Introduction: The Paragraph IV Filing as Corporate Weapon
A Paragraph IV certification is the most powerful offensive instrument in the generic pharmaceutical industry’s arsenal. It is also, without qualification, the most dangerous. The filing of an Abbreviated New Drug Application (ANDA) with a Paragraph IV certification is a statutory declaration that a brand-name drug’s patents are invalid, unenforceable, or will not be infringed by the proposed generic product. That declaration triggers a cascade of legal, regulatory, and commercial consequences that can generate hundreds of millions of dollars in profit or expose a company to catastrophic, bet-the-business litigation risk.
The 180-day exclusivity period granted to the first successful Paragraph IV filer is the mechanism that makes this risk economically rational. Operating as the sole generic entrant against a brand in a duopoly market structure, the first filer captures premium pricing before the market commoditizes. That window commonly represents 60% to 80% of a generic product’s total lifetime profit. For drugs with multi-billion-dollar brand revenues, the 180-day prize can exceed $500 million in net present value.
The legislative architecture that created this system, the Drug Price Competition and Patent Term Restoration Act of 1984, known universally as the Hatch-Waxman Act, was a deliberate compromise. Congress attempted to balance two objectives that are structurally in tension: sustaining the incentive for branded pharmaceutical companies to fund the $2-3 billion average cost of bringing a new drug to market, and accelerating the availability of low-cost generic alternatives once that investment has been recouped. Generic drugs now account for over 90% of all U.S. prescriptions filled, up from 19% in 1984. The Paragraph IV mechanism is the engine that drives that access.
But the reward structure is deliberately calibrated to attract only the most sophisticated and well-capitalized challengers. The filing of a Paragraph IV certification is deemed a statutory act of infringement, giving the patent holder the right to sue immediately. That lawsuit automatically imposes a 30-month stay on FDA approval. Litigation costs for high-value ANDA cases routinely exceed $5 million through trial and appeal. An at-risk launch ahead of a final court ruling exposes the generic company to potential treble damages if infringement is later found. The Hatch-Waxman framework is not designed for the faint of heart or the poorly prepared.
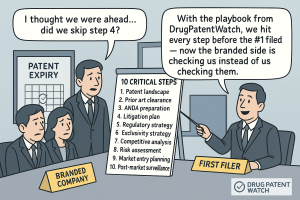
This guide provides a technically rigorous, 10-step operational framework for IP teams, portfolio managers, and R&D leads responsible for executing a winning Paragraph IV strategy. It covers product selection through commercial analysis, forensic patent landscape deconstruction, Freedom-to-Operate quantification, formulation and bioequivalence strategy, dual-track legal offense, cross-functional team architecture, pre-litigation preparation, notice letter tactics, and final ANDA dossier integrity review. Each section includes IP valuation context, investment strategy guidance, and specific data points that separate actionable intelligence from generic commentary.
Key Takeaways: The Big Picture
- The 180-day exclusivity period is the single most valuable regulatory asset in generic pharmaceuticals. Its financial value often exceeds the total development cost of the ANDA by a factor of 10 to 50 times, depending on the brand’s annual revenue.
- The Orange Book is both a map and a strategic minefield. The FDA’s ministerial role in accepting patent listings without independent legal review creates systematic opportunities for abuse by brand manufacturers and systematic leverage for aggressive generic challengers.
- The IPR process at the PTAB has permanently shifted the balance of power in Hatch-Waxman litigation. It gives challengers a faster, cheaper, and technically expert forum with a lower burden of proof than district court.
- Paragraph IV strategy is an organizational problem as much as a legal or scientific one. The companies that win are those that have built genuine cross-functional integration between their legal, R&D, regulatory, and commercial functions.
- The notice letter is a negotiation instrument. Its quality directly determines the brand manufacturer’s litigation risk calculus and therefore the probability and terms of an early settlement.
Step 1: Strategic Product Selection and Commercial Opportunity Analysis
Why the Target Choice Determines Everything
The most consequential decision in a Paragraph IV campaign is made before a single patent is read or a formulation is developed. Every subsequent investment of capital, time, and organizational capacity flows from the initial product selection. A suboptimal target selected through inadequate commercial analysis cannot be rescued by brilliant legal strategy or superior formulation science. The question ‘Is this product worth fighting for?’ must be answered rigorously, quantitatively, and honestly before the project consumes resources.
Sophisticated product selection requires a multi-dimensional commercial analysis that goes well beyond identifying drugs with large brand revenues. The analysis must model competitive intensity, price erosion dynamics, barrier profiles, and the brand manufacturer’s likely strategic response with the same rigor applied to the legal and scientific questions.
IP Valuation as a Core Selection Criterion
Before committing to a Paragraph IV target, the IP team must conduct a preliminary IP valuation of the brand asset being challenged. This is distinct from the detailed patent landscape analysis in Step 2. At the selection stage, the question is whether the brand’s IP portfolio, even if successfully challenged, leaves open a commercially viable and defensible market window for the generic entrant.
A brand drug’s IP value is a composite of several stacking protections. The primary compound patent, typically expiring 20 years from filing date and often extended by Patent Term Extension (PTE) under 35 U.S.C. § 156 by up to five years to compensate for regulatory delay, establishes the baseline exclusivity period. Layered on top are secondary patents covering specific salt forms, polymorphs, formulation methods, manufacturing processes, and method-of-use claims filed sequentially across the drug’s commercial life. This ‘patent thicket’ architecture is the primary mechanism through which branded companies extend effective market exclusivity well beyond the primary compound patent’s life.
The IP valuation process must map this thicket structure and estimate the realistic effective exclusivity duration, which is often the resolution date of litigation on the last credibly enforceable secondary patent rather than the nominal expiration date of the first patent. Historical data from DrugPatentWatch and IQVIA consistently shows that the actual date of generic market entry lags the primary patent expiration by two to five years on average for actively defended brands. For investment strategy purposes, this means the market entry window is frequently narrower than a naive reading of Orange Book expiration dates suggests.
Market Sizing, Price Erosion Modeling, and Competitive Entry Forecasting
The financial model for any Paragraph IV target must be built around three scenarios: sole generic entry during 180-day exclusivity, entry alongside two competitors, and entry into a fully commoditized market. These scenarios produce dramatically different economics and require distinct strategic assumptions.
With a single generic competitor, the market price for the generic typically settles 30-39% below the brand’s pre-expiry WAC. Add a second generic entrant and the price falls approximately 54%. With three to five entrants, prices erode 60-70% or more. In highly contested therapeutic categories with many ANDA filers, price erosion can reach 85% or greater within 24 months of initial generic entry. The 180-day exclusivity period is structurally designed to capture value during the window before this commoditization occurs. That window is where the economic case for a Paragraph IV challenge lives or dies.
The projection of competitive entry requires tracking current ANDA filing activity against the target, monitoring Paragraph IV certifications already in progress through FDA public databases, and modeling the patent challenge difficulty for competitors relative to the first filer. A target with five active Paragraph IV filers already competing for first-filer status is commercially different from a target where the IP complexity has deterred all but the most sophisticated challengers.
The Sweet Spot: High Barrier, Moderate Scale
The counterintuitive strategic insight embedded in successful Paragraph IV programs is that the optimal target is rarely the largest market. Blockbuster drugs with $5 billion or more in annual brand revenue consistently attract swarms of generic challengers, produce rapid commoditization after any entry, and generate the most aggressive and expensive brand defense campaigns. Litigation costs on these assets routinely exceed $10 million per defendant company through trial.
The ‘sweet spot’ in product selection is the moderate-scale drug, typically $100 million to $400 million in annual brand revenue, protected by a complex patent portfolio that deters less sophisticated challengers. A market of $150 million annual revenue with limited competition post-exclusivity, accessed through a Paragraph IV challenge that only two or three companies have the capability to execute, produces superior risk-adjusted returns compared to competing against 20 filers for a share of a $3 billion market.
The barriers to entry that create this favorable competitive dynamic include sterile injectable manufacturing requirements, complex formulation chemistry such as lipid nanoparticles or modified release systems, expensive and methodologically complex bioequivalence studies, and small patient populations that make the per-unit economics unattractive to low-cost generic manufacturers. Each of these barriers is a deterrent to undercapitalized competitors and a competitive moat for companies with the scientific and financial depth to clear them.
FDA Data Assets for Opportunity Identification
The FDA publishes two critical data sources that function as a forward-looking opportunity registry for generic drug business development. The ‘List of Off-Patent, Off-Exclusivity Drugs without an Approved Generic’ is updated biannually and identifies drug products where no generic has yet been approved despite the expiration of both patent protection and regulatory exclusivity. Part I covers products with apparently straightforward ANDA pathways. Part II flags products with potential scientific or regulatory complexity, which is a signal that barriers to entry are suppressing competition and therefore that a capable generic entrant may face a favorable market structure.
The FDA’s Orange Book itself, accessed programmatically through DrugPatentWatch’s integrated database platform, allows business development teams to screen the full landscape of expiring protections by therapeutic category, dosage form, and patent expiry timing. The most valuable analysis combines this patent expiry data with active ANDA tracking to identify products where expiry is imminent, no generic has been approved, and few or no Paragraph IV challenges are currently pending.
Investment Strategy Note
For institutional investors and portfolio managers tracking generic pharmaceutical companies, product selection quality is a leading indicator of future revenue. The number of first-filer-eligible Paragraph IV challenges in a company’s ANDA pipeline, weighted by the brand revenue of the target drugs and discounted for litigation risk, is a more predictive metric than total ANDA count. Companies with a concentration of first-filer positions in moderate-complexity, limited-competition targets consistently demonstrate superior IP monetization relative to those chasing the most-contested blockbusters.
Key Takeaways: Step 1
The initial product selection process should function as a formal investment gating mechanism, not an informal business development exercise. Quantitative models for price erosion, competitive entry probability, IP valuation, and litigation cost must all clear defined thresholds before a project receives development funding. Prioritize targets where complexity deters competitors more than it deters you. The FDA’s off-patent, no-generic list is an underutilized systematic source of low-competition opportunities.
Step 2: Comprehensive Patent Landscape Analysis and Orange Book Deconstruction
The Orange Book as Both Map and Minefield
The Orange Book, formally ‘Approved Drug Products with Therapeutic Equivalence Evaluations,’ is the statutory starting point for every Paragraph IV analysis. Every U.S. drug patent that the NDA holder submits as covering the approved drug product or method of use is listed here, along with its expiration date, any Patent Term Extensions, and use codes for method-of-use patents. Every ANDA applicant must certify against every listed patent with a Paragraph I (patent expired), Paragraph II (will not expire before approval), Paragraph III (waiting for expiry), or Paragraph IV (invalid or not infringed) certification.
What makes the Orange Book strategically complex is a structural feature of its administration that is frequently misunderstood even within the industry. The FDA operates in a purely ministerial capacity with respect to patent submissions from NDA holders. The agency lists what it receives without independently evaluating whether the submitted patents actually claim the approved drug product or any approved method of use. This posture, explicitly adopted because the FDA views patent law as beyond its regulatory expertise, creates a well-documented opportunity for anticompetitive manipulation.
Brand manufacturers have listed patents in the Orange Book that, on careful analysis, do not cover the approved product. These listings impose real costs on generic challengers regardless of their legal merit, because the mere act of listing a patent forces ANDA applicants to certify against it and subjects them to the possibility of a lawsuit and automatic 30-month stay. A brand’s willingness to file even a legally weak infringement suit within the 45-day window, knowing that the stay alone is worth hundreds of millions in protected revenue, creates a systematic incentive to list dubious patents.
This dynamic has attracted increasing regulatory scrutiny. The FTC issued a formal policy statement in September 2023 explicitly warning that improper Orange Book patent listings constitute unfair methods of competition under Section 5 of the FTC Act and that the Commission would pursue enforcement action. Concurrently, the Orange Book Transparency Act of 2020 directed the FDA to study its listing practices and report to Congress. Several prominent pharmaceutical companies, including AstraZeneca for its Symbicort inhaler patents and Amneal, have been the subjects of FTC challenges to specific Orange Book listings in recent years. These regulatory developments change the strategic calculus for generic challengers: a questionable listing is no longer merely a legal hurdle to clear in court. It is now also potential leverage for an FTC complaint, a formal patent listing dispute with the FDA, or an antitrust counterclaim in the ensuing Hatch-Waxman litigation.
Anatomy of an Orange Book Entry: What the Data Actually Tells You
Each Orange Book entry provides the NDA number, the brand product’s active ingredient, dosage form, route of administration, strength, and applicant name. Patent entries include the U.S. patent number, expiration date including any PTE, and a flag indicating whether the patent claims the drug substance, drug product, or a specific method of use.
Method-of-use patents carry ‘use codes,’ which are brief narrative descriptions of the patented indication submitted by the NDA holder. These use codes are critical for designing skinny label carve-out strategies (discussed in Step 5), but they are also opaque and variable in their precision. Some use codes are drafted narrowly, limiting the patent’s scope on the face of the Orange Book. Others are deliberately broad and vague, extending the apparent scope of the patent’s protection. Parsing use codes carefully, and cross-referencing them against the actual claim language of the underlying patent, is a distinct analytical step that cannot be skipped.
Patent Term Extensions awarded under 35 U.S.C. § 156 can extend patent protection by up to five years for time lost during the FDA regulatory review process, subject to a maximum of 14 years of post-approval protection. A patent expiring in 2027 with a two-year PTE does not effectively expire until 2029. Drug programs projected against nominal expiration dates without accounting for PTEs consistently underestimate the real IP protection horizon.
Beyond the Orange Book: Non-Listable Patent Threats
The FDA’s patent listing regulations explicitly exclude certain categories of patents from Orange Book eligibility. Process patents claiming a method of manufacturing the API or drug product cannot be listed. Patents covering packaging, metabolites, or synthetic intermediates are similarly ineligible. These exclusions mean that a complete Orange Book clearance does not establish freedom to operate.
A generic manufacturer that successfully challenges every listed Orange Book patent can still face a blocking infringement suit based on an unlisted process patent covering a critical step in the API synthesis route, or an unlisted metabolite patent that catches the active metabolite of the generic’s API. Several major generic launches have been delayed or disrupted by precisely this mechanism. Process patents for API synthesis are particularly common in small-molecule drugs where the innovator developed proprietary chemistry to achieve the target molecule. Identifying them requires a broader patent search that looks beyond the brand product itself to any patent held by the NDA holder or its API suppliers that relates to the underlying chemical or manufacturing technology.
IP Valuation of the Brand Asset
A rigorous patent landscape analysis must produce more than a list of patents to certify against. It must yield an IP valuation: a structured assessment of the brand drug’s total intellectual property portfolio, its commercial value to the innovator, and the probability-weighted timeline to generic entry accounting for challenge success rates.
The IP valuation framework for a typical small-molecule brand drug maps the patent thicket across four layers. The primary compound patent, typically expiring 7-12 years from approval date after PTE, is the foundational asset. Salt or polymorph patents, usually filed 2-5 years after the compound patent and claiming specific solid-state forms that may offer superior manufacturing or stability characteristics, constitute the first layer of secondary IP. Formulation patents, covering the specific excipient composition, release mechanism, or delivery technology of the approved product, are often the most commercially significant secondary patents because they directly protect the product that patients use. Method-of-use patents for new indications filed as the drug’s clinical profile expands provide incremental coverage, often extending effective exclusivity by three to seven years beyond the compound patent expiry.
The aggregate value of this IP stack to the brand manufacturer is measured in the net present value of the revenue it protects. A drug with $2 billion in annual revenues, where the IP stack delays generic entry by three years beyond the primary compound patent expiry, has an IP value attributable to those secondary patents of approximately $5-6 billion at standard pharmaceutical NPV discount rates. This figure quantifies what the brand manufacturer will spend defending those patents and what the first-filer challenger stands to gain by defeating them.
Key Takeaways: Step 2
The Orange Book is a legal tool administered by a non-legal agency. Treat every listing with analytical skepticism. Non-listable process and metabolite patents require a separate, broader search beyond Orange Book scope. IP valuation analysis at the landscape stage, quantifying the NPV of the brand’s secondary patent portfolio, tells you both what you stand to gain and what the innovator will spend to stop you.
Step 3: Freedom-to-Operate Analysis and Litigation Risk Quantification
What an FTO Actually Is
A Freedom-to-Operate analysis answers one question with precision: can the specific proposed product, manufactured by the specific proposed process and sold in the specific target jurisdictions, be brought to market without infringing any valid, enforceable patent claim held by a third party? It is not a general technology review, not a patentability assessment, and not an Orange Book certification exercise. It is a targeted infringement and validity analysis bounded by a precisely defined product and process description.
The distinction between FTO and patentability is worth stating clearly because the terms are often conflated. Patentability asks whether an invention is novel and non-obvious enough to merit patent protection. For a generic pharmaceutical product, this question is almost entirely irrelevant, because a generic is by definition a copy of an existing product and is not patentable. The FTO asks whether the specific product infringes someone else’s rights, which is entirely independent of whether the generic product could itself receive patent protection.
The integrity of an FTO depends completely on the precision of its scope definition. The analysis must be anchored to a documented description of the final product: the specific API, its physical form (salt, polymorph, amorphous), the complete formulation with all excipients and their concentrations, the step-by-step manufacturing process, and the complete list of intended uses that will appear on the product label. An FTO conducted against a preliminary formulation that is later modified due to stability findings is only as current as its last update. Any material change to the product triggers an FTO update requirement.
Structuring the FTO: Claim-by-Claim Risk Mapping
The analytical core of an FTO is a claim-by-claim infringement analysis for each patent identified in the landscape search. For every independent claim and every commercially significant dependent claim of every relevant patent, the analysis asks: does the proposed product or process meet each and every limitation of this claim, either literally or under the doctrine of equivalents?
Literal infringement requires an exact correspondence between the claim language and the accused product’s features. Infringement under the doctrine of equivalents applies where an element of the product, while not literally described by the claim language, performs substantially the same function, in substantially the same way, to achieve substantially the same result. The doctrine of equivalents analysis is highly fact-specific and varies significantly based on the patent’s prosecution history. Statements made by the applicant to the patent examiner during prosecution that narrow the scope of the claims in order to obtain allowance can create ‘prosecution history estoppel,’ which forecloses reliance on the doctrine of equivalents for the surrendered territory.
The output of the claim-by-claim analysis is a risk-tiered patent map. High-risk patents are those where the proposed product appears to literally infringe one or more claims, leaving no credible design-around path and making invalidity challenge the only viable approach. Moderate-risk patents are those where infringement depends on contested claim construction or where a plausible design-around exists. Low-risk patents are those with narrow or clearly inapplicable claims that the proposed product definitively avoids.
Quantifying Litigation Risk: The AIPLA Financial Model
The FTO risk map must be converted into a financial model to support the go/no-go business decision. The American Intellectual Property Law Association’s biennial Report of the Economic Survey provides the industry-standard benchmarks for Hatch-Waxman litigation costs by stakes level. The figures below, drawn from AIPLA survey data, represent median all-in legal fees and are routinely exceeded in the most aggressively litigated blockbuster patent disputes.
| Amount at Risk | Cost Through Discovery and Claim Construction | Cost Through Trial and Appeal |
|---|---|---|
| Below $1 million | $350,000 | $650,000 |
| $1 million to $10 million | $1,000,000 | $1,800,000 |
| $10 million to $25 million | $2,000,000 | $3,500,000 |
| Above $25 million | $3,000,000+ | $5,000,000+ |
For a drug with $1 billion in annual brand revenue, the ‘amount at risk’ for both sides in Hatch-Waxman litigation effectively starts at the upper tier. The brand is defending patent exclusivity worth billions. The generic challenger is pursuing a 180-day exclusivity prize worth hundreds of millions. Litigation costs at this level, while substantial, represent a single-digit percentage of the financial stakes, which is why both sides typically commit to full litigation rather than early capitulation.
The FTO financial model should apply these cost benchmarks to the risk-tiered patents identified in the landscape analysis, probability-weight them by the analyst’s estimated success rate for invalidity or non-infringement arguments, and integrate them into the overall project P&L model. A first-filer exclusivity prize of $300 million with $8 million in projected litigation costs and a 60% probability of winning represents a risk-adjusted expected value of $172 million. The same prize with a 25% probability of winning and $15 million in litigation costs has an expected value of $60 million. These are fundamentally different investment propositions.
The FTO as a Living Document
The FTO is not a report commissioned once and filed. It is an active risk management instrument that must be updated at defined project milestones: when the formulation is finalized, when the manufacturing process is locked, when a new patent is issued to the brand manufacturer that could cover the generic product, and when the litigation status of any relevant patent changes through district court rulings or PTAB decisions. A dynamic FTO maintained on a rolling basis provides the continuous risk signal that allows project leaders to detect emerging threats and redirect the scientific program before those threats become blocking events.
Investment Strategy Note
For portfolio managers evaluating generic pharmaceutical companies, the robustness of a company’s FTO process is a proxy for the quality of its risk management infrastructure. Companies that conduct FTO analysis iteratively and integrate legal feedback into R&D decision-making demonstrate the cross-functional maturity that predicts successful Paragraph IV execution. Companies that treat FTO as a one-time diligence step typically experience the cost of that shortcut at the litigation stage.
Key Takeaways: Step 3
Define the FTO scope precisely against a documented product and process description. Apply AIPLA litigation cost benchmarks to build a financial model that quantifies risk in dollar terms rather than qualitative categories. Treat the FTO as a living document updated at each development milestone. The ratio of FTO investment to potential litigation cost is consistently favorable, making rigorous pre-filing analysis the highest-return activity in the Paragraph IV development process.
Step 4: Formulation and Bioequivalence Strategy
The Formulation Enigma: Science, Law, and Strategy in Parallel
Generic drug formulation is a constrained optimization problem. The R&D team must develop a product that replicates the innovator’s RLD performance in vivo without access to the proprietary formula, while simultaneously avoiding infringement of any valid formulation patent, and while demonstrating that equivalence through clinical studies designed to the FDA’s current evidentiary standards. None of these constraints can be addressed sequentially. They must be solved in parallel, with constant communication between the laboratory, the legal team, and the regulatory affairs function.
The innovator’s formulation is a trade secret. What is publicly available is the product’s observed clinical pharmacokinetic profile, its dissolution behavior under standardized conditions, and its approved labeling. From these public data points, the formulation team must reverse-engineer the critical performance attributes and then achieve them through a novel combination of excipients and manufacturing methods that is both scientifically sound and legally non-infringing.
Excipients, though pharmacologically inactive, exert profound influence on drug performance. The choice of binder, filler, disintegrant, coating polymer, and plasticizer affects tablet hardness, dissolution rate, drug release kinetics, and physical stability under stress conditions. For modified-release systems, the release-controlling polymer matrix or membrane is the single most critical determinant of the drug’s in vivo absorption profile, and it is precisely the component most likely to be the subject of a formulation patent. Choosing excipients that achieve the target release profile while avoiding patented polymer systems is the central formulation challenge in most Paragraph IV development programs.
Polymorph and Salt Form Strategy
Many small-molecule APIs exist in multiple solid-state forms, including different crystalline polymorphs and various salt forms. Innovators routinely patent specific polymorphs or salt forms that offer commercial advantages in stability, solubility, or manufacturability. These patents, if valid, can block generic entry even after the compound patent expires.
The generic formulation strategy must therefore include systematic polymorph and salt form screening to identify whether the proposed API form is covered by any valid patent and, if so, whether a commercially viable alternative form exists that is outside the patent claims. This screening work is both scientific and legal. The scientific output is a characterization of the alternative form’s physical properties and suitability for the target formulation. The legal output is a claim analysis confirming that the alternative form falls outside the scope of the brand’s IP. Both outputs feed directly into the FTO and the non-infringement argument.
The Chemistry, Manufacturing, and Controls (CMC) Dossier
The ANDA’s CMC section is the scientific backbone of the entire application. It must comprehensively document the drug substance (API), the drug product formulation, the manufacturing process for both, and the analytical methods and specifications used to ensure consistent quality. The FDA’s review of the CMC section through its Question-Based Review (QbR) approach focuses on identifying and controlling the critical quality attributes (CQAs) and critical process parameters (CPPs) that determine product performance.
The CMC narrative must be internally consistent and must accurately describe the exact product used in the bioequivalence study. Any discrepancy between the CMC-described formulation and the clinical study formulation is a substantive deficiency that can delay approval, trigger a Complete Response Letter, or, in the context of litigation, provide the brand’s counsel with material to attack the scientific validity of the bioequivalence data.
Bioequivalence Study Design: Meeting the FDA’s Evidentiary Standard
Bioequivalence is demonstrated by showing that the 90% confidence interval for the test-to-reference ratio of the key pharmacokinetic parameters, Cmax (peak concentration) and AUC (total exposure), falls within the 80-125% acceptance range. This standard, based on the two-one-sided tests (TOST) procedure, is designed to ensure that any difference in drug exposure between the generic and the brand is clinically insignificant.
The FDA’s Office of Generic Drugs publishes Product-Specific Guidances (PSGs) for individual drugs, providing detailed methodological recommendations on study design, subject population, fed or fasted conditions, sampling schedules, and analytical approaches. PSGs are updated periodically and must be checked for each product to ensure compliance with the most current requirements. Following an outdated PSG is a common source of preventable study failure.
A regulation change implemented in 2009 requires ANDA applicants to submit data from all bioequivalence studies conducted on the final formulation, including failed studies. This ‘all studies’ requirement prevents cherry-picking of favorable results and means that a failed BE study is not simply discarded. It must be disclosed, and if the failure was due to the proposed formulation rather than study design factors, it raises substantive questions about the formulation’s suitability that must be addressed before the ANDA can be approved. The practical implication is that formulation optimization must be substantially complete before clinical BE studies are initiated, because the cost of a failed study includes not just the study itself but also the regulatory burden of explaining and resolving the failure.
Complex Products: Heightened BE Requirements
For products classified as complex by the FDA, including complex drug substances (peptides, polymeric molecules), complex formulations (liposomes, microspheres), complex routes of delivery (inhalation products, topicals), or complex drug-device combinations, simple PK-based bioequivalence is often insufficient. These products require additional evidence of equivalence that may include device characterization, in vitro aerosol studies, formulation equivalence testing, and in some cases clinical endpoint studies that are far more expensive than standard PK bioequivalence studies.
The development of a biosimilar, which is the biologics analogue of a Paragraph IV generic challenge, requires a comprehensive comparability exercise that includes extensive analytical characterization, functional assays, PK/PD studies in humans, and, in many cases, clinical efficacy trials. The regulatory pathway for interchangeability designation, which allows pharmacist-level substitution for the reference biologic, requires additional switching studies demonstrating that alternating between the biosimilar and the reference product does not produce safety or efficacy concerns. The IP strategy for biosimilar development is governed by the Biologics Price Competition and Innovation Act (BPCIA) and involves the ‘patent dance,’ a formal information exchange process that identifies which of the reference product sponsor’s patents are relevant, followed by either litigation or negotiated standstill. This is a distinct but structurally analogous process to Hatch-Waxman Paragraph IV litigation.
Key Takeaways: Step 4
Formulation development, IP navigation, and regulatory strategy must run in parallel, not sequentially. Failed bioequivalence studies must be disclosed. For complex products, bioequivalence standards are substantially more demanding and substantially more expensive. The polymorph and salt form screening program is both a scientific and legal exercise and must produce a documented record of the design-around rationale.
Step 5: Legal Strategy I: The Non-Infringement Pathway
Designing Around: The Philosophy and Practice
The non-infringement prong of a Paragraph IV certification is the assertion that the generic product, as specifically designed and manufactured, falls outside the legally protected scope of the innovator’s patents. This argument requires both a product that has been intentionally designed to avoid infringing claims and a legal team capable of demonstrating that design-around intention with precision and persuasion.
Claim construction is the threshold issue in every infringement analysis. Patent claims are interpreted by courts using intrinsic evidence, the patent specification and prosecution history, and extrinsic evidence including expert testimony, dictionaries, and technical treatises. The specific legal meaning assigned to key claim terms by the court can dramatically narrow or expand a patent’s effective scope, and the entire non-infringement case turns on those interpretations. The Markman hearing, where the court determines the meaning of disputed claim terms, is often the decisive event in ANDA litigation, occurring before trial and substantially shaping the parties’ relative positions.
Design-around strategies in pharmaceutical Paragraph IV cases take several forms. Formulation design-around is the most common, developing a non-infringing excipient system that achieves the same bioequivalent release profile through a different technical mechanism. Polymorph or salt form design-around replaces the innovator’s patented solid-state form with an alternative outside the claim scope, as discussed in Step 4. Process design-around develops an alternative synthetic route to the API that avoids unlisted process patents covering the innovator’s manufacturing chemistry.
The Skinny Label Strategy: Mechanics and Risk
The skinny label is a statutory carve-out mechanism codified in the Hatch-Waxman framework that allows an ANDA applicant to omit from its proposed labeling any indication or use that is covered by a valid method-of-use patent listed in the Orange Book. The generic product can be approved for and marketed in the non-patented indications, with the patented indication simply absent from the label.
The mechanics of a skinny label filing require careful cross-reference between the Orange Book use codes and the actual claim language of the underlying method-of-use patent. Not every use code perfectly tracks the patent claims. A use code can be broader than the patent claims, meaning that carving out the use code text may result in a label that is more restrictive than strictly necessary. A use code can also be narrower than the claims, creating risk that the label retains text that could support an induced infringement theory even after the carve-out.
The legal risk of the skinny label strategy is embodied in the GSK v. Teva line of cases, which attracted significant attention beginning with a 2020 Federal Circuit panel decision that found Teva liable for induced infringement of GSK’s carvedilol heart failure method-of-use patent despite having carved out the patented indication from its label. The court’s analysis focused on Teva’s marketing materials and communications that referenced the generic’s use for heart failure, finding evidence that Teva actively encouraged infringement even though its label did not include the patented indication. The Federal Circuit later vacated that decision and remanded for reconsideration, but the case established that skinny labels carry meaningful induced infringement risk when the generic manufacturer’s conduct, beyond the label text itself, includes promotional activities referencing the patented use.
The practical implication for any company pursuing a skinny label strategy is that the carve-out must be comprehensive and consistent, extending not just to the label text but to all marketing and promotional materials, medical affairs communications, and training programs. The label design and all commercial communications must be reviewed by patent counsel before launch.
Documentation as Trial Evidence
The legal value of a well-documented design-around process is substantial and frequently underestimated. Contemporaneous records in laboratory notebooks, R&D project reports, and internal memoranda that show the team identified specific patent claims, understood their scope, and deliberately developed a product to fall outside those claims constitute affirmative evidence of non-infringing intent. This evidence can be highly persuasive to a district court judge evaluating competing expert interpretations of complex formulation chemistry.
The absence of this documentation is equally telling. A generic manufacturer that cannot produce records showing its design-around rationale presents a weaker non-infringement case and exposes itself to the inference that any technical distinctions between its product and the patented claims are accidental rather than deliberate. Design-around documentation is the responsibility of the R&D team from the first day of the project, not a retrospective exercise conducted by the legal team in preparation for trial.
Key Takeaways: Step 5
Non-infringement is a design philosophy, not a legal argument constructed after the product is finalized. Claim construction at the Markman hearing is often the pivotal event in ANDA litigation. Skinny labels carry post-GSK v. Teva induced infringement risk that extends beyond the label text to all commercial conduct. Design-around documentation created contemporaneously is more persuasive than expert testimony reconstructed at trial.
Step 6: Legal Strategy II: Patent Invalidity Challenge
The Grounds for Attack
When designing around a patent is not scientifically feasible or commercially viable, the only path forward is a direct invalidity challenge. A patent claim is invalid if the USPTO should not have granted it in the first place. The Paragraph IV certification declares that the challenger intends to establish this invalidity, whether through lack of novelty, obviousness, insufficient disclosure, or other statutory grounds. Invalidity, unlike non-infringement, is an affirmative case that the challenger bears the burden of proving.
Lack of novelty, or anticipation under 35 U.S.C. § 102, requires showing that a single prior art reference discloses every element of the claimed invention exactly as claimed. The prior art reference need not use the same words as the claims, but every limitation must be present, either explicitly or through the doctrine of inherent anticipation. Inherent anticipation is particularly potent for pharmaceutical patents where a claimed property or result is the inevitable consequence of following a prior art teaching, even if the prior art author was unaware of that property. A reference that discloses a compound for one therapeutic purpose can anticipate a later patent claiming the same compound for a different purpose if the new use is the inherent pharmacological result of administering the same compound.
Obviousness under 35 U.S.C. § 103 is the most frequently litigated and most frequently successful invalidity ground in pharmaceutical patent challenges. The U.S. Supreme Court’s 2007 decision in KSR International Co. v. Teleflex Inc. liberalized the obviousness standard by rejecting the rigid ‘teaching, suggestion, or motivation’ test that had previously required an explicit suggestion in the prior art to combine references. Post-KSR, courts apply a more flexible, common-sense approach that asks whether a person of ordinary skill in the art would have had reason to combine prior art references with a reasonable expectation of achieving the claimed invention. For pharmaceutical secondary patents, the KSR standard makes it substantially easier to argue that selecting a known salt form, combining known formulation components, or applying a known drug delivery technology to a known compound was obvious to a medicinal chemist or formulation scientist in the field.
The Amgen v. Sanofi Impact on Enablement Challenges
The Supreme Court’s 2023 decision in Amgen Inc. v. Sanofi has created a significant new vulnerability for broad pharmaceutical patents, particularly those claiming biological molecules or large chemical genus claims. The Court unanimously held that a patent claiming a genus of antibodies defined by their function rather than their specific structure failed the enablement requirement of 35 U.S.C. § 112 because the specification did not teach those skilled in the art how to make and use the full scope of the claimed genus without undue experimentation.
The practical consequence of Amgen v. Sanofi is a heightened enablement bar for any patent claiming a broad class of compounds or molecules rather than a specifically identified structure. Innovators who secured broad genus claims in the pre-Amgen era now face significantly increased invalidity risk under § 112. For generic and biosimilar challengers, this opens a class of invalidity arguments that was less viable before 2023. Patent claims that cover thousands or millions of structural variants based on a common functional property, without providing enabling disclosure for more than a handful of specific examples, are now substantially more vulnerable to an enablement challenge in both district court and IPR proceedings.
IPR at the PTAB: The Changed Battlefield
The America Invents Act of 2011 created the Inter Partes Review (IPR) process at the Patent Trial and Appeal Board, and it has permanently altered the strategic landscape of pharmaceutical patent litigation. Before the AIA, district court was the only venue for an invalidity challenge in the context of ANDA litigation. The IPR creates a parallel administrative forum that offers generic challengers several structural advantages.
In district court, a patent is presumed valid, and the challenger must prove invalidity by ‘clear and convincing evidence,’ the highest civil standard of proof. In an IPR, the presumption of validity does not apply in the same way, and the challenger needs only prove invalidity by a ‘preponderance of the evidence,’ meaning more likely than not. This difference in burden is not a procedural technicality. In close cases involving competing expert interpretations of ambiguous prior art, the lower IPR burden can be the deciding factor between a finding of validity and a finding of unpatentability.
PTAB panels consist of three Administrative Patent Judges with technical and legal expertise in the relevant field. They evaluate prior art arguments with a depth of technical understanding that generalist district court judges typically cannot match. This makes PTAB an especially attractive forum for validity challenges that turn on subtle distinctions in organic chemistry, formulation science, or pharmacokinetic data.
IPR proceedings are designed to conclude within 18 months of institution. For a generic company whose 30-month stay is already running, this timeline creates the possibility of a PTAB invalidity ruling that arrives well before the district court trial, dramatically reshaping the litigation landscape and settlement dynamics. A PTAB institution decision alone, even before a final written decision, signals to the brand manufacturer that its patent faces serious invalidity risk and substantially increases its willingness to negotiate a settlement on favorable terms.
IPR does carry one significant strategic limitation: estoppel. Under 35 U.S.C. § 315(e), a petitioner who receives a final written decision in an IPR is estopped from raising in subsequent district court proceedings any invalidity ground that was raised or reasonably could have been raised in the IPR. Companies that file IPR petitions must therefore be comprehensive in the invalidity arguments they include, because any ground left out of the petition is potentially foreclosed in the concurrent district court case. This requires careful coordination between the IPR litigation team and the district court litigation team to ensure that the dual-track strategy is internally consistent and that no valuable argument is sacrificed.
Secondary Considerations as the Brand’s Defense
Brand manufacturers defending against obviousness challenges almost invariably rely on secondary considerations, also called objective indicia of non-obviousness. These include commercial success of the patented drug, evidence that others in the field tried and failed to solve the same problem, evidence of long-felt unmet need, unexpected results, and praise from others in the field. Secondary considerations can be powerful enough to overcome an otherwise strong obviousness case, and they must be specifically addressed and rebutted in the invalidity strategy.
The commercial success argument is the most frequently deployed and requires the most careful rebuttal. The brand manufacturer will argue that the drug’s commercial success proves the value of the invention. The generic challenger must demonstrate that the commercial success is attributable to the brand’s market position, pricing power, and promotional investment rather than to the specific technical features claimed in the patent. Nexus between the commercial success and the claimed invention is required for commercial success to constitute persuasive evidence of non-obviousness, and establishing that nexus can be difficult for the brand when the patent covers a secondary feature rather than the fundamental therapeutic mechanism.
Key Takeaways: Step 6
Post-KSR, obviousness is the most effective invalidity ground for secondary pharmaceutical patents. Post-Amgen v. Sanofi, broad genus claims face heightened enablement vulnerability. The IPR process provides a faster, cheaper, technically expert forum with a lower burden of proof than district court, making it a standard component of any sophisticated Paragraph IV invalidity strategy. IPR estoppel requires comprehensive, coordinated petition preparation to avoid foreclosing district court arguments.
Step 7: Building the Cross-Functional ‘War Room’
Why Organizational Structure Predicts Outcome
A Paragraph IV campaign is an organizational stress test. It requires a sustained, multi-year collaboration among professionals with fundamentally different training, incentive structures, and working styles: patent attorneys, medicinal and formulation chemists, clinical pharmacologists, regulatory affairs specialists, and financial analysts. In companies where these functions operate in departmental silos, the information dependencies between them generate systematic inefficiencies, delayed decisions, and missed threats. In companies where these functions are genuinely integrated under project-based leadership, the information flows in real time, decisions are made with complete situational awareness, and the entire organization performs as a single coordinated unit.
The difference in outcomes between siloed and integrated operating models is documented in practice. Teva Pharmaceutical Industries, which built and maintained the world’s largest portfolio of first-filer Paragraph IV positions at its peak, explicitly structured its generics pipeline around cross-functional project teams that brought legal, scientific, and commercial expertise together under a unified program management structure. Mylan (now Viatris) built its biosimilar pipeline around a similar integrated model. The organizational architecture was not incidental to the commercial success. It was constitutive of it.
Core Team Roles and Accountability
The cross-functional project team for a Paragraph IV filing requires genuine accountability, not just nominal representation from each department. Four functions must be genuinely integrated rather than periodically consulted.
The legal function owns the complete patent analysis, from initial landscape search through FTO, non-infringement argument development, invalidity strategy, notice letter drafting, and litigation management. The legal team must be present in R&D discussions where formulation decisions are made, because those decisions create or close legal options in real time.
The R&D and scientific function owns the formulation development, process development, bioequivalence study design and execution, and CMC documentation. The science team must understand the IP constraints identified by the legal team and must design the product to satisfy both scientific performance requirements and legal clearance requirements simultaneously.
The regulatory affairs function owns the ANDA dossier assembly, FDA correspondence, and regulatory strategy. Regulatory affairs must ensure that the scientific and legal components of the application are packaged in a format that meets the FDA’s current technical requirements and that the proposed labeling is consistent with both the scientific data and the patent certification strategy.
The commercial and business development function owns the financial modeling, competitive intelligence, market entry timing strategy, and ongoing revision of the business case as new information becomes available. The commercial team’s revenue models directly affect decisions about litigation investment levels, settlement terms, and at-risk launch timing.
Structural Principles for High Performance
Effective cross-functional integration requires structural support beyond simply holding joint meetings. Shared data platforms, including electronic lab notebooks connected to the patent analysis system and a common project management platform visible to all functions, eliminate the information asymmetries that cause coordination failures. Defined decision rights, specifying which team has authority to make which categories of decision and how interdisciplinary conflicts are resolved, prevent the paralysis that occurs when multiple functions believe they have veto power over each other’s decisions.
The project leader’s role is to maintain alignment on the shared commercial objective, manage pace against the ANDA submission timeline, identify cross-functional dependencies before they become bottlenecks, and escalate decisions to senior leadership when the team cannot resolve them internally. The project leader need not be a scientist, attorney, or regulatory specialist. The critical competency is the ability to translate across functional languages, maintain momentum under uncertainty, and make decisions with incomplete information.
Key Takeaways: Step 7
Organizational integration is a structural predictor of Paragraph IV success. Siloed functions produce the information delays and decision failures that compromise legal strategy, inflate development costs, and miss market entry windows. The cross-functional war room model requires genuine accountability, shared data infrastructure, clear decision rights, and a capable project leader, not just calendar invitations to joint meetings.
Step 8: Pre-Litigation Preparation and Evidence Fortification
The Pre-Filing Window as a Litigation Investment Period
The time between the decision to pursue a Paragraph IV challenge and the submission of the ANDA is not downtime. It is the most productive period available for litigation preparation, and the quality of work done in this window directly determines the quality of the litigation that follows. Companies that treat pre-filing preparation as an afterthought encounter the consequences in discovery, at Markman hearings, and at trial.
Expert witness identification and retention is a critical pre-filing activity. The best academic and industry experts in formulation science, medicinal chemistry, pharmacokinetics, and patent law are in finite supply and are routinely retained by the brand’s litigation team in major Paragraph IV cases. Early identification and retention of the most credible experts in the relevant technical areas is a genuine competitive advantage. An expert retained a year before the ANDA is filed can contribute to the legal and scientific strategy itself, not just testify to a strategy that has already been locked in. Their early input can identify weaknesses in the invalidity arguments or non-infringement narrative before those weaknesses become liabilities at trial.
Non-testifying consultants, working under attorney-client privilege as litigation consultants rather than as designated expert witnesses, provide a different and equally valuable function. Because their work product is privileged and not subject to discovery, they can provide frank, candid assessment of the case’s vulnerabilities that a designated testifying expert cannot. The pre-filing consultation with a non-testifying expert is the most honest risk assessment a company can get, and it is protected from disclosure to the opposing party.
Litigation Hold Implementation
The litigation hold obligation arises as soon as litigation is reasonably anticipated. For a Paragraph IV filing, that point is not when the notice letter is sent, but when the internal decision to pursue the strategy is made. A formal litigation hold must be implemented at that point, requiring the preservation of all documents that could be relevant to the anticipated litigation: laboratory notebooks, formulation development records, bioequivalence study protocols and data, regulatory submissions and correspondence, internal communications about the patent strategy, and financial projections related to the project.
Spoliation of evidence, whether through routine document destruction that continues after the litigation hold should have been implemented or through deliberate deletion, can result in severe court sanctions. In the Hatch-Waxman context, where the 30-month stay creates a defined litigation timeline, courts are unsympathetic to companies that claim they were unaware of the preservation obligation. The hold must be comprehensive, covering email, electronic files, and physical records, and it must be communicated to all relevant custodians with written confirmation of receipt.
Red Team Review: Testing the Strategy Against Itself
The single most valuable pre-litigation exercise is a structured adversarial review of the legal strategy by a team playing the role of brand’s counsel. The red team, ideally composed of outside counsel not involved in developing the primary strategy, is given access to the non-infringement arguments, the invalidity theories, the prior art references, and the factual record and is asked to attack them with maximum aggression.
The red team review identifies the weaknesses that the primary team, necessarily invested in its own strategy, is least likely to see clearly. It may reveal that a key prior art reference for the invalidity case is distinguishable on a technical basis that the primary team has overlooked. It may show that the non-infringement argument for a specific claim element is weaker than assumed, or that the design-around documentation has gaps that opposing counsel will exploit in deposition. These findings, however uncomfortable, are far less costly when discovered in a pre-filing review than when they emerge for the first time at the Markman hearing or in cross-examination of the company’s expert witness.
Key Takeaways: Step 8
Pre-litigation preparation is an investment, not overhead. Early expert retention secures the best available scientific credibility and allows expert input into strategy, not just strategy defense. The litigation hold obligation runs from the date of the internal decision to pursue the filing, not from the date of the lawsuit. The red team review is the most productive risk management tool available before the ANDA is submitted, and its findings must be acted upon, not filed away.
Step 9: The Paragraph IV Notice Letter: Strategic Drafting for Maximum Leverage
The Mechanics of a Valid Notice
The notice letter is governed by precise statutory and regulatory requirements that admit no flexibility. The ANDA applicant must send the letter to both the NDA holder and each patent owner of record within 20 days of receiving the FDA’s acknowledgment letter confirming the ANDA’s acceptance for filing review. The FDA acknowledgment letter is a prerequisite; sending notice before receiving it renders the notice invalid and can forfeit first-filer status, as established in SB Pharmco Puerto Rico Inc. v. Mutual Pharmaceutical Co.
The content requirements are equally specific. The notice must identify the ANDA applicant, the ANDA number, the reference listed drug, the specific Orange Book-listed patents being challenged, and the factual and legal basis for the certification. For a non-infringement assertion, this means a specific description of how the generic product’s design falls outside the patent claims. For an invalidity assertion, it means identification of the prior art and a claim-by-claim analysis of why that prior art renders the claims unpatentable.
The letter must be sent by registered or certified mail or by a method permitting return receipt, and the ANDA applicant must submit a copy of the notice to the FDA along with a statement certifying compliance with the notice requirements. The 45-day clock for the patent holder to file suit begins running from the date the NDA holder receives the notice.
Strategic Drafting: Detailed Statement Architecture
The detailed statement is where the notice letter shifts from a compliance document to a strategic instrument. The legal team faces a genuine tension: the statute requires substantive detail, but disclosing the complete litigation strategy in the notice letter gives the brand manufacturer a complete preview of the defense it will need to prepare. The resolution of this tension requires judgment about the specific case, the brand manufacturer’s litigation posture, and the commercial dynamics of the particular drug.
The notice letter’s primary strategic function in the 45-day window is to shape the brand manufacturer’s risk perception. A notice letter that presents well-developed invalidity arguments supported by specifically identified prior art, a clearly articulated non-infringement theory with precise claim mapping, and a professional, authoritative presentation signals that the challenger is a capable and prepared adversary. Brand manufacturers routinely conduct rapid assessments of their litigation exposure in the 45-day window, and those assessments are based almost entirely on the notice letter. A weak letter invites an aggressive lawsuit. A strong letter opens the door to early settlement discussions.
For invalidity arguments, the notice should specifically identify the prior art references relied upon and walk through the claim mapping clearly enough to demonstrate that the challenger understands the patent and the prior art. For non-infringement arguments, the notice should state the specific claim limitations that the generic product does not meet and explain why, without disclosing the precise proprietary composition of the generic formulation if that disclosure can be avoided.
The Offer of Confidential Access
When the notice letter includes a non-infringement assertion, it must be accompanied by an Offer of Confidential Access (OCA) allowing designated counsel for the NDA holder to review the confidential portions of the ANDA. The OCA terms, including who may receive access, the scope and duration of any prosecution bar, and the confidentiality protections applicable to reviewed information, are a matter of negotiation between the parties.
The OCA is both a legal requirement and a tactical tool. Its terms can be used to manage the risk that brand counsel, after reviewing the ANDA, will use confidential formulation information to develop new, targeted patent applications directed at the generic product’s specific design-around approach. A well-drafted prosecution bar in the OCA prevents counsel who reviews the confidential ANDA information from prosecuting related patents for a defined period, protecting the generic company’s proprietary formulation work from being reverse-engineered into new IP claims by the innovator.
The 45-Day Window as a Negotiation Period
The 45-day window is not a passive waiting period. It is an intense bilateral negotiation period, sometimes formal and sometimes through back channels, during which both parties are rapidly assessing their relative positions. The brand manufacturer is evaluating its probability of winning an injunction extending the 30-month stay if it sues, its probability of prevailing at trial, and the NPV of its patent exclusivity versus the cost and uncertainty of litigation. The generic challenger is evaluating the same calculation from the opposite side.
Settlement negotiations during this period, and in the early phase of litigation if suit is filed, are governed by the antitrust constraints established by the Supreme Court’s 2013 decision in FTC v. Actavis, Inc. The Court held that a ‘reverse payment’ settlement, in which a brand manufacturer pays a generic company to stay off the market, may be anticompetitive and subject to antitrust challenge. Settlements must be structured to fall within the safe harbor for reasonable litigation compromises rather than naked market allocation. The antitrust constraints on settlement are a constant background consideration in any negotiation and require careful review by antitrust counsel in addition to patent counsel.
Key Takeaways: Step 9
Notice letter timing is non-negotiable and legally precise. The detailed statement is a strategic document, not just a compliance exhibit. It must be substantive enough to demonstrate competence and shape risk perception without unnecessarily disclosing the complete litigation strategy. OCA terms require careful drafting to protect against prosecution bar risks. The 45-day window is a negotiation period governed by FTC v. Actavis antitrust constraints that counsel must integrate into any settlement discussions.
Step 10: ANDA Dossier Assembly and Final Pre-Submission Audit
The Substantially Complete Standard and Its Consequences
The 180-day exclusivity prize flows to the first applicant to file a substantially complete ANDA containing a Paragraph IV certification. The FDA applies its ‘substantially complete’ standard at the point of submission; an application that fails this standard receives a Refuse-to-Receive (RTR) letter, which denies the application a filing date. An RTR for a first-filer is commercially catastrophic. The company loses its place in line, its 180-day exclusivity claim evaporates, and any competitor who files a substantially complete application after the RTR becomes the new first filer.
The RTR risk is not theoretical. The FDA issues RTR letters with some regularity, and the causes are consistently preventable: missing certification for an Orange Book-listed patent, bioequivalence data that does not conform to current FDA guidance, CMC sections that are missing required elements, or labeling that does not comply with the ‘same as’ requirement. Each of these deficiencies reflects a breakdown in the project’s quality control process, and each is avoidable with a systematic pre-submission audit.
Pre-Submission Checklist: Critical Elements
The final dossier review must verify the presence and quality of each required ANDA component against the FDA’s current regulations and guidance documents, most directly 21 CFR § 314.94.
The basis for submission section must correctly identify the RLD and the ANDA applicant’s basis for relying on its safety and efficacy data. The CMC section must completely document the drug substance, drug product, manufacturing process, and quality control specifications. The bioequivalence data section must include all studies conducted on the final formulation, including any failed studies, with complete study reports and statistical analyses demonstrating that the 90% confidence interval for Cmax and AUC falls within the 80-125% acceptance range.
The patent certification section must include a certification for every single patent listed in the Orange Book for the RLD, with no omissions. This is the most common source of RTR-level deficiencies. Orange Book listings are updated continuously, and a patent listed after the ANDA development process began must be identified and certified against. For each Paragraph IV certification, the notice letter requirement and OCA requirement must be documented.
The proposed labeling section must present a version that is the ‘same as’ the most recently approved RLD labeling with all permissible differences annotated and justified in a side-by-side comparison. Where a skinny label carve-out has been employed, the proposed labeling must specifically exclude the patented indication’s language while remaining consistent with the non-carved-out portions of the approved label.
eCTD Format and FDA Electronic Submission Requirements
All ANDAs must be submitted in electronic Common Technical Document (eCTD) format through the FDA’s Electronic Submission Gateway (ESG). eCTD formatting requirements are technically specific and require validated submission software. Submission validation errors, while fixable, can delay the acceptance review and in competitive first-filer situations, even a short delay in receiving an FDA acknowledgment letter can be commercially significant.
The FDA encourages pre-submission meetings for complex products and provides a mechanism for resolving specific submission questions before the ANDA is filed. For complex products where BE methodology questions exist or where the patent certification strategy involves novel legal theories, a pre-ANDA meeting with the FDA’s Office of Generic Drugs can identify and resolve potential deficiencies before the submission clock starts.
Internal Consistency Audit: The Cross-Functional Final Check
Beyond the checklist verification, the final review must assess the dossier’s internal consistency as a whole. The scientific work in the CMC and BE sections must be consistent with each other: the formulation described in the CMC must be the exact formulation used in the BE study. The legal strategy in the patent certifications must be consistent with the scientific claims in the CMC: if the non-infringement argument relies on a specific formulation design-around, the CMC section must describe that exact design-around accurately and in sufficient detail to support the legal argument.
The regulatory labeling strategy must be consistent with the legal strategy: if a skinny label is being used to avoid a method-of-use patent, the label carve-out must accurately excise the patented indication in a way that is both legally correct and scientifically coherent with the remaining approved uses. Inconsistencies between sections of the ANDA are both regulatory deficiencies and litigation risks: the brand’s counsel will review the ANDA closely in discovery and any internal contradiction becomes ammunition for attacking the credibility of the generic’s scientific and legal arguments.
Investment Strategy Note
For institutional investors, the first-filer ANDA pipeline is a quantifiable asset. Each substantially complete Paragraph IV application with plausible invalidity or non-infringement arguments against a commercially significant drug represents a conditional revenue claim that can be probability-weighted and discounted for litigation risk. Companies that report pipeline metrics with specificity about first-filer status, brand revenue of the challenged product, and litigation stage provide enough information for analysts to build DCF-based valuations of the exclusivity option embedded in each filing.
Key Takeaways: Step 10
The RTR is a catastrophic and preventable outcome. The final pre-submission audit must be systematic, checklist-driven, and cross-functional. Internal consistency across CMC, bioequivalence, labeling, and patent certification sections is both a regulatory requirement and a litigation readiness criterion. The first-filer date is the single most valuable commercial milestone in the Paragraph IV process, and protecting it requires zero tolerance for avoidable submission deficiencies.
Investment Strategy: A Framework for Analysts Evaluating Paragraph IV Pipelines
For portfolio managers and institutional investors evaluating generic pharmaceutical companies, the Paragraph IV pipeline is the most operationally complex asset to value and the most consequential to get right.
The baseline valuation framework assigns each pending Paragraph IV application a conditional expected value derived from the brand product’s annual revenue, the estimated 180-day exclusivity prize as a percentage of that revenue (typically 15-25% for a sole generic entering at a 35% discount to brand WAC), the probability of success on the merits (historical first-filer success rates in contested litigation approximate 60% when challenged through trial), and the projected litigation cost as a reduction to gross expected value.
The quality of the Paragraph IV pipeline, as distinct from its size, turns on several specific attributes. First-filer status, meaning the company is confirmed as the first to file a substantially complete ANDA with a Paragraph IV certification for a given product, is a binary distinction that separates the 180-day prize from a commodity market position. Companies with a high concentration of first-filer positions relative to their total ANDA count demonstrate superior commercial intelligence and IP targeting capability.
The complexity profile of the challenged products matters because complex targets generate less competition. A company whose Paragraph IV pipeline is concentrated in complex injectable drugs, modified-release products, or specialty formulations faces fewer competitors upon entry than one concentrated in standard solid oral dose generics. The former profile supports higher gross margin and more durable market position post-exclusivity.
Litigation stage is a relevant financial indicator. A Paragraph IV challenge in the pre-litigation phase represents a contingent asset. One where the district court has ruled for the generic challenger on the primary patents, or where the PTAB has issued an institution decision in an IPR filed against the brand’s key patents, represents a materially derisked asset with significantly higher probability of commercial realization.
The brand manufacturer’s response posture is also predictive. A brand with a history of early settlement in Hatch-Waxman litigation, rather than fighting through trial, represents a lower-cost path to market entry for the generic challenger. Several mid-size branded companies have effectively signaled that their preference is authorized generic arrangements or early settlements over expensive, multi-year litigation, and those signals are visible in public litigation records and settlement disclosures.
Frequently Asked Questions
What disqualifies a company from 180-day exclusivity after filing?
Several post-filing events can forfeit or delay exclusivity. The most significant is failure to obtain tentative approval or final approval within 30 months of filing and maintaining first-filer status. The ‘forfeiture events’ under the Hatch-Waxman amendments include failing to market the drug within 75 days of receiving final approval or 30 months after filing, whichever is later; withdrawing the ANDA or converting the Paragraph IV certification to a Paragraph III; amending the ANDA to remove the Paragraph IV certification; entering into an agreement with the NDA holder, patent owner, or another ANDA applicant that is found to be a violation of antitrust law; and failing to obtain a court decision or settlement establishing invalidity or non-infringement within the exclusivity period.
How has the FDA’s Real-Time Oncology Review (RTOR) and other expedited programs affected Paragraph IV strategy?
Expedited FDA approval programs for brand products, including RTOR, Breakthrough Therapy Designation, and Accelerated Approval, can alter the timing and market structure that generic companies must model. An accelerated approval drug that receives full approval earlier than projected shortens the effective patent protection window differently than standard review timelines, requiring generic developers to revisit their competitive timing models when these designations are granted to brand competitors in their target therapeutic areas.
What is the current state of authorized generic strategy by brand manufacturers, and how should generic companies account for it?
Authorized generics, launched by brand manufacturers or their licensees during the first-filer’s 180-day exclusivity period, are a legitimate competitive response that the Hatch-Waxman Act does not prohibit. The authorized generic converts the first filer’s duopoly into a three-player market structure and materially reduces the 180-day prize value. Several major brand manufacturers, including Pfizer and AbbVie, have used authorized generics as a standard lifecycle management tool. Generic companies modeling the value of a first-filer position must explicitly scenario-plan for authorized generic entry as a downside case, with probability of occurrence estimated based on the specific brand manufacturer’s historical behavior and the commercial stakes involved.
How does multi-district litigation (MDL) consolidation affect Paragraph IV strategy?
When a brand manufacturer faces multiple ANDA filers simultaneously, the district court cases are often consolidated into multi-district litigation (MDL) proceedings, typically before a single federal judge in the district where the first suit was filed. MDL consolidation creates both efficiencies and strategic complications. Invalidity arguments made by one generic defendant can benefit all defendants if successful. Settlement negotiations become more complex when multiple defendants with potentially conflicting interests must coordinate their positions. Companies filing Paragraph IV challenges against heavily contested products should anticipate MDL consolidation and plan their litigation strategy accordingly.
Conclusion
The Paragraph IV process is the most demanding, highest-stakes strategy in pharmaceutical commerce. Every element of the 10-step framework described here, from the initial commercial analysis through the final pre-submission audit, is a necessary component of a complete campaign. No single step is dispensable, and no step can compensate for failures in the others.
What separates the companies that consistently convert Paragraph IV challenges into first-filer exclusivity prizes from those that absorb expensive failures is not primarily legal skill or scientific capability, though both are required in abundance. It is organizational integration, the sustained capacity to run a complex, multi-year, multi-disciplinary campaign with the coherence and internal consistency that a successful ANDA demands. The legal theory must match the scientific design-around. The scientific design-around must be reflected accurately in the regulatory dossier. The regulatory strategy must be consistent with the litigation strategy. The commercial model must accurately incorporate legal probability assessments and scientific development risks.
The regulatory landscape continues to shift. The FTC’s 2023 policy statement on improper Orange Book listings, the Supreme Court’s Amgen v. Sanofi enablement decision, and the continued evolution of IPR practice at the PTAB all represent material changes to the operating environment since the Hatch-Waxman framework’s last major legislative revision. Generic companies that treat these developments as operational updates rather than strategic signals will be structurally disadvantaged against those who recognize that the boundaries of the game itself are moving.
The companies that master this framework, that build the organizational infrastructure, data capabilities, and cross-functional culture required to execute all 10 steps at the level of excellence the Paragraph IV process demands, will capture the 180-day exclusivity positions that drive disproportionate value creation in the generic pharmaceutical industry for the next decade.
Data sources referenced throughout: DrugPatentWatch integrated patent and regulatory database; AIPLA Report of the Economic Survey; FDA Orange Book; FDA Office of Generic Drugs Product-Specific Guidances; IQVIA MIDAS market data; U.S. Supreme Court decisions KSR Int’l Co. v. Teleflex Inc. (2007), FTC v. Actavis, Inc. (2013), Amgen Inc. v. Sanofi (2023); U.S. Court of Appeals for the Federal Circuit decisions including GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc. (2020, 2021); 21 CFR Part 314; 35 U.S.C. §§ 102, 103, 112, 156, 315.