SOVALDI Drug Patent Profile
✉ Email this page to a colleague
When do Sovaldi patents expire, and what generic alternatives are available?
Sovaldi is a drug marketed by Gilead Sciences Inc and is included in two NDAs. There are nine patents protecting this drug and one Paragraph IV challenge.
This drug has three hundred and sixty-eight patent family members in forty-nine countries.
The generic ingredient in SOVALDI is sofosbuvir. There are nine drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the sofosbuvir profile page.
DrugPatentWatch® Generic Entry Outlook for Sovaldi
Sovaldi was eligible for patent challenges on December 6, 2017.
There have been four patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for SOVALDI?
- What are the global sales for SOVALDI?
- What is Average Wholesale Price for SOVALDI?
Summary for SOVALDI
| International Patents: | 368 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 49 |
| Clinical Trials: | 34 |
| Drug Prices: | Drug price information for SOVALDI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for SOVALDI |
| What excipients (inactive ingredients) are in SOVALDI? | SOVALDI excipients list |
| DailyMed Link: | SOVALDI at DailyMed |
Recent Clinical Trials for SOVALDI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Cairo University | Phase 3 |
| Ain Shams University | Phase 3 |
| Assiut University | Early Phase 1 |
Pharmacology for SOVALDI
| Drug Class | Hepatitis C Virus Nucleotide Analog NS5B Polymerase Inhibitor |
| Mechanism of Action | RNA Replicase Inhibitors |
Paragraph IV (Patent) Challenges for SOVALDI
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| SOVALDI | Tablets | sofosbuvir | 400 mg | 204671 | 2 | 2017-12-06 |
US Patents and Regulatory Information for SOVALDI
SOVALDI is protected by nine US patents and two FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | PELLETS;ORAL | 212480-001 | Aug 28, 2019 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | PELLETS;ORAL | 212480-002 | Aug 28, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-002 | Aug 28, 2019 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for SOVALDI
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Ireland UC | Sovaldi | sofosbuvir | EMEA/H/C/002798Sovaldi is indicated in combination with other medicinal products for the treatment of chronic hepatitis C (CHC) in adult and paediatric patients aged 3 years and above (see sections 4.2, 4.4 and 5.1).For hepatitis C virus (HCV) genotype specific activity, see sections 4.4 and 5.1.Sovaldi is indicated in combination with other medicinal products for the treatment of chronic hepatitis C (CHC) in adults and paediatric patients aged 3 years and above (see sections 4.2, 4.4 and 5.1).For hepatitis C virus (HCV) genotype specific activity, see sections 4.4 and 5.1. | Authorised | no | no | no | 2014-01-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for SOVALDI
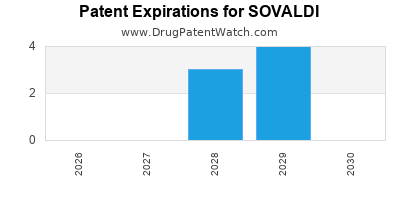
When does loss-of-exclusivity occur for SOVALDI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 15
Estimated Expiration: ⤷ Start Trial
Argentina
Patent: 0819
Estimated Expiration: ⤷ Start Trial
Patent: 0870
Estimated Expiration: ⤷ Start Trial
Patent: 1813
Estimated Expiration: ⤷ Start Trial
Patent: 2937
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10249481
Estimated Expiration: ⤷ Start Trial
Patent: 11235044
Estimated Expiration: ⤷ Start Trial
Patent: 11235112
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012024884
Estimated Expiration: ⤷ Start Trial
Patent: 2012024923
Estimated Expiration: ⤷ Start Trial
Patent: 2013004621
Estimated Expiration: ⤷ Start Trial
Patent: 2013007556
Estimated Expiration: ⤷ Start Trial
Patent: 1012781
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 63151
Estimated Expiration: ⤷ Start Trial
Patent: 94669
Estimated Expiration: ⤷ Start Trial
Patent: 94671
Estimated Expiration: ⤷ Start Trial
Patent: 19700
Estimated Expiration: ⤷ Start Trial
Patent: 49694
Estimated Expiration: ⤷ Start Trial
Patent: 15187
Estimated Expiration: ⤷ Start Trial
Patent: 88217
Estimated Expiration: ⤷ Start Trial
Patent: 77960
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 10000520
Estimated Expiration: ⤷ Start Trial
Patent: 11000716
Estimated Expiration: ⤷ Start Trial
Patent: 11000717
Estimated Expiration: ⤷ Start Trial
Patent: 11000718
Estimated Expiration: ⤷ Start Trial
Patent: 13000903
Estimated Expiration: ⤷ Start Trial
Patent: 13000904
Estimated Expiration: ⤷ Start Trial
China
Patent: 2459299
Estimated Expiration: ⤷ Start Trial
Patent: 2858790
Estimated Expiration: ⤷ Start Trial
Patent: 2906102
Estimated Expiration: ⤷ Start Trial
Patent: 4017020
Estimated Expiration: ⤷ Start Trial
Patent: 4292256
Estimated Expiration: ⤷ Start Trial
Patent: 5085592
Estimated Expiration: ⤷ Start Trial
Patent: 5198949
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 70789
Estimated Expiration: ⤷ Start Trial
Patent: 30166
Estimated Expiration: ⤷ Start Trial
Patent: 30167
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 120532
Estimated Expiration: ⤷ Start Trial
Patent: 120534
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0151075
Estimated Expiration: ⤷ Start Trial
Patent: 0160958
Estimated Expiration: ⤷ Start Trial
Patent: 0171267
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16976
Estimated Expiration: ⤷ Start Trial
Patent: 18045
Estimated Expiration: ⤷ Start Trial
Patent: 19273
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 52930
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 12012282
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 6341
Estimated Expiration: ⤷ Start Trial
Patent: 6731
Estimated Expiration: ⤷ Start Trial
Patent: 8709
Estimated Expiration: ⤷ Start Trial
Patent: 8742
Estimated Expiration: ⤷ Start Trial
Patent: 1171417
Estimated Expiration: ⤷ Start Trial
Patent: 1290988
Estimated Expiration: ⤷ Start Trial
Patent: 1290993
Estimated Expiration: ⤷ Start Trial
Patent: 1370186
Estimated Expiration: ⤷ Start Trial
Patent: 1592101
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 52930
Estimated Expiration: ⤷ Start Trial
Patent: 52931
Estimated Expiration: ⤷ Start Trial
Patent: 52933
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Patent: 10264
Estimated Expiration: ⤷ Start Trial
Patent: 52422
Estimated Expiration: ⤷ Start Trial
Patent: 10562
Estimated Expiration: ⤷ Start Trial
Patent: 13337
Estimated Expiration: ⤷ Start Trial
Patent: 90428
Estimated Expiration: ⤷ Start Trial
Patent: 21275
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 69414
Estimated Expiration: ⤷ Start Trial
Patent: 78171
Estimated Expiration: ⤷ Start Trial
Patent: 81775
Estimated Expiration: ⤷ Start Trial
Patent: 99645
Estimated Expiration: ⤷ Start Trial
Patent: 13571
Estimated Expiration: ⤷ Start Trial
Patent: 13572
Estimated Expiration: ⤷ Start Trial
Patent: 17494
Estimated Expiration: ⤷ Start Trial
Patent: 19106
Estimated Expiration: ⤷ Start Trial
Patent: 51578
Estimated Expiration: ⤷ Start Trial
Patent: 54977
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 26235
Estimated Expiration: ⤷ Start Trial
Patent: 31637
Estimated Expiration: ⤷ Start Trial
Patent: 34239
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 6492
Estimated Expiration: ⤷ Start Trial
Patent: 2099
Estimated Expiration: ⤷ Start Trial
Patent: 2174
Estimated Expiration: ⤷ Start Trial
Patent: 7471
Estimated Expiration: ⤷ Start Trial
Patent: 9115
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 72539
Estimated Expiration: ⤷ Start Trial
Patent: 85659
Estimated Expiration: ⤷ Start Trial
Patent: 09535
Estimated Expiration: ⤷ Start Trial
Patent: 58528
Estimated Expiration: ⤷ Start Trial
Patent: 06716
Estimated Expiration: ⤷ Start Trial
Patent: 55605
Estimated Expiration: ⤷ Start Trial
Patent: 12527477
Estimated Expiration: ⤷ Start Trial
Patent: 13523767
Estimated Expiration: ⤷ Start Trial
Patent: 13525277
Estimated Expiration: ⤷ Start Trial
Patent: 13527145
Estimated Expiration: ⤷ Start Trial
Patent: 15028060
Estimated Expiration: ⤷ Start Trial
Patent: 15205903
Estimated Expiration: ⤷ Start Trial
Patent: 16053045
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 6918
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 0725
Estimated Expiration: ⤷ Start Trial
Patent: 11012417
Estimated Expiration: ⤷ Start Trial
Patent: 12011171
Estimated Expiration: ⤷ Start Trial
Patent: 12011324
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 294
Estimated Expiration: ⤷ Start Trial
Patent: 846
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 6635
Estimated Expiration: ⤷ Start Trial
Patent: 3232
Estimated Expiration: ⤷ Start Trial
Patent: 3602
Estimated Expiration: ⤷ Start Trial
Patent: 9926
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 130151
Estimated Expiration: ⤷ Start Trial
Patent: 130183
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 014502684
Estimated Expiration: ⤷ Start Trial
Patent: 015502237
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 52930
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Patent: 90428
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 52930
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Patent: 52422
Estimated Expiration: ⤷ Start Trial
Patent: 13337
Estimated Expiration: ⤷ Start Trial
Patent: 90428
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01500285
Estimated Expiration: ⤷ Start Trial
Patent: 01600316
Patent: E PROCEDIMENTO PER LA PREPARAZIONE DELL'ESTERE 1-METILETILICO DI N-[(2'R)- 2'-DEOSSI-2'-FLUORO-2'-METIL-P-FENIL-5'- URIDILlL]-L-ALANINA
Estimated Expiration: ⤷ Start Trial
Patent: 01700412
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 368
Estimated Expiration: ⤷ Start Trial
Patent: 229
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201500835W
Estimated Expiration: ⤷ Start Trial
Patent: 201702025S
Estimated Expiration: ⤷ Start Trial
Patent: 201702294Q
Estimated Expiration: ⤷ Start Trial
Patent: 201708263S
Estimated Expiration: ⤷ Start Trial
Patent: 6197
Estimated Expiration: ⤷ Start Trial
Patent: 4323
Estimated Expiration: ⤷ Start Trial
Patent: 4324
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 32792
Estimated Expiration: ⤷ Start Trial
Patent: 52930
Estimated Expiration: ⤷ Start Trial
Patent: 09923
Estimated Expiration: ⤷ Start Trial
Patent: 90428
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1108749
Patent: N-[(2'R)-2'-DEOXY-2'-FLUORO-2'-METHYL-P-PHENYL-5'-URIDYLYL]-ALANINE 1 -METHYLETHYL ESTER AND PROCESS FOR ITS PRODUCTION
Estimated Expiration: ⤷ Start Trial
Patent: 1207799
Patent: NUCLEOSIDE PHOSPHORAMIDATES
Estimated Expiration: ⤷ Start Trial
Patent: 1207800
Patent: STEREOSELECTIVE SYNTHESIS OF PHOSPHORUS CONTAINING ACTIVES
Estimated Expiration: ⤷ Start Trial
Patent: 1400249
Patent: NUCLEOSIDE PHOSPHORAMIDATES
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1599183
Estimated Expiration: ⤷ Start Trial
Patent: 1603817
Estimated Expiration: ⤷ Start Trial
Patent: 1715981
Estimated Expiration: ⤷ Start Trial
Patent: 1759369
Estimated Expiration: ⤷ Start Trial
Patent: 120034662
Estimated Expiration: ⤷ Start Trial
Patent: 120138242
Estimated Expiration: ⤷ Start Trial
Patent: 130064064
Estimated Expiration: ⤷ Start Trial
Patent: 140147144
Estimated Expiration: ⤷ Start Trial
Patent: 150043553
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 16466
Estimated Expiration: ⤷ Start Trial
Patent: 51944
Estimated Expiration: ⤷ Start Trial
Patent: 86821
Estimated Expiration: ⤷ Start Trial
Patent: 38350
Estimated Expiration: ⤷ Start Trial
Patent: 44990
Estimated Expiration: ⤷ Start Trial
Patent: 48803
Estimated Expiration: ⤷ Start Trial
Patent: 00773
Estimated Expiration: ⤷ Start Trial
Patent: 27501
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1107341
Patent: Nucleoside phosphoramidates
Estimated Expiration: ⤷ Start Trial
Patent: 1136593
Patent: Nucleoside phosphoramidates
Estimated Expiration: ⤷ Start Trial
Patent: 1136945
Patent: Purine nucleoside phosphoramidate
Estimated Expiration: ⤷ Start Trial
Patent: 1139457
Patent: Stereoselective synthesis of phosphorus containing actives
Estimated Expiration: ⤷ Start Trial
Patent: 1518313
Patent: Nucleoside phosphoramidates
Estimated Expiration: ⤷ Start Trial
Patent: 1704249
Patent: Nucleoside phosphoramidates
Estimated Expiration: ⤷ Start Trial
Patent: 98117
Estimated Expiration: ⤷ Start Trial
Patent: 76352
Estimated Expiration: ⤷ Start Trial
Patent: 83692
Estimated Expiration: ⤷ Start Trial
Patent: 98358
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 2959
Patent: НУКЛЕОЗИДФОСФОРАМІДАТИ
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 310
Patent: SINTESIS ESTEREOSELECTIVA DE ACTIVOS QUE CONTIENEN FOSFORO
Estimated Expiration: ⤷ Start Trial
Patent: 311
Patent: FOSFORAMIDATOS DE NUCLEOSIDOS
Estimated Expiration: ⤷ Start Trial
Patent: 312
Patent: FOSFORAMIDATO DE NUCLEOSIDO DE PURINA
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering SOVALDI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| France | 14C0082 | ⤷ Start Trial | |
| European Patent Office | 2609923 | ⤷ Start Trial | |
| European Patent Office | 2792680 | ⤷ Start Trial | |
| France | 14C0082 | ⤷ Start Trial | |
| Mexico | 2012011324 | ⤷ Start Trial | |
| Taiwan | 201136945 | Purine nucleoside phosphoramidate | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2013066748 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SOVALDI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2203462 | PA2014040 | Lithuania | ⤷ Start Trial | PRODUCT NAME: SOFOSBUVIRUM; REGISTRATION NO/DATE: EU/1/13/894/001 - EU/1/13/894/002 20140116 |
| 2203462 | 14C0082 | France | ⤷ Start Trial | PRODUCT NAME: SOFOSBUVIR; REGISTRATION NO/DATE: EU/1/13/894/001-002 20140117 |
| 2203462 | C300704 | Netherlands | ⤷ Start Trial | PRODUCT NAME: SOVALDI (SOFOSBUVIR); REGISTRATION NO/DATE: EU/1/13/894/001-002 20140117 |
| 2203462 | 826 | Finland | ⤷ Start Trial | |
| 2203462 | 92600 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: SOVALDI (SOFOSBUVIR); AUTHORISATION NUMBER AND DATE: EU/1/13/894(001-002) - SOVALDI - SOFOSBUVIR, 20140117 |
| 2203462 | 67/2014 | Austria | ⤷ Start Trial | PRODUCT NAME: SOFOSBUVIR; REGISTRATION NO/DATE: EU/1/13/894 20140117 |
| 2203462 | C20140035 | Estonia | ⤷ Start Trial | PRODUCT NAME: SOFOSBUVIIR;REG NO/DATE: EU/1/13/894 17.01.2014 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of Sovaldi
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.