DESCOVY Drug Patent Profile
✉ Email this page to a colleague
When do Descovy patents expire, and what generic alternatives are available?
Descovy is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are two patents protecting this drug and two Paragraph IV challenges.
This drug has fifty-eight patent family members in forty-two countries.
The generic ingredient in DESCOVY is emtricitabine; tenofovir alafenamide fumarate. There are eighteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the emtricitabine; tenofovir alafenamide fumarate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Descovy
A generic version of DESCOVY was approved as emtricitabine; tenofovir alafenamide fumarate by MACLEODS PHARMS LTD on April 7th, 2026.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for DESCOVY?
- What are the global sales for DESCOVY?
- What is Average Wholesale Price for DESCOVY?
Summary for DESCOVY
| International Patents: | 58 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 19 |
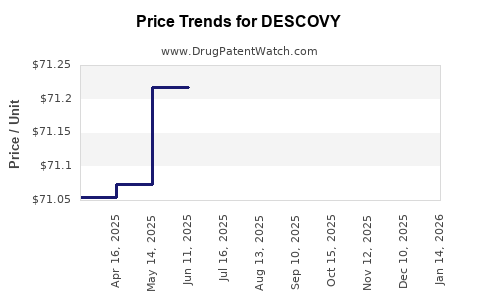
| Drug Prices: | Drug price information for DESCOVY |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DESCOVY |
| What excipients (inactive ingredients) are in DESCOVY? | DESCOVY excipients list |
| DailyMed Link: | DESCOVY at DailyMed |
Paragraph IV (Patent) Challenges for DESCOVY
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| DESCOVY | Tablets | emtricitabine; tenofovir alafenamide fumarate | 120 mg/15 mg | 208215 | 1 | 2022-10-31 |
| DESCOVY | Tablets | emtricitabine; tenofovir alafenamide fumarate | 200 mg/25 mg | 208215 | 6 | 2019-11-05 |
US Patents and Regulatory Information for DESCOVY
DESCOVY is protected by two US patents and two FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-002 | Jan 7, 2022 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-002 | Jan 7, 2022 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-002 | Jan 7, 2022 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for DESCOVY
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for DESCOVY
When does loss-of-exclusivity occur for DESCOVY?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 39
Estimated Expiration: ⤷ Start Trial
Argentina
Patent: 7546
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 12296622
Estimated Expiration: ⤷ Start Trial
Patent: 14271320
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2014003420
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 45553
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 14000370
Estimated Expiration: ⤷ Start Trial
China
Patent: 3732594
Estimated Expiration: ⤷ Start Trial
Patent: 0343135
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 80063
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 140072
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0161696
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18385
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 14013206
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 7768
Estimated Expiration: ⤷ Start Trial
Patent: 1490208
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Patent: 70088
Estimated Expiration: ⤷ Start Trial
Patent: 31832
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 99026
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 31253
Estimated Expiration: ⤷ Start Trial
India
Patent: 12DEN2014
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 0949
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 51275
Estimated Expiration: ⤷ Start Trial
Patent: 56537
Estimated Expiration: ⤷ Start Trial
Patent: 80162
Estimated Expiration: ⤷ Start Trial
Patent: 14528924
Estimated Expiration: ⤷ Start Trial
Patent: 15038149
Estimated Expiration: ⤷ Start Trial
Patent: 16169228
Estimated Expiration: ⤷ Start Trial
Patent: 18065870
Estimated Expiration: ⤷ Start Trial
Patent: 20040972
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 6627
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA. (TENOFOVIR ALAFENAMIDE HEMIFUMARATE.)
Estimated Expiration: ⤷ Start Trial
Patent: 14001549
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA. (TENOFOVIR ALAFENAMIDE HEMIFUMARATE.)
Estimated Expiration: ⤷ Start Trial
Moldova, Republic of
Patent: 08
Estimated Expiration: ⤷ Start Trial
Patent: 140011
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 612
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 350
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0421
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 141328
Patent: TENOFOVIR ALAFENAMIDA HEMIFUMARATO
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 014500349
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 44810
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01600476
Patent: TENOFOVIR ALAFENAMMIDE EMIFUMARATO
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 353
Patent: TENOFOVIR ALAFENAMID HEMIFUMARAT (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 14011548
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 44810
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1400582
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1612642
Estimated Expiration: ⤷ Start Trial
Patent: 140054068
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 08871
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1321396
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Start Trial
Patent: 16499
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 5311
Patent: ГЕМІФУМАРАТ ТЕНОФОВІРУ АЛАФЕНАМІДУ (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 262
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA, SUS COMPOSICIONES, MÉTODO DE PREPARACIÓN, MÉTODOS PARA TRATAR INFECCIONES VIRALES Y SU USO PARA PREPARAR MEDI CAMENTOS
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering DESCOVY around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 1065065 | ⤷ Start Trial | |
| Japan | 3844978 | ⤷ Start Trial | |
| World Intellectual Property Organization (WIPO) | 9111186 | ⤷ Start Trial | |
| Spain | 2627903 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DESCOVY
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0513200 | 7/2004 | Austria | ⤷ Start Trial | PRODUCT NAME: EMTRICITABIN; NAT. REGISTRATION NO/DATE: EU/1/03/261/001- EU/1/03/261/003 20031024; FIRST REGISTRATION: EU EU/1/03/261/003 |
| 1301519 | 1690012-8 | Sweden | ⤷ Start Trial | PRODUCT NAME: TENOFOVIR ALAFENAMIDE OR A SALT OR SOLVATE THEREOF, IN PARTICULAR TENOFOVIR ALAFENAMIDE FUMARATE; REG. NO/DATE: EU/1/15/1061/001-002 20151123 |
| 1663240 | 2015/053 | Ireland | ⤷ Start Trial | PRODUCT NAME: A COMBINATION OF RILPIVIRINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING THE HYDROCHLORIDE SALT, TENOFOVIR, IN PARTICULAR TENOFOVIR DISOPROXIL FUMARATE, AND EMTRICITABINE; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 1663240 | 1590055-8 | Sweden | ⤷ Start Trial | PRODUCT NAME: A COMBINATION OF RILPIVIRINE OR A PHARMACEUTICALLYACCEPTABLE SALT OF RILPIVIRINE, INICLUDING THE HYDROCHLORIDE SALT OF RILPIVIRINE, AND EMTRICITABINE; REG. NO/DATE: EU/1/11/737 20111128 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
DESCOVY: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.