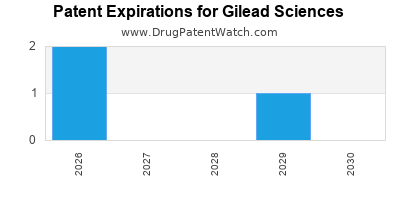
Gilead Sciences Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GILEAD SCIENCES
GILEAD SCIENCES has twenty-seven approved drugs.
There are eighty-one US patents protecting GILEAD SCIENCES drugs.
There are two thousand one hundred and fifty patent family members on GILEAD SCIENCES drugs in sixty-one countries and three hundred and forty supplementary protection certificates in twenty countries.
Summary for Gilead Sciences
| International Patents: | 2150 |
| US Patents: | 81 |
| Tradenames: | 21 |
| Ingredients: | 20 |
| NDAs: | 27 |
| Patent Litigation for Gilead Sciences: | See patent lawsuits for Gilead Sciences |
| PTAB Cases with Gilead Sciences as patent owner: | See PTAB cases with Gilead Sciences as patent owner |
Drugs and US Patents for Gilead Sciences
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-002 | Jan 18, 2012 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ||||
| Gilead Sciences Inc | TYBOST | cobicistat | TABLET;ORAL | 203094-002 | Jun 20, 2025 | DISCN | Yes | No | 10,039,718*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | EPCLUSA | sofosbuvir; velpatasvir | PELLETS;ORAL | 214187-002 | Jun 10, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Gilead Sciences Inc | SUNLENCA | lenacapavir sodium | TABLET;ORAL | 215974-001 | Dec 22, 2022 | RX | Yes | Yes | 11,944,611 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Gilead Sciences Inc | YEZTUGO | lenacapavir sodium | SOLUTION;SUBCUTANEOUS | 220018-001 | Jun 18, 2025 | RX | Yes | Yes | 10,071,985 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Gilead Sciences
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | COMPLERA | emtricitabine; rilpivirine hydrochloride; tenofovir disoproxil fumarate | TABLET;ORAL | 202123-001 | Aug 10, 2011 | 5,977,089*PED | ⤷ Start Trial |
| Gilead Sciences Inc | GENVOYA | cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 207561-001 | Nov 5, 2015 | 7,803,788 | ⤷ Start Trial |
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | 6,939,964*PED | ⤷ Start Trial |
| Gilead Sciences Inc | BIKTARVY | bictegravir sodium; emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 210251-001 | Feb 7, 2018 | 6,642,245 | ⤷ Start Trial |
| Gilead Sciences Inc | ODEFSEY | emtricitabine; rilpivirine hydrochloride; tenofovir alafenamide fumarate | TABLET;ORAL | 208351-001 | Mar 1, 2016 | 7,067,522 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GILEAD SCIENCES drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 600 mg/200 mg/300 mg | ➤ Subscribe | 2008-12-29 |
| ➤ Subscribe | Tablets | 150 mg, 200 mg, and 250 mg | ➤ Subscribe | 2012-05-17 |
| ➤ Subscribe | Tablets | 150 mg, 150 mg, 200 mg, 300 mg | ➤ Subscribe | 2018-10-04 |
| ➤ Subscribe | Tablets | 300 mg | ➤ Subscribe | 2010-01-26 |
| ➤ Subscribe | Tablets | 200 mg/25 mg/300 mg | ➤ Subscribe | 2015-05-20 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2015-12-09 |
International Patents for Gilead Sciences Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Ecuador | SP18033723 | ⤷ Start Trial |
| Japan | 6742403 | ⤷ Start Trial |
| Japan | 6360607 | ⤷ Start Trial |
| Netherlands | 301212 | ⤷ Start Trial |
| Mexico | 2022013596 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Gilead Sciences Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2487166 | 2016C/065 | Belgium | ⤷ Start Trial | PRODUCT NAME: COBICISTAT AND TENOFOVIR ALAFENAMIDE; AUTHORISATION NUMBER AND DATE: EU/1/15/1061 20151123 |
| 3347352 | CA 2022 00052 | Denmark | ⤷ Start Trial | PRODUCT NAME: LENACAPAVIR ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, ISAER NATRIUM-LENACAPAVIR; REG. NO/DATE: EU/1/22/1671 20220819 |
| 2203462 | 92600 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: SOVALDI (SOFOSBUVIR); AUTHORISATION NUMBER AND DATE: EU/1/13/894(001-002) - SOVALDI - SOFOSBUVIR 20140117 |
| 2822954 | SPC/GB18/031 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: BICTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BICTEGRAVIR SODIUM.; REGISTERED: UK EU/1/18/1289/001(NI) 20180625; UK EU/1/18/1289/002(NI) 20180625; UK PLGB 11972/0008 20180625 |
| 1663240 | 1590056-6 | Sweden | ⤷ Start Trial | PRODUCT NAME: A COMBINATION OF RILPIVIRINE, OR A PHARMACEUTICALLY ACCEPTABLE SALT OF RILPIVIRINE, INCLUDING THE HYDROCHLORIDE SALT OF RILPIVIRINE, AND TENOFOVIR DISOPROXIL, IN PARTICULAR TENOFOVIR DISOPROXIL FUMARATE; REG. NO/DATE: EU/1/11/737 20111128 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.