GILEAD SCIENCES INC Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GILEAD SCIENCES INC, and what generic alternatives to GILEAD SCIENCES INC drugs are available?
GILEAD SCIENCES INC has twenty-three approved drugs.
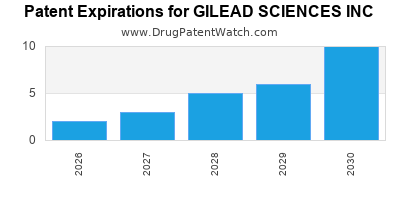
There are eighty-two US patents protecting GILEAD SCIENCES INC drugs.
There are two thousand two hundred and twenty-eight patent family members on GILEAD SCIENCES INC drugs in sixty-six countries and three hundred and seventy supplementary protection certificates in nineteen countries.
Summary for GILEAD SCIENCES INC
| International Patents: | 2228 |
| US Patents: | 82 |
| Tradenames: | 18 |
| Ingredients: | 18 |
| NDAs: | 23 |
| Drug Master File Entries: | 7 |
| Patent Litigation for GILEAD SCIENCES INC: | See patent lawsuits for GILEAD SCIENCES INC |
Drugs and US Patents for GILEAD SCIENCES INC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | 7,964,580*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | HARVONI | ledipasvir; sofosbuvir | PELLETS;ORAL | 212477-002 | Aug 28, 2019 | RX | Yes | Yes | 8,735,372*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | RX | Yes | Yes | RE44599 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Gilead Sciences Inc | GENVOYA | cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 207561-001 | Nov 5, 2015 | RX | Yes | Yes | 8,754,065*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | SOVALDI | sofosbuvir | TABLET;ORAL | 204671-001 | Dec 6, 2013 | RX | Yes | Yes | 9,085,573*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | HARVONI | ledipasvir; sofosbuvir | PELLETS;ORAL | 212477-001 | Aug 28, 2019 | RX | Yes | No | 9,393,256*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for GILEAD SCIENCES INC
Paragraph IV (Patent) Challenges for GILEAD SCIENCES INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 200 mg/25 mg/300 mg | ➤ Subscribe | 2015-05-20 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2015-12-09 |
| ➤ Subscribe | Tablets | 150 mg, 200 mg, and 250 mg | ➤ Subscribe | 2012-05-17 |
| ➤ Subscribe | Tablets | 150 mg, 150 mg, 200 mg, 300 mg | ➤ Subscribe | 2018-10-04 |
| ➤ Subscribe | Tablets | 300 mg | ➤ Subscribe | 2010-01-26 |
International Patents for GILEAD SCIENCES INC Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2014040471 | ⤷ Try a Trial |
| Norway | 20071745 | ⤷ Try a Trial |
| Philippines | 12016502499 | ⤷ Try a Trial |
| Iceland | 7977 | ⤷ Try a Trial |
| Japan | 6571256 | ⤷ Try a Trial |
| South Korea | 20200067930 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for GILEAD SCIENCES INC Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2595980 | 53/2020 | Austria | ⤷ Try a Trial | PRODUCT NAME: REMDESIVIR ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ ODER ESTER DAVON; REGISTRATION NO/DATE: EU/1/20/1459 (MITTEILUNG) 20200703 |
| 1564210 | 203 5022-2013 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR ALEBO JEHO HYDRAT, SOLVAT, TAUTOMER ALEBO FARMACEUTICKY PRIJATELNA SOL; NAT. REGISTRATION NO/DATE: EU/1/13/830/001 - EU/1/13/830/002 20130527; FIRST REGISTRATION: EU EU/1/13/830/001 - EU/1/13/830/002 20130527 |
| 2430014 | 2020046 | Norway | ⤷ Try a Trial | PRODUCT NAME: LEDIPASVIR - FORLENGET SPC; REG. NO/DATE: EU/1/14/958 20141203 |
| 1564210 | SPC/GB13/065 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR; REGISTERED: UK EU/1/13/830/001 20130527; UK EU/1/13/830/002 20130527 |
| 2937350 | CA 2020 00060 | Denmark | ⤷ Try a Trial | PRODUCT NAME: REMDESIVIR ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/20/1459 20200703 |
| 1564210 | 13C0061 | France | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR OU UN HYDRATE, SOLVATE, TAUTOMERE, OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CEUX-CI; REGISTRATION NO/DATE: EU/1/13/830/001-002 20130527 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.