Last updated: April 24, 2026
Where is sumatriptan succinate in the product lifecycle?
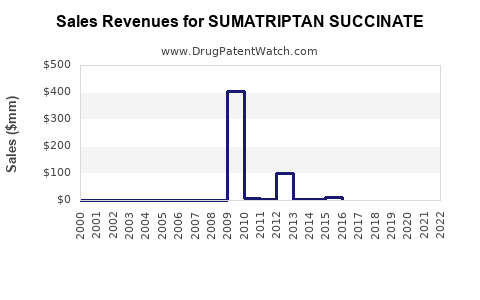
Sumatriptan succinate is a mature, long-established prescription migraine therapy with multiple dosage forms and strong historical brand penetration. The commercial landscape is dominated by generic competition in many markets, while branded and specialty-delivery variants (notably subcutaneous and transdermal/nasal formulations where available) maintain differentiated pricing in specific geographies and channels.
Lifecycle signal from supply structure and pricing reality
- Core active ingredient is off-patent in many jurisdictions, driving widespread generic penetration.
- Margin durability is linked to:
- formulation differentiation (delivery route, device/packaging, patient preference),
- label scope (acute migraine vs. other headache indications),
- distribution contracts and pharmacy reimbursement dynamics,
- local regulatory and patent-exclusivity remnants (where they exist for specific formulations).
Commercial implication: Broad-based generic substitution limits sustained revenue growth for the base compound in most countries. Financial trajectory therefore tracks incremental share shifts across brands, route-specific products, and price erosion cycles rather than new-molecule adoption.
What drives market demand for sumatriptan succinate?
Demand for sumatriptan is anchored to migraine incidence and the need for rapid acute symptom relief. Key market drivers operate through patient, prescriber, and payer pathways.
Patient and prescriber pull
- Migraine prevalence supports steady baseline demand for acute treatments.
- Triptans remain central acute therapy options; sumatriptan is frequently treated as a “default” triptan due to broad clinical familiarity.
- Rapid onset formulations increase attractiveness in patients with high disability from pain and nausea.
Payer and formulary dynamics
- Generics pressure net price.
- Formularies favor cost-effective triptan options, which typically leads to:
- switching to the lowest cost covered product,
- limiting branded use to specific criteria (step edits, prior authorization) in some markets.
Channel effects
- Pharmacy distribution favors availability and contracted wholesaler terms.
- Institutional tendering for hospitals can increase uptake of particular delivery routes (for example, injectable for emergency/acute settings).
How has competitive pressure shaped pricing and margins?
Generic competition is the central market force. Once the molecule and most formulations lose exclusivity, the market re-prices toward acquisition cost and contracted reimbursement ceilings.
Competitive pattern
- Price compression after generic entries tends to be steep initially, then stabilizes as supply consolidates among fewer manufacturers.
- Differentiated formulations can slow decline but usually cannot fully prevent margin compression if they also face generic substitution or if payer policies place them in the same therapeutic class with interchangeable switching.
Brand vs generic roles (typical structure)
- Branded originators: maintain higher net prices where exclusivity or route differentiation persists.
- Generics: expand share rapidly post-launch and establish price floors through competitive bidding.
- Route differentiation: can preserve relative pricing for delivery systems that require patient/clinician adoption of a specific regimen.
What are the main product and regulatory levers affecting revenues?
Revenue is driven by how route-specific products land within reimbursement and how regulatory labeling supports switching.
Dosage forms and route strategy
Sumatriptan is sold across multiple routes in different markets, commonly including:
- oral tablets,
- nasal sprays,
- subcutaneous injections,
- and other formulation variants depending on country approvals and market presence.
Route mix matters because:
- subcutaneous products tend to serve patients with faster onset needs and emergency/acute administration contexts,
- nasal products target patients who cannot tolerate oral intake,
- oral products face the strongest generic substitution pressure and class-level price competition.
Patent and exclusivity structure (what it does to the P&L)
Because the molecule is old, the “financial trajectory” primarily reflects:
- expiry of patents on specific formulations,
- any remaining exclusivity for newer variants,
- and the speed of generic market entry in each geography.
Net outcome:
- revenue growth is usually flat-to-declining at the base level,
- stability occurs when brand variants retain differentiated reimbursement positions longer than generics in core channels.
What does the long-run financial trajectory typically look like for sumatriptan succinate?
Sumatriptan’s financial trajectory follows a mature-drug curve:
- Early growth phase driven by brand launch and neurologist/primary care adoption.
- Plateau as competitors enter within the triptan class and as prescribers stabilize.
- Multi-year decline from generic substitution and payer price resets.
- Periodic stabilization when formulation mix, contracted supply, and route-specific demand offset some erosion.
Expected revenue shape over time (mature pattern)
- Net sales: generally declines after generic entry; any growth is modest and tied to route-specific brand resilience or incremental patient share.
- Gross margin: compresses due to lower net price; manufacturers that compete on cost and scale can partially defend margin.
- Operating income: tends to track sales declines unless cost structure scales down or the portfolio shifts to higher-margin variants.
Where does sumatriptan succinate sit versus other migraine therapies in market dynamics?
Sumatriptan competes in a class where newer preventive and acute therapies have expanded. However, for many payers and patients, triptans continue to be the default acute choice due to:
- long clinical familiarity,
- broad accessibility,
- oral simplicity and established prescribing workflows.
Class dynamics
- CGRP-targeting therapies (preventive and acute) have carved share in some settings, especially where payers cover them with fewer barriers or where patients prefer non-triptan options.
- In cost-sensitive segments, triptans retain strong relevance due to lower cost relative to newer branded agents.
Net effect: Newer acute options pressure share in selective payer pockets, but generic availability keeps sumatriptan anchored as an economic standard therapy.
How do safety, tolerability, and label coverage influence commercial outcomes?
Clinical positioning is stable: sumatriptan is established for acute migraine. Commercial outcomes track prescriber confidence and patient willingness to use triptans.
Safety profile as a commercial constant
- The safety profile drives long-term trust and steady prescribing patterns.
- Switching behavior occurs mainly when patients experience inadequate efficacy or intolerance, or when prescribers move patients to alternative triptans or newer non-triptan acute therapies.
Label and substitution behavior
- Where label supports interchangeability and payers treat triptans as class substitutes, switching accelerates.
- Where route-specific labeling or tolerability considerations matter, route-level differentiation can preserve revenue longer.
What are the practical market metrics to monitor going forward?
Because the drug is mature, the key to forecasting is not breakthrough clinical value but operational and reimbursement signals.
Market metrics (high-signal indicators)
- Number and timing of generic launches by route and geography.
- Average net price and discount intensity in contracted pharmacy channels.
- Formulary status and prior authorization criteria changes.
- Share movement across route types (oral vs nasal vs injection).
- Patient adherence and repeat prescription patterns in migraine populations.
Business implications for forecasting
- Expect recurring pricing pressure tied to generic supply updates and reimbursement resets.
- Monitor route-mix shifts; injectable and nasal routes can provide temporary stabilization even under generics if patient switching is slower.
What is the investment-relevant view of financial trajectory and risk?
Revenue resilience
- Base compound faces structural headwinds from generic substitution.
- Resilience comes from:
- route differentiation,
- channel contracting,
- and any lingering formulation exclusivity in specific markets.
Major risks
- Faster-than-expected generic penetration.
- Payer formulary tightening that pushes more switching.
- Contract losses that re-route volume to lower-cost alternatives.
Upside vectors
- Stronger adoption of route variants that avoid immediate class-based substitution.
- Consolidation among generic suppliers that can raise effective pricing.
- Market-specific reimbursement changes that favor specific presentations.
Key Takeaways
- Sumatriptan succinate is a mature migraine acute therapy with revenue dominated by generic-driven pricing cycles and route-level differentiation rather than new clinical uptake.
- Demand stays structurally supported by migraine incidence, but net sales trend is typically flat-to-declining at the base level as generics replace branded supply.
- Financial trajectory is largely a function of (1) timing and intensity of generic entry, (2) reimbursement and formulary position, and (3) product route mix (oral vs nasal vs injection).
- Newer CGRP acute and preventive options may shift some acute demand, but generic affordability keeps sumatriptan anchored in cost-sensitive segments.
- Forecasting should prioritize generic launch calendars, net price trends, formulary constraints, and route-mix movements.
FAQs
1. What most strongly determines sumatriptan succinate revenue in mature markets?
Generic entry timing, net price compression from payer reimbursement resets, and route mix across oral, nasal, and injectable presentations.
2. Does sumatriptan face competition primarily from within the triptan class or from newer non-triptan therapies?
Both. Within-class competition is intensified by generic substitution across triptans; non-triptan therapies can take share where payers cover them broadly and where clinicians/patients prefer alternatives.
3. Which dosage route typically offers the best revenue resilience?
In many markets, route-specific differentiated products (often nasal and injectable) can show more durability than oral tablets because switching can be slower and channel demand can be more segment-specific.
4. What is the typical long-run financial pattern for an old migraine active ingredient once generics dominate?
Sales decline or stagnation after generic launches, with periodic stabilization driven by contract dynamics, supply consolidation, and any remaining presentation-level advantages.
5. What business indicators best predict the next revenue turning point?
Upcoming generic launches by route and geography, changes to formulary status and prior authorization requirements, and observed net price trends in contracted pharmacy channels.
References
[1] European Medicines Agency. Sumatriptan: EPAR product information and assessment materials. EMA. https://www.ema.europa.eu/ (accessed via relevant product page for sumatriptan-containing medicinal products).