Last updated: February 19, 2026
Onzetra Xsail, a nasal spray formulation of the antiemetic ondansetron, targets the prevention of chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting (PONV). Its market position is defined by its rapid onset of action, non-invasive delivery, and the established efficacy of ondansetron. The drug's financial trajectory will be influenced by market penetration, competitor landscape, pricing strategies, and patent exclusivity.
What is the Current Market Landscape for Onzetra Xsail?
The market for antiemetics is substantial, driven by the high incidence of nausea and vomiting associated with cancer treatments and surgical procedures. Onzetra Xsail competes within the broader 5-HT3 receptor antagonist class, which includes well-established oral and intravenous formulations of ondansetron, as well as other agents like granisetron and palonosetron.
Key Market Drivers:
- Prevalence of Cancer and Surgeries: The global incidence of cancer and the volume of surgical procedures directly correlate with the demand for effective antiemetic therapies. In 2023, an estimated 1.9 million new cancer cases were diagnosed in the United States alone [1].
- Patient Preference for Convenience: Nasal drug delivery offers a non-invasive alternative to oral or intravenous routes, potentially improving patient compliance and comfort, particularly for patients experiencing severe nausea.
- Clinical Efficacy of Ondansetron: Ondansetron is a proven, first-line treatment for CINV and PONV, with decades of clinical data supporting its efficacy and safety profile.
Competitive Set Analysis:
Onzetra Xsail faces competition from:
- Intravenous (IV) Ondansetron: The standard for acute CINV and PONV, offering rapid and potent symptom control.
- Oral Ondansetron: Widely used for CINV and PONV, offering convenience for outpatient settings and delayed-onset symptom management.
- Other 5-HT3 Receptor Antagonists: Granisetron and palonosetron, available in various formulations (oral, IV, transdermal), offer comparable efficacy but may differ in onset, duration, and cost.
- Newer Antiemetic Classes: Neurokinin-1 (NK1) receptor antagonists and dopamine antagonists are increasingly used, often in combination with 5-HT3 antagonists, particularly for highly emetogenic chemotherapy regimens.
Market Share Considerations:
While specific market share data for Onzetra Xsail is proprietary, its success hinges on capturing a segment of patients who can benefit from its distinct delivery mechanism. Factors influencing adoption include physician prescribing habits, formulary inclusion by payers, and patient/caregiver demand for the nasal spray option.
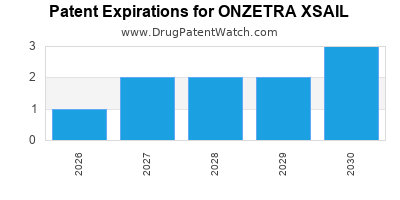
What are the Key Patents and Exclusivity Periods for Onzetra Xsail?
The patent landscape for pharmaceutical products is critical for determining market exclusivity and revenue potential. For Onzetra Xsail, patent protection is centered around the formulation, method of use, and potentially manufacturing processes.
Patent Types and Anticipated Expiry:
- Formulation Patents: These patents protect the specific composition of Onzetra Xsail, including the active pharmaceutical ingredient (ondansetron), excipients, and delivery device. These are typically the longest-lasting patents.
- Method of Use Patents: These patents cover the specific indication for which Onzetra Xsail is approved, such as the prevention of CINV or PONV via nasal administration.
- Manufacturing Process Patents: These can protect novel or efficient ways of producing the drug substance or the final drug product.
Information on specific patent numbers and their expiry dates is publicly available through patent databases such as the USPTO (United States Patent and Trademark Office) and the EPO (European Patent Office). For a comprehensive analysis, a detailed patent search is required. However, general patent terms are usually 20 years from the filing date, with potential extensions through mechanisms like the Patent Term Extension (PTE) in the U.S. to compensate for regulatory review delays.
Regulatory Exclusivity:
Beyond patent protection, regulatory exclusivities play a significant role. For instance, New Chemical Entity (NCE) exclusivity in the U.S. grants 5 years of market protection, while Orphan Drug exclusivity offers 7 years for rare disease treatments. While ondansetron itself is not a new chemical entity, novel formulations like Onzetra Xsail may be eligible for different forms of data exclusivity, depending on the specific regulatory pathway and the nature of the innovation.
Impact on Market Entry for Generics:
The expiry of key patents and loss of regulatory exclusivity will open the door for generic competition. Generic manufacturers will seek to replicate the bioequivalence of Onzetra Xsail, leading to price erosion and a potential decline in market share for the branded product. The timing of this generic entry is a crucial determinant of Onzetra Xsail's long-term financial viability.
What are the Projected Financial Trajectories and Revenue Drivers for Onzetra Xsail?
The financial trajectory of Onzetra Xsail will be shaped by a combination of sales volume, pricing strategy, market penetration, and the eventual impact of generic competition.
Revenue Generation Assumptions:
- Initial Market Penetration: Early revenue will depend on the success of launch strategies, physician adoption, and patient access. Marketing efforts targeting oncologists, anesthesiologists, and hospital pharmacies will be critical.
- Pricing Strategy: The price of Onzetra Xsail will be a key determinant of revenue. It is likely to be positioned at a premium to oral ondansetron due to the novel delivery system and perceived benefits, but competitive with other advanced antiemetic therapies.
- Sales Volume Growth: Sustained revenue growth will be driven by increasing physician comfort with the nasal spray, expansion into new clinical settings, and potential market expansion for ondansetron itself.
- Market Share Capture: The percentage of patients requiring antiemetics who utilize Onzetra Xsail will directly influence its financial performance.
Key Financial Metrics to Monitor:
- Net Sales: The total revenue generated from the sale of Onzetra Xsail, net of discounts, rebates, and returns.
- Gross Profit Margin: The profitability of the drug before operating expenses, indicating the efficiency of manufacturing and pricing.
- Earnings Before Interest, Taxes, Depreciation, and Amortization (EBITDA): A measure of operational profitability.
- Return on Investment (ROI): The profitability relative to the capital invested in research, development, manufacturing, and marketing.
Projected Scenarios:
- Optimistic Scenario: Rapid market adoption, strong formulary acceptance, and effective differentiation against competitors lead to significant market share gains and sustained high pricing, resulting in strong revenue growth through the patent-protected period.
- Base Case Scenario: Steady market penetration, a competitive pricing strategy, and a moderate level of differentiation lead to consistent revenue growth, with a predictable decline post-generic entry.
- Pessimistic Scenario: Slow physician adoption, payer restrictions, intense competition, and a more aggressive generic pricing strategy result in limited market penetration and a shortened period of robust revenue generation.
Impact of Generic Entry:
The introduction of generic versions of Onzetra Xsail will lead to a significant and rapid decline in its average selling price. The rate of this decline will depend on the number of generic manufacturers entering the market and their pricing strategies. While sales volume may initially remain high, revenue will contract. Manufacturers often focus on maintaining brand loyalty through value-added services or by developing next-generation products to mitigate this impact.
What are the Key Performance Indicators (KPIs) for Onzetra Xsail's Success?
Monitoring specific key performance indicators is essential for assessing the ongoing success and financial health of Onzetra Xsail. These metrics provide actionable insights for strategic adjustments and investment decisions.
Sales and Market Penetration KPIs:
- Total Prescription Volume: Tracks the number of prescriptions written for Onzetra Xsail, indicating overall market demand and physician prescribing patterns.
- New Prescription Growth Rate: Measures the rate at which new patients are starting Onzetra Xsail, reflecting market penetration and the effectiveness of marketing efforts.
- Market Share within the Ondansetron Segment: Compares Onzetra Xsail's sales volume to total ondansetron sales (all formulations), highlighting its share of the specific active ingredient market.
- Market Share within the Broader Antiemetic Market: Assesses Onzetra Xsail's position relative to all competing antiemetic products, demonstrating its overall competitive standing.
- Patient Adherence Rates: While more challenging to measure directly for a branded product, this can be inferred through refill rates and patient support program engagement, indicating the drug's long-term utility.
Financial and Commercial KPIs:
- Net Revenue Growth: Tracks the year-over-year increase or decrease in net sales.
- Average Selling Price (ASP): Monitors the revenue generated per unit sold, crucial for understanding pricing power and the impact of discounts/rebates.
- Gross Profit Margin: Assesses the profitability of each unit sold.
- Cost of Goods Sold (COGS) as a Percentage of Net Sales: Indicates manufacturing efficiency.
- Sales, General, and Administrative (SG&A) Expenses as a Percentage of Net Sales: Measures the efficiency of commercial operations and marketing spend.
- Return on Marketing Investment (ROMI): Evaluates the effectiveness of marketing campaigns in driving sales.
Clinical and Patient-Centric KPIs:
- Physician Adoption Rate: Tracks the percentage of target physicians (oncologists, anesthesiologists) who have prescribed Onzetra Xsail.
- Hospital/Clinic Formulary Status: Monitors the ease of access and reimbursement for Onzetra Xsail within healthcare institutions.
- Patient Reported Outcomes (PROs) Post-Launch: While not a direct commercial KPI, ongoing data collection on patient satisfaction and symptom control can inform marketing and clinical support strategies.
Timing of Generic Entry:
- Days to Generic Launch: The number of days from the expiry of key patents or loss of regulatory exclusivity until the first generic competitor enters the market. This directly impacts the duration of brand-level revenue.
What are the Risks and Opportunities for Onzetra Xsail?
Onzetra Xsail operates within a dynamic pharmaceutical market, presenting both significant opportunities for growth and inherent risks that could impact its commercial success and financial performance.
Opportunities:
- Unmet Needs in Specific Patient Populations: Certain patient groups may exhibit poor adherence to oral medications due to severe nausea or difficulty swallowing. Onzetra Xsail's nasal delivery offers a targeted solution.
- Expansion into New Indications or Settings: While currently approved for CINV and PONV, further research could explore its efficacy in other conditions involving nausea and vomiting, or in different clinical settings (e.g., emergency departments, palliative care).
- Combination Therapies: The potential for synergistic effects with other antiemetic classes (e.g., NK1 antagonists) could lead to improved patient outcomes and expanded prescribing.
- Geographic Market Expansion: Introducing Onzetra Xsail into international markets, where ondansetron is approved and demand for advanced antiemetics exists, presents significant growth potential.
- Advancements in Delivery Technology: Future iterations or related products could leverage improved nasal spray technology for enhanced absorption, patient experience, or to address specific pharmacokinetic profiles.
Risks:
- Intense Competition: The antiemetic market is crowded with established generic and branded products. Onzetra Xsail must demonstrate clear clinical and economic advantages to gain market share.
- Payer Restrictions and Reimbursement Challenges: Insurance providers and healthcare systems may impose utilization management controls, prior authorization requirements, or offer lower reimbursement rates, limiting patient access and impacting profitability.
- Physician Prescribing Inertia: Healthcare providers may be resistant to adopting new formulations or delivery methods, preferring to stick with familiar and less expensive options.
- Manufacturing and Supply Chain Disruptions: Any issues with the manufacturing process, raw material sourcing, or distribution could lead to stock-outs and damage brand reputation.
- Adverse Event Profile: Although ondansetron has a well-established safety profile, any unexpected or severe adverse events associated with the nasal formulation could lead to prescribing restrictions or product withdrawal.
- Generic Competition Erosion: The eventual entry of generic versions will lead to significant price erosion and a decline in market share, necessitating a robust post-exclusivity strategy.
- Patent Litigation: Competitors may challenge existing patents, potentially leading to early loss of exclusivity or significant legal expenses.
Strategic Mitigation:
Companies developing and marketing Onzetra Xsail must proactively address these risks through rigorous clinical development, comprehensive health economics and outcomes research (HEOR) to support value propositions, strong payer engagement, targeted physician education, and robust supply chain management.
Key Takeaways
Onzetra Xsail's market viability is anchored in its differentiated nasal delivery of the proven antiemetic ondansetron. Success hinges on effectively penetrating the CINV and PONV markets against established oral and intravenous competitors. Key financial drivers include initial pricing, market adoption rates, and the timing of patent expiry. While significant opportunities exist in addressing patient preferences for non-invasive delivery and potential market expansion, risks such as intense competition, payer access limitations, and the inevitable erosion from generic entry must be actively managed. Continuous monitoring of sales volume, market share, net revenue, and ASP will be critical for assessing performance.
Frequently Asked Questions
What is the primary clinical advantage of Onzetra Xsail over traditional oral ondansetron?
Onzetra Xsail's primary clinical advantage is its nasal spray delivery, offering a non-invasive route that may provide a faster onset of action and improved patient compliance, particularly for individuals experiencing severe nausea or difficulty swallowing.
How does Onzetra Xsail compare in terms of cost to intravenous ondansetron?
Onzetra Xsail is likely priced at a premium to generic oral ondansetron and may fall within a similar price range or slightly higher than intravenous formulations, reflecting the novel delivery system and associated development costs. Direct cost comparisons require specific pricing data from manufacturers and payers.
What is the typical duration of market exclusivity for a novel drug formulation like Onzetra Xsail?
Market exclusivity for a novel formulation like Onzetra Xsail is primarily determined by its patent protection. Patents typically last 20 years from the filing date, with potential extensions through mechanisms like Patent Term Extension (PTE) in the U.S. Regulatory exclusivities may also apply depending on the specific regulatory pathway followed during approval.
What specific types of chemotherapy regimens is Onzetra Xsail indicated for the prevention of nausea and vomiting?
Onzetra Xsail is indicated for the prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. The specific regimens it is approved for are detailed in its official prescribing information, which should be consulted for precise indications.
What are the potential challenges in achieving broad physician adoption for Onzetra Xsail?
Challenges to physician adoption include inertia preferring established treatments, the need for physician education on the benefits of nasal delivery, formulary restrictions imposed by payers, and the competitive landscape offering multiple antiemetic options at various price points.
Citations
[1] National Cancer Institute. (2023, October 26). Cancer statistics. Retrieved from https://www.cancer.gov/about-cancer/understanding/statistics