Introduction: Beyond Inert—The Strategic Imperative of Excipient Selection
In the intricate and high-stakes world of pharmaceutical development, the journey from a promising active pharmaceutical ingredient (API) to a successful, marketable drug product is fraught with scientific, regulatory, and commercial hurdles. For decades, the focus of this journey has understandably been on the API—its efficacy, its safety, its mechanism of action. The other components of the final dosage form, the excipients, were often relegated to a secondary, almost passive role. Derived from the Latin excipere, meaning ‘to except’ or ‘other than’, these substances were traditionally viewed as inert fillers, binders, and lubricants, necessary for manufacturing but pharmacologically inconsequential.1 On average, these “inactive” ingredients constitute about 90% of a given drug product, a silent majority whose influence was profoundly underestimated.2
Today, this paradigm has been irrevocably shattered. The modern understanding, born from decades of hard-won experience, clinical setbacks, and advanced analytical science, is that excipients are highly functional, critically important components that profoundly influence a drug’s entire lifecycle.3 They are the unsung architects of a drug product’s performance, governing everything from manufacturing efficiency and shelf-life stability to clinical bioavailability and, ultimately, patient adherence and safety.1 The selection of excipients is no longer a downstream technical task but a foundational and strategic element of drug development, a complex dance of chemistry, biology, and engineering where the competitive advantage is often found in the details.3
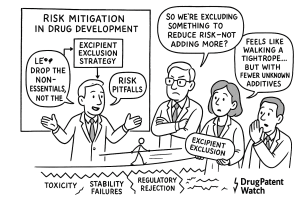
This report introduces and elaborates on a proactive, risk-based framework for navigating this complexity: the Excipient Exclusion Filter. This is a systematic, strategic decision-making process where formulators, R&D leaders, and business development teams proactively screen and eliminate potentially problematic excipients at the earliest stages of development. This approach moves beyond reactive troubleshooting—addressing an unexpected adverse event in a clinical trial or a failed stability batch—to a state of proactive, intelligent risk mitigation. By applying filters such as “lactose-free,” “gelatin-free,” “alcohol-free,” or “dye-free” based on a deep understanding of the API, the target patient population, and the commercial strategy, companies can de-risk their development pipelines, accelerate timelines, and create safer, more effective, and more valuable medicines.
The business case for this patient-centric approach to formulation is no longer theoretical; it is a commercial imperative. In an increasingly competitive market where patients are more informed and empowered, a drug’s formulation is a key differentiator. A product that meets the specific needs of sensitive populations—whether due to allergies, intolerances, ethical beliefs, or age-related physiological differences—is not just a medical treatment but a complete therapeutic solution. As industry analysis underscores, misalignment with patient needs can directly impact commercial success.
“Poor patient centricity has a direct impact on company success — not listening to the “Voice of the…source businesses.”
— Arthur D. Little, “Why patient centricity is key to long-term pharma company success,” Prism, 2024 6
Studies have shown that drugs developed using patient-centric processes are significantly more likely to succeed; one analysis found they were 20% more likely to launch successfully.7 “Free-from” formulations are one of the most tangible and impactful expressions of this patient-centric philosophy. The Excipient Exclusion Filter framework provides the technical and strategic roadmap to achieve this. By applying this framework, pharmaceutical companies transform excipient selection from a potential liability into a strategic asset. It forces an early alignment of R&D with commercial, regulatory, and intellectual property (IP) strategy, preventing costly late-stage reformulations and creating differentiated products with a stronger value proposition.8 This report will provide an exhaustive guide to understanding, building, and implementing this framework to mitigate risk, enhance patient safety, and secure a decisive competitive advantage in the modern pharmaceutical landscape.
The Excipient Risk Landscape: Identifying Hidden Liabilities in Your Formulation
The foundational shift in modern pharmaceutics is the acknowledgment that no excipient is truly inert. While these substances are included for their functional properties and are not intended to have a therapeutic effect, their chemical and physical nature means they can, and often do, interact with the API, the patient’s body, and the manufacturing environment in unexpected and sometimes detrimental ways.1 Understanding the full spectrum of these potential risks is the first step in building an effective mitigation strategy. The excipient risk landscape can be broadly categorized into three primary domains: adverse patient reactions, API-excipient incompatibilities, and the dangers posed by impurities and concomitant components.
Deconstructing the “Inert” Myth: Common Adverse Reactions and Hypersensitivities
The most direct and concerning risk posed by excipients is their potential to cause adverse reactions in patients. These reactions are not pharmacological effects in the traditional sense but are rather intolerances or true immunological hypersensitivities. The clinical manifestations can range from mild, transient discomfort to severe, life-threatening events.2 With one study noting that a staggering 92.8% of oral medicines contain at least one ingredient with the potential to act as an allergen, the scope of this issue is vast.12 Formulation scientists must be acutely aware of the most common culprits.
- Fillers and Diluents: These excipients provide bulk to the dosage form. Lactose, the most common filler in the world, is a primary concern for the large portion of the global population with lactase deficiency, which can lead to gastrointestinal symptoms like diarrhea, flatulence, and abdominal pain.14
Wheat starch, while less common, contains trace amounts of gluten and must be avoided by patients with celiac disease or a true wheat allergy.14 - Preservatives: Added to multi-dose liquid and semi-solid formulations to prevent microbial growth, preservatives are frequent provocateurs of hypersensitivity. Parabens (methylparaben, propylparaben) used in creams and oral liquids are linked to delayed hypersensitivity reactions like contact dermatitis.3
Benzyl alcohol, used in injectable and inhalant formulations, has been implicated in both local and systemic allergic reactions and is particularly dangerous in neonates.11
Sodium metabisulphite, an antioxidant, is known to cause hypersensitivity reactions, including bronchospasm and anaphylaxis in susceptible individuals.2 - Dyes and Colorants: Used for aesthetics and product identification, colorants are a well-documented source of adverse reactions. Tartrazine (FD&C Yellow No. 5) has been associated with hypersensitivity reactions, urticaria (hives), and the aggravation of asthma.2 Other synthetic azo dyes have also been implicated.19
Carmine, a red dye derived from insects, has been reported to cause anaphylaxis.18 - Stabilizers and Surfactants: These are crucial for complex formulations, especially biologics and injectables. Polyethylene glycol (PEG) and structurally related polysorbates (e.g., Polysorbate 80) have gained significant attention for their potential to cause hypersensitivity reactions, including anaphylaxis, as highlighted during the rollout of certain COVID-19 vaccines.11
- Other Sensitizers: A variety of other excipients can cause reactions in specific populations. Lanolin (wool fat) in topical products can cause skin hypersensitivity.2
Sesame oil and arachis (peanut) oil, used as vehicles in some injections, are potent allergens that must be clearly labeled and avoided in allergic patients.2
API-Excipient Incompatibility: A Threat to Stability and Bioavailability
Beyond direct patient harm, excipients can pose a significant risk to the drug product itself. Due to the intimate and prolonged contact between the API and excipients within a dosage form, the potential for chemical and physical interactions is immense.22 Such incompatibilities can lead to the degradation of the API, a reduction in its potency, the formation of toxic byproducts, or altered physical properties that negatively impact bioavailability and therapeutic performance.23
The mechanisms of these interactions are diverse and often subtle, requiring deep chemical knowledge to anticipate and prevent.
- Maillard Reaction: This is a classic and well-understood incompatibility. It is a chemical reaction between the amino group of an API (specifically, primary or secondary amines) and a reducing sugar, such as lactose. This reaction leads to the formation of brown-colored degradation products (melanoidins) and a loss of API potency. The presence of heat and moisture accelerates this reaction, making it a critical concern during both manufacturing and long-term storage.23 The presence of reducing sugars as an impurity in other excipients, like microcrystalline cellulose (MCC), can also trigger this reaction.25
- Oxidation: Many APIs are susceptible to oxidative degradation. This risk can be exacerbated by impurities within certain excipients. For example, residual peroxides or phenolic impurities commonly found in polymers like povidone and crospovidone can act as oxidizing agents, leading to the degradation of the API and compromising the product’s shelf life.23 The choice of excipient grade, with specified low levels of such impurities, becomes critical for oxidation-prone drugs.27
- Aldehyde Interactions: Trace levels of aldehydes, such as formaldehyde, can be found as impurities in a wide range of common excipients, including lactose, MCC, starch, and polyethylene glycols (PEGs). These highly reactive aldehydes can interact with the API, leading to the formation of adducts and other degradation products, ultimately impacting the drug’s stability and safety profile.23
- Physical Interactions: Interactions are not limited to chemical reactions. An excipient can physically interact with an API to alter its performance. For instance, the extended mixing of a lubricant like magnesium stearate can lead to “over-lubrication,” where the hydrophobic lubricant coats the API particles, impeding water penetration and delaying dissolution.28
Impurities and Concomitant Components: The Unseen Dangers
The risk profile of an excipient is not defined solely by its primary chemical entity. Excipients are often complex mixtures that include what the International Pharmaceutical Excipients Council (IPEC) refers to as “concomitant components”—unavoidable substances that arise from the raw materials, catalysts, solvents, or side reactions during the manufacturing process.30 While some of these components may be benign or even beneficial to an excipient’s functionality, others can be harmful impurities that pose a direct risk to patient safety or product stability.
A landmark example of this risk is the case of hydrazine in Povidone. In the late 1980s, it was discovered that Povidone, a widely used binder and polymer, contained trace amounts of hydrazine, a toxic and potentially carcinogenic substance. Investigation revealed that hydrazine was formed as a byproduct of the polymerization reaction. This discovery necessitated a modification of the manufacturing process to reduce hydrazine levels to what was deemed acceptable for safe use in pharmaceuticals. Subsequently, a specific test method and upper limit for hydrazine were established and incorporated into the official pharmacopeial monograph for Povidone.30
This case highlights a critical principle: formulators must look beyond the name on the label and understand the potential impurity profile of their excipients. Other potential impurities of concern that can be introduced during excipient manufacturing include:
- Residual Solvents: Organic volatile chemicals that are not completely removed during manufacturing.31
- Heavy Metals or Elemental Impurities: Catalysts and environmental contaminants that may be present in raw materials or leach from processing equipment.32
- Filter Aids and Charcoal: Materials used during purification that may leave trace residues.32
The World Health Organization (WHO) explicitly notes that because excipients are often used in large quantities and may contain such impurities, they can significantly affect the quality and safety of the finished pharmaceutical product.33 This reality necessitates a holistic, context-dependent approach to risk assessment. An excipient’s risk is not a fixed property but a dynamic variable influenced by the specific API it is paired with, the intended patient population, the dosage form, and the manufacturing process. A simple list of “good” and “bad” excipients is therefore inadequate. An effective risk mitigation strategy requires a dynamic framework that considers the entire system—drug, patient, and process—to identify and filter out potential liabilities before they compromise a development program.
Building Your Exclusion Framework: A Systematic Approach to De-Risking
Transitioning from a reactive to a proactive stance on excipient risk requires a structured, systematic framework that is integrated into the very fabric of the drug development process. The Excipient Exclusion Filter is not an ad-hoc checklist but a disciplined methodology aligned with the principles of Quality by Design (QbD). It transforms excipient selection from an empirical, trial-and-error art into a rational, science-based process. This framework consists of four key steps, beginning with the patient and ending with a robust control strategy.
Step 1: Define the Target Product Profile (TPP) and Patient Population
Every formulation decision must originate from a clear understanding of the final product’s goals and the patients it is intended to serve. The Target Product Profile (TPP) is the foundational document that outlines these objectives, and it must be the starting point for applying any exclusion filters.
First, the specific patient population must be clearly defined. Is the drug intended for pediatric use, particularly neonates or infants? If so, a host of excipients known to be toxic or problematic in this group—such as benzyl alcohol, ethanol, propylene glycol, and parabens—must be immediately flagged for exclusion or severe restriction due to the immaturity of metabolic and clearance functions in young children.17 Is the target demographic
geriatric? This population often suffers from dysphagia (difficulty swallowing) and polypharmacy, which may necessitate filters against large or hard-to-swallow fillers and a preference for orally disintegrating or liquid formulations.3
Second, the intended route of administration dictates a fundamental set of permissible excipients. Parenteral (injectable) formulations bypass the body’s natural defense barriers and demand the highest standards of purity, sterility, and safety. This route automatically excludes a vast number of excipients that are acceptable for oral use and requires the use of ultra-high purity grades with specified low levels of bioburden and endotoxins.1 Similarly, topical and ophthalmic formulations have their own unique sets of approved and prohibited excipients to avoid irritation and ensure safety.1
Step 2: Characterize the API and Identify Inherent Risks
With the patient and product context established, the next step is a deep dive into the physicochemical nature of the Active Pharmaceutical Ingredient (API). Thorough pre-formulation studies are non-negotiable, as the API’s inherent properties will dictate its potential for negative interactions with excipients.22
This characterization must identify any “red flags” in the API’s structure and stability profile. For example:
- Does the API contain a primary or secondary amine group? This immediately activates a “reducing sugar” exclusion filter. To prevent the destructive Maillard reaction, excipients like lactose, maltose, and glucose should be avoided in favor of non-reducing sugars like sucrose or sugar alcohols like mannitol.23
- Is the API known to be sensitive to oxidation? This triggers a filter for excipients with low or controlled levels of peroxide impurities. A formulator should seek out specialized grades of polymers like povidone or polysorbates that are specified to have low peroxide values, or consider adding an antioxidant to the formulation.27
- Is the API highly sensitive to moisture (hygroscopic)? This would necessitate a filter against highly hygroscopic excipients and a preference for those with low water activity and bound moisture, which is less likely to participate in degradation reactions.5
By mapping the API’s vulnerabilities, the formulator can proactively build a “negative space” of excipients to avoid, dramatically narrowing the field of potential candidates and reducing the likelihood of late-stage stability failures.
Step 3: Conduct Initial Screening Using Regulatory and Public Databases
Once the initial filters based on patient and API risks are in place, the next step is to screen potential excipient candidates against established regulatory and safety databases. These resources provide invaluable information on the precedent of an excipient’s use, which is a key factor in assessing its regulatory risk.
The most critical tool for this purpose, particularly for products intended for the US market, is the FDA’s Inactive Ingredient Database (IID).36 The IID is a public repository of all inactive ingredients present in FDA-approved drug products. For each excipient, it lists the dosage forms and routes of administration in which it has been used, and crucially, the maximum potency (amount per unit dose) previously approved for that context.
The IID serves several functions in the exclusion framework:
- Establishing Precedent: If a formulator wishes to use a specific excipient (e.g., polysorbate 80) in an oral tablet, they can consult the IID to confirm it has been previously approved for that use.
- De-risking Dosage Levels: If the intended amount of the excipient in the new formulation is at or below the maximum potency listed in the IID for that route, it is not considered “novel.” This generally means it will require a less extensive safety review by the agency, significantly de-risking the regulatory pathway.39
- Identifying Red Flags: If an excipient is not listed in the IID for the intended route of administration, or if the intended dose exceeds the listed maximum potency, it is considered a “novel excipient” from a regulatory perspective. This would require the submission of extensive preclinical safety and toxicology data, a significant barrier to development.42
Similarly, the European Medicines Agency (EMA) provides a guideline with an annex listing excipients known to have a recognized action or effect. If a medicine contains one of these excipients, it must be declared on the label, and specific warning information must be included in the package leaflet.44 This database helps formulators understand the labeling implications of their excipient choices from the outset.
While these databases are invaluable for establishing a safety baseline, they should not be seen as an innovation ceiling. Novel APIs and advanced drug delivery systems often require novel excipients or new uses of existing ones that lack precedent.1 A purely IID-driven strategy could stifle innovation and lead to suboptimal formulations for challenging drugs. The expert formulator, therefore, uses these databases to quickly filter out high-risk choices for standard formulations but also understands when the therapeutic benefit of a novel excipient justifies the increased regulatory burden of moving beyond the established database. The exclusion framework serves to document this critical risk-benefit analysis.
Step 4: Integrate Exclusion Filters into a Quality by Design (QbD) Strategy
The final step is to formalize the exclusion framework within the globally accepted Quality by Design (QbD) paradigm. QbD is a systematic approach to development that begins with predefined objectives and emphasizes product and process understanding and process control, based on sound science and quality risk management.43
Within this paradigm, the outputs of the exclusion filter process become direct inputs for the QbD framework:
- Quality Target Product Profile (QTPP): The decisions made in Step 1 become part of the QTPP. For example, “lactose-free” or “suitable for pediatric use (ethanol-free)” become critical quality attributes (CQAs) that the final product must possess.48
- Critical Material Attributes (CMAs): The properties of the chosen alternative excipients become potential CMAs. If lactose is excluded and mannitol is chosen as the replacement filler for a direct compression tablet, then the particle size distribution, flowability, and compressibility of the mannitol become CMAs. These are physical properties that must be controlled within an appropriate limit, range, or distribution to ensure the final product consistently meets its CQAs (e.g., content uniformity, hardness, dissolution).48
By integrating the exclusion framework into QbD, the selection of excipients is elevated from a simple screening exercise to a foundational element of the product’s control strategy. It ensures that the risks associated with excipients are not just identified, but systematically understood, controlled, and monitored throughout the product’s lifecycle, leading to a more robust, safe, and effective medicine.
The Lactose-Free Filter: Navigating the Industry’s Most Common Hurdle
Among the myriad of exclusion filters a formulator might apply, the “lactose-free” filter is by far the most common and, in many ways, the most challenging. Lactose is the undisputed workhorse of the oral solid dosage world, a ubiquitous filler and diluent prized for its versatility and excellent manufacturing properties.3 However, its use is complicated by two major factors: the high global prevalence of lactose intolerance and its chemical reactivity with certain APIs. Navigating the decision to formulate without lactose requires a deep understanding of the patient population, the technical complexities of replacement, the properties of available alternatives, and the significant biopharmaceutical risks involved.
The Scale of the Problem: Lactose Intolerance by the Numbers
Lactose intolerance, the inability to digest the primary sugar in milk due to a deficiency of the enzyme lactase, is a widespread global health consideration. While nearly all infants produce lactase, its production drops off after weaning in a majority of the world’s population.49
- Global Prevalence: Estimates suggest that approximately 65% to 80% of the global adult population experiences some degree of lactase non-persistence.50
- Ethnic and Geographic Variation: The prevalence of lactose intolerance varies dramatically among different ethnic groups. It is lowest in populations with a long history of dairy farming, such as Northern Europeans, where as few as 5% of individuals are affected. In stark contrast, it affects 70-100% of people of East Asian descent, around 80% of African-Americans and Native Americans, and up to 90% of adults in some parts of Asia.50 In the United States alone, an estimated 30 to 50 million people are lactose intolerant.52
While the amount of lactose used as an excipient in most tablets and capsules is relatively small—daily exposure from medicines is unlikely to exceed 2 grams, whereas symptoms typically appear after ingesting around 12 grams—patients with severe intolerance may need to avoid it completely.15 A randomized, double-blind, controlled study involving 77 lactose-intolerant individuals found that a 400 mg dose of lactose (a common amount in a single tablet) did not cause a significant increase in symptoms compared to a placebo.55 Despite this evidence, growing patient awareness, the “clean label” trend, and the desire to provide therapeutic options for all patient groups have created a strong market pull for lactose-free pharmaceutical products.56
Technical Challenges of Lactose Replacement
The reason lactose is so pervasive in pharmaceutical manufacturing is that it possesses a nearly ideal combination of functional properties. It is chemically stable (for non-amine drugs), physically robust, readily available, cost-effective, and exhibits excellent powder flow and compressibility, especially in its spray-dried and anhydrous forms.3 Replacing it, therefore, is not a simple matter of substitution. The chosen alternative must replicate these critical functionalities to ensure the final product can be manufactured consistently and will perform as intended.
Key challenges in replacing lactose include:
- Matching Compactibility: Lactose provides excellent binding properties, leading to tablets with high mechanical strength (hardness) and low friability (tendency to chip or break). An alternative must provide similar tablet strength to withstand the rigors of manufacturing, coating, packaging, and transport.26
- Ensuring Flowability: In high-speed tablet manufacturing, the powder blend must flow uniformly and consistently from the hopper into the die cavity to ensure consistent tablet weights. Spray-dried lactose has excellent flow properties. A replacement excipient must also flow well to prevent weight variation and content uniformity issues.61
- Maintaining Stability: Lactose is relatively non-hygroscopic, meaning it does not readily absorb moisture from the air. This contributes to the stability of both the formulation and the API. Any replacement must also exhibit low hygroscopicity to prevent issues like tablet softening, sticking, or API degradation.59
- Replicating Dissolution Profile: The excipient matrix plays a critical role in how quickly a tablet disintegrates and the drug dissolves. A replacement must not negatively impact the drug’s release profile, as this could alter its bioavailability and therapeutic effect.63
A Formulator’s Toolkit: Key Alternatives to Lactose
Fortunately, formulation scientists have a range of alternative excipients at their disposal. The selection of the right substitute depends heavily on the specific requirements of the API and the manufacturing process, particularly whether the formulation is intended for direct compression or wet granulation.
| Excipient | Primary Function | Key Advantages | Key Disadvantages/Challenges | Best For… |
| Lactose (Baseline) | Filler, Diluent, Binder | Excellent compressibility, good flow, low cost, well-characterized, multiple grades available 3 | Maillard reaction with amine drugs, unsuitable for intolerant patients 25 | General-purpose direct compression and wet granulation for non-amine APIs. |
| Mannitol | Filler, Diluent | Excellent for direct compression, non-hygroscopic, pleasant taste/mouthfeel, good for chewable/ODTs 35 | Higher cost than lactose, can have lower compactibility in some grades. | Direct compression, orally disintegrating tablets (ODTs), moisture-sensitive APIs. |
| Dicalcium Phosphate (DCP) | Filler, Diluent | Excellent flow, non-hygroscopic, low cost. | Poor tablet hardness (low compactibility), can be abrasive to tooling 60 | Formulations where flow is a major challenge and high tablet strength is not critical. |
| Microcrystalline Cellulose (MCC) | Binder, Filler, Disintegrant | Excellent binder, produces very hard tablets, has disintegration properties 4 | Poor flowability (especially fine grades), can be lubricant-sensitive, can be hygroscopic 61 | Wet granulation; direct compression when blended with a glidant or free-flowing excipient. |
| Dextrates (e.g., Emdex®) | Filler, Binder | Water-soluble, similar particle morphology, bulk density, and flowability to spray-dried lactose 67 | Can be more expensive than standard lactose. | Direct 1:1 replacement for spray-dried lactose in direct compression formulations. |
| Co-processed Excipients | Multifunctional | Engineered for superior flow and compressibility, simplifies formulation, reduces excipient count 24 | Higher cost, proprietary nature can lead to single-supplier dependency. | High-performance direct compression tablets, especially for challenging APIs or when streamlining manufacturing. |
Biopharmaceutical Implications: Managing Bioequivalence (BE) Risk
Perhaps the most critical consideration when reformulating to be lactose-free is the potential impact on the drug’s bioavailability. Regulatory agencies require that a reformulated product be bioequivalent to the original, meaning it must release the API into the bloodstream at the same rate and to the same extent.71 Changing a major excipient like lactose can significantly alter the dissolution profile, putting bioequivalence at risk.63
The level of this risk is strongly correlated with the API’s properties, as defined by the Biopharmaceutics Classification System (BCS):
- BCS Class I (High Solubility, High Permeability): For these well-behaved drugs, absorption is not typically limited by dissolution. Therefore, the probability of a change in lactose content causing bioinequivalence is considered low.63
- BCS Class III (High Solubility, Low Permeability): Absorption is limited by the drug’s ability to cross the intestinal wall, not by its dissolution. The risk of bioinequivalence is considered medium.63
- BCS Class II (Low Solubility, High Permeability): Here, the dissolution rate is the rate-limiting step for absorption. Any change in the formulation that affects how quickly the drug dissolves—which lactose replacement certainly can—poses a significant risk. The probability of bioinequivalence is therefore classified as medium.63
- BCS Class IV (Low Solubility, Low Permeability): These are the most challenging drugs, with both poor solubility and poor permeability. Their absorption is highly sensitive to formulation changes. Consequently, the probability of bioinequivalence resulting from lactose replacement is high.63
This framework underscores that the decision to reformulate must be accompanied by a thorough biopharmaceutical risk assessment, with the level of scrutiny and testing increasing dramatically for BCS Class II and IV drugs.
Case Studies in Lactose-Free Reformulation
Successful reformulation is achievable with a rational, data-driven approach, as demonstrated by several published case studies.
Case Study 1: Cetirizine Tablets
A study focused on reformulating the popular antihistamine cetirizine, whose marketed version (Reactine) contained lactose. The objective was to create a bioequivalent, lactose-free version suitable for intolerant patients.
- Challenge: Replace spray-dried lactose without compromising the excellent flow and compression properties required for direct compression manufacturing.
- Solution: The formulators selected dextrates (Emdex®), an excipient known to have very similar physical properties to spray-dried lactose.
- Outcome: The dextrates-based formulation was found to be fully equivalent to both a lactose-based control and the original marketed product. It exhibited identical powder flow characteristics, tablet hardness, friability, and, most importantly, a virtually superimposable dissolution profile. This case study represents a successful, low-risk 1:1 replacement strategy.67
Case Study 2: Rivaroxaban (BCS Class II) Tablets
A more challenging project involved developing lactose-free formulations of rivaroxaban (Xarelto®), an anticoagulant. As a BCS Class II drug, its bioavailability is dependent on its dissolution rate, making the choice of excipients critical.
- Challenge: Replace lactose in a formulation for a poorly soluble drug while maintaining or improving the dissolution rate to ensure bioavailability.
- Solution: The formulators chose anhydrous dibasic calcium phosphate (DCPA) as the primary filler/diluent. They developed both traditional tablets and a Multiple-Unit Pellet System (MUPS).
- Outcome: The resulting lactose-free formulations demonstrated excellent mechanical strength and chemical stability. Crucially, the in vitro dissolution profiles were comparable to the original lactose-containing Xarelto® product. The study successfully demonstrated a viable pathway for creating effective, stable, and patient-friendly lactose-free versions of a challenging BCS Class II drug.72
These cases illustrate that while replacing lactose is a significant technical undertaking, a systematic approach grounded in understanding excipient functionality and biopharmaceutical risk can lead to successful, patient-centric products. This very challenge is a major force driving innovation in the excipient industry, pushing manufacturers to develop advanced co-processed materials that offer turnkey solutions for high-performance, lactose-free formulations.24 For drug developers, mastering these new materials is no longer just a technical exercise but a pathway to creating differentiated products that meet a clear and growing market need.
The Animal-Free Filter: Formulating for Vegan, Halal, and Kosher Markets
Beyond physiological intolerances, a rapidly growing driver for excipient exclusion is the alignment of pharmaceutical products with the ethical, religious, and lifestyle choices of patients. The global rise in vegetarianism, veganism, and adherence to dietary laws such as Halal and Kosher presents both a significant challenge and a substantial market opportunity for the pharmaceutical industry.73 Applying an “animal-free” filter requires formulators to look beyond common allergens and scrutinize the origin of every ingredient in their formulation, with the most prominent and complex component being gelatin.
Gelatin’s Dominance and Its Limitations
For nearly a century, gelatin has been the gold standard for capsule manufacturing, particularly for soft gelatin capsules (softgels). Derived from the collagen of animal sources—primarily bovine (cow) and porcine (pig) skin and bones—gelatin possesses a unique combination of properties that make it exceptionally well-suited for encapsulation.75 It forms strong, flexible films, has excellent machinability on high-speed equipment, and dissolves quickly at body temperature to release the drug, making it both cost-effective and highly functional.75
However, its animal origin is its greatest liability in the modern market, creating several limitations:
- Dietary and Ethical Exclusions: Gelatin is inherently unsuitable for the millions of consumers who follow vegetarian or vegan diets. Furthermore, its typical sourcing from pork or non-ritually slaughtered beef makes it non-compliant with Halal (Islamic) and Kosher (Jewish) dietary laws.77
- Technical Challenges: Despite its strengths, gelatin is not without technical flaws. It has a relatively high moisture content (13-16%), making it unsuitable for highly moisture-sensitive or hygroscopic drugs.81 It is also susceptible to
cross-linking, a chemical reaction that can occur over time, especially in the presence of certain reactive fill ingredients (like those containing aldehydes) or under conditions of high heat and humidity. Cross-linking can make the gelatin shell less soluble, delaying or preventing drug release and compromising the product’s efficacy.81 - Safety and Supply Chain Concerns: While rigorously controlled, the animal origin of gelatin has historically raised concerns about the potential for transmission of diseases like Bovine Spongiform Encephalopathy (BSE), requiring stringent sourcing and certification.80 Moreover, the complex global supply chain for gelatin was shown to be vulnerable to disruptions during the COVID-19 pandemic, prompting manufacturers to seek more resilient alternatives.83
The Rise of HPMC and Other Vegetarian Capsules
In response to gelatin’s limitations, the industry has developed several plant-based alternatives, with Hydroxypropyl Methylcellulose (HPMC) emerging as the clear market leader.75 HPMC, also known as hypromellose, is a polymer derived from plant cellulose, typically from pine or spruce trees.84 It directly addresses many of gelatin’s key weaknesses:
- Vegetarian and Vegan Source: Being plant-derived, HPMC is suitable for all consumers, regardless of dietary or religious restrictions, and can be certified Halal and Kosher.85
- Low Moisture Content: HPMC capsules have a significantly lower moisture content (typically 3-8%) compared to gelatin. This makes them the ideal choice for formulating moisture-sensitive and hygroscopic APIs, enhancing product stability and shelf-life.81
- No Cross-Linking Risk: As a cellulose polymer, HPMC contains no proteins or amino acids and is therefore not susceptible to the cross-linking reactions that can plague gelatin. This ensures consistent, predictable dissolution performance over the product’s lifetime.81
Other vegetarian options include Pullulan capsules, which are derived from tapioca starch through a natural fermentation process.79
This switch is not without trade-offs. HPMC is a more expensive raw material, and its manufacturing process can be more complex, resulting in a final capsule that is typically three to four times more costly than its gelatin counterpart.75 Furthermore, HPMC can exhibit lower machinability on certain high-speed encapsulation machines, sometimes requiring process adjustments or specialized equipment. The dissolution profile can also differ; while gelatin dissolves rapidly at body temperature, HPMC dissolves at room temperature and may exhibit a slower release profile for some drugs, a factor that must be carefully evaluated during development.86
The adoption of HPMC capsules represents a strategic convergence of multiple industry drivers. It began as a response to a niche market demand from vegetarians but was then embraced by formulators as a technical solution for challenging APIs. This trend was further accelerated by supply chain security concerns, transforming HPMC from an alternative into a mainstream strategic choice for new drug development. This convergence explains the high projected compound annual growth rate (CAGR) for the HPMC capsules market, with estimates ranging from 8.5% to 9.7%.89
Beyond Capsules: Identifying Other Animal-Derived Excipients
Applying a comprehensive animal-free filter requires scrutiny beyond just the capsule shell. Many other common excipients can be derived from animal sources, and formulators must be diligent in verifying their origin.
- Lactose: Derived from cow’s milk, it is unsuitable for vegans.3
- Magnesium Stearate/Stearic Acid: A common lubricant, stearates can be derived from either animal fats (tallow) or vegetable oils (palm oil). For an animal-free formulation, a vegetable-grade source must be explicitly specified and certified.91
- Glycerin/Glycerol: Used as a plasticizer and humectant, glycerin can be produced from animal fats, vegetable oils, or synthetically. Again, a vegetable or synthetic source is required for animal-free products.91
- Shellac: A resin used for tablet coating, it is secreted by the female lac bug, making it unsuitable for vegans.28
- Carmine: A vibrant red colorant derived from crushed cochineal insects, which is unacceptable in vegan formulations.20
- Lanolin: An emollient used in topical preparations, lanolin is derived from the wool fat of sheep.2
Navigating Vegan, Halal, and Kosher Requirements
While there is significant overlap, formulating for Vegan, Halal, and Kosher standards involves distinct requirements for ingredient sourcing, processing, and prevention of cross-contamination. It is crucial to understand these nuances to ensure full compliance. While religious authorities may grant exemptions for essential, life-saving medicines if no compliant alternative exists, patient preference is overwhelmingly in favor of formulations that respect their beliefs without forcing an ethical compromise.91
| Criteria | Vegan Compliance | Halal Compliance | Kosher Compliance |
| Ingredient Restrictions | Prohibits all animal-derived ingredients, including meat, poultry, fish, dairy (lactose), eggs, honey, and insect products (carmine, shellac).77 | Prohibits pork, blood, alcohol, and meat from animals not slaughtered according to Islamic law. Gelatin must be from Halal-certified sources.74 | Prohibits pork, shellfish, and meat from non-Kosher animals. Meat and dairy cannot be mixed. Gelatin must be from Kosher-certified sources.77 |
| Prohibited Substances | Animal products, animal by-products, ingredients tested on animals.74 | Pork, alcohol, carrion, blood.77 | Pork, shellfish, mixing of meat and dairy.77 |
| Processing Requirements | No animal testing. No use of animal-derived processing aids (e.g., bone char for sugar refining).77 | Requires specific purification protocols. Dedicated production lines may be necessary to prevent contact with non-Halal substances.77 | Requires specific purification protocols (“Kosherization”). Strict separation of meat and dairy processing, including separate equipment and utensils.77 |
| Cross-Contamination Rules | Strict protocols to prevent any cross-contamination with animal products.74 | Strict protocols to prevent cross-contamination with prohibited substances, especially pork and alcohol.77 | Strict protocols to prevent cross-contamination between meat and dairy products.77 |
| Certification Body Examples | The Vegan Society, Vegan Action.77 | IFANCA (USA), JAKIM (Malaysia), MUIS (Singapore).77 | Orthodox Union (OU), Star-K, OK Kosher Certification.77 |
(Data sourced from 74)
Successfully navigating these requirements allows pharmaceutical companies to access large and growing market segments, demonstrating a commitment to patient-centricity that extends beyond clinical needs to encompass personal values and beliefs.
The Alcohol-Free Filter: Solving the Solubility Challenge in Liquid Formulations
Liquid dosage forms are indispensable in pharmaceuticals, particularly for pediatric and geriatric patients who may have difficulty swallowing solid tablets or capsules. For decades, ethanol (alcohol) has been a cornerstone of liquid formulations, valued for its dual functionality as both a powerful solvent for poorly water-soluble APIs and an effective antimicrobial preservative.93 However, a growing body of evidence on the potential risks of ethanol, especially in vulnerable populations, has made the “alcohol-free” exclusion filter a critical consideration in modern formulation development. Removing ethanol presents a significant technical challenge, forcing formulators to find alternative solutions for both drug solubilization and product preservation.
The Dual Role of Ethanol: A Necessary Evil?
Ethanol’s utility in liquid formulations is undeniable. Many APIs are hydrophobic organic molecules with very low solubility in water. Ethanol, as a semi-polar solvent, can effectively bridge the polarity gap between water and these drugs, allowing for the creation of clear, stable solutions.93 This is often achieved through
cosolvency, where ethanol is mixed with water to create a solvent system with the ideal polarity to dissolve the API.
Beyond its role as a solvent, ethanol is also a potent preservative. At concentrations of 15% or higher, it effectively inhibits the growth of bacteria and fungi, ensuring the microbial stability of multi-dose liquid products throughout their shelf life. For preparations with a neutral or slightly alkaline pH, a concentration of 18% is often considered necessary for adequate preservation.93 This dual-action capability has made it an efficient and cost-effective choice for formulators.
The Risks of Ethanol, Especially in Vulnerable Populations
Despite its functional benefits, the inclusion of ethanol in medicines is a significant safety concern, particularly for children, but also for several other patient groups.
- Pediatric Patients: This is the population at highest risk. The enzymes responsible for metabolizing ethanol—alcohol dehydrogenase (ADH) and cytochrome P450 2E1 (CYP2E1)—are not fully developed in infants and young children. ADH concentrations in neonates can be less than 20% of adult levels, with full capacity not reached until around age five.96 This reduced metabolic capacity can lead to the accumulation of ethanol in the blood, increasing the risk of acute and chronic toxicity.
- Adverse Effects: In children, signs of ethanol intoxication include central nervous system (CNS) depression, hypothermia, and severe hypoglycemia, which can lead to seizures and coma.97 Chronic exposure, even at low levels from medication, raises concerns about long-term effects on neurological and cognitive development.95 The related compound,
benzyl alcohol, has been infamously linked to “gasping baby syndrome,” a fatal condition in neonates characterized by metabolic acidosis and respiratory depression, underscoring the extreme vulnerability of this population to certain alcohols.17 - Regulatory Guidance: Recognizing these risks, global regulatory bodies have issued strict guidance. The FDA recommends that over-the-counter (OTC) products for children under six be alcohol-free, with limits of <5% for children 6-12 and <10% for those over 12.96 The EMA similarly advises that ethanol should be avoided in pediatric medicines unless its use is necessary and unavoidable.97
- Other Vulnerable Groups: The risks are not confined to children. Ethanol should be avoided or used with extreme caution in:
- Pregnant Women: Due to the known risk of fetal alcohol syndrome.97
- Patients with Liver Disease: Impaired liver function reduces the ability to metabolize alcohol.
- Patients with a History of Alcoholism: To avoid triggering a relapse.
- Patients Taking Interacting Medications: Ethanol can have synergistic effects with other CNS depressants, such as opioids, benzodiazepines, and certain antihistamines, leading to profound sedation, respiratory depression, and potentially death.99
Strategies for Alcohol-Free Solubilization
Removing ethanol requires formulators to find alternative methods to achieve drug solubility. This often involves a multi-pronged approach, selecting a strategy based on the API’s specific physicochemical properties.
- Cosolvency with Alternative Solvents: The most direct replacement strategy is to use other water-miscible cosolvents. Common choices include glycerin, propylene glycol, and polyethylene glycols (PEGs) of various molecular weights.93 These solvents work similarly to ethanol by reducing the polarity of the aqueous vehicle. However, it is crucial to note that these alternatives are not without their own safety considerations. Propylene glycol, for instance, can also cause CNS effects at high doses and its use is cautioned in children under four years old.17
- pH Adjustment and Salt Formation: For APIs that are weak acids or weak bases (ionizable drugs), solubility is highly dependent on pH. By adjusting the pH of the formulation with a suitable buffering agent, the drug can be converted into its more soluble ionized (salt) form. This is one of the most effective methods for increasing aqueous solubility but is only applicable to ionizable compounds.100
- Surfactants (Micellar Solubilization): Surfactants are amphiphilic molecules that, above a certain concentration (the critical micelle concentration or CMC), form colloidal aggregates called micelles. The hydrophobic cores of these micelles can encapsulate poorly soluble drug molecules, effectively dissolving them in the aqueous medium. Common pharmaceutical surfactants include polysorbates (e.g., Tween 80) and poloxamers.100
- Hydrotropy: This technique involves the addition of high concentrations of substances known as hydrotropes (e.g., sodium benzoate, nicotinamide, urea) which can increase the solubility of poorly soluble drugs through various mechanisms, including the formation of soluble complexes.100
- Complexation with Cyclodextrins: Cyclodextrins are cyclic oligosaccharides with a hydrophilic exterior and a hydrophobic inner cavity. Poorly soluble drug molecules can fit into this cavity to form a “host-guest” inclusion complex. This complex is much more water-soluble than the drug molecule alone, significantly enhancing solubility.27
Ensuring Stability and Preservation in Alcohol-Free Systems
When ethanol is removed, its preservative function must be replaced to ensure the microbial safety of the product. This introduces a new set of challenges, as the formulator must select an effective antimicrobial preservative system that is also safe for the target patient population. Common choices include parabens (methyl-, propyl-), sodium benzoate, and sorbic acid.11
This creates a potential “formulation cascade.” The primary decision to apply an alcohol-free filter necessitates a secondary decision on an alternative preservative. However, many of these preservatives, such as parabens, are themselves known sensitizers and are on pediatric caution lists, potentially triggering another exclusion filter.11 This cascade effect highlights the interconnectedness of excipient risks. Replacing a single, multifunctional excipient like ethanol is rarely a simple swap; it requires a holistic re-evaluation of the entire formulation system to ensure both solubility and stability are achieved without introducing new safety concerns.
For some drugs, creating a stable solution may not be feasible. In these cases, developing a stable suspension is a viable alternative. A study on the diuretic furosemide demonstrated the successful preparation of stable, alcohol-free suspensions using commercially available suspending vehicles like Ora-Blend and Ora-Blend SF. These formulations remained physically and chemically stable for 30 days, providing an important alcohol-free option for patients who need it.101
The Allergen & Sensitizer Filter: Addressing Dyes, Preservatives, and More
Beyond the major categories of lactose, animal-derived products, and alcohol, a host of other excipients can trigger adverse reactions in sensitive individuals. Applying a broad “allergen and sensitizer” filter is crucial for developing truly patient-centric formulations. This involves a careful review of colorants, preservatives, and other excipients known to cause hypersensitivity. The decision to formulate a “dye-free” product, in particular, presents a complex trade-off between mitigating allergen risk and maintaining the critical safety function of medication identification.
The Color Conundrum: Patient Preference vs. Health Risks
Pharmaceutical colorants serve several important and legitimate functions. They enhance the aesthetic appeal of a product, which can improve patient acceptance and adherence, especially in children.28 More critically, they provide a vital safety feature: allowing for easy identification and differentiation of drugs, strengths, and dosages, thereby helping to prevent potentially fatal medication errors.2 Certain colorants, such as titanium dioxide, also provide opacity, which can protect light-sensitive APIs from degradation and improve shelf-life stability.2
Despite these benefits, colorants—particularly synthetic ones—are a well-documented source of adverse reactions.
- Synthetic Azo Dyes: This class of dyes is most frequently implicated in hypersensitivity. Tartrazine (FD&C Yellow No. 5) is the most notorious example, with numerous reports linking it to allergic-type reactions, including urticaria (hives), angioedema, and the exacerbation of asthma, particularly in individuals with aspirin sensitivity.2 The FDA acknowledges this risk and requires that any product containing tartrazine explicitly state its presence on the label.105 Other azo dyes, such as
Sunset Yellow (FD&C Yellow No. 6), have been associated with similar adverse effects.19 One analysis suggests that as many as one-third of all medications contain at least one chemical dye that has been associated with allergic reactions.107 - Natural Dyes: Even colorants from natural sources are not without risk. Carmine, a red dye extracted from the cochineal insect, is known to cause allergic reactions, including rare cases of anaphylaxis.18
This has led to a growing consumer and regulatory push towards “dye-free” medications, driven by patient preference for “clean labels” and a desire to avoid any potentially unnecessary chemical exposure.108
Formulating Dye-Free: Technical and Practical Challenges
While seemingly straightforward, the decision to remove colorants from a formulation introduces several significant challenges that must be carefully managed.
- Medication Safety and Identification: The most significant risk is the loss of a key visual identifier. In a hospital setting or for a patient taking multiple medications (polypharmacy), the color and shape of a tablet are critical cues to ensure the right drug is administered at the right dose. A collection of “little white tablets” dramatically increases the potential for medication errors.2
- API Stability: If a colorant like titanium dioxide was providing opacity to protect a light-sensitive API, its removal may compromise the drug’s stability. This would necessitate either a complete reformulation with light-protectant excipients or a switch to more expensive, opaque packaging (e.g., amber bottles, blister packs with foil backing).2
- Patient Acceptance and the Placebo Effect: The color of a medication can influence a patient’s perception of its effect. Studies have shown associations between color and expected action (e.g., red for stimulants, blue for sedatives).111 An uncolored tablet might be perceived as less potent or less appealing, potentially impacting adherence.
- Alternative Colorants: Formulators can explore alternatives to problematic synthetic dyes. Natural colorants, derived from sources like beetroot (red), turmeric (yellow), and spinach (green), are an option. However, they present their own challenges: they are generally more expensive, less stable to light and pH changes, and provide a more limited and less vibrant palette of colors compared to their synthetic counterparts.20
This creates a complex risk-benefit trade-off. A truly patient-centric approach does not necessarily mean eliminating all dyes blindly. It involves a nuanced strategy: first, applying an exclusion filter to eliminate dyes with a known high incidence of hypersensitivity, like tartrazine. Second, using the minimum effective concentration of any colorant. Third, carefully weighing the risk of a potential allergic reaction (which is rare for most dyes) against the very real and more frequent risk of a medication error for a specific drug and its target patient population.
Excluding Other Problematic Excipients
The allergen and sensitizer filter extends beyond colorants to include several other classes of excipients that warrant careful consideration.
- Preservatives: As mentioned previously, many antimicrobial preservatives can cause hypersensitivity. Parabens, widely used in oral liquids and topicals, are known contact sensitizers.11
Sulfites, such as sodium metabisulphite used as an antioxidant, can trigger severe reactions including bronchospasm and anaphylaxis, especially in asthmatics.2
Benzalkonium chloride, a common preservative in nebulizer solutions and eye drops, can cause bronchoconstriction and ocular toxicity, respectively.2 The need for preservative-free formulations, particularly for ophthalmic and single-dose products, is a growing trend. - Oils and Fats: Arachis (peanut) oil is a potent allergen that can cause severe, life-threatening anaphylactic reactions. Although pharmaceutical-grade arachis oil is highly refined to remove most of the allergenic proteins, regulatory bodies like the UK’s MHRA advise as a precaution that medicines containing it should not be used by patients with a known peanut allergy.14 The potential for cross-sensitivity with
soya also necessitates caution.14 - Sweeteners: While primarily used to improve palatability, certain sweeteners must be excluded for specific metabolic disorders. Aspartame, which is metabolized to phenylalanine, is strictly contraindicated for patients with phenylketonuria (PKU), a genetic disorder where phenylalanine cannot be metabolized and accumulates to toxic levels.2
Applying this broad filter requires diligence and a deep knowledge of both the excipients themselves and the vulnerabilities of the target patient population. It is a critical step in minimizing adverse drug reactions and ensuring the development of medications that are safe for the widest possible range of patients.
Manufacturing and Scale-Up: From Lab-Scale Reformulation to Commercial Reality
The decision to apply an exclusion filter and replace a standard excipient is far more than a simple change in a recipe. It is a decision that sends ripples through the entire manufacturing and supply chain ecosystem. The journey from a successful lab-scale reformulation to a robust, scalable, and commercially viable product is fraught with technical, regulatory, and logistical challenges. A seemingly minor substitution on paper can fundamentally alter the behavior of the formulation, demanding a complete re-evaluation of the manufacturing process and a significant investment in validation and regulatory affairs.
The Ripple Effect: How One Change Impacts the Entire Process
Replacing a well-characterized, multifunctional excipient like lactose or gelatin can have a cascade of unforeseen consequences on the physical properties of the drug formulation and its processability.64
- Impact on Powder Properties and Manufacturability: In solid oral dosage forms, the bulk physical properties of the powder blend are paramount for successful manufacturing.
- Flowability: High-speed tablet presses rely on the consistent and uniform flow of powder from a hopper into the die. Replacing a free-flowing excipient like spray-dried lactose with a material known for poor flow, such as a fine grade of microcrystalline cellulose (MCC), can lead to inconsistent die filling. This results in significant tablet weight variation and, consequently, failure to meet content uniformity specifications.61 To overcome this, a manufacturer might be forced to abandon a simple and cost-effective
direct compression (DC) process in favor of more complex and expensive granulation methods (wet or dry) to improve the powder’s flow characteristics.114 - Compressibility: The ability of a powder to form a strong, robust tablet under pressure is critical. Lactose is known for its excellent compressibility. An alternative like dicalcium phosphate, while free-flowing, is known to produce tablets with poor hardness and high friability.60 This may require the addition of a stronger binder or an increase in compression forces, which can in turn introduce other problems like excessive tooling wear.
- Lubricant Sensitivity: The interaction between the powder blend and the lubricant (e.g., magnesium stearate) is a delicate balance. A new primary excipient may be more or less sensitive to the lubricant. Increased sensitivity can lead to “over-lubrication” even with short blend times, resulting in softer tablets and slower dissolution rates.28
- Impact on Product Performance: The ultimate goal is to produce a product that is bioequivalent to the original. The new excipient matrix can profoundly affect this.
- Disintegration and Dissolution: The porosity, wettability, and solubility of the excipient matrix control how quickly a tablet breaks apart and the API dissolves. Replacing a soluble filler like lactose with an insoluble one like MCC will fundamentally change the mechanism of disintegration from dissolution-based erosion to swelling-based rupture, which can alter the drug release profile.64
- Stability: The new excipient may have a different hygroscopicity or contain different levels of reactive impurities (peroxides, aldehydes), potentially creating new stability challenges for the API that were not present in the original formulation.22
Navigating the Regulatory Gauntlet: FDA and EMA Perspectives
From a regulatory standpoint, changing an excipient in an approved drug product is a major event that requires rigorous justification and documentation. It is not considered a minor change that can be made without notifying the authorities.
In the United States, such a change would typically require a formal submission to the FDA. Depending on the potential impact on the product’s safety and efficacy, this could be a “Changes Being Effected in 30 Days” (CBE-30) supplement or, for more significant changes, a “Prior Approval Supplement” (PAS). A PAS requires FDA approval before the modified product can be marketed, a process that can be lengthy and resource-intensive.118 The manufacturer must provide a comprehensive data package including:
- Updated formulation details.
- New manufacturing batch records.
- Comparative physicochemical characterization data.
- Full stability data for the new formulation.
- Comparative in vitro dissolution data.
- In some cases, new in vivo bioequivalence studies to prove the reformulated product performs identically to the approved version.
Regulatory bodies like the FDA and EMA have stringent guidelines on excipient quality, sourcing, and the labeling of known allergens.44 Any reformulation must demonstrate not only that the new product is equivalent but also that the new excipient meets all quality and safety standards for its intended use.
Supply Chain Integrity and Excipient Variability
The challenges extend beyond the manufacturing floor and into the global supply chain. When replacing a commodity excipient like lactose, which is available from numerous high-quality suppliers, with a more specialized or novel alternative, supply chain risks can increase.
- Excipient Variability: A critical tenet of QbD is understanding and controlling sources of variability. Excipients sourced from different suppliers, or even different batches from the same supplier, can have subtle differences in physical properties (e.g., particle size distribution, surface area, moisture content) that can significantly impact the manufacturing process and final product performance.24 This is a well-documented phenomenon; interchange between different suppliers can lead to final products with different quality attributes.120 Managing this requires a robust supplier qualification program, multi-source strategies where possible, and stringent incoming material testing to ensure consistency.43
- Supply Chain Resilience: Niche or proprietary co-processed excipients may be available from only a single supplier. While these materials can offer superior performance, relying on a single source introduces significant supply chain risk. Any disruption—whether from manufacturing issues, natural disasters, or geopolitical events—could halt production of the drug product.121
The immense technical, regulatory, and financial barriers associated with post-approval reformulation create a powerful “formulation lock-in” effect. Once a drug is approved, the inertia to remain with the existing formulation is enormous, as the cost and complexity of change are often prohibitive, especially for generic products with narrow profit margins. This reality underscores the paramount importance of applying the Excipient Exclusion Filter framework during initial development. The highest leverage point for de-risking a formulation and aligning it with patient needs is before the first pivotal clinical trials and regulatory submissions. Getting the formulation right the first time avoids the monumental downstream costs, delays, and risks of attempting to fix it after it is already on the market.
Strategic Formulation: Using Exclusion Filters for Competitive Advantage
While the primary function of the Excipient Exclusion Filter framework is to mitigate risk, its strategic application extends far beyond a defensive posture. In the modern pharmaceutical landscape, a well-designed, patient-centric formulation is a powerful offensive tool. By proactively using exclusion filters to create “free-from” products, companies can achieve significant market differentiation, execute effective lifecycle management strategies, and build a robust intellectual property portfolio that creates a durable competitive advantage.
Patient-Centricity as a Driver of Market Share and Brand Loyalty
In an era of empowered patients and crowded therapeutic landscapes, the clinical efficacy of an API is often just the entry ticket to the market. The winning product is frequently the one that offers the best overall patient experience. A formulation that addresses the specific needs, preferences, and values of a target patient group can be a decisive factor in physician prescribing habits and patient choice.10
- Meeting Unmet Needs: A “lactose-free” formulation directly targets the large global population with lactose intolerance, offering them a safe and comfortable treatment option that competitors may not provide. A “vegan” or “Halal-certified” capsule appeals to patients whose lifestyle or religious beliefs are a core part of their identity. These are not minor considerations; they are powerful drivers of brand loyalty and market share.
- Aligning with Consumer Trends: The broader consumer market trend is overwhelmingly towards “clean labels,” transparency, and products that are perceived as natural or free from unnecessary additives.108 A “dye-free” or “preservative-free” medication aligns with this powerful zeitgeist, creating a positive brand perception that can influence choice even among patients without a specific allergy. This aligns with the evolution of patients from passive recipients of care to active consumers who expect healthcare experiences to be as frictionless and personalized as those they encounter in other industries.6
Lifecycle Management and IP Strategy
The Excipient Exclusion Filter framework is also a cornerstone of sophisticated lifecycle management and intellectual property (IP) strategy. The finite lifespan of a composition of matter patent for an API means that innovator companies must constantly seek ways to extend the commercial viability of their key products. Reformulation is a primary vehicle for achieving this.
By developing a new, improved, patient-centric version of a drug—for example, by switching from a gelatin capsule to a novel HPMC-based enteric-release capsule—a company can launch a “line extension.” This new product is not merely a replacement but a value-added offering that can defend market share against the eventual entry of generics for the original formulation.
Crucially, this new formulation may itself be patentable. Formulation patents, which protect the specific combination of an API with various excipients to create a final drug product, are a key tool for building a “multi-layered fortress” of IP around a successful drug.124 These secondary patents can extend a product’s market exclusivity for years beyond the expiration of the primary API patent, providing a significant return on the investment in formulation development.125
Leveraging Patent Intelligence for Strategic Formulation
To effectively use formulation as a strategic tool, companies must have a clear view of the competitive and IP landscape. This is where patent intelligence platforms, such as DrugPatentWatch, become indispensable.3 By systematically analyzing the patent filings of competitors, a company can “crack the code” of their formulation strategies and identify both threats and opportunities.
Patents are a treasure trove of technical information, often disclosing specific excipients used, their concentration ranges, manufacturing processes (e.g., wet granulation, direct compression), and performance data.124 This intelligence can be leveraged in two primary ways:
- “Design-Around” Strategy: This is a critical tactic for generic and 505(b)(2) developers. By carefully studying the claims of an innovator’s formulation patents, a company can intentionally design a new formulation that is bioequivalent to the branded product but does not literally infringe on the patent’s claims. For example, if an innovator’s patent claims a specific combination of sucrose and arginine as stabilizers, a generic developer can use patent intelligence to understand the core problem (e.g., aggregation) and then focus its R&D on finding a non-infringing solution, such as using trehalose and proline instead. The patent provides the problem definition for free, allowing for a highly focused and efficient R&D effort.124
- “White Space” Analysis: Patent landscape mapping can reveal areas of unmet need or unprotected technological territory. By analyzing the existing patents in a therapeutic area, a company can identify formulation approaches that no competitor has yet claimed. For instance, if all existing products for a condition are oral solid tablets, but the target patient population includes many with difficulty swallowing, a “white space” exists for a patient-friendly oral liquid, sublingual film, or orally disintegrating tablet. A company can then direct its R&D into this unprotected space, knowing that a successful formulation will be both clinically meaningful and commercially protectable with new IP.124
This strategic approach reveals that the constraints imposed by an exclusion filter can be a powerful catalyst for innovation. When a formulator is told they cannot use a standard, go-to excipient like lactose, they are forced to think creatively and explore more advanced solutions. This can lead to the development of novel co-processed excipients, unique polymer blends, or new manufacturing techniques. These solutions, born out of the necessity to circumvent a constraint, are often novel and non-obvious in their own right, making them patentable inventions. In this way, the exclusion filter transforms from a simple risk-mitigation tool into a veritable engine of innovation, driving the creation of new intellectual property that strengthens the company’s competitive position.
The Future of Excipient Selection: Smart Excipients and Personalized Medicine
The evolution of excipient science is far from over. The trends shaping the future of pharmaceutical formulation point towards an era of even greater sophistication, where excipients are no longer just functional aids but are integral, intelligent components of the drug delivery system. This future will be driven by advances in material science, the transformative power of data analytics and artificial intelligence, and the ultimate pursuit of truly personalized medicine. The Excipient Exclusion Filter framework, as we know it today, will evolve in tandem with these advancements.
The Rise of Multifunctional and Co-processed Excipients
The industry is rapidly moving beyond the traditional approach of creating simple physical mixtures of individual excipients. The future belongs to highly engineered, multifunctional and co-processed excipients. These are single-particle materials created by combining two or more standard excipients using advanced processes like spray drying or granulation.4
A single co-processed particle might, for example, combine the functions of a filler (like mannitol), a binder (like PVP), and a superdisintegrant (like crospovidone).126 This offers several profound advantages:
- Improved Performance: These excipients are engineered to have superior physical properties, such as enhanced flowability and compressibility, which are critical for robust and efficient manufacturing processes like direct compression.127
- Simplified Formulations: By combining multiple functions into one ingredient, the total number of excipients in a formulation can be reduced, simplifying development, quality control, and supply chain management.
- Overcoming Challenges: They are often designed specifically to solve common formulation problems, such as providing a ready-to-use, high-performance, lactose-free alternative for direct compression.70
As drug molecules become more complex and challenging to formulate, the demand for these high-functionality excipients will continue to grow, making them a cornerstone of future oral solid dosage form development.47
AI-Driven Formulation Development and Risk Prediction
Perhaps the most disruptive force on the horizon is the integration of Artificial Intelligence (AI) and Machine Learning (ML) into the formulation development process. The traditional trial-and-error approach to formulation is slow, resource-intensive, and often inefficient. AI promises to revolutionize this paradigm.121
AI algorithms can analyze vast, multidimensional datasets encompassing API physicochemical properties, the material attributes of thousands of excipients, manufacturing process parameters, and historical stability and clinical outcome data. By identifying complex, non-linear relationships within this data, AI models can:
- Predict Optimal Formulations: Instead of manually screening dozens of excipient combinations, an AI can predict a small number of optimal formulations with the highest probability of meeting the Target Product Profile.130
- Identify Incompatibilities Early: AI can flag potential API-excipient incompatibilities before the first physical mixture is even made, saving significant time and resources.121
- De-risk Scale-Up: By modeling the impact of process parameters, AI can help predict how a formulation will behave when scaled from a lab-scale press to a full-scale commercial manufacturing line, reducing the risk of costly batch failures.
This data-driven approach will make the application of exclusion filters more precise and predictive, allowing formulators to screen out problematic excipients with a much higher degree of confidence early in development.
Smart Excipients and Personalized Medicine
The ultimate frontier in excipient science is the development of “smart” excipients. These are advanced, stimuli-responsive polymers and materials that can actively modulate drug release in response to specific physiological signals within the body.3
Imagine a drug product built with excipients that can:
- Remain inert in the acidic environment of the stomach but rapidly dissolve at the specific pH of the upper small intestine for targeted release (pH-responsive polymers).
- Exist as a liquid at room temperature for easy injection, but instantly form a solid gel depot upon reaching body temperature to provide sustained release over weeks or months (thermo-responsive polymers).3
- Release their drug payload only in the presence of a specific enzyme that is overexpressed in tumor tissue (enzyme-responsive systems).
These intelligent materials are the key enablers of true personalized medicine. In the future, it may be possible to design a formulation not just for a broad patient population, but for an individual. The current Excipient Exclusion Filter framework, which is largely a human-driven, logic-based process of elimination (e.g., “IF patient is lactose intolerant, THEN exclude lactose”), will evolve. It will transform into a generative “Inclusion Algorithm.”
In this future state, a formulator will input the API’s structure, the desired therapeutic profile, and the patient’s specific biological data—such as their genetic profile (e.g., lactase persistence status), their gut microbiome composition, or biomarkers for their specific disease state. A generative AI will then not just exclude problematic excipients but will algorithmically design a bespoke formulation from a library of conventional, multifunctional, and smart excipients, selecting the optimal combination with the highest predicted probability of success for that unique individual. The process will shift from reactive exclusion to proactive, personalized, and generative inclusion, marking the next great leap in pharmaceutical formulation science.
Key Takeaways
- Excipients Are Not Inert: The traditional view of excipients as inactive fillers is obsolete. They are active, functional components that critically impact a drug product’s stability, manufacturability, bioavailability, and patient safety. Their selection is a strategic decision with far-reaching consequences.
- Proactive Risk Mitigation is Essential: The “Excipient Exclusion Filter” provides a systematic framework for moving beyond reactive troubleshooting. By proactively identifying and excluding problematic excipients based on API characteristics, patient populations, and commercial goals, companies can significantly de-risk their development pipelines.
- Patient-Centricity Drives Commercial Success: Formulating for sensitive populations is no longer a niche activity but a core business strategy. “Free-from” products (e.g., lactose-free, gelatin-free, dye-free) meet a clear and growing market demand, serving as powerful differentiators that enhance patient adherence, build brand loyalty, and capture market share.
- Reformulation is a Complex, High-Stakes Endeavor: Replacing a common excipient like lactose or gelatin is not a simple substitution. It poses significant technical challenges related to manufacturability (flow, compression), stability, and, most critically, bioequivalence. The risk of failure is high, especially for drugs with poor solubility or permeability (BCS Class II & IV).
- Early Application of Filters is Critical: The high technical, regulatory, and financial barriers to post-approval reformulation create a “formulation lock-in.” The highest-leverage opportunity to create a safe and patient-centric formulation is during the initial development of a new drug, before pivotal clinical trials begin.
- Constraints Breed Innovation and Create Value: The necessity of formulating without standard excipients forces scientific creativity. This can lead to the development of novel, high-performance co-processed excipients or unique drug delivery systems that are themselves patentable, turning a formulation constraint into a source of new intellectual property and competitive advantage.
- Leverage Intelligence Tools for a Strategic Edge: The effective use of regulatory databases like the FDA’s Inactive Ingredient Database (IID) is crucial for de-risking. Furthermore, competitive intelligence platforms such as DrugPatentWatch are essential for analyzing competitor formulation strategies, enabling “design-around” approaches for generics and identifying “white space” opportunities for new, patentable formulations.
- The Future is Engineered and Personalized: The pharmaceutical industry is moving towards highly engineered, multifunctional excipients and the use of Artificial Intelligence to predict and design optimal formulations. The ultimate goal is the development of “smart” excipients that enable truly personalized medicine, where drug products are tailored to the unique biological profile of an individual patient.
Frequently Asked Questions (FAQ)
1. Q: What is the first step I should take when tasked with developing a “lactose-free” version of an existing tablet?
A: The first and most critical step is to perform a thorough characterization of both the existing lactose-based product and the Active Pharmaceutical Ingredient (API). You must understand the precise functional role lactose is playing in the current formulation. Is it primarily a filler in a wet granulation process, or is it a crucial binder and flow aid in a direct compression tablet? Concurrently, determine the API’s Biopharmaceutics Classification System (BCS) class. This will define the target physical properties (e.g., particle size, flowability, compressibility) that your replacement excipient system must match and, more importantly, will quantify the level of biopharmaceutical risk. For a BCS Class I drug, the risk of bioinequivalence is low, but for a BCS Class II or IV drug, the risk is medium to high, demanding a much more rigorous development and testing program.
2. Q: Is switching from gelatin to HPMC capsules always the better choice for a new product?
A: Not necessarily. The choice should be a data-driven, risk-based decision tailored to your specific product profile. HPMC capsules offer undeniable advantages for moisture-sensitive or hygroscopic APIs, for formulations with ingredients prone to cross-linking with gelatin, and for products targeting vegetarian, vegan, Halal, or Kosher markets. However, these benefits come at a higher cost and may require adjustments to manufacturing processes. For a robust, non-sensitive API intended for a broad market where cost of goods is a primary driver, traditional gelatin capsules may still be a perfectly viable and more economical option. The key is to weigh the technical benefits and market advantages of HPMC against its higher cost and potential for different dissolution behavior.
3. Q: How can I justify the higher cost of using alternative or multifunctional excipients to management?
A: The justification should be framed in terms of total value and risk reduction, not just the unit cost of the excipient. A more expensive, high-performance excipient can deliver value in several ways:
- Process Efficiency: It may enable a simpler and cheaper manufacturing process, such as shifting from multi-step wet granulation to single-step direct compression, leading to significant savings in time, energy, and capital expenditure.
- Risk Reduction: It can reduce the risk of costly batch failures, stability issues, or failed bioequivalence studies, which can save millions of dollars and months of delays in a development program.
- Market Differentiation: It allows for the creation of a differentiated, patient-centric product (e.g., lactose-free, smaller tablet size) that can command a premium price, capture greater market share, or improve patient adherence, leading to better outcomes and higher revenue.
- IP Creation: The use of a novel or co-processed excipient can be part of a new, patentable formulation, extending the product’s commercial life.
4. Q: My API is poorly soluble. How can I formulate an alcohol-free liquid for pediatric use?
A: This is a classic and significant formulation challenge. Since ethanol is not an option, you must turn to alternative solubilization techniques. The strategy will depend on the API’s properties:
- Cosolvency: Create a solvent system using other water-miscible solvents like glycerin or polyethylene glycols (PEGs). However, be mindful of the safety limits for these excipients in pediatric populations as well.
- pH Adjustment: If your API is a weak acid or base, adjusting the pH of the solution to form a soluble salt is often the most effective approach.
- Surfactant Systems: Use a suitable, low-irritation surfactant to create a micellar solution or a self-emulsifying drug delivery system (SEDDS).
- Complexation: Employ cyclodextrins to form a soluble inclusion complex with the API.
- Suspension: If achieving a stable solution is not feasible, the alternative is to develop a well-formulated suspension. This requires careful selection of wetting agents to ensure the API particles are properly dispersed and suspending agents (thickeners) to prevent settling, along with an effective, pediatric-safe preservative system.
5. Q: We are a generic company. How can we use the “exclusion filter” concept to our advantage?
A: The exclusion filter framework is a powerful strategic tool for generic companies in two key ways:
- For ANDA (Abbreviated New Drug Application) “Design-Around” Strategies: Carefully analyze the innovator’s formulation patents to identify the specific excipients and combinations they have claimed. You can then apply an exclusion filter to deliberately avoid these patented components. This guides your R&D to develop a formulation that is bioequivalent but non-infringing, creating a clear legal path to market entry upon expiration of the primary API patent.
- For 505(b)(2) “Value-Added” Generic Strategies: Analyze the innovator’s product to identify a formulation weakness or an unmet need in a specific patient subpopulation. For example, if the blockbuster branded drug is a lactose-containing tablet, you can apply a “lactose-free” filter to develop a new version for lactose-intolerant patients. This creates a differentiated product with its own market exclusivity and allows you to compete on clinical value and patient preference, not just on price.
Works cited
- Definition of Pharmaceutical Excipients, accessed October 6, 2025, https://www.pharmaexcipients.com/pharmaceutical-excipients-some-definition/
- Pharmaceutical excipients – where do we begin? – Australian Prescriber, accessed October 6, 2025, https://australianprescriber.tg.org.au/articles/pharmaceutical-excipients-where-do-we-begin.html
- How Excipients Shape Drug Effectiveness – DrugPatentWatch – Transform Data into Market Domination, accessed October 6, 2025, https://www.drugpatentwatch.com/blog/how-excipients-shape-drug-effectiveness/
- What is an excipient? – Pharmaceutical Press, accessed October 6, 2025, https://www.pharmaceuticalpress.com/resources/article/what-is-an-excipient/
- The Role of Pharma Excipients in Enhancing Drug Stability and Absorption – Colorcon, accessed October 6, 2025, https://www.colorcon.com/education-insights/the-role-of-pharma-excipients-in-enhancing-drug-stability-and-absorption
- Why patient centricity is key to long-term pharma company success …, accessed October 6, 2025, https://www.adlittle.com/en/insights/prism/why-patient-centricity-key-long-term-pharma-company-success
- The Business Case for Patient Experience – ACRP, accessed October 6, 2025, https://acrpnet.org/2022/10/18/the-business-case-for-patient-experience
- Drug reformulations and repositioning in the pharmaceutical industry and their impact on market access: regulatory implications – PMC – PubMed Central, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC4865781/
- Challenges of Pharmaceutical Industry: Key Issues in 2025 – Viseven, accessed October 6, 2025, https://viseven.com/pharmaceutical-industry-challenges/
- Patient-Centricity in Drug Development – Getting it Right the First Time – Eradigm Consulting, accessed October 6, 2025, https://eradigm.com/patient-centricity-in-drug-development-getting-it-right-the-first-time/
- Excipients in pharmaceuticals: mechanisms of hypersensitivity and …, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC12237841/
- Biologic excipients: Importance of clinical awareness of inactive ingredients – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC7316246/
- Biologic excipients: Importance of clinical awareness of inactive ingredients | PLOS One, accessed October 6, 2025, https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0235076
- Allergy and intolerance | Medicines Learning Portal, accessed October 6, 2025, https://www.medicineslearningportal.org/2016/10/excipients-and-patients-part-2.html
- Medicines, excipients, and dietary intolerances | The BMJ, accessed October 6, 2025, https://www.bmj.com/content/358/bmj.j3468
- Medicines, excipients and dietary intolerances | Drug and Therapeutics Bulletin, accessed October 6, 2025, https://dtb.bmj.com/content/54/8/93
- THE PCCA BLOG | Challenges in Pediatric Compounding: Excipients and Dosage Forms, accessed October 6, 2025, https://www.pccarx.com/Blog/challenges-in-pediatric-compounding-excipients-and-dosage-forms
- Food Additives and Allergies/Intolerances – FoodAllergy.org, accessed October 6, 2025, https://www.foodallergy.org/resources/food-additives-and-allergies/intolerances
- (PDF) Toxicity of Azo Dyes in Pharmaceutical Industry – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/333023104_Toxicity_of_Azo_Dyes_in_Pharmaceutical_Industry
- Natural vs Synthetic Food Colors: Which Is Safer? – Hridhan Chem, accessed October 6, 2025, https://hridhanchem.com/natural-food-colors-vs-synthetic-food-colors/
- Allergy or Hypersensitivity to Pharmaceutical Excipients – TAYSIDE PRESCRIBER, accessed October 6, 2025, https://www.nhstaysideadtc.scot.nhs.uk/approved/bulletin/taypres/2022/Issue%20165%20-%20Allergy%20or%20Hypersensitivity%20to%20Pharmaceutical%20Excipients.pdf
- Drug-Excipient Compatibility Studies in Formulation Development: Current Trends and Techniques – Fisher Digital Publications, accessed October 6, 2025, https://fisherpub.sjf.edu/cgi/viewcontent.cgi?article=1213&context=pharmacy_facpub
- Drug‑Excipient Compatibility Study Through a Novel Vial‑in‑Vial Experimental Setup: A Benchmark Study, accessed October 6, 2025, https://www.pharmaexcipients.com/news/novel-vial%E2%80%91in%E2%80%91vial/
- Excipients Limitations: Challenges in Drug Formulation and Delive – Open Access Journals, accessed October 6, 2025, https://www.openaccessjournals.com/articles/excipients-limitations-challenges-in-drug-formulation-and-delivery-18459.html
- Drug-Excipient Compatibility Study Through a Novel Vial-in-Vial Experimental Setup, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC10169295/
- Pharmaceutical lactose: Making the right choice – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/286494704_Pharmaceutical_lactose_Making_the_right_choice
- Overcoming Excipient Risks and Challenges for Parenteral Formulations – Sigma-Aldrich, accessed October 6, 2025, https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/pharmaceutical-and-biopharmaceutical-manufacturing/classical-pharma-manufacturing/excipients-parenteral-formulations
- Excipient – Wikipedia, accessed October 6, 2025, https://en.wikipedia.org/wiki/Excipient
- Pharmaceutical Excipients – Types and Functions of Excipients – Pharmatech – Rx, accessed October 6, 2025, https://pharmatech-rx.com/pharmaceutical-excipients/
- Excipient Quality and Selection – PharmTech, accessed October 6, 2025, https://www.pharmtech.com/view/excipient-quality-and-selection
- Guidance for Industry #169 – Drug Substance – FDA, accessed October 6, 2025, https://www.fda.gov/media/69923/download
- 1086 IMPURITIES IN DRUG SUBSTANCES AND DRUG PRODUCTS – USP-NF, accessed October 6, 2025, https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/usp-nf-notices/c1086_m99805_PF436.pdf
- WHO Expert Committee on Specifications for Pharmaceutical Preparations: fifty – World Health Organization (WHO), accessed October 6, 2025, https://cdn.who.int/media/docs/default-source/medicines/norms-and-standards/guidelines/production/trs1052_annex2.pdf?sfvrsn=4729bcf0_6&download=true
- The Current States, Challenges, Ongoing Efforts, and Future …, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC9026161/
- Excipient Selection In Parenteral Formulation Development, accessed October 6, 2025, https://www.pharmaexcipients.com/wp-content/uploads/attachments/257115883-Excipient-for-parenteral.pdf?t=1425059372
- Inactive Ingredients Database Download – FDA, accessed October 6, 2025, https://www.fda.gov/drugs/drug-approvals-and-databases/inactive-ingredients-database-download
- Inactive ingredient Search for Approved Drug Products | HealthData.gov, accessed October 6, 2025, https://healthdata.gov/FDA/Inactive-ingredient-Search-for-Approved-Drug-Produ/7zx4-ci7k
- Inactive ingredient Search for Approved Drug Products – Dataset – Catalog, accessed October 6, 2025, https://catalog.data.gov/dataset/inactive-ingredient-search-for-approved-drug-products
- FDA Inactive Ingredient List – pharma excipients, accessed October 6, 2025, https://www.pharmaexcipients.com/excipient-sources/inactive-ingredient-search-for-approved-drug-products-in-the-usa/
- Search Databases – FDA, accessed October 6, 2025, https://www.fda.gov/industry/fda-basics-industry/search-databases
- Drug Approvals and Databases | FDA, accessed October 6, 2025, https://www.fda.gov/drugs/development-approval-process-drugs/drug-approvals-and-databases
- Do Excipients Matter to Clinical Pharmacology and Therapeutics? – ASCPT, accessed October 6, 2025, https://www.ascpt.org/Portals/28/docs/Annual%20Meetings/Annual%20Meeting%20Archive/2017%20Annual%20Meeting/Friday%20March%2017/How%20Inert%20are%20Excipients_Robert%20Lionberger.pdf?ver=2018-04-18-093622-043
- Review of Current Issues in Pharmaceutical Excipients – PharmTech, accessed October 6, 2025, https://www.pharmtech.com/view/review-current-issues-pharmaceutical-excipients
- Policy document Guideline on the excipients in the label and the package leaflet of medicinal products for human use and MEB clarification of the Annex, accessed October 6, 2025, https://www.cbg-meb.nl/binaries/college-ter-beoordeling-van-geneesmiddelen/documenten/beleidsdocumenten/2023/01/01/meb-8/MEB08-180911EN-Guideline+on+the+excipients+and+MEB+clarification.pdf
- Annex to the European Commission guideline on ‘Excipients in the labelling and package leaflet of medicinal products for human use’, accessed October 6, 2025, https://www.ema.europa.eu/en/annex-european-commission-guideline-excipients-labelling-package-leaflet-medicinal-products-human-use
- Excipients labelling | European Medicines Agency (EMA), accessed October 6, 2025, https://www.ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/product-information-requirements/excipients-labelling
- The Future of Pharmaceutical Excipients: A $14 Billion Market Driven by Global Demand, accessed October 6, 2025, https://www.americanpharmaceuticalreview.com/Featured-Articles/618347-The-Future-of-Pharmaceutical-Excipients-A-14-Billion-Market-Driven-by-Global-Demand/
- USP-NF – 〈1059〉 Excipient Performance., accessed October 6, 2025, https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/revisions/gc-1059-excipient-performance-pf-46-1.pdf
- The curious evolution of lactose intolerance – American Chemical Society, accessed October 6, 2025, https://www.acs.org/pressroom/tiny-matters/the-curious-evolution-of-lactose-intolerance.html
- Lactose intolerance – Genetics – MedlinePlus, accessed October 6, 2025, https://medlineplus.gov/genetics/condition/lactose-intolerance/
- en.wikipedia.org, accessed October 6, 2025, https://en.wikipedia.org/wiki/Lactose_intolerance#:~:text=Epidemiology,-An%20estimate%20of&text=Worldwide%2C%20about%2080%25%20of%20people,of%20Asia%20are%20lactose%20intolerant.
- Lactose Intolerance | Boston Children’s Hospital, accessed October 6, 2025, https://www.childrenshospital.org/conditions/lactose-intolerance
- Lactose Intolerance – StatPearls – NCBI Bookshelf, accessed October 6, 2025, https://www.ncbi.nlm.nih.gov/books/NBK532285/
- Assessing the clinical impact of lactose in medicines – SPS – Specialist Pharmacy Service, accessed October 6, 2025, https://www.sps.nhs.uk/articles/assessing-the-clinical-impact-of-lactose-in-medicines/
- Low-dose lactose in drugs neither increases breath hydrogen excretion nor causes gastrointestinal symptoms | Request PDF – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/51430205_Low-dose_lactose_in_drugs_neither_increases_breath_hydrogen_excretion_nor_causes_gastrointestinal_symptoms
- Lactose-Free Products Market Growth, Analysis | Industry Insights & Statistics | MarketsandMarkets, accessed October 6, 2025, https://www.marketsandmarkets.com/Market-Reports/lactose-free-products-market-4457397.html
- Pharmaceutical Lactose Market Size, Share, Trends & Forecast, accessed October 6, 2025, https://www.verifiedmarketresearch.com/product/pharmaceutical-lactose-market/
- Lactose: Characteristics, Food and Drug-Related Applications, and Its Possible Substitutions in Meeting the Needs of People with Lactose Intolerance – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC9141425/
- Lactose Technical manual for oral solid dose – by Kerry – Pharma Excipients, accessed October 6, 2025, https://www.pharmaexcipients.com/news/lactose-technical-manual-kerry/
- Influence of different types of lactose on tablets compactibility – SciELO México, accessed October 6, 2025, https://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1870-01952015000200041
- Insight Into a Novel Strategy for the Design of Tablet Formulations Intended for Direct Compression | Request PDF – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/314296177_Insight_Into_a_Novel_Strategy_for_the_Design_of_Tablet_Formulations_Intended_for_Direct_Compression
- FORMULATION FORUM – Manufacturing of Solid Oral Dosage Forms by Direct Compression – Drug Development and Delivery, accessed October 6, 2025, https://drug-dev.com/formulation-forum-manufacturing-of-solid-oral-dosage-forms-by-direct-compression/
- Novel Insights Into Excipient Effects on the Biopharmaceutics of APIs …, accessed October 6, 2025, https://www.researchgate.net/publication/261734855_Novel_Insights_Into_Excipient_Effects_on_the_Biopharmaceutics_of_APIs_from_different_BCS_classes_Lactose_in_Solid_Oral_Dosage_Forms
- The Effects of Lactose, Microcrystalline Cellulose and Dicalcium Phosphate on Swelling and Erosion of Compressed HPMC Matrix Tablets: Texture Analyzer – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC3870058/
- Understanding the Different Types of Excipients – Ankit Pulps, accessed October 6, 2025, https://www.celluloseankit.com/different-types-of-excipients/
- Potential alternatives to lactose in spray-dried powders for pulmonary drug delivery | Request PDF – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/295384623_Potential_alternatives_to_lactose_in_spray-dried_powders_for_pulmonary_drug_delivery
- Eye on Excipients: Formulating for Lactose Intolerance | Tablets …, accessed October 6, 2025, https://www.tabletscapsules.com/3641-Technical-Articles/592426-Eye-on-Excipients-Formulating-for-Lactose-Intolerance/
- The Use of in a Lactose-free Reformulation of Cetirizine Tablets EMDEX – Pharma Excipients, accessed October 6, 2025, https://www.pharmaexcipients.com/wp-content/uploads/2020/02/The-Use-of-EMDEX%C2%AE-in-a-Lactose-free-Reformulation-of-Cetirizine-Tablets.pdf
- Pharmaceutical design of a new lactose-free coprocessed excipient: Application of hydrochlorothiazide as a low solubility drug model | Request PDF – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/225046146_Pharmaceutical_design_of_a_new_lactose-free_coprocessed_excipient_Application_of_hydrochlorothiazide_as_a_low_solubility_drug_model
- WO2022180444A1 – Lactose free formulation of empagliflozin using direct compression process – Google Patents, accessed October 6, 2025, https://patents.google.com/patent/WO2022180444A1/en
- Overcoming Formulation Challenges in Generic Drug Development – DrugPatentWatch – Transform Data into Market Domination, accessed October 6, 2025, https://www.drugpatentwatch.com/blog/overcoming-formulation-challenges-in-generic-drug-development/
- Development and Evaluation of Lactose-Free Single-Unit and …, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11597082/
- Animal-derived ingredients in medicines: a framework for ethical prescribing practices, accessed October 6, 2025, https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1693059/full
- Halal Vegan Supplements | ISA, accessed October 6, 2025, https://www.isahalal.com/news-events/blog/vegan-supplements-and-halal-criteria
- What are gelatin capsules? And what are the alternatives …, accessed October 6, 2025, https://www.darlingii.com/rousselot/resources/blogs/2019/03/rousselot-what-are-gelatin-capsules-and-what-are-the-alternatives
- Shell Formulation in Soft Gelatin Capsules: Design and Characterization – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11468233/
- Lifestylopathy: Ethical Pharmaceuticals and Halal Compliance …, accessed October 6, 2025, https://esmed.org/lifestylopathy-ethical-pharmaceuticals-and-halal-compliance/
- What Patients On a Vegan Diet Need to Know About Medications | Pharmacy Times, accessed October 6, 2025, https://www.pharmacytimes.com/view/what-patients-with-a-vegan-diet-need-to-know-about-medications
- Vegan, Vegetarian and Gelatin Capsules | What is the Difference, accessed October 6, 2025, https://www.lfacapsulefillers.com/articles/vegan-vegetarian-and-gelatin-capsules
- Gelatin vs. Veg Caps: Which Capsule is better? (Acne Help) | MDacne, accessed October 6, 2025, https://www.mdacne.com/questions/gelatin-vs-veg-caps-which-capsule-is-better
- The strategic advantage of HPMC capsules in modern … – ACG, accessed October 6, 2025, https://www.acg-world.com/blogs/strategic-advantage-hpmc-capsules-modern-pharmaceutical-manufacturing
- Modified pea starch as an alternative to gelatin in capsule shell, accessed October 6, 2025, https://www.roquette.com/innovation-hub/pharma/expert-opinion/lycagel-premix-vegetal-alternative-to-gelatin-in-capsule-shell
- HPMC Capsules: Why They Are Gaining Popularity in the Supplement Industry, accessed October 6, 2025, https://adragos-pharma.com/hpmc-capsules-why-they-are-gaining-popularity-in-the-supplement-industry/
- The Rise of HPMC Capsules: A Sustainable Alternative to Gelatin …, accessed October 6, 2025, https://mikazone.com/the-rise-of-hpmc-capsules/
- Differences between Gelatin and HPMC capsules – Blog CapsCanada, accessed October 6, 2025, https://blog.capscanada.com/differences-between-gelatin-capsules-and-hpmc-capsules
- Drug release behavior of polymeric matrix filled in capsule – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC5094431/
- Evaluation of hard gelatin capsules and hydroxypropyl methylcellulose containing ampicillin, accessed October 6, 2025, https://www.researchgate.net/publication/262780188_Evaluation_of_hard_gelatin_capsules_and_hydroxypropyl_methylcellulose_containing_ampicillin
- Evaluation of hard gelatin capsules and hydroxypropyl methylcellulose containing ampicillin, accessed October 6, 2025, https://www.scielo.br/j/qn/a/bTZTPnsTX8zvDmkZQ3jZPFd/
- Vegetarian Capsule Material Market – Global Industry Analysis and Forecast (2024-2030) by Type, Application, and Region, accessed October 6, 2025, https://www.maximizemarketresearch.com/market-report/global-vegetarian-capsule-material-market/106092/
- Vegetable Capsules Market Analysis an Forecast to 2031 – InsightAce Analytic, accessed October 6, 2025, https://www.insightaceanalytic.com/report/vegetable-capsules-market/2465
- The unseen animal behind medicine: exploring considerations of animal-derived medications and anaesthetics in today’s landscape – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11719331/
- Prescribing advice on animal derived medicines in within Primary Care, accessed October 6, 2025, https://ggcmedicines.org.uk/media/uploads/prescribing_resources/animal_derived_medicines_-_1609.pdf
- Pharmaceutical Solutions II: Nonaqueous Solutions | Pharmlabs, accessed October 6, 2025, https://pharmlabs.unc.edu/labexercises/compounding/pharmsol2/
- Development and Chemical Stability Studies of Alcohol-Free Phenobarbital Solution for Use in Pediatrics: a Technical Note – PubMed Central, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC2977013/
- Alcohol use in paediatric medication: potential impact on the brain and the current regulation, accessed October 6, 2025, https://www.researchgate.net/publication/392268928_Alcohol_use_in_paediatric_medication_potential_impact_on_the_brain_and_the_current_regulation
- Ethanol Content of Medications and Its Effect on Blood Alcohol Concentration in Pediatric Patients – PMC – PubMed Central, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11001219/
- Questions and Answers on Ethanol in the context of the revision of the guideline on ‘Excipients in the label and package leaflet – European Medicines Agency, accessed October 6, 2025, https://www.ema.europa.eu/en/documents/scientific-guideline/questions-and-answers-ethanol-context-revision-guideline-excipients-label-and-package-leaflet-medicinal-products-human-use_en.pdf
- 9 A Guide to Pharmaceutical Excipients (Inert Ingredients) – Pediatric Pharmacotherapy – The University of Virginia, accessed October 6, 2025, https://med.virginia.edu/pediatrics/wp-content/uploads/sites/237/2015/12/199609.pdf
- Alcohol, ethyl (ethanol) dosing, indications, interactions, adverse effects, and more, accessed October 6, 2025, https://reference.medscape.com/drug/alcohol-ethyl-ethanol-343730
- Solubilization techniques used for poorly water-soluble drugs – PMC – PubMed Central, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11628819/
- Alcohol-free Extemporaneous Formulations of Furosemide Are Chemically and Physically Stable in Ora-Blend Products for 30 Days – PubMed, accessed October 6, 2025, https://pubmed.ncbi.nlm.nih.gov/32401745/
- Pharmaceutical Natural and Synthetic Colorants, Pigments, Dyes, and Lakes: Applications, Perspectives, and Regulatory Aspects – Taylor & Francis eBooks, accessed October 6, 2025, https://www.taylorfrancis.com/chapters/edit/10.1201/9781003057505-6/pharmaceutical-natural-synthetic-colorants-pigments-dyes-lakes-applications-perspectives-regulatory-aspects-debarshi-kar-mahapatra-sanjay-kumar-bharti
- Medication Dyes as a Source of Drug Allergy – JDDonline – Journal of Drugs in Dermatology, accessed October 6, 2025, https://jddonline.com/articles/medication-dyes-as-a-source-of-drug-allergy-S1545961613P0099X/?_page=3
- Safety of ingestion of yellow tartrazine by double-blind placebo controlled challenge in 26 atopic adults | Allergologia et Immunopathologia – Elsevier, accessed October 6, 2025, https://www.elsevier.es/en-revista-allergologia-et-immunopathologia-105-articulo-safety-ingestion-yellow-tartrazine-by-S0301054609001426
- Food Allergies | FDA, accessed October 6, 2025, https://www.fda.gov/food/nutrition-food-labeling-and-critical-foods/food-allergies
- Toxicity of tartrazine: Scientific review report – Therapeutic Goods Administration (TGA), accessed October 6, 2025, https://www.tga.gov.au/sites/default/files/tartrazine-toxicity-1402.pdf
- Medication dyes as a source of drug allergy | Request PDF – ResearchGate, accessed October 6, 2025, https://www.researchgate.net/publication/235397567_Medication_dyes_as_a_source_of_drug_allergy
- Why Pharma Is Going Direct to Consumers – IQVIA, accessed October 6, 2025, https://www.iqvia.com/locations/united-states/blogs/2025/08/why-pharma-is-going-direct-to-consumers
- Red Dye: The Silent Red Flag in Your Medicine Cabinet – NewBeauty, accessed October 6, 2025, https://www.newbeauty.com/red-dye-in-medicine/
- The Challenge for Manufacturers in Tablet Splitting – Pharmaceutical Technology, accessed October 6, 2025, https://www.pharmtech.com/view/challenge-manufacturers-tablet-splitting
- Colour of Medicines and Children’s Acceptability? A Systematic Literature Review of Children’s Perceptions about Colours of Oral Dosage Forms – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC10384887/
- 2024 – Pharmacoating Dyes & Pigments – Pharmaceutical Colorants, accessed October 6, 2025, https://pharmacoating.com/english/markets/pharmaceutical-colorants/
- A review of emerging technologies enabling improved solid oral dosage form manufacturing and processing, accessed October 6, 2025, https://pure.ulster.ac.uk/files/91447171/1_s2.0_S0169409X21002325_main.pdf
- Direct Compression Versus Granulation – Pharmaceutical Technology, accessed October 6, 2025, https://www.pharmtech.com/view/direct-compression-versus-granulation
- COMBILOSE: A novel lactose-based co-processed excipient for direct compression, accessed October 6, 2025, https://japsonline.com/abstract.php?article_id=3892&sts=2
- The impact of lactose type on disintegration: An integral study on porosity and polymorphism, accessed October 6, 2025, https://www.researchgate.net/publication/364465373_The_impact_of_lactose_type_on_disintegration_an_integral_study_on_porosity_and_polymorphism
- Impact of Pharmaceutical Excipients on Drug Bioavailability and Stability – JOCPR, accessed October 6, 2025, https://www.jocpr.com/articles/impact-of-pharmaceutical-excipients-on-drug-bioavailability-and-stability-10277.html
- FDA Color Additive Scrutiny Expands to Pharmaceuticals – Latham & Watkins LLP, accessed October 6, 2025, https://www.lw.com/en/insights/fda-color-additive-scrutiny-expands-to-pharmaceuticals
- Guidance for Industry: Questions and Answers Regarding Food Allergen Labeling (Edition 5) | FDA, accessed October 6, 2025, https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-questions-and-answers-regarding-food-allergen-labeling-edition-5
- Impact Of Excipient Variability On Drug Product Processing And Performance, accessed October 6, 2025, https://www.researchgate.net/publication/282761319_Impact_Of_Excipient_Variability_On_Drug_Product_Processing_And_Performance
- Pharmaceutical Excipients Market Projects USD 15.49 Billion by 2034 – Towards Healthcare, accessed October 6, 2025, https://www.towardshealthcare.com/insights/pharmaceutical-excipients-market-sizing
- Addressing the Challenges of Patient-Centric Drug Development – Lubrizol, accessed October 6, 2025, https://www.lubrizol.com/company/insights/2021/10/addressing-the-challenges-of-patient-centric-drug-development
- Evolution of Patient-Centricity in the Multi-Indication Pharmaceutical Era – IQVIA, accessed October 6, 2025, https://www.iqvia.com/blogs/2025/03/evolution-of-patient-centricity-in-the-multi-indication-pharmaceutical-era
- Cracking the Code: Using Drug Patents to Reveal Competitor …, accessed October 6, 2025, https://www.drugpatentwatch.com/blog/cracking-the-code-using-drug-patents-to-reveal-competitor-formulation-strategies/
- The Predictive Pipeline: Structuring Drug Development Timelines with AI-Driven Patent Intelligence – DrugPatentWatch – Transform Data into Market Domination, accessed October 6, 2025, https://www.drugpatentwatch.com/blog/the-predictive-pipeline-structuring-drug-development-timelines-with-ai-driven-patent-intelligence/
- SPECIAL FEATURE – Excipients: Advanced Biologics Require Innovative Excipient Science, accessed October 6, 2025, https://drug-dev.com/special-feature-excipients-advanced-biologics-require-innovative-excipient-science/
- CHALLENGES AND APPROACHES ON FORMULATION AND MANUFACTURING OF ORALLY DISSOLVING TABLET DOSAGE FORMS – IJCRT.org, accessed October 6, 2025, https://www.ijcrt.org/papers/IJCRT2404591.pdf
- Spray Drying for Direct Compression of Pharmaceuticals – Pharma Excipients, accessed October 6, 2025, https://www.pharmaexcipients.com/news/spray-drying-direct-compression/
- Emerging Artificial Intelligence (AI) Technologies Used in the Development of Solid Dosage Forms – PMC, accessed October 6, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC9694557/
- Pharmaceutical Excipients Industry – Future Trends and Their Impact on Global Growth, accessed October 6, 2025, https://www.marketsandmarkets.com/ResearchInsight/pharmaceutical-excipients-industry.asp
- Pharmaceutical Excipients Market Size to Hit USD 15.43 Bn by 2034, accessed October 6, 2025, https://www.precedenceresearch.com/pharmaceutical-excipients-market