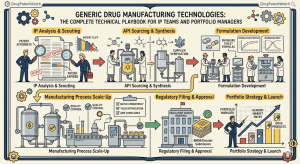
1. The Market Structure Forcing Manufacturing Innovation
The global generic drug market generated approximately $245 billion in 2020 and is projected to reach $435 billion by 2025 at a compound annual growth rate of roughly 9.5%. That growth rate sounds healthy. The underlying economics are less comfortable.
Generic manufacturers operate in a structurally adversarial pricing environment. Group purchasing organizations extract aggressive rebates. Markets with four or fewer manufacturers, which account for over 75% of current drug shortages, generate margins too thin to justify stockpiling inventory or maintaining spare manufacturing capacity. The result is a market where the mechanism that makes drugs affordable, intense price competition, directly degrades the supply reliability that makes them medically useful.
The American Progress data on shortages is instructive here. More than 58% of current shortages have lasted two years or longer. The most clinically consequential cases, such as the carcinogenic nitrosamine contamination that triggered the valsartan recall cascade in 2018 and 2019, trace back not to regulatory negligence but to manufacturing processes operating with no economic buffer for quality investment.
This is the core structural problem that advanced manufacturing technologies (AMTs) address. A continuous manufacturing line with integrated Process Analytical Technology (PAT) does not just cut cost per unit. It breaks the causal chain between price pressure and quality degradation by making real-time quality monitoring economically integrated rather than economically optional.
The FDA’s own economic analyses confirm the dynamic: greater generic competition lowers prices and improves access, but those gains are time-limited if the thin-margin structure eventually collapses the manufacturing base. Agency guidance on supply chain resilience, published through 2023 and 2024, names AMTs explicitly as the preferred structural intervention.
Key Takeaways for Segment 1
Generic pricing competition creates a self-defeating cycle: it lowers margins, which reduces investment in manufacturing quality and redundancy, which causes shortages, which undermines the access gains that price competition was meant to deliver. AMTs break that cycle by shifting the cost structure rather than tolerating it.
2. IP Valuation Through a Manufacturing Lens
Portfolio managers and IP counsel typically value pharmaceutical intellectual property through three lenses: composition-of-matter patents, method-of-treatment patents, and formulation patents. Manufacturing process patents are frequently underweighted relative to their actual commercial contribution.
That is a mistake. Consider the following: a composition-of-matter patent on an API typically expires 20 years from filing, and Orange Book listing does not protect the molecule from Paragraph IV challenges after that term. A manufacturing process patent, particularly one covering continuous manufacturing integration or a novel nanoparticle production method, can generate independent IP protection that survives the primary patent cliff by years.
How Manufacturing IP Compounds Valuation
When a brand manufacturer implements a proprietary continuous manufacturing process, it files process patents that are distinct from the Orange Book-listed formulation patents. A Paragraph IV filer challenging the Orange Book patent list must also clear any relevant process IP before manufacturing at commercial scale. This creates what practitioners sometimes call a ‘manufacturing moat,’ a set of process and equipment patents that extend de facto exclusivity even after the composition-of-matter patent expires.
Janssen’s implementation of continuous manufacturing for its antiretroviral darunavir (Prezista) illustrates this precisely. Janssen publicly targeted manufacturing 70% of its highest-volume products via continuous manufacturing within an 8-year window, with explicit goals of a 33% reduction in waste and 80% reduction in manufacturing and testing cycle time. The process IP surrounding that continuous manufacturing platform does not appear in the Orange Book, but it is highly relevant to any generic manufacturer’s freedom-to-operate analysis.
For ANDA filers, the question is not just ‘when does the listed patent expire?’ but ‘what process patents must we design around, license, or challenge to manufacture at commercial scale using modern methods?’
Manufacturing Process Patent Categories Worth Tracking
Process patents in this space cluster into several categories that IP teams should monitor separately. First are unit operation integration patents, which cover the specific way discrete steps are combined into a continuous or semi-continuous flow. Second are control system patents, which cover the algorithms, sensors, and feedback loops used to maintain Critical Quality Attributes in real time. Third are excipient and nano-carrier formulation patents, which cover the composition of delivery vehicles like liposomes, polymeric nanoparticles, or albumin-bound formulations. Fourth are equipment design patents, which cover novel reactor or extruder configurations used in hot-melt extrusion or spray drying for amorphous solid dispersions.
A full IP valuation of any generic product opportunity should map each of these patent clusters separately and assess freedom-to-operate for each manufacturing approach under consideration.
Investment Strategy Note for Analysts
When screening generic manufacturers for portfolio allocation, ask whether their ANDA pipeline overlaps with products whose reference listed drugs (RLDs) are manufactured using patented continuous manufacturing or advanced formulation processes. A company that has done that clearance work, or has developed its own manufacturing process IP as a defensive moat, carries materially lower litigation risk per ANDA than one that has not. Manufacturing IP due diligence is not a technical footnote; it is a direct input to ANDA litigation probability, which is the dominant valuation driver for generic pipelines.
Key Takeaways for Segment 2
Manufacturing process patents extend competitive protection well beyond Orange Book-listed formulation and composition patents. IP teams should conduct parallel freedom-to-operate analysis for continuous manufacturing, PAT control systems, and nano-carrier formulation IP. Analysts should weight this analysis in pipeline valuation models.
3. Continuous Manufacturing: Technical Architecture and ANDA Strategy
3.1 How Continuous Manufacturing Works at the Unit Operation Level
Continuous manufacturing (CM) eliminates the stop-start structure of batch production by integrating discrete unit operations, granulation, drying, blending, compression, and coating, into a single uninterrupted flow. Raw materials enter one end; finished solid oral dosage forms exit the other without intermediate hold steps.
The technical backbone of a CM line for solid oral dosages typically includes a twin-screw wet or dry granulator, a continuous dryer (often a plug-flow fluid bed), an in-line blender with API and excipient loss-in-weight feeders, and a tablet press with integrated coating. Sensors at each transition point, typically near-infrared (NIR), Raman, or laser diffraction instruments, measure Critical Quality Attributes in real time. Those measurements feed into automated control loops that adjust processing parameters, feed rates, temperature, or compression force, before an out-of-specification condition propagates downstream.
The FDA’s definition of a CM batch is not the fixed-volume batch of traditional processing. It is defined by time or by a specified quantity of material processed through the system. This distinction has significant ANDA implications, as discussed below.
3.2 Cost Structure and Footprint Analysis
A continuous manufacturing facility can occupy 70% less floor space than an equivalent batch facility. That number, from Drug Development & Delivery’s analysis of the economics, reflects the elimination of intermediate hold tanks, drying rooms, and inter-step quality testing suites. The capital cost of a CM tablet line runs approximately $5 million per line, with integrated PAT sensors adding $200,000 to $300,000 per line.
For a generic manufacturer with 10 to 15 product changeovers per line per year, that capital cost is harder to justify than for an innovator running a single high-volume product for years. This is not a trivial obstacle. It is the central reason no generic drug had received FDA approval through a CM process as of 2024. The economics favor innovators, which creates a competitive gap that well-capitalized generic manufacturers can exploit if they approach CM with a platform strategy rather than a single-product justification.
A platform approach means designing the CM line to accommodate multiple APIs with similar physicochemical properties, using the same granulation and compression equipment with validated cleaning and changeover procedures. The unit economics change substantially when capital is amortized across a product cluster rather than a single ANDA.
3.3 Regulatory Framework: ANDA Implications of Continuous Manufacturing
The FDA issued its draft guidance on CM in 2019, with the finalized guidance acknowledging that its traditional batch-based regulatory framework does not map cleanly onto CM processes. Key issues for ANDA filers include how to define the batch for quality purposes, what in-process sampling frequency is required to validate uniformity, and how to handle material that was manufactured during a process upset.
The agency’s Quality by Design (QbD) framework, ICH Q8, Q9, and Q10, is the operative regulatory lens for CM submissions. Under QbD, the manufacturer defines a Design Space, the multidimensional set of processing parameters within which product quality is assured. CM facilitates an expanded Design Space because real-time sensor data provides continuous evidence of conformance rather than end-of-batch testing. This means a CM-enabled ANDA can potentially support broader post-approval change flexibility under the FDA’s CMC post-approval change pathways, a significant commercial advantage during product lifecycle management.
For ANDA filers specifically, the absence of an approved generic CM pathway as of 2024 creates both a barrier and an opportunity. The first generic manufacturer to establish FDA precedent for a CM-based ANDA in a particular dosage form will benefit from the regulatory learning that accrues to a first mover, including clearer dialogue with the agency on what data packages satisfy CM validation requirements.
3.4 Competitive Intelligence: Which Generic Manufacturers Are Investing
The Pharmtech analysis of CM in generics identifies contract manufacturers and CMOs as particularly reluctant adopters due to multi-product line requirements. However, a small number of generic manufacturers are actively building CM capabilities. Tracking patent filings in IPC class B01J (chemical or physical processes) combined with pharmaceutical company assignee searches at USPTO is a practical method for identifying which organizations are building proprietary CM process IP.
The Brookings Institution’s 2023 technology solutions report on generic drug manufacturing resilience explicitly names CM as a priority for supply chain strengthening and calls for regulatory and financial incentives to close the adoption gap. This signals that regulatory relief or financial support programs for CM adoption may emerge from legislative activity in 2025 and 2026.
Key Takeaways for Segment 3
CM lines cost approximately $5 million per installation plus $200K to $300K in sensors, and no generic has cleared an FDA approval through CM as of 2024. The economic case requires a platform strategy across multiple ANDAs. QbD-based Design Space submissions for CM-enabled ANDAs can support broader post-approval change flexibility, which is a lifecycle management advantage that repays the upfront capital investment over time.
Investment Strategy
For analysts evaluating generic manufacturers, a company that files CM process patents and demonstrates active FDA dialogue on CM pathways is de-risking its manufacturing cost structure for the next decade. Flag any generic manufacturer that discloses CM line installation in SEC filings or facility inspection records; that is a leading indicator of pipeline differentiation, not a sunk cost.
4. 3D Printing: Process IP, Spritam’s Patent Estate, and the Distributed Manufacturing Endgame
4.1 Spritam’s IP Architecture as a Case Study
Aprecia Pharmaceuticals’ Spritam (levetiracetam), the first 3D-printed drug approved by the FDA (August 2015), is the reference point for understanding how 3D printing intersects with pharmaceutical IP strategy. Spritam uses Aprecia’s ZipDose technology, a binder jet printing process that produces a highly porous, rapidly dissolving tablet capable of delivering doses up to 1,000 mg in a single wafer that disintegrates with a sip of water.
Aprecia’s patent estate around Spritam clusters around three categories. The first is the ZipDose process itself, covering the specific binder jetting parameters, powder bed characteristics, and aqueous binder formulations that produce a mechanically stable but rapidly dissolving structure. The second is the dosage form design, covering the layered architecture that achieves rapid disintegration without compromising dose uniformity. The third is quality control methods, covering NIR and vision system-based inline inspection protocols specific to the high-porosity tablet structure.
Any generic manufacturer seeking to develop a levetiracetam equivalent using 3D printing must either design around each of these patent clusters or wait for term expiration. The formulation freedom-to-operate analysis is substantially more complex than for a conventional compressed tablet, because the manufacturing process and the final product characteristics are inseparable in binder jet printing.
4.2 3D Printing Methods: Technical Differentiation and IP Landscape by Process
Pharmaceutical 3D printing encompasses at least six distinct process categories, each with its own IP landscape and regulatory considerations.
Binder jet printing (Aprecia’s approach) deposits liquid binder onto powder beds layer by layer. It is fast and capable of producing high-dose porous structures. Fused deposition modeling (FDM) extrudes drug-loaded polymer filaments through a heated nozzle. It produces dense, controlled-release geometries and is widely used in academic research but generates brittle, high-temperature-sensitive output. Selective laser sintering (SLS) uses a laser to fuse drug-polymer powder particles without solvents, avoiding stability problems associated with aqueous or solvent-based processes. Stereolithography (SLA) uses UV-cured photopolymers and is limited to UV-compatible drug molecules. Hot-melt extrusion-coupled printing combines continuous HME with a printing head, integrating amorphous solid dispersion formation and shaping in a single step. Inkjet printing deposits picoliter-scale droplets of drug solution onto substrates, useful for very low-dose precision applications including pediatric dosing strips.
Each process generates a distinct patent landscape. FDM filament composition patents, SLS sintering parameter patents, and SLA photoinitiator-drug compatibility patents are all independently searchable and require separate clearance. IP teams evaluating 3D printing for a given product must first select the process on technical grounds, then conduct IP clearance specific to that process category.
4.3 FABRX Economic Data: The 3D Printing Business Case
The FABRX study on 3D-printed minoxidil capsules provides the most granular public cost-benefit data available. At the 2.5 mg dose, 3D printing generated a 35% reduction in production cost versus conventional capsule filling. At 5 mg, the reduction was 20%. Manual labor fell by 55% due to automated quality control that replaces manual capsule weight sampling. Production time dropped by approximately 10%. These are not projected savings; they are reported production data from a commercial operation.
The mechanism behind the labor reduction is significant for quality professionals. Automated inline vision systems and weight monitoring in the 3D printing process replace the statistical sampling protocols required by conventional encapsulation, which rely on periodic manual pulls and offline testing. The 3D printing system generates a complete in-process quality record for every unit, not a sample-based inference, which has direct implications for quality system design and FDA submission data packages.
4.4 The Distributed Manufacturing Endgame: Regulatory and Commercial Architecture
The 2025 regulatory development cited by PharmTech, legislation enabling 3D printing within a distributed manufacturing framework, changes the commercial geometry of pharmaceutical production fundamentally. The model works as follows: a centralized Pharmaceutical Quality System (PQS) site holds the validated master batch record, process parameters, and quality specifications. Smaller satellite printing facilities, potentially at hospital pharmacies, specialty pharmacies, or regional distribution hubs, print patient-specific doses on demand under the authority of the central PQS.
For generic manufacturers, this architecture creates a new competitive vector. A company that establishes the central PQS site and licenses or operates satellite printing units controls the production relationship even in a decentralized network. The IP protection around that architecture includes the master batch record itself as a trade secret, the validated printing process parameters as process IP, and potentially the software platform managing dose specification transmission and quality release as a separate software IP asset.
The EMA’s Regulatory Science to 2025 framework explicitly endorses digital tools and data-driven development, including distributed manufacturing models, as strategic priorities. Manufacturers building distributed 3D printing networks should plan their regulatory strategy around simultaneous FDA and EMA engagement, given the size of both markets and the value of harmonized approval.
Key Takeaways for Segment 4
Spritam’s IP architecture demonstrates that 3D printing process patents, dosage form design patents, and quality control method patents form independent protection layers. Generic entry requires clearance across all three. The FABRX data puts hard numbers on the business case: 35% cost reduction, 55% labor reduction. The distributed manufacturing framework developing in regulation through 2025 and 2026 turns 3D printing from a manufacturing technology into a distribution network architecture.
5. Modular Manufacturing: CapEx Structures, Facility IP, and Pandemic-Resilience Economics
5.1 Modular Architecture: What It Actually Means for GMP Facilities
Modular manufacturing in pharmaceuticals refers to pre-engineered, factory-assembled facility components that are shipped to site and interconnected to form a complete GMP manufacturing suite. The modules are self-contained: they include HVAC, utility connections, cleanroom paneling, and equipment mounting points. They arrive at site meeting ISO classification requirements and FDA/EMA cGMP facility design standards.
The practical implications are substantial. Traditional stick-built pharmaceutical facilities require 24 to 36 months from groundbreaking to regulatory approval. Modular facilities can reduce that timeline to 12 to 18 months, with some manufacturers reporting reductions of 30 to 50% off traditional construction schedules. Capital certainty is higher because modular components are manufactured under controlled factory conditions, reducing the weather, labor, and materials variability that inflates stick-built construction budgets.
Bancroft’s design documentation for generic drug manufacturers illustrates the practical application. Their configurable modular designs specify validated materials and finishes that withstand frequent washdowns, which is essential for generic manufacturers running 20 to 30 product changeovers per line per year. Passive solar orientation and efficient HVAC design reduce energy costs, which in a thin-margin business are not cosmetic sustainability choices but material line items.
5.2 Volume Flexibility: The Pandemic-Resilience Economic Case
The PMC study on modular continuous drug product manufacturing quantifies what practitioners mean by ‘volume flexibility.’ A modular system provides the ability to rapidly scale production up or down by adding or removing production modules without rebuilding fixed infrastructure. For a conventional batch facility, scaling up means either underutilizing a larger fixed facility or running additional shifts on existing equipment with diminishing quality control returns. Neither option is commercially clean.
The COVID-19 pandemic made the cost of inflexibility explicit. Manufacturers producing critical generics on fixed, dedicated lines could not rapidly shift capacity to shortage-affected products. Modular systems, by contrast, allow production to follow demand in real time. The PMC mini-tablet study demonstrated real-time quality assurance integration within a modular continuous production system, generating an in-process data stream that allowed release decisions to be made continuously rather than at batch end.
Pfizer’s public commitment to portable, continuous, miniature, and modular (PCMM) technology for its product portfolio, demonstrated through a filgrastim (Neupogen) pilot using FDA-approved drug with modular equipment, establishes both technical proof of concept and regulatory precedent. Neupogen’s use in the Pfizer PCMM demonstration is strategically significant: by using an already-approved biologic as the demonstration molecule, Pfizer generated a regulatory data package showing that modular manufacturing does not compromise product quality versus conventional processes. Generic manufacturers can reference that precedent in their own modular facility justifications to FDA.
5.3 Modular Cleanroom IP: A Frequently Overlooked Asset Class
The IP associated with modular cleanroom design is less visible than drug product patents but commercially meaningful. Patents covering proprietary panel connection systems, integrated HEPA filtration mounting, electrostatic discharge (ESD) flooring systems for high-potency manufacturing suites, and reconfigurable airflow management systems are all independently protectable innovations.
For generic manufacturers building or licensing modular cleanroom systems, these equipment patents matter in two ways. First, they determine which modular cleanroom vendors can be used without licensing disputes. Second, manufacturers that develop proprietary modular cleanroom configurations can generate licensing revenue from CMO partners who want to replicate the design at their own facilities.
Key Takeaways for Segment 5
Modular construction reduces pharmaceutical facility construction timelines by 30 to 50% and improves capital cost certainty. The Pfizer PCMM demonstration with Neupogen provides regulatory precedent for modular manufacturing equivalence. Volume flexibility in modular systems translates directly to supply chain resilience, which is now a measurable factor in FDA supply chain risk assessments.
Investment Strategy
Analysts should evaluate whether a generic manufacturer’s facility investment pipeline uses modular construction. A company that can deploy a new manufacturing suite in 12 to 18 months rather than 36 months has a structural time-to-market advantage when ANDA approval windows open. That advantage compounds across a multi-ANDA portfolio.
6. Nanotechnology and Super Generics: From Abraxane’s IP Playbook to the Next Generation
6.1 Defining the Super Generic Category
The term ‘super generic’ lacks a formal FDA regulatory definition, which is itself commercially meaningful. It refers to reformulated versions of approved small molecules that use advanced delivery technologies to improve bioavailability, reduce adverse effects, lower dosing frequency, or enable new routes of administration. These products typically file under the 505(b)(2) NDA pathway rather than the standard ANDA pathway, because they demonstrate clinical differentiation from the RLD rather than simple bioequivalence.
The 505(b)(2) pathway allows the applicant to rely on published literature and the FDA’s prior findings of safety and effectiveness for the RLD, while submitting its own clinical data demonstrating the enhanced profile of the reformulated product. This is categorically different from an ANDA, where the goal is therapeutic equivalence. A 505(b)(2) super generic seeks to demonstrate superiority or differentiation on specific attributes, enabling a premium price point that ANDA-based generics cannot command.
6.2 Abraxane’s IP Architecture: The Albumin-Bound Nanoparticle Model
Abraxane (nab-paclitaxel), developed by American BioSciences and later acquired by Celgene (now Bristol Myers Squibb), is the most commercially successful super generic built on nanoparticle technology. Conventional paclitaxel (Taxol) requires Cremophor EL (polyethoxylated castor oil) as a solubilizing agent, which causes severe hypersensitivity reactions requiring premedication with corticosteroids and antihistamines. Abraxane uses a proprietary albumin-bound nanoparticle (nab) platform that eliminates Cremophor entirely. The resulting formulation achieves higher intratumoral paclitaxel concentrations and reduced peripheral neuropathy at equivalent doses.
Abraxane’s IP estate covers the nab technology itself, the specific albumin:paclitaxel ratio and nanoparticle size distribution specifications, the manufacturing process using high-pressure homogenization to form nanoparticles in situ, and the absence of conventional solvent excipients as a product characteristic. A generic challenger must address each of these layers. Generic nab-paclitaxel development requires demonstrating bioequivalence not just for total drug exposure but for the nanoparticle characteristics that drive the differentiated safety and efficacy profile.
This IP architecture illustrates why nanoparticle-based super generics are defensible for longer than conventional small molecule generics. The product’s therapeutic differentiation is physically inseparable from its manufacturing process. You cannot replicate Abraxane’s clinical profile without replicating its nab manufacturing process, which is itself protected by process patents.
6.3 Nanotechnology Platform Categories and Their IP Characteristics
The four most commercially developed nanoparticle platforms each have distinct IP characteristics that IP teams should map separately.
Lipid nanoparticles (LNPs), now best known from Moderna and Pfizer-BioNTech’s mRNA COVID vaccines, have an IP landscape dominated by Alnylam Pharmaceuticals, Arbutus Biopharma, and MIT/Whitehead Institute licensed patents. Any generic or biosimilar developer working with LNPs must conduct detailed freedom-to-operate analysis against Alnylam’s lipid composition patents and ionizable lipid synthesis method patents, which remain active through the early 2030s in most major markets.
Polymeric nanoparticles, typically using PLGA (poly lactic-co-glycolic acid) or PEGylated block copolymers, have a more fragmented IP landscape. Many foundational PLGA patents have expired, but second-generation patents covering specific particle size distributions, surface functionalization methods, and targeted delivery ligand attachment chemistry remain active.
Albumin-bound nanoparticles (nab technology) are primarily covered by Abraxis/BMS patents. The core technology patents begin to expire in the mid-2020s, which opens a development window for nab-based super generics in indications beyond breast cancer and pancreatic cancer.
Liposomes and lipid-based nano-carriers, including liposomal doxorubicin (Doxil), have the oldest IP landscape in the nanoparticle space. Doxil’s core patents have long expired, but the manufacturing process for sterile, long-circulating PEGylated liposomes remains technically challenging. The FDA’s draft guidance on liposomal product development (2018, updated 2023) requires demonstration of physicochemical equivalence beyond standard bioequivalence, including particle size distribution, drug encapsulation efficiency, and membrane bilayer characteristics.
6.4 SUBACAP and the Itraconazole Model: A Smaller-Scale Blueprint
SUBACAP, an improved itraconazole formulation developed to address the high inter- and intra-patient variability of conventional itraconazole, demonstrates the commercial logic of super generic development at a smaller scale than Abraxane. Conventional itraconazole has notoriously unpredictable oral bioavailability, ranging from near-zero to high absorption depending on fed versus fasted state, gastric pH, and individual patient metabolism. SUBACAP’s reformulation, using nanotechnology to produce a more soluble and consistently absorbed form, reduces that variability and allows a lower dose to achieve therapeutic blood levels.
The commercial result is a product that commands a premium over generic itraconazole despite being based on an off-patent molecule, because the enhanced formulation provides genuinely better clinical predictability. For a 505(b)(2) applicant, demonstrating that reduced variability in a pharmacokinetics study generates a period of FDA-granted exclusivity under the new clinical studies exclusivity provisions, typically three years, before competitors can reference the clinical data.
Key Takeaways for Segment 6
Nanoparticle-based super generics are defensible at the IP level because their therapeutic differentiation is inseparable from their manufacturing process. Abraxane’s nab platform, Doxil’s liposomal architecture, and SUBACAP’s solubility-enhanced itraconazole each illustrate how process IP and clinical differentiation reinforce each other. LNP patents from Alnylam and Arbutus remain the most significant freedom-to-operate constraint for any developer working with lipid nanoparticle delivery systems through the early 2030s.
Investment Strategy
Track the expiration timeline for Abraxane’s nab-technology process patents. The opening of that development window will generate a wave of 505(b)(2) nab-formulation applications that could produce significant market opportunity for first-to-file applicants. Analysts should also monitor Alnylam’s licensing posture on LNP patents, as the terms of those licenses will determine how many companies can economically develop LNP-based super generics in the second half of this decade.
7. Process Analytical Technology: PAT Patents, Real-Time Release, and Regulatory Leverage
7.1 PAT Technical Framework and Critical Quality Attribute Monitoring
Process Analytical Technology encompasses the instruments, analytical methods, and control algorithms used to measure Critical Quality Attributes (CQAs) in real time during manufacturing. The FDA’s PAT guidance, issued in 2004 and still operative, defines the framework as a system for designing, analyzing, and controlling manufacturing through timely measurements of critical quality and performance attributes of raw and in-process materials and processes.
The specific instruments used in PAT deployments cover a wide technical range. Near-infrared (NIR) spectroscopy measures drug concentration, moisture content, and polymorphic form in solid dosage forms without sample destruction. Raman spectroscopy provides complementary molecular fingerprinting with higher chemical specificity for API identification. Focused beam reflectance measurement (FBRM) monitors particle size distribution in wet granulation in real time. X-ray powder diffraction (XRPD), typically offline but increasingly inline for high-value products, monitors crystalline form transitions. Acoustic emission sensors detect mixing endpoint and granulation endpoint without physical contact with the batch.
The control architecture sitting above these sensors is where significant proprietary IP accumulates. A PAT system that integrates multi-sensor data into a real-time process control decision, adjusting granulation water addition rate based on a combination of NIR moisture readings and FBRM particle chord length distribution, requires a process control algorithm that is separately patentable from the sensors themselves.
7.2 Real-Time Release Testing: The Regulatory and Commercial Payoff
Real-Time Release Testing (RTRT) is the regulatory mechanism that converts PAT investment into commercial return. Under RTRT, the manufacturer uses in-process sensor data to certify product quality and release finished product without end-of-batch laboratory testing. The FDA’s Quality by Design framework explicitly supports RTRT as a natural outcome of a well-designed PAT-enabled process.
The commercial implications for generic manufacturers are significant. Traditional batch release in a well-run facility takes 14 to 21 days, incorporating laboratory testing, analyst review, and quality unit disposition. RTRT compresses that to hours. For a generic manufacturer managing a large portfolio of fast-moving products, reducing finished goods inventory holding time by two to three weeks per SKU generates substantial working capital benefits. For products with tight shelf lives, such as short-dated sterile injectables, RTRT can enable production-to-shipment cycles previously impossible with conventional testing.
The FDA’s CDER has published product-specific guidances for generic drug development that increasingly reference PAT methods as acceptable or preferred methodologies for demonstrating bioequivalence and CQA conformance. Manufacturers that invest in PAT infrastructure gain a regulatory alignment benefit: their processes generate the data types the FDA’s guidance documents describe, which streamlines the bioequivalence study design and ANDA technical sections.
7.3 PAT IP Strategy: Patents, Trade Secrets, and Competitive Advantage
PAT systems generate two categories of intellectual property. The first is patentable: novel sensor configurations, proprietary chemometric models trained on large datasets of production runs, and control algorithms that achieve demonstrably better process control than prior art. The second is trade secret: the specific calibration datasets, the sensor calibration protocols, and the trained multivariate models that take years of production data to develop and are not reconstructable by a competitor without that same production history.
For a generic manufacturer that invests in PAT over a decade, the trained chemometric models represent a significant competitive moat even after process patents expire. A competitor entering the same product space must build its own models from scratch, typically requiring 18 to 24 months of production data collection and validation before RTRT-ready model performance is achieved. This is a genuine, durable competitive advantage that does not appear on a patent dossier but is commercially real.
Key Takeaways for Segment 7
PAT investment generates two types of return: immediate cost reduction through cycle time compression and waste reduction, and durable competitive advantage through trained chemometric models that cannot be replicated quickly. RTRT-enabled operations reduce finished goods holding time by weeks, with direct working capital benefits. FDA product-specific guidances increasingly reference PAT methods, rewarding manufacturers that have aligned their process design with agency preferences.
8. AI and Machine Learning: Process Optimization IP and Regulatory Filing Considerations
8.1 AI in Process Control: What Pfizer and Moderna Have Actually Built
Pfizer’s generative AI platform for manufacturing process monitoring targets two specific metrics: a 10% improvement in product yield and a 25% reduction in cycle time. These are not aspirational projections. Pfizer’s manufacturing data science team has deployed AI-based anomaly detection across multiple production lines, with the system flagging deviations from golden batch process trajectories in real time and suggesting corrective actions to operators before out-of-specification conditions develop.
Moderna’s application of AI in COVID-19 vaccine production focused on automating quality control analyses that had previously required hours of manual review per batch. The automation reduced human analyst time and improved throughput for a production process that was scaling from zero to billions of doses in under 12 months. The technical architecture was not primarily AI for discovery; it was AI for quality data processing at manufacturing scale.
These two examples establish a pattern. AI’s highest near-term value in pharmaceutical manufacturing is not in design space exploration or formulation prediction. It is in reducing the cognitive load on quality analysts processing large volumes of in-process and end-of-batch data, and in flagging deviation patterns that human review would catch too slowly.
8.2 Digital Twins: Process Simulation IP and Its ANDA Implications
A digital twin is a computational model of a manufacturing process that is calibrated against real production data and runs in parallel with the physical process, predicting output quality from input parameter changes in real time. Siemens, Emerson, and a small number of specialized pharma technology vendors offer digital twin platforms. Pharmaceutical manufacturers that build proprietary digital twins for their specific processes own the model as a trade secret, even when the underlying simulation platform is licensed.
For ANDA filers, digital twin models have an emerging regulatory application. The FDA’s 2023 and 2024 guidance documents on model-informed drug development (MIDD) and in silico manufacturing models create a pathway for using validated computational models as supporting evidence in regulatory submissions. A generic manufacturer with a validated digital twin of its continuous manufacturing process can potentially support expanded Design Space claims in an ANDA with simulation data supplementing physical process characterization data.
This is early-stage regulatory science. FDA has not established a standardized acceptance criteria for digital twin models in ANDA submissions, but the direction of regulatory thinking is clear. Manufacturers building digital twin capabilities now are positioning for a regulatory environment that will reward them within three to five years.
8.3 AI/ML for Supply Chain and Sustainability
AI-driven supply chain optimization addresses two of the generic market’s most persistent vulnerabilities: API sourcing concentration and demand forecasting accuracy. Predictive analytics models that integrate supplier quality history, geopolitical risk indicators, and order lead time variability can identify API supply risk months before a physical shortage develops, allowing manufacturers to pre-position inventory or qualify alternative suppliers.
On the sustainability side, AI optimization of chemical dosing in manufacturing processes, energy load shifting to off-peak periods, and waste stream analysis for solvent recovery generate measurable cost savings that are also environmentally meaningful. These are not primarily marketing claims; they are operational cost reductions. For thin-margin generic manufacturers, AI-enabled energy and waste optimization can contribute meaningfully to EBITDA without requiring new product launches.
Key Takeaways for Segment 8
Pfizer’s documented AI targets, 10% yield improvement and 25% cycle time reduction, establish a benchmark for evaluating AI investment ROI in manufacturing contexts. Digital twins generate trade secret IP that will have growing regulatory value as FDA’s MIDD framework matures. AI supply chain tools address API concentration risk, the most frequently cited cause of generic drug shortages, with predictive lead time of months rather than days.
9. Manufacturing Technology Selection: Decision Framework for Portfolios
9.1 Product-Technology Matching Matrix
No single advanced manufacturing technology applies equally well to all generic product types. IP teams and portfolio managers benefit from a structured matching process that maps product characteristics against technology capabilities.
For high-volume solid oral dosage forms with stable demand profiles, continuous manufacturing generates the clearest return. The economics improve with production volume and product tenure; a product sold in hundreds of millions of units annually amortizes a $5 million CM line investment faster than one with volatile or fragmented demand.
For poorly water-soluble APIs, defined as belonging to BCS Class II or IV (low aqueous solubility), nanotechnology formulation approaches generate the largest clinical differentiation opportunity. These molecules cannot achieve therapeutic exposure through simple compression without formulation intervention. Nanoparticle encapsulation, amorphous solid dispersion via hot-melt extrusion, or lipid-based formulation can transform a commercially marginal compound into a viable super generic with 505(b)(2) exclusivity.
For pediatric and geriatric formulations requiring dose flexibility, 3D printing matches the clinical need precisely. Patient populations that cannot swallow standard tablets, or that require doses not available in commercial strengths, are natural target markets for on-demand 3D printing at pharmacy or hospital level.
For emergency-use products and pandemic-response drugs, modular manufacturing provides the fastest path to flexible capacity. The ability to deploy a GMP-compliant manufacturing suite in 12 months rather than 36 is the relevant variable in a public health emergency.
9.2 Build, License, or Partner: Capital Allocation Logic
Generic manufacturers face a genuine capital allocation tension. Building internal CM capability requires approximately $5 million per line plus the cost of regulatory validation and workforce training. Licensing access to a CMO’s existing CM infrastructure avoids the capital outlay but cedes process control and introduces supply dependency on the CMO’s scheduling and capacity priorities.
The optimal structure depends on product volume, product tenure, and the strategic importance of process IP ownership. For a product with 10 or more years of anticipated generic market tenure and annual revenue above $50 million, internal CM investment generates positive NPV with reasonable discount rates. For smaller or shorter-tenure products, CMO partnerships are more capital-efficient.
A hybrid model is emerging in the market: generic manufacturers invest in PAT and AI/ML process control capabilities internally while using CMO infrastructure for the physical manufacturing steps. This preserves the most defensible IP (the process control algorithms and chemometric models) internally while outsourcing capital-intensive equipment. The CMO relationship requires careful contractual management to ensure the internally developed process IP does not transfer to the CMO.
Key Takeaways for Segment 9
Technology selection should match product volume, API physicochemical class, patient population, and supply urgency. CM suits high-volume, stable oral solids. Nanoparticle formulation suits BCS Class II/IV super generics. 3D printing suits dose-flexible pediatric and geriatric products. Internal versus outsourced manufacturing decisions should center on which IP layer is most commercially durable, and that layer should be retained internally regardless of where physical production occurs.
10. Supply Chain Resilience: How AMTs Restructure Risk
10.1 The Concentration Problem in Generic Manufacturing
The American Progress data on drug shortages identifies market concentration as the primary structural driver. Over 75% of current shortages occur in markets with four or fewer manufacturers. When one of those manufacturers has a quality failure, a natural disaster affecting a facility, or an API supplier disruption, there is no reserve capacity to absorb the gap.
The FDA’s reporting on generic drug shortages consistently shows that quality and manufacturing problems, not raw demand spikes, cause the majority of clinically consequential shortages. The valsartan nitrosamine contamination, the heparin API concentration in Chinese facilities, and sartan API supply disruptions in 2018 and 2019 all trace to manufacturing quality failures or geographic API concentration, not demand volatility.
AMTs address this structurally. CM with integrated PAT reduces the probability of quality failures because the process generates real-time evidence of CQA conformance rather than batch-end sampling. Modular facilities reduce geographic concentration risk because they can be deployed rapidly in new regions. 3D printing’s distributed manufacturing model eliminates the single-facility concentration problem entirely for products where it is technically applicable.
10.2 API Sourcing Concentration: The Upstream Vulnerability
The United States depends on Indian and Chinese API manufacturers for approximately 80% of the APIs used in generic drugs. This concentration is the upstream analog of the facility concentration problem. A geopolitical disruption, a regulatory enforcement action by FDA or EMA at a major Indian or Chinese API facility, or a single contamination event can cascade into shortages across dozens of finished dosage form products that share the same API source.
AI-enabled supply chain monitoring tools can identify concentration risks by mapping API supplier networks and flagging situations where multiple finished products depend on a single API facility. But monitoring is not a solution; it is an early warning system. The structural solution is API supply diversification, which requires economic incentives for domestic or near-shore API production that do not currently exist at scale in the United States.
The Biosecure Act, introduced in 2024, represents a legislative signal that Congress is prepared to constrain Chinese API sourcing for national security reasons. Manufacturers with diversified API supply chains, or those investing in domestic API production partnerships, carry lower regulatory and geopolitical risk than those that have optimized purely for lowest-cost Chinese API sourcing.
10.3 Regulatory Risk Mapping: FDA Warning Letters and Facility Risk Scores
The FDA’s Pharmaceutical Quality System (PQS) and its Site Selection Model for routine inspections generate risk scores for manufacturing facilities based on compliance history, inspection frequency, and product criticality. Generic manufacturers whose facilities carry high FDA risk scores face elevated probability of Warning Letter issuance, import alerts, or consent decree requirements, any of which can cause supply disruption.
AMT adoption directly improves FDA facility risk scores in two ways. First, the real-time monitoring data generated by PAT systems provides the data integrity and process control evidence that FDA investigators prefer to see. Second, CM processes with built-in quality controls are inherently less vulnerable to the human error in manual process steps that accounts for a significant fraction of 483 observation categories.
Key Takeaways for Segment 10
Supply chain risk for generic drugs operates at three levels: API sourcing concentration, facility geographic concentration, and manufacturing process quality risk. AMTs address all three: CM and PAT reduce process quality risk, modular manufacturing reduces geographic concentration risk, and AI supply chain monitoring provides advance warning on API sourcing concentration. Regulatory risk scores are materially influenced by process control quality, making PAT investment a direct input to FDA inspection outcomes.
11. Investment Strategy for Institutional Analysts
11.1 Manufacturing Technology as a Portfolio Screening Variable
Institutional investors in generic pharmaceutical companies typically screen on ANDA pipeline depth, Paragraph IV challenge history, and gross margin trends. Manufacturing technology capability is underweighted in most screening models despite being a primary driver of the variables that are screened.
A generic manufacturer with a proprietary continuous manufacturing platform, a validated PAT infrastructure, and a digital twin capability has a structurally lower cost of goods sold trajectory than a batch manufacturer operating equivalent volumes. It also carries lower FDA inspection risk, lower probability of batch failure-driven inventory write-downs, and higher capacity utilization because CM equipment does not require the interstitial downtime of batch changeovers.
These operational advantages compound over a multi-year horizon. A company that reduces cost of goods by 8% through CM adoption in year one, reduces batch failure rates by 40% through PAT in year two, and reduces supply chain disruption risk through AI monitoring in year three generates EBITDA improvement that is not captured by ANDA count or generic pricing models.
11.2 Specific Metrics to Track
Analysts evaluating manufacturing technology maturity in generic companies should track the following specific indicators. Capital expenditure on AMT infrastructure as a percentage of total CapEx, disclosed in SEC filings or earnings calls, signals management commitment. FDA 483 observation categories at each facility, available through the FDA’s Establishment Inspection Report summaries under FOIA, indicate whether manufacturing quality issues are trending toward or away from process control deficiencies that AMTs address. Patent filing activity in IPC classes covering pharmaceutical manufacturing processes (A61K manufacturing methods, B01J process operations) indicates proprietary process IP development. CMO partnership announcements specifying technology types indicate whether a manufacturer is building CMO relationships around CM or 3D printing infrastructure.
11.3 The 505(b)(2) Super Generic Catalyst
For analysts tracking the super generic 505(b)(2) space, nab-technology patent expirations and the LNP patent licensing landscape are the two most consequential variables for the next five years. As Abraxane’s nab-technology process patents begin expiring in the mid-2020s, the development window for nab-based reformulations of other oncology APIs opens. The first companies to file 505(b)(2) applications using the nab platform for new cancer indications will benefit from three years of new clinical studies exclusivity, during which competitors cannot reference their clinical data.
On the LNP side, Alnylam’s settlement of its LNP patent disputes with Moderna (2022 settlement, Moderna paying royalties on COVID vaccine sales) established the commercial value of LNP IP. As those settlements mature and some LNP formulation patents begin expiring, a second-generation LNP super generic opportunity will develop. Companies that have built LNP manufacturing capabilities will be positioned to exploit that window faster than those starting from zero.
Key Takeaways for Segment 11
Manufacturing technology capability is an underweighted variable in generic company investment screening. AMT adoption reduces CoGS, FDA inspection risk, and supply chain disruption probability, all of which are direct drivers of the margin and pipeline variables that analysts do screen. Track CapEx allocation, FDA 483 trends, and process patent filings as forward indicators. The nab-technology and LNP patent expiration timelines are the most significant 505(b)(2) super generic catalysts on a five-year horizon.
12. Frequently Asked Questions
Q: What is the most immediate ROI opportunity in advanced manufacturing technology for a mid-sized generic manufacturer?
PAT implementation generates the fastest payback for most manufacturers. The combination of cycle time reduction through RTRT, batch failure rate reduction through real-time monitoring, and FDA inspection risk reduction through data integrity improvement typically delivers positive ROI within two to three years on most high-volume product lines. CM is higher-impact but requires longer payback periods and larger upfront capital commitments.
Q: How does a Paragraph IV filer analyze freedom-to-operate against manufacturing process patents?
The analysis requires searching independently for process patents in USPTO class A61K (pharmaceutical formulations) and B01J (chemical or physical processes), filtering by the brand manufacturer as assignee and by IPC codes relevant to the specific process category, such as continuous manufacturing, nano-carrier formation, or hot-melt extrusion. Standard Orange Book patent analysis misses this layer entirely. Legal counsel should conduct a separate process patent clearance opinion before committing capital to a CM or nano-formulation development program.
Q: What regulatory submission pathway applies to a super generic developed using nanotechnology?
Most nanotechnology-reformulated super generics file under Section 505(b)(2) of the FD&C Act rather than the ANDA pathway, because they seek to demonstrate clinical superiority or differentiation rather than simple bioequivalence. The 505(b)(2) pathway allows reliance on existing safety data for the RLD while requiring new clinical data for the differentiated attributes. FDA’s product-specific guidances for complex drug products, including liposomal and nanoparticle formulations, specify the physicochemical and clinical data requirements for each product category.
Q: How does the FDA define a ‘batch’ for a continuous manufacturing process?
The FDA’s draft CM guidance allows the manufacturer to define the batch as either a fixed time period of production or a specified quantity of material. The definition must be stated in the ANDA or NDA and validated through process characterization studies demonstrating that the defined batch size produces uniform product throughout the continuous run. This is a significant regulatory flexibility compared to the fixed-quantity batch definition required for conventional batch processes.
Q: What is the competitive significance of a trained PAT chemometric model as a trade secret?
A chemometric model for NIR or Raman spectroscopy that is trained on 18 to 24 months of production data from a specific manufacturing process cannot be replicated by a competitor without that same production history. The model contains embedded information about the specific API lot-to-lot variability, equipment response characteristics, and excipient interactions encountered during that production period. A competitor building a new production line must collect its own calibration dataset from scratch, a process that typically takes one to two years and requires ongoing refinement. This is a durable competitive advantage that does not depend on patent protection and does not expire.
Summary of Key Takeaways Across All Segments
The generic drug manufacturing landscape is shifting from a commodity competition on price to a multi-dimensional competition on process quality, supply chain resilience, and therapeutic differentiation. Continuous manufacturing, 3D printing, modular facilities, nanotechnology, PAT, and AI each address a distinct dimension of that competition.
Manufacturing process patents, trained chemometric models, digital twin IP, and distributed manufacturing network architectures are the asset classes that will define competitive positioning in this market through the 2030s. Companies that begin building those assets now, through internal development, strategic CMO partnerships, or targeted acquisitions, will hold structural advantages that commodity competitors cannot replicate on a short timeline.
The regulatory environment at FDA and EMA is moving in the direction that AMT-invested manufacturers want: QbD-aligned submissions, RTRT pathways, digital twin acceptance, and distributed manufacturing frameworks are all in active regulatory development. First movers in each technology category will benefit from deeper FDA dialogue, more refined guidance, and cleaner approval processes than late followers.
This pillar page references data from the FDA, EMA, Brookings Institution, PMC, American Progress, PharmTech, Drug Development & Delivery, MDPI, and DrugPatentWatch’s own research on ANDA strategy and generic market dynamics. For patent expiration intelligence, ANDA pipeline tracking, and Paragraph IV litigation history, visit DrugPatentWatch.com.