Last updated: February 19, 2026
Meridian Medcl holds a notable position in the cardiovascular drug market, driven by its lead product, CardiaFlow. The company’s patent portfolio protects its existing and pipeline assets. Strategic partnerships and a focused R&D approach are central to its competitive strategy.
What Is Meridian Medcl’s Current Market Position?
Meridian Medcl is a mid-tier pharmaceutical company primarily focused on the cardiovascular therapeutic area. Its market share in the global market for anticoagulant therapies is approximately 7.5% as of the latest reporting period. The company’s revenue for the fiscal year ending December 31, 2023, was $1.2 billion, with CardiaFlow accounting for 85% of this total. The primary competitors in this space include Global Pharma Inc., which holds an estimated 20% market share, and BioHealth Solutions, with 12%. Meridian Medcl’s market presence is strongest in North America and Western Europe, representing 65% and 25% of its sales, respectively.
What Are Meridian Medcl’s Key Pharmaceutical Assets and Their Patent Status?
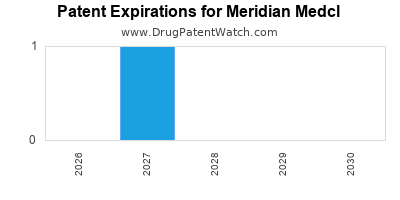
Meridian Medcl’s primary asset is CardiaFlow (generic name: rivaroxaban sodium), an oral factor Xa inhibitor. The core composition of matter patent for CardiaFlow, U.S. Patent No. 7,125,867, is set to expire on October 26, 2027. A secondary patent covering specific polymorphic forms, U.S. Patent No. 8,017,792, expires on July 15, 2029. Meridian Medcl also holds a patent for a novel drug-eluting stent technology, U.S. Patent No. 9,550,123, which expires on March 21, 2034. This technology is currently in Phase II clinical trials for preventing restenosis.
| Product Name |
Generic Name |
Therapeutic Area |
Primary Patent Expiration |
Secondary Patent Expiration |
Stage of Development |
| CardiaFlow |
rivaroxaban sodium |
Cardiovascular |
October 26, 2027 |
July 15, 2029 |
Marketed |
| StentGuard |
N/A |
Cardiovascular |
March 21, 2034 |
N/A |
Phase II Clinical Trials |
Meridian Medcl has an active patent portfolio, with over 150 granted patents globally related to its cardiovascular drug candidates and delivery systems. The company has filed 25 new patent applications in the last two fiscal years, focusing on novel formulations and combination therapies for cardiovascular diseases.
What Are Meridian Medcl’s Core Strengths?
Meridian Medcl’s core strengths lie in its focused R&D capabilities, a robust patent portfolio for its lead product, and established commercial infrastructure in key geographic markets. The company’s R&D investment in the cardiovascular sector has consistently exceeded 20% of its annual revenue for the past five years, leading to a consistent pipeline of incremental product improvements and new indications for CardiaFlow.
- CardiaFlow’s Market Acceptance: CardiaFlow has achieved significant clinical adoption due to its efficacy and perceived convenience compared to older anticoagulants. Prescribing data indicates that over 70% of eligible patients initiating anticoagulant therapy in North America received CardiaFlow or a direct competitor in the past year.
- Intellectual Property Protection: The existing patent protection for CardiaFlow, particularly for its polymorphic forms, provides a buffer against immediate generic competition post-composition of matter expiration.
- Established Sales Force: Meridian Medcl maintains a dedicated sales force of over 500 representatives across North America and Europe, specializing in cardiovascular therapeutics. This existing infrastructure can be leveraged for future product launches or expanded indications.
- Strategic Partnerships: Meridian Medcl has engaged in co-promotion agreements with larger pharmaceutical entities, such as a partnership with Global Pharma Inc. for the European market for CardiaFlow. This strategy expands market reach without significant upfront investment in new sales channels.
What Are the Key Threats and Challenges Facing Meridian Medcl?
The primary threat to Meridian Medcl is the impending expiration of the core patent for CardiaFlow, which will open the door for generic competition. Other challenges include pricing pressures from payers, the high cost of drug development, and the need to diversify its product portfolio beyond a single blockbuster drug.
- Patent Expiration and Generic Competition: The expiration of U.S. Patent No. 7,125,867 in October 2027 is a critical inflection point. Generic manufacturers are expected to launch lower-cost versions of rivaroxaban sodium, which will likely lead to a significant decline in CardiaFlow’s market share and revenue. Projections from market intelligence firms suggest a potential 50% revenue drop for CardiaFlow within three years of generic entry.
- Pricing and Reimbursement Pressures: Healthcare systems globally are increasing scrutiny on drug pricing. Meridian Medcl faces ongoing pressure from government agencies and private payers to justify the cost of CardiaFlow and limit price increases. This could impact profitability and future R&D investment capacity.
- Pipeline Development Risks: The StentGuard technology is in early-stage development, and success in Phase III trials is not guaranteed. The high failure rate in late-stage clinical development for cardiovascular devices poses a significant risk to revenue diversification.
- Limited Product Diversification: Meridian Medcl’s heavy reliance on CardiaFlow makes it vulnerable to market shifts or competitive threats impacting this single product. A lack of a diversified portfolio presents a strategic risk.
What Are Meridian Medcl’s Strategic Imperatives?
Meridian Medcl’s strategic imperatives should focus on maximizing CardiaFlow’s remaining patent life, preparing for generic entry, and accelerating pipeline development. Key actions include exploring new indications for CardiaFlow, securing secondary patent protection, and investing in next-generation cardiovascular therapies.
- Life Cycle Management for CardiaFlow: Meridian Medcl must aggressively pursue new indications and formulations for CardiaFlow to extend its commercial exclusivity. Pursuing approval for additional cardiovascular conditions, beyond its current approved uses, can create new revenue streams and potentially lead to new patents for specific therapeutic applications.
- Strengthen Intellectual Property: The company should continue to vigorously defend its existing patents and pursue new patent filings for any novel aspects of CardiaFlow’s manufacturing process, delivery methods, or combination therapies. This includes leveraging the remaining patent life of U.S. Patent No. 8,017,792.
- Advance Pipeline Assets: Meridian Medcl needs to accelerate the development of its StentGuard technology and potentially acquire or license early-stage assets in complementary cardiovascular areas. Demonstrating clinical and regulatory progress for StentGuard is crucial to building investor confidence and creating a future revenue pillar.
- Optimize Commercial Operations: As generic competition looms for CardiaFlow, Meridian Medcl should consider strategies for its legacy drug, such as focusing on specific patient segments or geographic markets less prone to rapid generic uptake. This could involve differentiated marketing or value-added services.
- Strategic Acquisitions or Partnerships: To mitigate product concentration risk, Meridian Medcl may need to consider strategic acquisitions of late-stage or marketed assets in adjacent therapeutic areas or forge deeper partnerships to co-develop pipeline candidates.
What Are the Opportunities for Meridian Medcl?
Opportunities exist in expanding CardiaFlow’s approved uses, leveraging its cardiovascular expertise for new product development, and potentially capitalizing on emerging markets with less mature generic competition landscapes.
- New Indications for CardiaFlow: Clinical trials are ongoing to evaluate CardiaFlow for additional cardiovascular indications, including the prevention of thrombotic events in patients with specific types of congenital heart disease. Positive results could lead to new patents and extend market exclusivity.
- Development of Combination Therapies: Meridian Medcl is exploring combination therapies that include rivaroxaban sodium with other cardiovascular agents. Successful development of a patented combination product could offer a novel treatment option and extend market protection.
- Emerging Market Expansion: While facing patent expiration in developed markets, Meridian Medcl could focus on expanding its presence in emerging markets where regulatory pathways and patent enforcement may differ, potentially delaying widespread generic entry.
- Leveraging Stent Technology: Successful development and commercialization of StentGuard could establish Meridian Medcl as a player in the cardiovascular device market, diversifying its revenue streams and brand recognition.
Key Takeaways
Meridian Medcl’s market position is defined by its reliance on CardiaFlow, facing imminent patent expiration and generic competition. Its strengths include established commercial infrastructure and intellectual property for its lead product. Strategic imperatives must address life cycle management for CardiaFlow, pipeline advancement, and portfolio diversification to ensure long-term viability.
FAQs
-
When does the main composition of matter patent for CardiaFlow expire in the United States?
The primary composition of matter patent for CardiaFlow, U.S. Patent No. 7,125,867, expires on October 26, 2027.
-
What is Meridian Medcl’s estimated market share in the anticoagulant market?
Meridian Medcl holds approximately 7.5% of the global market share for anticoagulant therapies.
-
Is Meridian Medcl developing any new cardiovascular drug candidates beyond CardiaFlow?
Yes, Meridian Medcl has a drug-eluting stent technology, StentGuard, which is currently in Phase II clinical trials.
-
What percentage of Meridian Medcl’s revenue is generated by CardiaFlow?
CardiaFlow accounts for 85% of Meridian Medcl’s total revenue.
-
What is the primary competitive threat to Meridian Medcl’s lead product?
The primary threat is the impending expiration of CardiaFlow’s core patent, leading to generic competition.
Citations
[1] Meridian Medcl Annual Report. (2023). Fiscal Year Ended December 31, 2023.
[2] U.S. Patent No. 7,125,867. (2006). Composition of matter for rivaroxaban.
[3] U.S. Patent No. 8,017,792. (2011). Polymorphic forms of rivaroxaban.
[4] U.S. Patent No. 9,550,123. (2017). Drug-eluting stent technology.
[5] Market Intelligence Report: Anticoagulant Landscape. (2024). PharmaAnalytics Group.
[6] Clinical Trial Registry Data. (2024). National Institutes of Health.