland | 7977 | ⤷ Try a Trial |
| Japan | 6571256 | ⤷ Try a Trial |
| South Korea | 20200067930 | ⤷ Try a Trial |
| Taiwan | 201446755 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for GILEAD SCIENCES INC Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2595980 | 53/2020 | Austria | ⤷ Try a Trial | PRODUCT NAME: REMDESIVIR ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ ODER ESTER DAVON; REGISTRATION NO/DATE: EU/1/20/1459 (MITTEILUNG) 20200703 |
| 1564210 | 203 5022-2013 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR ALEBO JEHO HYDRAT, SOLVAT, TAUTOMER ALEBO FARMACEUTICKY PRIJATELNA SOL; NAT. REGISTRATION NO/DATE: EU/1/13/830/001 - EU/1/13/830/002 20130527; FIRST REGISTRATION: EU EU/1/13/830/001 - EU/1/13/830/002 20130527 |
| 2430014 | 2020046 | Norway | ⤷ Try a Trial | PRODUCT NAME: LEDIPASVIR - FORLENGET SPC; REG. NO/DATE: EU/1/14/958 20141203 |
| 1564210 | SPC/GB13/065 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR; REGISTERED: UK EU/1/13/830/001 20130527; UK EU/1/13/830/002 20130527 |
| 2937350 | CA 2020 00060 | Denmark | ⤷ Try a Trial | PRODUCT NAME: REMDESIVIR ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/20/1459 20200703 |
| 1564210 | 13C0061 | France | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR OU UN HYDRATE, SOLVATE, TAUTOMERE, OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CEUX-CI; REGISTRATION NO/DATE: EU/1/13/830/001-002 20130527 |
| 1632232 | 93385 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE CHLORHYDRATE DE RILPIVIRINE OU UNE FORME THERAPEUTIQUE EQUIVALENTE QUI EN DERIVE TELLE QUE PROTEGEE PAR LE BREVET DE BASE, EMTRICITABINE, ET TENOFOVIR ALAFENAMIDE, OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI, EN PARTICULIER LE FUMARATE DE TENOFOVIR ALAFENAMIDE; AUTHORISATION NUMBER AND DATE: EU/1/16/1112 ODEFSEY - EMTRICITABINE |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.