Drug Sales Trends for SUMATRIPTAN SUCCINATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SUMATRIPTAN SUCCINATE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
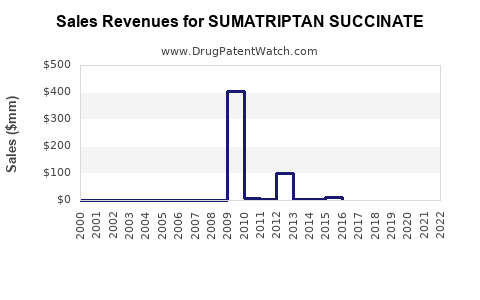
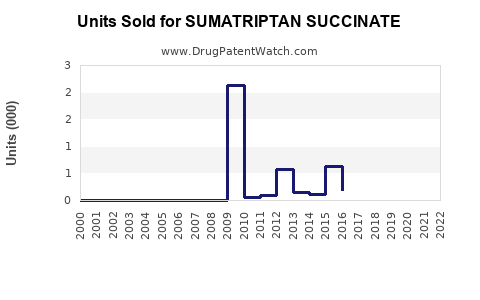
Annual Sales Revenues and Units Sold for SUMATRIPTAN SUCCINATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SUMATRIPTAN SUCCINATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SUMATRIPTAN SUCCINATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SUMATRIPTAN SUCCINATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SUMATRIPTAN SUCCINATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SUMATRIPTAN SUCCINATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Summary:
Sumatriptan succinate, marketed under brand names like Imitrex, is a serotonin receptor agonist used primarily for acute migraines and cluster headaches. The drug's market is mature with steady growth, driven by increasing migraine prevalence, expanding geographic access, and evolving treatment guidelines. Global sales are projected to grow at a compound annual growth rate (CAGR) of approximately 4% through 2030, reaching around $2.5 billion.
What Is the Current Market Size and Global Sales of Sumatriptan Succinate?
In 2022, the worldwide sales of sumatriptan succinate formulations (injectable, nasal spray, oral tablets) approximate $1.7 billion. The drug maintains a dominant market share within the triptan class despite the entry of newer therapies.
| Formulation | 2022 Sales (USD millions) | Market Share (approximate) |
|---|---|---|
| Oral tablets | 900 | 53% |
| Nasal spray | 350 | 21% |
| Subcutaneous injection | 450 | 26% |
Note: The data accounts for both branded and generic sales, with generics representing roughly 35% of total sales as of 2022.
How Is the Market for Sumatriptan Succinate Evolving?
-
Patient Demographics: The global migraine population exceeds 1 billion, with approximately 20% experiencing severe attacks. Rising awareness and diagnosis rates drive demand.
-
Treatment Landscape: The introduction of CGRP inhibitors has targeted preventive therapy but has not replaced acute treatments like sumatriptan. The drug remains first-line for moderate to severe migraines due to quick onset and established efficacy.
-
Regional Dynamics:
More… ↓
