How generic and 505(b)(2) developers mine expired and lapsed pharmaceutical patents for low-cost market entry
The drug development world has a peculiar blind spot. Thousands of pharmaceutical patents lapse, expire, or get abandoned every year, often representing years of research, validated chemistry, and clinical proof-of-concept. Most of these assets sit in public records, unclaimed, while generic and 505(b)(2) developers spend millions chasing the same crowded blockbuster targets everyone else is watching.
This article is a practical guide for drug developers, business development professionals, and IP strategists who want to work a less-traveled path. We will cover what abandoned patents actually are, why they go unnoticed, how to find them systematically, what legal traps to avoid, and how to build a development strategy around them.
The opportunity is real. But it requires methodical research, solid freedom-to-operate analysis, and a clear understanding of the distinction between patent protection and regulatory exclusivity, two concepts that trip up even experienced developers.
Part One: The Anatomy of an Abandoned Pharmaceutical Patent
What “Abandoned” Actually Means
The word “abandoned” covers several distinct legal situations in patent law, and conflating them creates expensive mistakes. An abandoned patent is not automatically a free asset. The circumstances of abandonment determine your strategic options.
Under United States patent law, a patent application or issued patent can become abandoned through four primary mechanisms.
The first is failure to pay maintenance fees. Issued utility patents require maintenance fee payments at 3.5 years, 7.5 years, and 11.5 years from the grant date. Miss any of these payments, and the patent lapses. The United States Patent and Trademark Office (USPTO) publishes these lapses. A six-month grace period follows each deadline during which the patentee can pay a surcharge and revive the patent, so the window between a missed payment and a permanently abandoned patent runs roughly six months. After that window closes without payment, the patent enters the public domain, and the claimed invention becomes available for anyone to use.
The second mechanism is prosecution abandonment. A patent application can be abandoned during examination if the applicant fails to respond to an office action within the required period, typically three months extendable to six months with fees. Applications can also be abandoned expressly, when the applicant files a formal statement of abandonment. In pharmaceutical prosecution, express abandonment often happens when a company restructures its pipeline, loses funding, pivots to a different formulation, or decides the patent landscape does not support a viable commercial position.
The third mechanism is terminal disclaimer abandonment. This is subtle. A patent owner may disclaim the remaining term of a patent, often to overcome obviousness-type double patenting rejections. The practical effect is that the patent dies on a date earlier than its statutory term. Developers who rely only on the nominal expiration date without checking terminal disclaimers can miscalculate freedom to operate.
The fourth mechanism is abandonment by expiration of the statutory term. A patent has a maximum term of 20 years from its earliest effective filing date, subject to patent term adjustments and patent term extensions. When the term runs out, the patent expires. This is the most common form of “abandonment” developers encounter, and it is the least complicated legally, though it still comes with caveats around related patents.
Understanding which category applies to the patent you are evaluating is the first filter in any due diligence process.
Why Pharmaceutical Patents Get Abandoned Early
The pharmaceutical industry has an unusually high rate of early patent abandonment relative to other sectors, for reasons rooted in the economics of drug development.
Drug development takes time, money, and a tolerance for failure that few industries match. The probability of a drug candidate successfully moving from Phase I trials to approval sits at roughly 9.6 percent across all therapeutic areas, according to a 2019 MIT study published in Biostatistics [1]. When a candidate fails, the patents that protect it become liabilities rather than assets. Maintenance fees continue accruing even when there is no product generating revenue. Companies facing capital constraints routinely cull their patent portfolios to cut costs.
Pipeline restructuring is another driver. Large pharmaceutical companies regularly acquire smaller biotechs, bringing in patent portfolios that partially overlap with existing assets. Duplicative or strategically misaligned patents get abandoned. In some cases, the acquiring company already has a superior compound in the same therapeutic class and sees no reason to maintain a competitor’s older, weaker patents.
Regulatory failure is a third driver. A compound may have solid patent protection but a fatal flaw uncovered during clinical development. The company abandons the clinical program and stops paying maintenance fees on the relevant patents. The chemistry and the IP disappear into public records, but the scientific work that generated those patents, including published clinical data, remains accessible.
Licensing failure rounds out the picture. Small biotechs often patent compounds speculatively, hoping to license them to larger companies. When licensing efforts fail and the company lacks the capital to develop the drug independently, the patents lapse. The underlying science is often sound. <blockquote> “Of the roughly 7,000 to 9,000 pharmaceutical patent applications filed annually in the United States, a significant portion will never see a commercial product. Industry estimates suggest that fewer than 15 percent of patented pharmaceutical compounds ever reach the market.” — DrugPatentWatch analysis of USPTO pharmaceutical patent filings, 2023 [2] </blockquote>
The Difference Between Abandoned Patents and Expired Regulatory Exclusivity
This distinction cannot be overstated. Developers who confuse patent protection with regulatory exclusivity make errors that can delay or derail a product launch.
Patent protection and regulatory exclusivity are separate legal frameworks that often overlap but do not always align. A patent is a property right granted by the USPTO. Regulatory exclusivity is a market protection right granted by the FDA. They can coexist, one can outlast the other, or neither can apply.
The FDA grants several types of exclusivity for pharmaceutical products. Five-year new chemical entity (NCE) exclusivity applies when a drug contains an active moiety never previously approved by the FDA. Three-year exclusivity applies when a new clinical investigation is essential to approval. Orphan drug exclusivity runs seven years. Pediatric exclusivity adds six months to existing patents and exclusivities. None of these exclusivities depend on whether a patent is active or abandoned.
This means a drug compound can have all its patents abandoned while still sitting inside an active regulatory exclusivity period. A generic developer who identifies an abandoned patent on a drug still in its NCE exclusivity window cannot launch a product based on that patent without waiting for exclusivity to expire. The abandoned patent is irrelevant to that particular barrier.
The reverse also occurs. A compound’s regulatory exclusivities may have long since expired while continuation patents or method-of-use patents on the same drug remain active. Developers who focus only on regulatory exclusivity dates can walk into patent infringement.
Both analyses must run in parallel.
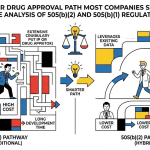
Part Two: The 505(b)(2) Pathway and Why Abandoned Patents Fit It Perfectly
A Quick Anatomy of 505(b)(2)
The 505(b)(2) regulatory pathway, established under the Hatch-Waxman Act and codified in 21 U.S.C. § 355(b)(2), allows an applicant to rely on published literature or on the FDA’s prior findings of safety and/or effectiveness for an approved drug, rather than conducting entirely new clinical trials.
This pathway sits between a traditional New Drug Application (NDA) and an Abbreviated New Drug Application (ANDA). A traditional NDA requires the applicant to provide complete clinical data from its own studies demonstrating safety and efficacy. An ANDA relies on bioequivalence to an existing approved reference listed drug (RLD). A 505(b)(2) NDA can rely on a combination of new studies and existing data, published or from the FDA’s review of a prior approval.
Products commonly developed through 505(b)(2) include new formulations of existing drugs, new dosage forms, new routes of administration, new combinations, and new indications. The pathway is also used for drugs where the active ingredient has been previously studied but not necessarily approved, or where the applicant wants to rely on published clinical literature rather than conducting its own Phase I through III trials.
The strategic leverage is significant. A 505(b)(2) NDA can reach approval with a fraction of the clinical investment required for a full NDA. According to analysis from the Tufts Center for the Study of Drug Development, 505(b)(2) approvals routinely occur three to five years faster than traditional NDA approvals [3]. The applicant still needs to characterize the product, conduct bridging studies, and address any safety signals, but the evidentiary burden is substantially lower.
Why Abandoned Patents Are Ideal 505(b)(2) Feedstock
Abandoned pharmaceutical patents have a specific attribute that makes them particularly valuable for 505(b)(2) development: they frequently come with a body of published data attached.
When a pharmaceutical company pursues a patent, it is typically because it has done the work to characterize a compound, validate a mechanism, or demonstrate a formulation advantage. That work generates data. Some of it ends up in the patent specification itself. Some of it ends up in published journal articles, conference presentations, and clinical trial disclosures. Some of it ends up in regulatory submissions that later become accessible through FDA’s summary basis of approval documents and drug approval packages.
A 505(b)(2) developer can use all of this. The published clinical data becomes the evidentiary foundation that the FDA considers. The chemistry in the patent specification helps characterize the compound. The clinical trial results help define the dose range and safety profile. All of this reduces the data generation burden on the developer.
The typical 505(b)(2) opportunity built around an abandoned patent works like this: a drug company patents a compound, conducts Phase II trials, publishes results, and then abandons the program because Phase III was too expensive, the market was too small, or the company went through a restructuring. The patents lapse. The clinical data sits in the literature. A 505(b)(2) developer can pick up the compound, rely on the published Phase II data, conduct the additional studies needed to fill FDA’s evidentiary requirements, and submit an NDA for approval.
This is not hypothetical. Developers like Supernus Pharmaceuticals, Assertio Therapeutics, and numerous others have built significant businesses using this approach. Noven Pharmaceuticals built its entire franchise on reformulating compounds backed by published literature.
The Hatch-Waxman Framework Around Abandoned Patents
Hatch-Waxman created an elaborate patent certification framework that governs generic entry. When a generic applicant files an ANDA, it must certify its relationship to each patent listed in the Orange Book for the reference drug. A Paragraph I certification states the patent information has not been filed. A Paragraph II certification states the patent has expired. A Paragraph III certification states the generic will not launch until the patent expires. A Paragraph IV certification states the patent is invalid, unenforceable, or will not be infringed.
Paragraph IV certifications trigger the possibility of 30-month stays and patent litigation. They are expensive and risky for generic applicants.
Abandoned patents eliminate much of this complexity. If a patent has lapsed due to non-payment of maintenance fees, a generic applicant who otherwise would have faced that patent can now file a Paragraph II certification, representing the patent has expired. No 30-month stay. No automatic litigation trigger.
For 505(b)(2) applicants, the analysis differs slightly. A 505(b)(2) applicant must also certify with respect to Orange Book-listed patents on the reference drug, but because the 505(b)(2) product is often meaningfully different from the RLD, the question of which patents apply and how they apply can be complex. An abandoned patent on the compound that the 505(b)(2) applicant wants to develop removes one potential barrier while the applicant’s own new formulation patents may provide a fresh period of exclusivity.
This is the full stack of the opportunity: an old compound with abandoned patents, validated by existing clinical data, reformulated or repositioned for a new indication or dosage form, submitted as a 505(b)(2) NDA, with fresh Orange Book-listable patents on the new formulation or method of use.
Part Three: Finding Abandoned Pharmaceutical Patents Systematically
The Databases You Need
Effective abandoned patent research requires working across multiple databases in a structured workflow. No single database captures the entire picture.
The USPTO Patent Center is the starting point for United States patent status. Patent Center replaced the older Patent Application Information Retrieval (PAIR) system and provides access to patent prosecution histories, current legal status, maintenance fee payment records, and related application chains. For any patent you are evaluating, the legal status section of Patent Center will show whether maintenance fees have been paid and whether the patent is currently active.
The USPTO Patent Full-Text and Image Database (PatFT) allows full-text searching of issued patents and published applications. You can search by assignee, inventor, classification code, and keywords. The CPC classification system is particularly useful for pharmaceutical searches. The A61K class covers pharmaceutical preparations, with numerous subclasses for specific drug types, formulations, and routes of administration.
Espacenet, maintained by the European Patent Office, provides access to patents from over 100 countries. For pharmaceutical development, European patent status matters because many compounds have both US and EP patents, and the abandoned-in-one-jurisdiction, active-in-another scenario creates geographic variation in your freedom to operate.
Google Patents provides a reasonably good interface for initial screening and cross-referencing, though it should not be relied on for definitive legal status determinations.
DrugPatentWatch specifically addresses the pharmaceutical patent analysis problem by integrating FDA Orange Book data with USPTO patent data, clinical trial information, and regulatory history. For developers focused on approved drugs, their generics pipeline, and upcoming patent cliffs, DrugPatentWatch provides a structured view of patent expiration timelines, abandoned patents on specific drugs, and the overall IP landscape around a given active ingredient. Analysts working in pharma competitive intelligence routinely use DrugPatentWatch as a first-pass tool before going deeper into USPTO records.
The FDA’s Drugs@FDA database provides access to approved drug products, their regulatory histories, and their approval packages. This is where you find the clinical data the original developer submitted and the FDA’s summary basis of approval, which can be a rich source of published pharmacokinetic, efficacy, and safety data for 505(b)(2) purposes.
ClinicalTrials.gov captures registered clinical trials, including studies on compounds that were later abandoned. A compound with failed patents but a registered clinical trial has a minimum floor of pharmacological and safety data that was generated under rigorous conditions.
Search Strategies That Actually Work
Keyword searching alone produces unreliable results in pharmaceutical patent research. Better approaches use a combination of strategies.
Start with the active ingredient. If you are interested in a specific therapeutic area and have identified a compound of interest, search the compound’s INN (International Nonproprietary Name) and its synonyms in USPTO PatFT. Review the result set for assignments to entities that no longer operate or have been acquired. Cross-reference those patent numbers against Patent Center to check maintenance fee status.
Work backward from known generic approvals. When the FDA approves a generic drug, it means the relevant patents on the reference listed drug have expired or have been successfully challenged. DrugPatentWatch maintains a database of Orange Book patent expirations that lets you see which active ingredient patents have recently expired and which have been abandoned before their nominal expiration dates. The gap between a patent’s filing date and its actual abandonment date tells you whether the patent was abandoned early, which is a signal of commercial failure or pipeline restructuring.
Search assignee abandonment patterns. Some pharmaceutical companies are known to aggressively prune their patent portfolios. If you identify a company that has undergone bankruptcy, merger, or therapeutic area exit, systematically search their patent portfolio against Patent Center to find which patents have lapsed. Portfolio-level searches can surface clusters of related abandoned patents representing an entire development program that a company shut down.
Search by CPC classification in concert with legal status filters. The USPTO’s classification system lets you narrow to specific drug types and formulations. Running a classification search with a filter for abandoned status produces a manageable, targeted list rather than an overwhelming result set.
Use citation analysis. When you find one relevant abandoned patent, review both the patents it cites and the patents that cite it. Patents in the same citation network are likely to cover related compounds, formulations, or methods of use. Some of these will be abandoned. Some will be active. The citation network gives you a map of the IP landscape around the compound.
Reading the Orange Book for Abandoned Patent Intelligence
The Orange Book, formally titled “Approved Drug Products with Therapeutic Equivalence Evaluations,” is a primary tool for pharmaceutical patent intelligence. The FDA updates it monthly and lists patents submitted by NDA holders for approved products.
The Orange Book does not list all patents related to a drug. It only lists patents that the NDA holder has certified as claiming the approved drug product or method of using it. This means the Orange Book is a conservative view. There may be additional patents related to the compound that are not listed, either because the NDA holder chose not to list them or because they are related to upstream chemistry not covering the final product.
The gap between the Orange Book and the full USPTO record is where abandoned patent opportunities often live. A drug may have one Orange Book-listed composition patent that expires in five years, but three additional composition patents that were abandoned five years ago. Those abandoned patents may have covered the compound in forms that could be useful for a 505(b)(2) reformulation, and they are now free for use.
Reading the Orange Book for abandoned patent intelligence requires several steps. First, identify the active ingredient and the approved products. Second, look at all listed patents for those products and their expiration dates. Third, go to USPTO and search for all patents assigned to the NDA holder covering the same compound, not just Orange Book-listed patents. Fourth, check maintenance fee status on each identified patent. Fifth, compare the Orange Book patent list against the full USPTO result set to identify patents covering the compound that are not Orange Book-listed and that have lapsed or been abandoned.
This comparison often surfaces abandoned formulation patents, abandoned method-of-use patents, and abandoned process patents. Each represents a different type of opportunity.
Part Four: The Legal Minefield — What Abandoned Does Not Mean
Freedom to Operate Is Not Automatic
An abandoned patent gives you freedom from that specific patent. It does not give you freedom to operate in the broader field. This distinction matters enormously, and professionals who treat it casually invite litigation.
Freedom to operate (FTO) is a legal analysis that determines whether practicing a particular invention would infringe any valid, unexpired patent held by a third party. An FTO analysis on a compound you identified through an abandoned patent search must examine the full patent landscape around that compound, not just the abandoned patent itself.
The most common mistake is treating the abandoned patent as the entire story. Consider a scenario: a pharmaceutical company patents a compound in 2005, conducts Phase II trials, abandons the patents in 2012 when the program fails, and publishes the clinical results in a peer-reviewed journal. A 505(b)(2) developer in 2024 identifies those abandoned patents and begins development. What else is out there?
Other companies may have patented formulations of the same compound after seeing the published clinical data. Other companies may hold method-of-use patents covering the indication the developer wants to pursue. The compound’s metabolites may be separately patented. Polymorphic forms of the compound may be protected. Process patents covering manufacturing methods may be active. Any of these can constrain development even when the original composition patents are abandoned.
FTO analysis requires a systematic search of all potentially relevant patents, not just the abandoned ones, followed by a claim-by-claim analysis of active patents that intersects with your proposed product and manufacturing process. This work requires a qualified patent attorney with pharmaceutical chemistry background. The cost of thorough FTO analysis is significantly less than the cost of a patent infringement lawsuit.
The Continuation Patent Problem
Continuation patents are a specific hazard in pharmaceutical patent landscapes, and they are frequently misunderstood by non-patent professionals.
When a patent is filed, the applicant can file one or more continuation applications claiming the same invention with different or narrower claims, or continuation-in-part (CIP) applications adding new matter while continuing to claim earlier priority. These continuation applications share the same priority date as the parent application, but they can be filed at any point while the parent or a related case is pending.
This creates a practical problem: a drug company can file a parent patent application in 2000, let that patent expire in 2020, and simultaneously keep continuation applications pending in prosecution throughout that period. The continuation cases inherit the 2000 priority date but have their own 20-year term from their own filing dates. A continuation filed in 2018 from a parent filed in 2000 would not expire until 2038.
In pharmaceutical patent prosecution, this practice is common. Large pharma companies often maintain “patent thickets” around their commercial products by keeping a chain of continuation applications alive throughout the product’s commercial life. Even when the original compound patents expire, continuation patents covering specific formulations, dosage ranges, patient populations, or methods of administration may remain active for years.
The FTO analysis for a compound with a complex prosecution history must trace every continuation and divisional application in the family. USPTO Patent Center’s continuity data shows the parent-child relationships between related applications. This tracing exercise often surfaces active patents that casual searchers miss.
Orphan Drug and Other Exclusivities That Outlast Patents
Regulatory exclusivities from the FDA operate independently of the patent lifecycle and can block generic or 505(b)(2) entry long after all relevant patents have expired or been abandoned.
Orphan drug exclusivity deserves particular attention. When the FDA designates a drug as an orphan drug (for a disease or condition affecting fewer than 200,000 persons in the United States), the approved product receives seven years of market exclusivity for that indication. No generic or 505(b)(2) product can be approved for the same indication during that period, regardless of patent status. A developer who identifies an abandoned compound and wants to pursue the same orphan indication must check orphan drug exclusivity dates carefully.
Pediatric exclusivity adds six months to the term of any existing patent or exclusivity. When the FDA awards pediatric exclusivity, it attaches to all listed Orange Book patents for the product. An abandoned patent cannot receive pediatric exclusivity, but active patents on the same product can be extended by six months in ways that affect when generic entry is possible.
New chemical entity exclusivity blocks any ANDA or 505(b)(2) NDA from being submitted for four years, and prevents approval for five years, from the date of original approval. If the abandoned compound you want to develop was approved as an NCE, check the approval date and the NCE exclusivity period.
Three-year exclusivity (from clinical investigation) applies when approval of a new application is based on new clinical investigations essential to approval. Even reformulations of existing drugs can receive three-year exclusivity. If the original developer received three-year exclusivity before abandoning the compound, that exclusivity period affects your timeline.
Data Exclusivity Under 505(b)(2) and the Reference Listed Drug Problem
505(b)(2) applicants face a specific regulatory exclusivity issue around their reference listed drug. When a 505(b)(2) NDA relies on the FDA’s findings of safety and effectiveness for an approved drug, that reliance is tied to the reference listed drug (RLD). If the RLD has active regulatory exclusivities, the 505(b)(2) approval can be blocked.
For abandoned compound opportunities where the compound was never approved as an RLD, the situation is different. A developer pursuing a 505(b)(2) based on published literature for a compound that was studied but never approved does not face RLD exclusivity barriers in the same way. The FDA’s review of the published data is the basis for approval, not the FDA’s prior findings for an approved product.
This distinction makes “studied but not approved” compounds particularly attractive for 505(b)(2) developers. There is no Orange Book, no listed patents, no active NCE exclusivity, and often a robust published literature base. The main IP risk is the continuation patent problem described above.
Part Five: Case Studies in Abandoned Patent Exploitation
Case Study 1: Carbomer Formulations and the Ophthalmic Market
The ophthalmic drug market offers a clear illustration of how 505(b)(2) developers mine abandoned or expired formulation patents for commercial opportunity. Carbomer-based gels for ophthalmic use were extensively patented in the 1980s and 1990s. As those patents aged and maintenance fees lapsed on secondary formulation patents, developers began combining the abandoned formulation IP with new clinical data to support 505(b)(2) NDAs.
Akorn, Inc. built a significant portion of its ophthalmic portfolio by identifying compounds and formulations whose original IP had lapsed and whose clinical data was available in the literature. This approach required meticulous FTO analysis to ensure no relevant continuation patents remained active, but the reward was a defensible market position with significantly lower development costs than a traditional NDA would require.
The pattern demonstrates a principle: even in crowded therapeutic areas, formulation patents age and lapse at different rates, creating windows where a developer with strong formulation chemistry and an FDA regulatory team can enter the market profitably.
Case Study 2: Old Antifungals, New Life
Amphotericin B is a classic antifungal compound first developed in the 1950s. Its composition patents expired decades ago. But the formulation story is more recent. Conventional amphotericin B has significant nephrotoxicity. In the 1990s and early 2000s, developers created lipid-based formulations, including liposomal, lipid complex, and colloidal dispersion versions, to reduce toxicity while maintaining efficacy.
By the mid-2010s, some of those lipid formulation patents began reaching the end of their terms or lapsing for non-payment. Developers looking at lipid formulation technologies for antifungal drugs had an aging but substantive clinical literature to draw on, and the original formulation patents were either expired or abandoned.
This created a 505(b)(2) development opportunity. A developer who could establish bioequivalence or pharmacokinetic comparability to AmBisome or Abelcet, the approved liposomal and lipid complex formulations, could potentially use the published clinical literature on those products as the evidentiary foundation for a 505(b)(2) NDA on a new lipid formulation.
The competitive advantage came not from chemistry but from regulatory strategy. Understanding which formulation patents were abandoned, which were active, and how the FDA would view a new lipid formulation versus the approved RLDs required intensive analysis of both the patent landscape and the regulatory history.
Case Study 3: CNS Compounds and the Attrition Pipeline
Central nervous system drug development has one of the highest attrition rates in the industry. Phase II failure rates for CNS compounds are particularly high. The result is an unusually large inventory of compounds with abandoned patents but published Phase II data.
Consider a hypothetical scenario that reflects a real pattern: a biotech company in 2008 patents a glutamate modulator for major depressive disorder. Phase II results show modest efficacy with an acceptable safety profile. The company cannot raise the capital for Phase III, and investors exit. The patents lapse by 2014. The Phase II data is published in a peer-reviewed journal in 2015.
A 505(b)(2) developer reviewing that published data in 2024 would see a compound with ten years of post-publication literature, no active composition patents, and clinical proof-of-concept for an indication with substantial unmet medical need. If the FTO analysis is clean, the path to a 505(b)(2) NDA involves additional Phase II work to address gaps in the FDA’s evidentiary requirements, formulation development, and potentially a Phase III study if the published data does not meet the FDA’s efficacy standard for the proposed indication.
The cost of that development program is substantially lower than starting from scratch. The compound’s pharmacokinetics, safety profile in humans, and preliminary efficacy signal are already characterized. The developer is compressing the risk curve.
This is the CNS attrition pipeline opportunity in concrete terms. Tools like DrugPatentWatch, combined with PubMed and ClinicalTrials.gov searches, can identify specific compounds fitting this pattern. The database work is not glamorous, but it is productive.
Case Study 4: The Reformulation of Colchicine
Colchicine is not a textbook case of abandoned patent exploitation, but it is a case study in how regulatory strategy around old drugs creates IP opportunity. Colchicine had been used for gout for centuries and was marketed in the US without formal FDA approval. URL Pharma conducted clinical trials on a specific formulation and dosage regimen, obtained FDA approval for Colcrys under a 505(b)(2) pathway, and received three-year exclusivity based on those new clinical investigations.
The lesson for developers is not to replicate URL Pharma’s specific play, but to recognize the principle: a drug with no active patents can still generate significant commercial value and IP protection through a 505(b)(2) NDA backed by new clinical data. The clinical investigation generates the exclusivity. The old compound provides the development head start.
Developers who identified similar “marketed without approval” compounds in the wake of FDA’s enforcement action against Colcrys saw an entire market segment of opportunity open up. The FDA’s drug efficacy study implementation (DESI) reviews and unapproved drug enforcement policies created a steady stream of compounds that needed formal approval but had substantial literature behind them.
Part Six: Building a Systematic Abandoned Patent Development Strategy
The Screening Funnel
Effective abandoned patent hunting works through a series of filters that progressively narrow a large universe of patents to a small set of viable development opportunities. Running this funnel systematically produces better results than ad hoc searching.
The first filter is therapeutic area focus. Developers with manufacturing capabilities, regulatory expertise, and commercial relationships in oncology will find more value in abandoned oncology patents than in abandoned cardiology patents, regardless of the abstract opportunity in cardiology. Matching abandoned patent opportunity to your organization’s capabilities is a prerequisite for rational prioritization.
The second filter is patent status verification. Once you have a target list of compounds in your therapeutic area with potentially lapsed or abandoned IP, verify patent status in USPTO Patent Center for each relevant patent. Do not rely on secondary sources or databases alone for definitive status determination. Patent Center is the authoritative source.
The third filter is exclusivity status. Cross-reference against FDA’s Drugs@FDA and the Orange Book to check for active regulatory exclusivities. A compound with abandoned composition patents but active NCE exclusivity is not available for generic or 505(b)(2) development until exclusivity expires.
The fourth filter is literature depth. A compound with abandoned patents but minimal published clinical data will require more development investment to reach a 505(b)(2) NDA. Use PubMed to search for published clinical studies on the compound. Check ClinicalTrials.gov for registered trials, including completed ones with results. The richer the literature base, the lower your development cost.
The fifth filter is FTO assessment. Commission a preliminary FTO analysis on the short list of compounds that survive the first four filters. This is where you invest professional time and money because it determines whether the opportunity is legally accessible. A preliminary FTO analysis typically costs $10,000 to $25,000. A thorough FTO analysis with formal opinion costs more. Both are orders of magnitude cheaper than discovering an infringement problem after development investment.
The sixth filter is market assessment. An abandoned compound with clean IP, solid literature, and achievable development costs still needs a commercial market. Evaluate the competitive landscape, the unmet medical need, reimbursement prospects, and the competitive response likely from existing market participants.
Compounds that pass all six filters are genuine development candidates. In practice, the funnel will be quite tight. You may screen hundreds of abandoned patents to find five to ten viable candidates.
Due Diligence Checklist for 505(b)(2) Candidates from Abandoned Patents
Patent due diligence for a 505(b)(2) candidate from an abandoned patent has specific components that differ from standard pharmaceutical M&A patent due diligence.
First, identify all patents in the same family as the abandoned patent. This means searching for parent applications, divisional applications, continuation applications, and CIP applications using the continuity data in Patent Center. Check the status of each.
Second, identify patents held by third parties that cover the same compound or related compounds. Search both by the compound’s INN and by its chemical structure using SciFinder, Reaxys, or STN. Chemical structure searches surface patents that use different nomenclature for the same compound.
Third, identify any active patents on the specific formulation you plan to develop. If you are formulating an old compound as an extended-release product, search for active patents on extended-release formulations of that compound held by any party.
Fourth, review the prosecution history of the abandoned patent for any file history estoppel issues. During prosecution, applicants sometimes make statements or amendments narrowing claim scope. These statements can create prosecution history estoppel that affects what claims the patent covers and what arguments a developer can make regarding their own formulation’s relationship to the patented technology.
Fifth, search for any inter partes review (IPR) or post-grant review proceedings related to the abandoned patent or related patents. PTAB proceedings create an additional layer of prior art and claim limitation analysis.
Sixth, check for any existing court decisions involving the abandoned patent or related patents. A patent may have been invalidated, or claims may have been construed in litigation in ways that affect your analysis.
Seventh, review the FDA’s drug approval package for any compound that was approved and is related to your target compound. Approval packages sometimes contain information about manufacturing methods or formulation characteristics that affects patent analysis.
Working with FDA on Abandoned Compound 505(b)(2) Applications
The FDA has specific expectations for 505(b)(2) NDAs that rely on published literature. Understanding those expectations before you begin development saves time and avoids costly surprises during review.
The most important document for this purpose is FDA’s guidance on 505(b)(2) applications. The current version, “Guidance for Industry: Applications Covered by Section 505(b)(2),” was issued in October 1999 and supplemented by subsequent guidance documents on specific issues [4]. FDA has also issued topic-specific guidance on bioequivalence studies, drug-drug interaction studies, and other clinical components that affect 505(b)(2) content.
For compounds with published Phase II data, the FDA’s typical expectation is that the 505(b)(2) applicant either replicate the pivotal efficacy finding in a new controlled trial or explain why the published evidence is adequate to establish substantial evidence of effectiveness. The FDA has accepted literature-based 505(b)(2) applications in some cases, but the bar is higher when the published literature consists of small, single-center studies rather than multi-center randomized controlled trials.
A pre-submission meeting (Type B meeting) with FDA is advisable before investing in pivotal studies. In this meeting, you can present your proposed development plan, your reliance on published data, and your proposed clinical endpoint strategy. The FDA’s response will tell you whether your proposed evidence package is likely to be sufficient or whether you need additional studies.
The FDA’s position on bioavailability and bioequivalence for modified formulations of abandoned compounds has evolved. For formulations that are sufficiently different from the original compound (different route of administration, significantly different pharmacokinetic profile), the FDA may not require bioequivalence to the original formulation and may instead evaluate the product on its own clinical data.
Part Seven: Generic Drug Development Through Abandoned Patents
ANDA Development and the Abandoned Patent Advantage
For generic drug developers, the most direct benefit of an abandoned patent is the simplified Paragraph II certification path. When a composition patent on the RLD has expired or lapsed, the generic applicant can certify Paragraph II for that patent, avoiding the 30-month stay and automatic patent litigation risk that a Paragraph IV certification triggers.
The commercial strategy for ANDA development around abandoned patents focuses on identifying drugs where the last relevant Orange Book-listed patents have lapsed before their expected expiration date. This can happen when an NDA holder acquires a product from another company and determines that maintaining all the listed patents is not worth the cost, particularly for products with declining sales. The NDA holder stops paying maintenance fees, the patents lapse, and the path for generic entry opens years ahead of schedule.
Tracking this requires monitoring maintenance fee payment records for Orange Book-listed patents. The USPTO publishes maintenance fee due dates and records payments. A development team that monitors Orange Book patents for missed maintenance fee payments can identify early-entry opportunities before competitors. DrugPatentWatch provides this type of patent cliff and expiration tracking, which is particularly useful for competitive intelligence teams scanning for early generic entry windows.
The first generic applicant to file an ANDA for an unpatented drug product with an unapproved application can receive 180-day generic exclusivity under specific circumstances. Understanding the precise Hatch-Waxman rules governing when this exclusivity applies in the context of abandoned patents requires regulatory counsel with Hatch-Waxman expertise.
Para IV Settlements and Their Effect on the Landscape
One phenomenon that creates abandoned patent opportunity for subsequent generic applicants is the Paragraph IV settlement. When a first generic applicant files a Para IV certification and the branded company sues, settlement negotiations sometimes result in agreements where the branded company licenses the generic to enter the market before patent expiration in exchange for the generic dropping its patent challenge.
These settlements are scrutinized by the FTC under pay-for-delay or reverse payment analyses following the Supreme Court’s 2013 decision in FTC v. Actavis. But a less-noticed effect of these settlements is that they sometimes include provisions where the branded company abandons specific patents as part of the deal. When that happens, those abandoned patents open the door for subsequent generic applicants who did not participate in the settlement.
A second generic applicant who files an ANDA after the first-filer settles needs to certify against all Orange Book-listed patents. If some patents were abandoned as part of the settlement, the second-filer’s certification path is simpler. Tracking patent abandonment events in the aftermath of Para IV settlements is a specific competitive intelligence activity that can reveal entry opportunities for second-wave generic applicants.
Authorized Generics and Abandoned Patent Gaps
Brand pharmaceutical companies sometimes launch authorized generics, manufactured by the brand but sold at generic prices, to compete with the first-filing generic applicant during its 180-day exclusivity period. Authorized generics do not require FDA approval separate from the NDA and do not face patent certification requirements the same way ANDAs do.
When the brand’s patents on its own product have been abandoned, the authorized generic strategy becomes more complicated for the brand. Without valid patents to assert, the brand cannot automatically trigger 30-month stays against generic applicants, and its ability to use patent-based legal strategy to manage generic entry is reduced. The abandonment of composition patents by the brand company, whether strategic or accidental, can shift negotiating leverage toward generic applicants.
Developers building an ANDA strategy should map not just the composition patents but the entire patent portfolio of the NDA holder, checking for abandoned patents that the brand might have intended to use as litigation leverage. A brand company that has inadvertently abandoned a key patent through maintenance fee failure has weakened its defensive position even if it does not immediately recognize the consequences.
Part Eight: Market Sizing the Opportunity
How Many Abandoned Pharmaceutical Patents Are Out There?
The universe of abandoned pharmaceutical patents in the United States is substantial. As of 2024, the USPTO’s pharmaceutical patent database (CPC class A61K) contains hundreds of thousands of issued patents, a significant fraction of which have been abandoned, either through maintenance fee lapse, term expiration, or express abandonment.
A 2022 analysis by Quinn Emanuel, drawing on USPTO data, estimated that approximately 30 to 40 percent of pharmaceutical patents that reach grant are abandoned before their full 20-year term, either due to maintenance fee non-payment or express abandonment [5]. This suggests that tens of thousands of pharmaceutical composition, formulation, and method-of-use patents have entered the public domain in the last decade alone.
The pharmaceutical patent filing rate in the US runs between 7,000 and 9,000 new pharmaceutical patent applications per year [2]. Even if the abandonment rate for applications is lower than for issued patents, the cumulative abandoned application universe represents an enormous potential source of prior art and development opportunity.
Not all of these abandoned patents represent viable development opportunities. Many cover compounds that failed for reasons unrelated to business or capital constraints, including genuine safety failures, insurmountable toxicity, or lack of efficacy that no reformulation will fix. But even a small fraction of that universe, say 5 to 10 percent of abandoned patents representing compounds with viable development profiles, translates to thousands of potential opportunities.
Financial Leverage of the Abandoned Patent Model
The financial case for developing products from abandoned pharmaceutical patents rests on two drivers: lower development costs and compressed timelines.
Development cost advantages come from multiple sources. The compound’s chemistry and initial preclinical toxicology are already characterized. This eliminates or significantly reduces the cost of discovery and early-stage preclinical studies. Published clinical data reduces the Phase I and Phase II burden. For a 505(b)(2) based on a compound with solid Phase II literature, the developer may be able to go directly to Phase III with a more focused clinical program, bypassing some or all of the early clinical work.
Tufts Center for the Study of Drug Development estimates the fully loaded cost of developing a new pharmaceutical compound through approval at approximately $2.6 billion, including the cost of capital and accounting for failed programs [6]. A 505(b)(2) NDA based on an abandoned compound with published Phase II data can potentially reduce the clinical development portion of that cost by 60 to 80 percent, because the developer is not funding the discovery, preclinical, and early clinical phases from scratch.
Timeline compression compounds the financial advantage. A shorter development timeline means earlier market entry, more years of commercial exclusivity, and lower carrying costs on the capital invested in development. For a product that takes seven years rather than twelve years to reach approval, the commercial return per year of exclusivity is substantially higher.
Risk-adjusted return comparisons between the abandoned patent model and the traditional NDA model consistently favor the abandoned patent approach, provided the FTO analysis is clean and the regulatory strategy is well-designed. The attrition rates are lower because the compound has already demonstrated a human proof-of-concept. The development costs are lower. The timeline is shorter. The trade-off is that the competitive barriers, particularly patent protection on the final product, must be engineered into the 505(b)(2) application through new formulation or method-of-use patents rather than relying on composition-of-matter patents.
Therapeutic Area Distribution of Opportunity
Abandoned pharmaceutical patent opportunity is not evenly distributed across therapeutic areas. The concentration of abandoned patents follows the inverse of commercial success: therapeutic areas with high failure rates produce the most abandoned patents.
Central nervous system disorders top the list. CNS drug development has the lowest probability of success of any therapeutic area, with Phase II success rates below 50 percent for most indications. The result is a large inventory of abandoned CNS patents covering compounds that demonstrated biological activity in preclinical models or early clinical trials. Many of these compounds have been validated for central nervous system penetration, safety margins, and receptor engagement, making them attractive starting points for repositioning.
Oncology follows closely. Cancer drug development has high Phase III failure rates, particularly for solid tumor indications. But oncology also has an unusually high rate of companion diagnostic development, biomarker characterization, and mechanistic research that surrounds each failed clinical program. The result is that abandoned oncology patents often come with a rich body of mechanistic literature that can support new clinical strategies.
Anti-infectives, particularly antibiotics, have seen substantial patent abandonment as commercial incentives for antibiotic development have deteriorated. The antimicrobial resistance crisis creates unmet medical need, but the commercial model for antibiotics is broken enough that companies have abandoned antibiotic programs regardless of clinical promise. This creates an opportunity for developers who can access government incentives, including BARDA funding and advanced market commitments, to pick up abandoned antibiotic patents and advance development with non-traditional funding structures.
Cardiovascular and metabolic disease abandonment patterns reflect both the high cost of cardiovascular outcomes trials and the competitive intensity of the market. Many cardiovascular compounds patented in the 1990s and 2000s lapsed as clinical programs ran into efficacy or safety issues in large trials. Some of these compounds may have utility in specific patient subpopulations that were not the primary focus of the original clinical program.
Part Nine: Common Mistakes and How to Avoid Them
Mistaking the Abandoned Patent for the Cleared Field
The single most expensive mistake in abandoned patent development is treating the abandoned patent as proof of a clear field. It is not. An abandoned patent removes one specific IP barrier. It does not remove continuation patents, related patents held by third parties, active method-of-use patents, or any other IP constraint on the field.
Developers who skip or shortcut FTO analysis in favor of a quick abandoned patent screen are taking a risk that can materialize as expensive post-commitment litigation. The discovery that a key manufacturing step is covered by an active patent held by a competitor, after development investment, is a painful and avoidable outcome.
The right approach is to run the abandoned patent search and the FTO analysis as parallel workstreams. The abandoned patent search tells you where development opportunity might exist. The FTO analysis tells you whether that opportunity is actually accessible.
Misreading Maintenance Fee Records
Maintenance fee records in Patent Center require careful interpretation. A missed maintenance fee deadline does not immediately mean the patent is abandoned. The USPTO allows a grace period of six months after each maintenance fee due date during which the patent can be revived by paying a surcharge. A patent that missed its 7.5-year maintenance fee deadline on January 1 remains revivable through June 30 of the same year.
If you identify a pharmaceutical patent with a missed maintenance fee payment during the grace period, you cannot rely on that patent being permanently abandoned until the grace period expires. Check the dates carefully. If the grace period is still open, the patent may be revived by the owner at any time.
Revived patents are another hazard. The USPTO allows petitions for revival of unintentionally abandoned patents even after the grace period in limited circumstances. A developer who begins investing in a program based on an abandoned patent should monitor USPTO records for any revival petition. While revival after significant time is relatively uncommon, it does occur, particularly for patents that were abandoned due to clerical errors or law firm mistakes rather than deliberate business decisions.
Ignoring Foreign Patent Rights
A pharmaceutical compound can have US patents that have lapsed while corresponding foreign patents remain active. If your product is intended for export, or if your manufacturing takes place in another country, active foreign patents can create infringement exposure that the abandoned US patents would not have created.
European Patent Office patents covering the same compound may have different term dates than US patents due to differences in filing dates, supplementary protection certificates (SPCs), and national patent law variations across EU member states. An SPC can extend a European patent’s effective term by up to five years. A US patent that has expired may correspond to a European compound patent still protected by an SPC.
Conduct parallel patent status analysis in each geographic market where you plan to develop, manufacture, or commercialize the product. This is particularly important for API manufacturing. If you source your active ingredient from a manufacturer in India, but the compound is covered by an active Indian patent, your manufacturing supply chain may have an IP problem regardless of US patent status.
Underestimating Regulatory Exclusivity Complexity
Developers occasionally calculate their market entry date based solely on patent expiration without checking regulatory exclusivity dates. The regulatory due diligence should always run in parallel with IP due diligence.
Check Drugs@FDA for every related approved product to identify the original approval date, formulation history, and any subsequent supplemental approvals that may have generated new exclusivity periods. Check the Orange Book for currently listed exclusivities. Contact FDA’s Center for Drug Evaluation and Research (CDER) if there is any ambiguity about exclusivity status.
One specific trap is the three-year exclusivity attached to supplemental NDAs. If the original developer filed a supplemental NDA adding a new indication or a new patient population shortly before abandoning its patents, the three-year exclusivity for that supplement may still be active even though the compound patents have lapsed. Depending on what your 505(b)(2) application relies on for approval, that supplemental NDA exclusivity could delay your approval.
Treating Published Phase II Data as Sufficient for Phase III
The published Phase II data that makes an abandoned compound attractive for 505(b)(2) development may not satisfy FDA’s requirements for substantial evidence of efficacy. Phase II trials are typically designed to establish proof of concept, explore dose-response relationships, and generate hypotheses about efficacy, not to provide the definitive evidence FDA requires for approval.
FDA’s standard for substantial evidence of effectiveness requires “adequate and well-controlled investigations.” For most indications, this means at least two adequate and well-controlled trials, typically Phase III randomized controlled trials, demonstrating effectiveness. The FDA has approved applications based on a single trial in some cases, usually when the trial is particularly robust or when the unmet medical need is compelling, but this is the exception rather than the rule.
Before committing to a development program based on published Phase II data, engage with FDA in a pre-IND or Type B meeting to discuss the adequacy of the published data as the evidentiary foundation for your proposed application. The FDA’s response will tell you whether you need one or two Phase III trials, whether the published Phase II data can substitute for Phase II work you would otherwise need to conduct, and what specific study designs and endpoints FDA will find persuasive.
Part Ten: Competitive Intelligence and Real-Time Monitoring
Setting Up a Patent Monitoring System
Building a systematic monitoring system around abandoned pharmaceutical patents requires infrastructure and process discipline. One-time searches produce one-time results. A monitoring program produces ongoing competitive intelligence.
The core components of an effective monitoring system are patent status alerts, competitive assignee tracking, and therapeutic area surveillance.
Patent status alerts can be set up through USPTO’s Patent Center notification system and through commercial patent monitoring services. Configure alerts for maintenance fee due dates and payment events for specific patents, for new publication events related to compounds in your target therapeutic areas, and for assignment changes that might indicate patent portfolio restructuring.
Competitive assignee tracking monitors the patent filing and abandonment activity of specific pharmaceutical companies. When a competitor files a cluster of new continuation applications in a therapeutic area you are watching, that is a signal the competitor is trying to extend coverage beyond an aging patent family. When a competitor stops paying maintenance fees on a cluster of related patents, that is a signal of portfolio pruning and potential opportunity.
Therapeutic area surveillance uses CPC classification-based searches to monitor new publications and status changes in specific drug categories. Configure alerts to notify you when patents in specific CPC subclasses transition to lapsed or abandoned status.
DrugPatentWatch’s monitoring features are specifically designed for pharmaceutical competitive intelligence, providing alerts on patent expiration, regulatory exclusivity changes, and Orange Book updates. For teams without the resources to build a custom monitoring system, DrugPatentWatch provides a structured pharmaceutical-specific framework that combines multiple data sources into a single view.
Analyzing Competitor Portfolio Abandonment Signals
When a large pharmaceutical company abandons a cluster of related patents in a specific therapeutic area, it is typically a signal of one of several strategic developments: therapeutic area exit, clinical program failure, pipeline rationalization post-merger, or capital constraints.
Each of these scenarios creates different development opportunities. A therapeutic area exit by a major company often means the compound or compounds it was developing will not be advanced by the original developer or by any well-capitalized successor. This is the highest-probability scenario for viable development by a smaller company or a 505(b)(2) developer.
Clinical program failure requires more investigation. If a compound failed Phase III for safety reasons, the published data will reveal those safety issues, and development should not proceed without understanding whether the safety issue is compound-specific or formulation-specific, addressable or fundamental. If the compound failed for efficacy reasons in a heterogeneous patient population, published data may support a narrower indication in a biomarker-defined subpopulation where the compound’s mechanism of action is most relevant.
Post-merger rationalization often creates the cleanest opportunities. When two large companies merge, their combined patent portfolios include redundancies. The patents covering compounds that did not fit the merged company’s commercial strategy get abandoned. These compounds were not development failures; they were strategic deprioritizations. The underlying chemistry and clinical data are often solid.
Monitor M&A activity in your target therapeutic areas and systematically review the patent portfolios of acquired companies three to five years post-merger. This is when maintenance fee decisions are typically made and when abandoned patents from rationalized programs begin appearing in USPTO records.
Part Eleven: Building the Business Case
Financial Modeling for Abandoned Patent Programs
A financial model for an abandoned patent development program needs to capture four drivers that distinguish it from a traditional NDA program: lower input costs, shorter timeline, different risk profile, and thinner patent moat on the output.
The lower input cost comes from the combination of eliminated discovery spending, reduced preclinical spending, and potentially reduced early clinical spending. A compound with published Phase II data can often enter the 505(b)(2) development program at the Phase IIb or Phase III stage, avoiding the cost of Phase I and Phase IIa trials.
Quantifying this reduction requires a bottom-up comparison of the development activities needed for the abandoned compound program versus a traditional full NDA program. The cost of compound discovery typically runs $5 million to $15 million per compound that reaches clinical development. Phase I trials cost $1 million to $10 million. Phase IIa trials cost $5 million to $15 million. Eliminating or reducing these components has meaningful financial impact.
The shorter timeline compresses the period during which capital is tied up in development before it can generate returns. A program that reaches approval in six years rather than twelve years generates the same annual revenue stream but starts it six years earlier. The net present value of that earlier start, discounted at a rate that reflects pharmaceutical development risk, can be substantial.
The different risk profile is both an advantage and a risk factor. The advantage is that a compound with published Phase II data demonstrating proof-of-concept has a lower probability of Phase III failure due to lack of efficacy. The FDA’s analysis of the published data in a Type B meeting can give you an early warning of regulatory concerns that would otherwise surface in Phase III. The risk is that the published data was generated by another organization under protocols you did not design, and replicating those results may be harder than it appears.
The thinner patent moat on the output is a genuine competitive disadvantage. A drug developed through a 505(b)(2) pathway from an abandoned compound patent cannot be protected with composition-of-matter patents because the compound is in the public domain. Protection relies on formulation patents, method-of-use patents, manufacturing process patents, and regulatory exclusivity. These are weaker protections than composition-of-matter patents and are more susceptible to design-around by competitors.
Model the competitive response. How long before a well-capitalized competitor can bring an alternative product to market using the same abandoned compound? What regulatory exclusivity periods protect your product? What formulation advantages can you patent that would create a barrier to competitive entry?
Partnership and Licensing Structures
Not every organization developing an abandoned patent opportunity needs to take the program all the way to approval and commercialization independently. Licensing and partnership structures can allow smaller developers to advance programs to a value inflection point and then partner with a larger company for Phase III funding and commercialization.
For abandoned compound programs, the natural value inflection point is Phase II completion. A developer who takes an abandoned compound through formulation development, IND filing, Phase I safety study, and Phase IIb efficacy study has substantially de-risked the program from where it started. The Phase IIb results either confirm or refute the original developer’s proof-of-concept. Either outcome is informative.
If Phase IIb confirms efficacy and safety, the program is at a point where it can attract Phase III partnership from a company with the clinical operations capacity and commercial infrastructure to take it to approval. The license value at that stage reflects the de-risked clinical data, the clean IP position, the regulatory exclusivity available on approval, and the commercial market size.
The valuation framework for these partnerships typically uses a risk-adjusted NPV approach, applying probability-of-success rates for Phase III completion and FDA approval to the projected commercial value of the approved product. Royalty rates, milestone payments, and upfront payments are then negotiated relative to that adjusted value.
For generic ANDA programs from abandoned patents, the partnership structure is different. The value driver is manufacturing cost and speed to market, not clinical data. ANDA partnerships often involve contract manufacturing organizations (CMOs) that manufacture the drug product and marketing companies that distribute it. The IP position is not a source of partnership value the way it is in a 505(b)(2) context; instead, the value is in being first to file and first to market.
Part Twelve: Regulatory Exclusivity as the New Moat
Designing Exclusivity Into 505(b)(2) Programs
When composition-of-matter patents are not available because the compound is in the public domain, 505(b)(2) developers need to design regulatory exclusivity strategically. This means understanding which regulatory pathways generate exclusivity and structuring the development program to qualify.
Three-year new clinical investigation exclusivity is the most accessible form of exclusivity for 505(b)(2) programs. The FDA grants three-year exclusivity when the approval of an NDA or supplement is based on new clinical investigations, other than bioavailability studies, that were essential to approval. For a 505(b)(2) NDA that includes new clinical data to support a new formulation, new dosage strength, or new route of administration, three-year exclusivity applies.
The scope of three-year exclusivity is narrower than NCE exclusivity. Three-year exclusivity only blocks FDA approval of a generic application for the same product (same drug, same conditions of use). It does not prevent another 505(b)(2) applicant from conducting its own clinical trials and obtaining approval for a similar product. In a market where the underlying compound is free for anyone to develop, three-year exclusivity protects your specific approved product from direct copies but does not prevent a competitor from developing a different formulation of the same compound.
New chemical entity (NCE) exclusivity is not available for abandoned compound programs, because the compound has previously been studied in humans and the active moiety has been characterized. The NCE exclusivity definition requires that no previously approved drug contained the active moiety.
Orphan drug designation is available for conditions affecting fewer than 200,000 US patients and can generate seven-year exclusivity for the designated indication. If the abandoned compound you are developing targets an orphan disease, applying for orphan drug designation early in the development program can layer a seven-year market exclusivity on top of any three-year new clinical investigation exclusivity. This combination creates a meaningful competitive barrier.
Pediatric exclusivity is available when the FDA requests pediatric studies and the applicant conducts them and submits a pediatric report. This adds six months to existing patents and exclusivities. For a 505(b)(2) program with limited patent protection, the pediatric exclusivity extension on three-year exclusivity adds six months to your protected window.
Patent Strategy for 505(b)(2) Developers
A 505(b)(2) developer working from an abandoned compound needs to generate new patentable innovations during the development process. The formulation itself, the drug delivery system, specific manufacturing processes, methods of treating specific patient populations, combination products, and specific dosage regimens can all be patentable.
The goal is to list these new patents in the Orange Book at the time of approval. Orange Book-listed patents trigger the Hatch-Waxman certification process for any subsequent generic applicant, requiring them to file Paragraph III certifications (wait for your patents to expire) or Paragraph IV certifications (challenge your patents as invalid or not infringed) before they can get an ANDA approved.
Well-crafted formulation patents are among the most defensible in this context. A specific polymeric matrix that produces a controlled release profile, a particular lipid formulation that improves bioavailability, or a specific salt form that offers manufacturing advantages can be patented regardless of the compound being in the public domain. The key is that the formulation innovation must be genuinely non-obvious, not merely a routine adaptation of existing formulation technology.
Method-of-use patents covering specific dosing regimens, patient selection criteria, or combination use are another layer of protection. These patents are weaker than formulation patents because they can be circumvented by prescribers through off-label use, but they still trigger the Hatch-Waxman process and require a generic applicant to challenge them.
Process patents covering specific manufacturing steps can be listed in the Orange Book if they claim the “approved drug substance or drug product.” These are the narrowest category but can be important when the manufacturing process provides a quality or cost advantage that competitors cannot easily replicate.
Part Thirteen: The Regulatory Submission Package
Structure of a 505(b)(2) NDA for an Abandoned Compound
Building the 505(b)(2) NDA submission package for an abandoned compound has specific characteristics that distinguish it from a traditional NDA submission.
Module 2 of the Common Technical Dossier (CTD), which contains summaries of the quality, nonclinical, and clinical data, must explain clearly what data comes from your own studies and what data comes from published literature or from prior FDA findings. This transparency is a regulatory requirement and allows the FDA to evaluate the adequacy of the published data as part of the evidentiary package.
For published literature, the FDA expects the applicant to provide copies of the published studies and an analysis of their quality and relevance. Published studies conducted under Good Clinical Practice (GCP) standards are weighted more heavily than older studies conducted before GCP requirements were widespread. Studies with appropriate placebo controls, adequate sample sizes, pre-specified primary endpoints, and published results from a complete study (not just a conference abstract) provide a stronger evidentiary foundation.
Module 5, the clinical data module, will contain your own new studies alongside published study reports. The clinical overview in Module 2.5 must integrate the published data with your own data to present a coherent picture of safety and efficacy.
The pharmacokinetic bridging data is often the most critical element of a 505(b)(2) submission. If your product’s formulation is different from the formulations used in the published clinical studies, you need to demonstrate the pharmacokinetic relationship between your formulation and the formulation for which efficacy data exists. This bridging may involve relative bioavailability studies comparing your formulation to the formulation used in the pivotal published trials.
CMC Considerations for Abandoned Compound Programs
Chemistry, manufacturing, and controls (CMC) requirements for an abandoned compound program have both advantages and challenges compared to a traditional NDA.
The advantage is that the compound’s chemical properties are likely well-characterized in the literature. Solubility, stability, polymorphic forms, degradation pathways, and spectroscopic characteristics may already be published, reducing the characterization work you need to conduct. Published patent specifications sometimes contain detailed synthetic routes and analytical characterization data that jump-starts CMC development.
The challenge is that the original developer’s proprietary manufacturing process is not available to you. If the original company used a specific synthetic route or a specific crystallization method that produced optimal particle size characteristics, you need to develop your own process that achieves equivalent or superior product quality without access to the original developer’s trade secrets.
This is where process patents held by the original developer, even if abandoned, remain important references. The patent specification describes the process the original developer used. You can use this information to understand the technical requirements for the process and to design a different approach that achieves the same quality outcomes.
Bioanalytical method development is another CMC-adjacent consideration. You will need validated analytical methods to support your pharmacokinetic studies. Published literature may describe analytical methods for measuring the compound in biological matrices, which can serve as a starting point for your bioanalytical method development.
Part Fourteen: The Future of Abandoned Patent Opportunity
AI-Driven Patent Mining and What It Changes
The application of artificial intelligence to pharmaceutical patent analysis is changing both the scale and the efficiency of abandoned patent discovery. Machine learning models trained on patent text, chemical structure databases, and clinical outcome data can screen large patent databases for abandoned patents with specific characteristics far faster than human analysts.
Tools that combine natural language processing with chemical structure recognition can identify semantic similarities between compound claims in abandoned patents and compounds being studied in current clinical programs, surfacing potentially relevant prior art that would be difficult to find through keyword searches alone.
The practical effect for abandoned patent mining is that the competitive landscape for identifying these opportunities is changing. Organizations with AI-assisted patent analysis capabilities can screen the entire pharmaceutical abandoned patent universe in a fraction of the time it would take manual analysts. This compresses the window in which an organization can identify and act on an abandoned patent opportunity before competitors find the same target.
For smaller developers without dedicated AI capabilities, the shift means relying more heavily on specialized databases that have incorporated analytical tools, including platforms like DrugPatentWatch that provide structured pharmaceutical patent and regulatory data with analytical features, or partnering with IP analytics firms that have built machine learning tools for pharmaceutical patent analysis.
The fundamentals of the opportunity do not change with AI. You still need FTO analysis, regulatory strategy, clinical development, and manufacturing. But the front-end discovery and prioritization work, the database mining and initial screening, is becoming faster and more systematic. Organizations that invest in that front-end capability will find opportunities earlier in the cycle.
International Harmonization and Cross-Border Abandoned Patent Opportunities
The global pharmaceutical market creates abandoned patent opportunities that span jurisdictions in ways that domestic-only analysis misses. A compound whose US patents have lapsed but whose EP patents remain active can still be developed and commercialized in the US market. The US market exclusivity is available regardless of what happens in Europe, as long as US patent analysis supports development.
Similarly, a compound with active US patents but lapsed EP patents might be manufacturable in the EU for export to markets where the compound is unprotected, subject to applicable import and patent law analysis in each relevant jurisdiction.
The increasing use of Patent Cooperation Treaty (PCT) applications and the harmonization of patent office examination procedures across major jurisdictions has created a more interconnected global patent landscape. When a company abandons a PCT application during the national phase, it typically abandons patent rights simultaneously in multiple jurisdictions. Tracking PCT application abandonment can surface global development opportunities in a single research step.
Key Takeaways
Abandoned pharmaceutical patents represent a real and systematically accessible source of low-risk development opportunity, but they require disciplined analysis to exploit correctly.
An abandoned patent means that specific patent is no longer enforceable. It does not mean the compound is free of all IP constraints. Freedom-to-operate analysis across all related patents, including continuations, divisionals, and third-party patents, is mandatory before any development commitment.
Regulatory exclusivity is a separate legal framework from patent protection. Check Drugs@FDA and the Orange Book for active exclusivities on every compound you evaluate, regardless of patent status.
505(b)(2) developers benefit from abandoned compound patents most when the compound has a published clinical literature base. The published data reduces development cost and timeline by replacing clinical studies the developer would otherwise need to conduct from scratch.
Generic developers benefit most from abandoned Orange Book-listed patents, which convert Paragraph IV certification hurdles into Paragraph II certifications and reduce the litigation risk associated with ANDA filings.
The financial model for abandoned patent programs is differentiated by lower input costs, shorter timelines, and a thinner patent moat on the output. The regulatory exclusivity strategy must compensate for the inability to secure composition-of-matter patent protection.
Database tools including USPTO Patent Center, DrugPatentWatch, Drugs@FDA, ClinicalTrials.gov, and PubMed form the core research infrastructure for systematic abandoned patent identification. No single database is sufficient.
The opportunity set is large and underworked, particularly in CNS, oncology, and anti-infectives. Organizations that build systematic monitoring programs and combine IP analysis with regulatory and clinical development expertise are positioned to access this opportunity more effectively than those conducting one-off searches.
FAQ
Q1: How do you know if a missed maintenance fee payment has permanently abandoned a patent versus just being in the grace period?
Check the maintenance fee due date in USPTO Patent Center and compare it to today’s date. The USPTO’s maintenance fee page for each patent shows the due date for each payment period. A patent misses its deadline on the due date, but the USPTO allows a six-month grace period after each deadline during which the patentee can pay a surcharge and revive the patent. If you are within six months of the original due date, the patent may still be revived. If more than six months have passed since the due date without payment or a revival petition, the patent is permanently abandoned absent extraordinary circumstances. Always verify in Patent Center’s transaction history to confirm no surcharge payment or revival petition has been filed.
Q2: Can a 505(b)(2) application rely on clinical data from a failed Phase III trial that was published in the literature?
Yes, but the FDA will scrutinize the failed trial carefully. A trial that failed its primary endpoint does not constitute evidence of effectiveness for that endpoint. However, a failed Phase III may still contain useful safety data, pharmacokinetic data, or evidence of activity in a subpopulation. The 505(b)(2) applicant can rely on published Phase III data for the portions of the evidence package where that data is informative, while providing separate studies or published data to address the indication’s efficacy requirement. A pre-IND or Type B meeting with FDA is essential to determine how the failed trial data will be weighed and what additional evidence FDA requires before the applicant commits to a clinical program.
Q3: What is the risk that an original patent owner can revive an abandoned pharmaceutical patent after we have already started development based on its abandoned status?
The risk is real but manageable. The USPTO allows petitions for revival of unintentionally abandoned patents. The standard for revival is that the abandonment must have been unintentional, meaning the patentee did not deliberately choose to abandon the patent. If a patent was abandoned because the patentee’s attorney missed a deadline due to a clerical error, the patentee can petition for revival even years later. If a patent was abandoned because the company decided not to pay the maintenance fee as a business decision, revival as “unintentional” is much harder to establish. For development programs based on abandoned patents, monitor USPTO records for revival petitions on the specific patents you are relying on. If the abandonment appears to have been a deliberate business decision rather than an administrative error, the revival risk is low.
Q4: How does the orphan drug designation strategy interact with the abandoned compound development approach, and what are the timing considerations?
Orphan drug designation can be applied for at any point before NDA submission, making it compatible with abandoned compound programs. The key timing consideration is that orphan drug exclusivity attaches to the approved drug for the designated orphan indication. If you develop an abandoned compound for an orphan indication and obtain both orphan drug exclusivity (seven years) and three-year new clinical investigation exclusivity, you have a meaningful combined exclusivity window. Apply for orphan drug designation early in the development program, before your IND is filed if possible, to establish your designation date. If a competitor is developing the same abandoned compound for the same orphan indication, the first company to obtain approval for that indication receives the seven-year orphan exclusivity, which blocks approval of the competitor’s application for the same indication.
Q5: What documentation should a developer preserve when it begins working with an abandoned pharmaceutical patent to protect its position against future IP disputes?
Document the date on which you confirmed the patent’s abandoned status through USPTO Patent Center, and retain a screenshot or printed record of the Patent Center transaction history showing lapsed maintenance fee status. Document your FTO analysis process and conclusions, including the identity of the attorney who conducted the analysis and the date of the opinion. If you rely on the patent specification for technical information about the compound’s chemistry, document that the patent is in the public domain and that your use of the technical disclosure is legally authorized. If you develop improvements or modifications to the compound or formulation that you subsequently patent, document the timeline of your innovation relative to the date of abandonment of the original patent, establishing that your innovations postdate the original patent and were independently developed. This documentation supports your position in any subsequent dispute about whether your development activity infringed active IP or relied properly on public domain material.
Citations
[1] Wong, C. H., Siah, K. W., & Lo, A. W. (2019). Estimation of clinical trial success rates and related parameters. Biostatistics, 20(2), 273-286. https://doi.org/10.1093/biostatistics/kxx069
[2] DrugPatentWatch. (2023). Pharmaceutical patent filing and abandonment analysis: USPTO pharmaceutical patent trends 2013-2023. DrugPatentWatch Intelligence Reports. https://www.drugpatentwatch.com
[3] Tufts Center for the Study of Drug Development. (2017). Optimizing the path forward: 505(b)(2) new drug applications. Tufts CSDD Impact Report, 19(1). https://csdd.tufts.edu
[4] U.S. Food and Drug Administration. (1999, October). Guidance for industry: Applications covered by section 505(b)(2). FDA Center for Drug Evaluation and Research. https://www.fda.gov/media/72419/download
[5] Quinn Emanuel Urquhart & Sullivan, LLP. (2022). Pharmaceutical patent portfolio management: Abandonment trends and strategic implications. Quinn Emanuel Patent Practice Group Report.
[6] Tufts Center for the Study of Drug Development. (2016). Cost to develop and win marketing approval for a new drug is $2.6 billion. Tufts CSDD Impact Report, 18(1). https://csdd.tufts.edu