Executive Summary
The global CDMO market hit USD 238.92 billion in 2024 and is on track to reach USD 465.24 billion by 2032, growing at a 9.0% CAGR. That number understates what is actually happening strategically. Pharmaceutical and biotech companies are not merely outsourcing tasks they find inconvenient — they are restructuring their entire operating models around external manufacturing partners. CDMOs now carry IP, regulatory relationships, process know-how, and often the only commercially viable route to clinical-grade material for advanced modalities like viral vectors, antibody-drug conjugates (ADCs), and mRNA therapeutics.
This guide exists because most CDMO selection content is written for procurement teams, not IP strategists, portfolio managers, or R&D leads who understand that the wrong partner can destroy a composition-of-matter patent’s competitive runway, expose a sponsor to third-party process IP, or leave a molecule sitting on the shelf while a viral vector CDMO works through an 18-month backlog. The selection criteria here are sequenced to serve those stakeholders specifically.
The piece covers: market structure and IP valuation of the major CDMO platforms; a methodology for mapping internal needs before entering any RFP process; technology roadmaps for small molecules, biologics, CGT, ADCs, and mRNA; the regulatory anatomy of FDA 483 observations and Warning Letters; supply chain dual-sourcing strategy; contract architecture including background IP versus foreground IP ownership; Paragraph IV implications of CDMO-generated process patents; financial red flags; and real-world case studies where these factors determined outcomes.
Part I: The CDMO Market in 2026 — Size, Structure, and IP Landscape
The Market by the Numbers
The CDMO industry generated roughly USD 238.92 billion in revenue in 2024. North America held the largest regional share at 38.59%, driven by a dense concentration of biotech sponsors in the Boston-Cambridge corridor, San Francisco Bay Area, and Research Triangle Park who lack in-house manufacturing infrastructure. Fortune Business Insights projects the global market reaches USD 465.24 billion by 2032. The U.S. segment alone is forecast to hit USD 83.86 billion by 2034.
Those figures, however, obscure the structural bifurcation happening inside the industry. The top five CDMO players — Lonza, Samsung Biologics, Catalent (now part of Nova Holdings after the 2024 acquisition by Novo Holdings), WuXi Biologics, and Recipharm — control only approximately 15% of the total market. The remaining 85% is split across hundreds of specialized mid-market and niche players. That fragmentation creates both opportunity and risk for sponsors: more choice in specialized capabilities, but also more due diligence burden and a higher probability of selecting a financially fragile partner.
The mRNA therapeutics CDMO segment grew from USD 4.62 billion in 2024 to an estimated USD 5.15 billion in 2025, with forecasts projecting a jump to USD 13.63 billion by 2034. Cell and gene therapy (CGT) CDMOs are growing faster still, though reliable market sizing remains difficult because a significant portion of viral vector manufacturing still happens in-house at large sponsors or at academic medical centers.
CDMO IP as a Core Asset Class
Every pharma IP team conducting CDMO due diligence should analyze the CDMO’s own patent portfolio before signing a master services agreement. A CDMO’s patent filings reveal what it can actually do at scale, where its genuine competitive moats sit, and — critically — whether any of its proprietary processes will generate foreground IP that could complicate your downstream patent strategy.
Lonza Group (SIX: LONN) holds one of the deepest IP portfolios in the large-molecule CDMO space. Its Ibex Solutions platform carries multiple granted patents covering single-use bioreactor configurations, perfusion cell culture processes, and downstream purification integration. As of 2025, Lonza’s process-related IP in mammalian cell culture covers several CHO-based high-density perfusion methods that can directly affect the freedom-to-operate analysis for any biologics sponsor using its Visp (Visp, Switzerland) or Portsmouth, NH facilities. Lonza’s biologics segment revenue reached approximately CHF 3.2 billion in 2024, and analysts at Jefferies estimate that its IP-embedded process know-how commands a 15-20% price premium over generic biologics CDMOs for novel mAb programs. For portfolio managers: Lonza trades at a premium to its CDMO peers partly because that IP makes customer switching costs very high — a dynamic that benefits Lonza’s revenue visibility but requires sponsors to negotiate IP carve-outs explicitly in contract language.
Samsung Biologics (KRX: 207940) has built its competitive position on scale rather than a dense patent thicket, but its bioreactor engineering IP — specifically its fed-batch process optimization methods filed through Korean and PCT filings — is increasingly relevant for sponsors targeting global supply chains. Samsung Biologics reported KRW 4.46 trillion (~USD 3.3 billion) in revenue in 2024 and operates approximately 604,000 liters of bioreactor capacity across its Incheon campus. Its Camp 4 facility added 180,000 liters of capacity in 2023, and Camp 5 is scheduled to add another 180,000 liters. IP valuation here is straightforward for analysts: Samsung’s asset value is primarily expressed through its capital base and its regulatory track record (the company holds FDA, EMA, PMDA, and MFDS approval for its sites), not a defensive patent portfolio. That means lower IP-transfer risk for sponsors — but also less process-specific differentiation.
Catalent (post-Novo Holdings acquisition) had a different IP profile: a sprawling portfolio covering drug delivery technologies including OptiMelt (hot-melt extrusion), OptiShell (capsule formats), and SmartBurst (immediate-release microspheres), plus biologics filling capabilities and gene therapy manufacturing at its Harmans, Maryland, and Bloomington, Indiana facilities. When Novo Holdings acquired Catalent for approximately USD 16.5 billion in early 2024 — primarily to secure capacity for Novo Nordisk’s GLP-1 supply chain — the transaction embedded those drug delivery IP assets within a vertically integrated ownership structure. Pharma sponsors working with former Catalent sites now operate within a context where their CDMO’s ultimate parent is also a direct commercial competitor. IP isolation agreements in that context require additional scrutiny.
WuXi Biologics (HKEX: 2269) — and the broader WuXi AppTec ecosystem — carries significant IP in its WuXiBody platform (a proprietary bispecific antibody technology) and its WuXia continuous bioprocessing system. Its process development patents cover CHO cell line engineering, fed-batch optimization, and integrated continuous manufacturing approaches. However, the passage of the BIOSECURE Act (H.R. 8333) into U.S. law in late 2024 has fundamentally changed the risk calculation for U.S.-based sponsors. The act restricts U.S. government contracts with companies affiliated with specified Chinese biotechnology entities, including WuXi AppTec and WuXi Biologics, and creates a compliance sunset that extends to commercial relationships for U.S.-listed companies seeking government reimbursement. Sponsors who were mid-program with WuXi Biologics when the law passed now face technology transfer timelines of 12-24 months to migrate to alternative CDMOs, a process that involves not just equipment qualification but renegotiation of cell line ownership and process IP agreements. For IP teams: any program that generated foreground IP at WuXi Biologics requires a formal IP ownership audit before transfer, with particular attention to whether WuXi’s WuXiBody platform or WuXia patents cover any process steps that became embedded in your product’s manufacturing history.
Recipharm (acquired by EQT in 2021, private) holds a diversified small-molecule manufacturing IP portfolio concentrated in inhalation drug delivery — particularly dry powder inhalers (DPIs) and pressurized metered-dose inhalers (pMDIs) — through its Aesica and RxElite subsidiaries. The inhalation IP is relevant for sponsors developing respiratory biologics or combination products where the device and formulation are co-developed.
The BioSecure Act: Reshaping the Global CDMO Map
The BIOSECURE Act is the most consequential regulatory event to hit the CDMO industry since the FDA’s cGMP modernization initiative in the early 2000s. Its core mechanism restricts U.S. federal contracts and, by extension, access to federal reimbursement programs for companies using prohibited biotechnology vendors. The named entities include WuXi AppTec, WuXi Biologics, BGI Genomics, MGI Tech, and Complete Genomics, though the Act includes a mechanism for expanding the list.
The practical effect for sponsors is a bifurcation of global CDMO capacity. European and U.S.-based CDMOs — Lonza, Samsung Biologics, Boehringer Ingelheim Biopharmaceuticals (contract business), AGC Biologics, and Rentschler Biopharma — are absorbing displaced programs. Wait times for commercial-scale biologics slots at these facilities are extending to 18-24 months in some modalities. Sponsors who had counted on WuXi’s cost advantage (typically 15-25% below Western equivalents for standard mAb programs) now need to rebuild cost models with Western pricing — a meaningful hit to projected margin on biosimilar and biologics pipeline assets.
For portfolio managers assessing biotech stocks: companies with heavy WuXi Biologics or WuXi AppTec exposure that lack contractual technology transfer rights face the highest write-down risk on pipeline valuations. IP teams at those companies should immediately audit their contracts for: (a) cell line ownership provisions, (b) process development IP assignment clauses, (c) batch record access rights, and (d) regulatory filing data ownership — all of which affect whether a technology transfer to an alternative CDMO is feasible within a clinically meaningful timeline.
Key Takeaways: Part I
The CDMO market is large, fragmented, and undergoing structural compression at the top driven by the BioSecure Act and Novo Holdings’ Catalent acquisition. CDMO IP portfolios are real assets — Lonza’s process patents create switching costs, Samsung’s scale creates regulatory certainty, and WuXi’s BioSecure Act exposure creates transfer risk. Sponsors who treat CDMO selection as a procurement exercise rather than an IP and portfolio strategy event do so at measurable financial risk.
Investment Strategy: Part I
Portfolio managers should screen CDMO-dependent biotech positions for BioSecure Act exposure by reviewing Q3 and Q4 2024 10-Q and 20-F filings, where most companies disclosed WuXi dependencies. Companies with active IND-stage programs at WuXi Biologics and no executed technology transfer agreements face 12-24 months of timeline risk — material for any program within 3 years of a projected NDA submission. Lonza and Samsung Biologics remain the clearest beneficiaries of the capacity displacement. Lonza’s forward bookings through 2027 are reported as fully committed at its Swiss and U.S. biologics facilities, which means new capacity demand from displaced WuXi clients may be pricing into its 2027-2028 revenue line.
Part II: Before You Shop — Internal Diligence You Must Complete
Development Stage Mapping
The most expensive CDMO selection error is mismatching the partner’s capabilities to the drug’s actual development stage. A CDMO optimized for Phase I clinical manufacturing — typically characterized by small batch sizes, flexible multi-product facilities, and rapid analytical method development — operates differently from one configured for Phase III pivotal trial supply or commercial launch. The differences are not cosmetic. A Phase I-focused CDMO may lack the validated, campaign-scale production systems that FDA reviewers expect to see documented in a commercial BLA or NDA submission.
Before issuing an RFP, map your program against the following stage-specific requirements:
Discovery to IND: You need pre-formulation capability, salt-form screening, polymorph characterization (for small molecules), or cell line development and titer optimization (for biologics). The CDMO must have analytical development infrastructure to support forced degradation studies, method development, and ICH Q1-compliant stability protocols.
Phase I to Phase II: The priority shifts to GMP clinical batch manufacturing, regulatory starting material (RSM) qualification, and the ability to support a first-in-human (FIH) IND package. For biologics, this means a CDMO with a track record of Phase I biologics batch manufacturing under 21 CFR Part 211 and a quality system that has survived at least one FDA pre-approval inspection.
Phase II to Phase III: Technology transfer, process validation (Stages 1-3 under FDA’s 2011 Process Validation Guidance), and scale-up are the key risks here. A CDMO that manufactures Phase II clinical supply but cannot demonstrate commercial-scale process performance qualification (PPQ) data will create a regulatory gap that delays BLA/NDA submission.
Commercial launch and lifecycle management: You need a CDMO with approved commercial manufacturing sites, established annual product review (APR) systems, and a track record of managing post-approval changes (PACs) through Prior Approval Supplements (PAS), Changes Being Effected (CBE-30), or Annual Report categories. CDMOs that lack experience managing PACs introduce significant risk at the commercialization stage because even minor manufacturing changes require regulatory submissions that can delay supply.
Molecule Complexity Matrix
Not every molecule requires the same manufacturing infrastructure. The choice of CDMO should reflect the molecule’s complexity class:
Standard small molecules (BCS Class I/III, oral solid dosages) can be manufactured by a wide range of CDMOs with conventional wet granulation, direct compression, or roller compaction equipment. The differentiation here is in the CDMO’s regulatory track record and its ability to support Paragraph IV ANDA filings if the product is a generic — relevant because the CDMO’s process IP may interact with the Orange Book-listed patents.
Highly potent APIs (HPAPIs), generally defined as compounds with occupational exposure limits (OELs) below 1 µg/m³, require containment infrastructure at Safebridge Category 3 or 4 levels (or equivalent ISPE band 4/5). CDMOs with genuine HPAPI capability include Lonza’s Visp facility, Curia Global (formerly Albany Molecular Research/AMRI), and CordenPharma. For oncology programs where the active compound or its intermediates are classified as Category 1A/1B carcinogens under GHS, the CDMO’s containment validation documentation will be reviewed in both IMPD submissions (for EU clinical trials) and IND packages.
Monoclonal antibodies (mAbs), bispecific antibodies, and Fc-fusion proteins require mammalian cell culture (typically CHO, NS0, or HEK293 host cells), chromatographic downstream purification (Protein A capture, ion exchange polishing, size-exclusion chromatography), and viral inactivation/clearance steps validated under ICH Q5A. The biologics CDMO market for standard mAbs is the most commoditized segment — multiple large platforms offer equivalent technical capabilities — so IP and financial diligence become the differentiating variables.
Antibody-drug conjugates (ADCs) require a CDMO with both biologic upstream capability (for the antibody or antibody fragment) and specialized conjugation chemistry infrastructure for high-potency cytotoxic payloads. The conjugation step typically involves Category 4-level containment for the cytotoxin (often an auristatin, maytansinoid, or calicheamicin derivative), an inert, well-characterized linker chemistry, and specialized analytical methods to characterize the drug-antibody ratio (DAR) and the resulting heterogeneous conjugate mixture. CDMOs with validated ADC conjugation platforms include Lonza (through its biologics and synthesis businesses), Abzena, Almac Group, and Piramal Pharma Solutions. The IP landscape in ADC conjugation is highly contested: Seattle Genetics (now Seagen, acquired by Pfizer in 2023 for USD 43 billion) holds key linker-payload patents covering vcMMAE conjugates, while ImmunoGen holds patents on DM1/DM4 maytansinoid payloads. A sponsor developing a novel ADC needs to map its planned linker-payload chemistry against the freedom-to-operate landscape before selecting a CDMO whose proprietary conjugation processes might incorporate patented methodologies.
Cell and gene therapies (CGT) represent the most infrastructure-intensive and capacity-constrained CDMO category. Viral vector manufacturing — specifically AAV and lentiviral vector production for in vivo and ex vivo gene therapies respectively — requires S2-level biosafety containment, ultracentrifugation or ion-exchange chromatography-based purification for AAV, and transiently transfected HEK293 or stable producer cell lines depending on the vector system. The CDMO capacity shortage for viral vectors is real: wait times at CDMOs like Andelyn Biosciences (formerly Nationwide Children’s Hospital), Charles River Laboratories’ gene therapy unit, and Spark Therapeutics’ Brammer Bio (acquired by Thermo Fisher Scientific in 2019) have run at 6-24 months for multi-batch programs. mRNA therapeutics require lipid nanoparticle (LNP) formulation capability, with the Moderna and BioNTech/Pfizer vaccine programs having demonstrated that LNP encapsulation is technically demanding, IP-rich (Arbutus Biopharma and Genevant Sciences control key ionizable lipid patents that were the subject of litigation with Moderna), and sensitive to process parameters that affect particle size, encapsulation efficiency, and in vivo transfection activity.
CAPEX-to-OPEX Conversion: The Financial Logic of Outsourcing
A GMP biologics manufacturing suite — bioreactors, downstream chromatography columns, clean-in-place (CIP) systems, water-for-injection (WFI) generation, and HVAC with ISO 5/7 classified areas — costs between USD 150 million and USD 500 million to build from greenfield, depending on scale, location, and modality. A gene therapy facility with appropriate biosafety containment and ultracentrifuge capacity runs USD 50-200 million at small scale. The timeline from construction start to first GMP batch at a new facility routinely exceeds 3-4 years when regulatory qualification, equipment commissioning, and process validation are included.
Outsourcing to a CDMO converts that capital expenditure into a variable operating expense tied to campaign-specific production runs. For a Series B biotech with USD 150-300 million in cash, the choice is not theoretical: spend 50-100% of the balance sheet on a manufacturing facility or retain capital for clinical operations. The financial model is compelling. But it creates a dependency that has to be managed with the same rigor as any other material asset.
The CAPEX-to-OPEX conversion is also relevant for large pharma in specific contexts: companies preparing assets for divestiture, spinning off generics businesses, or managing a pipeline transition after a major patent cliff. In each case, retaining in-house manufacturing infrastructure for a legacy product line while outsourcing next-generation pipeline production to CDMOs represents an intelligent capital allocation.
Key Takeaways: Part II
Stage-specific CDMO matching is not optional — it determines regulatory package quality and commercial readiness. Molecule complexity class directly determines the CDMO shortlist: most CDMOs cannot handle HPAPIs, ADC conjugation, viral vectors, or mRNA LNP formulation at commercial scale. The CAPEX-to-OPEX argument for outsourcing is financially compelling, but it creates strategic dependencies that must be managed through contract architecture and IP protections.
Part III: Quality and Regulatory Compliance — The Non-Negotiables
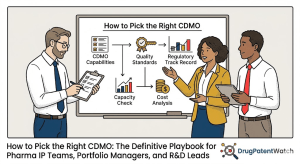
Building a CDMO Quality Audit Framework
Quality evaluation for a CDMO is a technical audit, not a supplier questionnaire. IP teams and R&D leads who have never conducted a GMP facility audit should engage a qualified person (QP) under EU GMP regulations, a regulatory affairs professional, or an experienced quality consultant to lead the process. The deliverable is a gap analysis against 21 CFR Part 211 (for U.S.-marketed products) and EU GMP Annex 1 (revised 2023), Annex 6, Annex 13, and, for biologics, ICH Q7, Q8, Q9, Q10, and Q12.
A structured CDMO quality audit framework covers the following areas in sequence:
Facility and equipment qualification: Review Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) documentation for critical equipment. For biologics CDMOs, bioreactor characterization studies and hold time validations are particularly important. For aseptic fill-finish operations, review Environmental Monitoring (EM) data trends and sterility testing failure rates over the past 24 months.
Analytical method validation: Confirm that the CDMO has validated analytical methods per ICH Q2(R2) for all critical quality attributes (CQAs) of your molecule. For biologics, this includes potency assays, glycan profile analysis, charge variant profiling, and sub-visible particle quantification per USP <787> or <788>. For small molecules, review the validated HPLC methods for assay and related substances.
Deviation and CAPA management: Request the CDMO’s deviation and corrective and preventive action (CAPA) log for the past 12-24 months, redacted for other client confidentiality but with classification, root cause category, and closure timing visible. A CDMO with a high proportion of “repeat” deviations — i.e., the same root cause appearing multiple times — has a systemic quality failure that no remediation promise will fix quickly.
Batch failure rates and product impact assessments: Ask for the historical batch failure rate by product type and manufacturing stage. A biologics CDMO with batch failure rates above 3-5% for standard mAbs warrants a detailed explanation. Any batch failure that resulted in a product impact assessment reaching the regulator should be disclosed and reviewed.
Data integrity: Since FDA’s renewed enforcement focus on data integrity starting in 2019, data integrity failures have become one of the most common sources of Warning Letters at CDMO facilities. Audit systems for audit trail completeness, raw data traceability, and computer system validation (CSV) status. A CDMO that cannot provide unbroken audit trail documentation for a given batch record has a data integrity vulnerability that could invalidate clinical trial batches or commercial product.
Decoding FDA 483 Observations vs. Warning Letters
FDA Form 483 observations are issued at the conclusion of an inspection when the investigator identifies conditions that may constitute a violation of the FD&C Act. A 483 requires a written response within 15 working days and does not, by itself, constitute a finding of non-compliance. Warning Letters are issued when the FDA determines that 483 observations were not adequately addressed, or when violations are severe enough to warrant immediate regulatory action. A Warning Letter is a public document and carries enforceable consequences.
For due diligence purposes, the character of the 483 observations matters more than the count. Observations related to data integrity (21 CFR 211.68), investigation adequacy (21 CFR 211.192), and written procedures (21 CFR 211.22) indicate systemic quality system failures. Observations related to facility maintenance or environmental monitoring trends indicate operational issues that are more likely to be remediated quickly. A CDMO that received two 483s in five years for facilities-related observations and addressed both with documented remediation is materially less concerning than one that received a 483 for data integrity and subsequently received a Warning Letter.
The FDA’s CDER Quality Management Maturity (QMM) program, launched in 2020, provides a framework for assessing a manufacturer’s quality culture beyond mere compliance. CDMOs that have participated in QMM assessments and can document their maturity level represent a higher-quality pool of partners. The program evaluates four domains: quality culture, quality planning, organizational structure, and knowledge management. CDMOs at QMM Level 3 or above demonstrate proactive quality management that significantly reduces the probability of inspection-driven supply disruptions.
Warning Letters at a CDMO should be treated as near-disqualifying for any program with a projected regulatory submission within 24 months. The FDA’s Application Integrity Policy (AIP) allows the agency to place holds on pending NDAs and BLAs from facilities with outstanding Warning Letters, directly blocking market entry.
Quality by Design (QbD) and Process Analytical Technology (PAT)
ICH Q8(R2) defines Quality by Design as a systematic approach to development that begins with predefined objectives and emphasizes product and process understanding based on sound science and quality risk management. QbD in practice means a CDMO conducts Design of Experiments (DoE) studies to identify critical process parameters (CPPs) and their relationship to CQAs, defines the Design Space within which those parameters can vary without affecting product quality, and establishes a Control Strategy that maintains the process within that Design Space.
CDMOs that implement QbD generate a much richer process understanding document (PUD) than those relying on traditional one-factor-at-a-time development. That richer PUD translates directly into a stronger regulatory package — FDA reviewers reward QbD submissions with fewer information requests and, in some cases, more flexible post-approval change protocols under ICH Q12. For sponsors, a CDMO’s QbD capability is not merely a quality marker; it is a commercial asset that accelerates regulatory timelines and reduces post-approval compliance burden.
Process Analytical Technology (PAT), as defined in FDA’s 2004 PAT Guidance, refers to real-time measurement and control of critical quality and performance attributes of raw materials and in-process materials. In practice, this means CDMOs using near-infrared (NIR) spectroscopy for blend uniformity monitoring, Raman spectroscopy for polymorph identification, or in-line particle size analysis. For biologics CDMOs, PAT applications include on-line bioreactor metabolite monitoring (glucose, lactate, dissolved oxygen), automated fed-batch control, and real-time viral clearance monitoring. PAT reduces the reliance on end-of-process release testing, accelerates batch release, and generates continuous process data streams that support ongoing process verification under FDA’s Stage 3 process validation requirements.
CDER QMM Program and Its Practical Implications
The FDA’s Quality Management Maturity Program is worth understanding in detail because it directly predicts inspection outcomes. Under QMM, FDA evaluates pharmaceutical manufacturers on four domains: organizational quality culture (senior leadership engagement, quality metrics review, continuous improvement infrastructure), quality planning (risk management, change management, annual product review rigor), organizational structure (quality unit independence, resource adequacy, training infrastructure), and knowledge management (process understanding documentation, technology transfer capability, regulatory intelligence).
CDMOs that score high on QMM assessments tend to have shorter inspection durations, fewer 483 observations, and faster post-inspection closeout. For sponsors, a CDMO’s QMM participation and self-reported maturity scores should be requested during due diligence. A CDMO that cannot describe its QMM posture has not invested in this dimension of regulatory relationship management — a real signal in the current enforcement environment.
Key Takeaways: Part III
Quality evaluation requires a structured technical audit, not a questionnaire. Data integrity is the highest-risk quality domain in the current FDA enforcement climate. Warning Letters near or during a regulatory submission window are near-disqualifying. QbD and PAT capabilities generate real commercial value by accelerating regulatory timelines. CDER QMM participation signals a mature quality organization.
Part IV: Expertise and Track Record — What ‘Experience’ Actually Means
Modality-Specific Expertise: The Verification Checklist
A CDMO’s claim to “expertise” in a given modality must be verified through specific technical evidence, not marketing materials. For each modality class, the verification questions differ.
For biologics (mAbs, bispecifics, Fc-fusions): Request the number of IND submissions supported in the past 5 years where the CDMO was the clinical manufacturer of record, the number of BLA/MAA submissions where the CDMO’s batch records were included, and the outcome of any FDA pre-approval inspections (PAI) at their facility in the past 5 years. Request the titer range achieved in CHO fed-batch processes across the portfolio (a credible large-scale biologics CDMO should achieve 3-8 g/L in standard programs and can articulate strategies for low-titer programs). Request data on their downstream recovery yields for standard mAb programs.
For HPAPIs and highly potent formulations: Request the OEL range of compounds handled, the containment classification of each manufacturing suite, and the industrial hygiene monitoring data demonstrating containment performance. Ask for the ISPE Baseline Guide for Risk-Based Manufacture of Pharmaceutical Products (the “ISPE Risk-MaPP” guide) classification of each suite. A CDMO claiming HPAPI capability must provide air monitoring data demonstrating that their containment infrastructure achieves the required performance on actual manufacturing campaigns, not just engineering calculations.
For cell and gene therapy: The key technical questions are platform-specific. For AAV programs: What producer cell system does the CDMO use (transient transfection in HEK293 vs. stable producer cell lines vs. baculovirus/insect cell systems)? What downstream purification methods are used (ultracentrifugation vs. AVB Sepharose affinity chromatography vs. ion-exchange)? What is the typical yield (genome copies per mL) and full-to-empty capsid ratio achieved? What analytical methods are in place for potency (transduction efficiency), purity (SDS-PAGE, SEC-MALS, TEM), and safety (replication-competent AAV, residual host cell proteins)? For lentiviral vectors: What is the manufacturing scale (flask-based vs. multilayer Cell Factory vs. bioreactor suspension)? What is the typical titer (infectious units/mL) and the vector copy number range achieved in transduction studies?
For mRNA: What is the in vitro transcription (IVT) scale? What LNP formulation technology is in use? Is the ionizable lipid proprietary or licensed (Genevant Sciences and Arbutus Biopharma’s patent family, or Precision NanoSystems/Bio-Techne’s formulation platforms)? What is the encapsulation efficiency typically achieved? What analytical methods are used for mRNA integrity (capillary gel electrophoresis), LNP particle size (DLS), and potency (luciferase or antigen expression assays in cell-based systems)?
IP-Rich CDMOs: Patent Portfolios as Proxies for Technical Depth
A CDMO’s patent activity in its core technical areas is a proxy for technical depth that is more reliable than its marketing claims. A company that has filed 20 process patents in CHO cell culture optimization over 10 years has genuinely worked through the problems that generate intellectual property. A company claiming CHO expertise with no granted patents in the area may be relying on know-how that is thinner than presented.
For IP teams, this analysis is operational: pull the CDMO’s patent portfolio from Espacenet, the USPTO patent full-text database, or a commercial patent analytics platform. Filter for IPC codes relevant to the manufacturing modality (e.g., C12P 21/00 for recombinant protein production, C12N 5/00 for cell culture, A61K 9/51 for nanoparticle formulations, C12N 15/86 for viral vectors). Review the assignee and co-assignee structure: does the CDMO own the patents outright, or do co-development arrangements mean a sponsor’s IP might be entangled with a third party’s estate?
Critically: determine whether any of the CDMO’s granted process patents could read on your manufacturing process as currently designed. If a CDMO’s patented perfusion cell culture method is the only commercially viable way to achieve the required titer for your biologic, and the CDMO’s patent covers that method, your sponsor organization’s freedom-to-operate is partially dependent on the CDMO relationship continuing. That creates leverage for the CDMO in contract renegotiations and significantly raises the cost of switching.
Reference-Checking Protocol
Client references from a CDMO are not a courtesy — they are a structured due diligence activity. Request a minimum of four references: two current clients with programs in the same modality as your program, one former client (a company that has transferred a program out), and one client whose program encountered a significant technical or quality challenge.
The question set for references should cover: How did the CDMO communicate problems — proactively or reactively? Did the CDMO’s batch failure rate match what they disclosed in the RFP? When a deviation occurred that affected the regulatory filing timeline, how did the CDMO’s project management respond? Would you use them again, and at what development stage? How did the CDMO handle IP ownership for process improvements developed during your program?
The reference from a former client — a company that transferred a program out — is the most valuable. That reference will reveal whether the CDMO facilitated the technology transfer professionally or created obstacles (e.g., delays in providing batch record documentation, disputes over ownership of process improvements, slow release of cell banks). A CDMO that makes technology transfer difficult is a CDMO that has structurally embedded itself as a dependency — a posture that benefits the CDMO and disadvantages the sponsor.
Key Takeaways: Part IV
Expertise verification requires modality-specific technical questions, not general inquiries about years of operation. A CDMO’s patent portfolio is a credible proxy for genuine technical depth. IP entanglement through CDMO-owned process patents is a real risk that must be assessed before program start. References from former clients and clients who experienced technical problems reveal more than references from satisfied current clients.
Investment Strategy: Part IV
Institutional investors evaluating biotech companies with CDMO-dependent pipelines should request copies of the CDMO master services agreements (or at minimum the contract summary terms) as part of due diligence on any acquisition or significant investment. The key terms to verify: IP ownership of CDMO-generated improvements, technology transfer provisions including the right to receive all batch records and analytical methods within 30 days of request, and termination provisions that do not impose penalties on the sponsor for regulatory-driven transitions.
Part V: Manufacturing Technology Roadmaps
Small Molecule Process Chemistry: Continuous Flow Chemistry
Traditional batch chemistry for small molecule APIs generates step-by-step reaction sequences in stirred-tank reactors, with each step requiring work-up, isolation, and purification before the next step begins. Continuous flow chemistry (also called flow chemistry or microreactor chemistry) performs reactions in a continuously flowing stream through a series of fixed reactor modules, enabling precise temperature control, rapid mixing, and real-time analytical monitoring at each step.
The regulatory pathway for continuous manufacturing (CM) of small molecules has matured significantly since FDA’s 2016 guidance on continuous manufacturing. Vertex Pharmaceuticals’ Orkambi and Symdeko were manufactured using CM processes, and FDA has approved multiple NDA submissions with CM manufacturing processes in the past decade. The key regulatory documents are FDA’s 2019 guidance on quality considerations for continuous manufacturing, which establishes expectations for real-time release testing (RTRT), design space definition, and control strategy under a CM paradigm, and ICH Q13 on continuous manufacturing of drug substances and drug products, finalized in 2023.
For CDMOs, the capital investment in continuous flow chemistry infrastructure — flow reactors, in-line analytics (NIR, Raman, UPLC), and process control systems — is substantial, typically USD 10-50 million per manufacturing train depending on scale. CDMOs with genuine CM capability include Pfizer CentreOne (which has commercialized its own CM platform through its CDMO business), Lonza’s small molecule segment, Cambrex, and Quotient Sciences. The IP landscape around specific flow chemistry reactor designs and in-line analytical methods is active, with Corning (for glass microreactors) and Chemtrix (for ceramic flow reactors) holding key hardware patents, while process-specific chemistry adaptations are typically owned by the CDMO or, where developed collaboratively, shared under co-ownership agreements.
The business case for a sponsor choosing a CM-capable CDMO rather than a batch manufacturer is: reduced API inventory risk (smaller in-process batch sizes), faster development-to-commercial scale-up (CM processes scale by run time, not by reactor volume), lower solvent consumption, and enhanced regulatory defensibility through continuous real-time quality data. For programs where the API synthesis involves hazardous intermediates (e.g., azides, peroxides, diazonium salts), flow chemistry is often the only viable commercial manufacturing approach because the small reactor volumes reduce the quantity of hazardous material present at any given time.
Biologics Manufacturing Technology Roadmap
The biologics manufacturing technology stack has been evolving for 40 years, from the original mouse-ascites mAb production systems of the 1980s through stainless-steel stirred-tank bioreactors in the 1990s and 2000s, to the single-use bioreactor revolution of the 2010s, and now toward perfusion-based continuous bioprocessing and intensified fed-batch processes in the 2020s.
Upstream cell culture evolution: The dominant platform for commercial mAb production remains fed-batch CHO cell culture in bioreactors ranging from 500L to 25,000L. Titers in optimized fed-batch processes now routinely reach 4-8 g/L for standard IgG1 mAbs, compared to 0.5-1 g/L in the early 2000s. The next generation of upstream technology is perfusion cell culture, which runs continuously by retaining cells through a cell retention device (alternating tangential flow filtration, acoustic separation, or hollow fiber systems) while continuously harvesting clarified cell culture fluid. Perfusion processes can achieve cell densities 5-10x higher than fed-batch, with product titers of 2-5 g/L per day on a volumetric basis — enabling production of equivalent mass in smaller bioreactors with shorter campaigns. CDMOs offering commercial-scale perfusion manufacturing include Lonza (through its Ibex platform), Boehringer Ingelheim, and AGC Biologics.
Downstream processing: Purification of mAbs typically follows a three-column sequence: Protein A affinity capture (removing >99% of host cell proteins in a single step), cation-exchange chromatography (removing charge variants and aggregates), and anion-exchange chromatography in flow-through mode (removing DNA, endotoxin, and remaining process-related impurities). This platform process has been regulatory-validated across hundreds of approved biologics. Innovation in downstream processing is now focused on continuous chromatography (periodic counter-current chromatography, twin-column continuous chromatography), which reduces buffer consumption and column resin investment per gram of product.
Viral clearance studies — required by ICH Q5A(R2), revised in 2024 — must be conducted by the CDMO and demonstrate that two orthogonal, mechanistically distinct viral inactivation/removal steps achieve a total viral reduction factor of at least 12 log10 for relevant and model viruses. CDMOs must have viral clearance study capabilities in-house or through qualified subcontract labs, and the study design must reflect the specific process parameters of the sponsor’s process, not generic platform data.
Single-use systems: The adoption of single-use bioreactors (SUBs), single-use chromatography systems, and single-use filtration assemblies has transformed the economics of biologics manufacturing. Single-use systems eliminate the need for CIP/SIP (clean-in-place/sterilize-in-place) validation, reduce cross-contamination risk in multi-product facilities, and reduce changeover time between product campaigns. CDMOs operating primarily single-use platforms — which includes most mid-scale biologics CDMOs with bioreactor capacities up to 2,000L — can offer more flexible scheduling and faster startup timelines for new programs. The tradeoff is cost at very large scale: a 20,000L SUB for a high-volume commercial product generates significant single-use consumable costs that exceed the CIP/SIP operating costs of stainless steel above certain thresholds. For most clinical-stage programs, single-use is economically superior.
Cell and Gene Therapy: The Viral Vector Bottleneck
The viral vector manufacturing capacity shortage is the most acute supply constraint in the CDMO industry. AAV manufacturing at commercial scale remains technically challenging for reasons that are fundamental to the biology: producer cell transfection efficiency, capsid assembly fidelity, full-to-empty capsid ratio, and downstream purification yield collectively limit what is achievable per batch in a way that has no parallel in traditional mAb manufacturing.
The technology roadmap for AAV manufacturing capacity expansion runs through three parallel tracks:
First, scaling transient transfection: Most Phase I/II AAV manufacturing uses transient transfection of HEK293 cells in cell factories, wave bioreactors, or suspension bioreactors using PEI-mediated triple transfection of plasmids encoding the cap, rep, and transgene sequences. This process is well-understood but difficult to scale beyond 200L suspension bioreactors without significant loss of consistency. CDMOs that have invested in high-density suspension HEK293 transfection at 200-500L scale (Andelyn Biosciences, Genezen, and VGXI) represent the current state of the art for transient transfection-based manufacturing.
Second, stable producer cell lines: Several groups are developing stable AAV producer cell lines that constitutively express rep and cap and can be induced to produce virus. BioMarin Pharmaceutical has published data on HeLa-based stable producer lines; Spark Therapeutics (Roche) uses a baculovirus/insect cell system for commercial production of Luxturna. The regulatory pathway for stable producer cell lines is more complex (requires cell line characterization studies under ICH Q5B), but the upstream yields and scalability improvements justify the additional regulatory investment for commercial programs.
Third, process intensification: Downstream purification of AAV — removing empty capsids and residual process impurities — is the rate-limiting step for many programs. AVB Sepharose affinity chromatography (developed by Cytiva) has become the near-universal first-step capture resin for most AAV serotypes. Subsequent ion-exchange polishing and analytical ultracentrifugation (AUC) for empty/full capsid characterization have become industry standards. CDMOs that have invested in AUC analytical capability, multiple-column downstream processing, and in-line monitoring for full/empty ratio are better positioned to deliver commercially viable AAV at scale.
The IP landscape in viral vector manufacturing is intensely competitive. Spark Therapeutics (Roche) holds key patents on AAV5 production methods. Applied Genetic Technologies Corp (AGTC) holds patents on AAV packaging methods. University of Pennsylvania’s Gene Therapy Program (Wilson lab) holds broad patents on AAV serotype capsid sequences (licensed through Penn’s Office of Technology Transfer). For any CGT program, a freedom-to-operate analysis specific to the viral vector’s capsid serotype, production method, and purification process is mandatory before CDMO selection.
ADC Manufacturing: Linker Chemistry and Conjugation IP
The ADC market has grown rapidly: FDA approved 14 ADCs between 2000 and 2024, with 7 of those approvals occurring between 2019 and 2024. The manufacturing of ADCs requires coordinating three distinct technical domains: antibody production (a biologics manufacturing problem), cytotoxic payload synthesis (a high-potency small molecule manufacturing problem), and conjugation (a specialized bioconjugation chemistry problem).
The conjugation step is where the most significant IP complexity lies. The two dominant conjugation chemistries are: cysteine-based conjugation (reducing interchain disulfide bonds to generate free thiols, then reacting with maleimide-bearing linkers), which produces a heterogeneous mixture of DAR 0, 2, 4, 6, and 8 species; and lysine-based conjugation (reacting NHS-ester activated linkers with lysine residues), which produces an even more heterogeneous mixture. Site-specific conjugation technologies — including unnatural amino acid incorporation (Sutro Biopharma), enzymatic conjugation (Synaffix’s GlycoConnect technology), and engineered cysteine insertion (Genentech’s THIOMAB technology) — generate more homogeneous DAR distributions that may improve the therapeutic window and pharmacokinetics of the final ADC.
CDMOs with validated ADC conjugation infrastructure must have: cytotoxin handling capability at Category 4 containment (OEL < 0.03 ng/m³ for auristatins), DAR characterization by HIC-HPLC or SEC-HPLC, aggregation testing by SEC, and potency testing (typically cell-based cytotoxicity assays). Lonza’s bioconjugation facility in Visp, Switzerland is one of the few sites globally with both large-scale antibody production and high-potency conjugation in proximity, reducing the risk of antibody handling and shipping to a separate conjugation site.
The IP ownership of the conjugation process — specifically, any novel process parameters or analytical methods developed during the CDMO engagement — must be addressed in the MSA before work begins. Synaffix’s GlycoConnect and HydraSpace technologies are licensed (Synaffix was acquired by OcuGen’s spin-off entity, then acquired by Lonza in 2021), meaning sponsors using Lonza’s GlycoConnect platform are operating under Lonza-controlled IP with licensing terms that affect the freedom-to-operate of the final ADC product.
Evergreening Tactics: How CDMO Process IP Intersects with Patent Lifecycle Management
Evergreening refers to a brand pharmaceutical company’s use of secondary patents — covering formulations, dosage forms, delivery devices, metabolites, and manufacturing processes — to extend de facto market exclusivity beyond the expiration of the original composition-of-matter patent. Manufacturing process patents are a legitimate and frequently used evergreening tool.
When a brand company develops a novel commercial manufacturing process — typically in collaboration with a CDMO — that process can be patented. If the process is listed in the FDA Orange Book as a method-of-manufacturing patent, generic filers must certify against it in their ANDA using a Paragraph IV certification (challenging the validity or non-infringement of the patent) or, if they cannot design around it, wait for the process patent to expire. Process patents typically have a filing date several years later than the composition-of-matter patent, providing meaningful additional exclusivity — sometimes 5-8 years beyond the original compound patent expiration.
The CDMO’s role in this strategy is significant. A CDMO that develops a novel crystallization process, a novel polymorphic form, a novel solvate, or a novel purification method during the commercial manufacturing development program generates IP that the sponsor should ensure is assigned to the brand company through the MSA. If the MSA is silent on foreground IP ownership, or if it defaults to CDMO ownership for “improvements to the CDMO’s background processes,” the sponsor may lose the ability to file and enforce a process patent that could represent years of additional exclusivity.
For R&D leads managing a late-stage pipeline asset, this is a direct commercial value question. A process patent with a 20-year term from filing that is filed 8 years after the composition-of-matter patent and survives Paragraph IV litigation could be worth hundreds of millions of dollars in extended exclusivity — or nothing, if the IP was not captured correctly at the CDMO.
AI and Agentic Automation in CDMO Operations
Artificial intelligence applications in pharmaceutical manufacturing have moved from pilot projects to production deployment at the leading CDMOs. The current implementation landscape, as of early 2026, includes several distinct use cases.
Predictive batch failure modeling: Machine learning models trained on historical batch records, process sensor data, and environmental monitoring data can predict with increasing accuracy which in-process batches are at elevated risk of failing specification before the batch is complete. PCI Pharma Services has published on AI-driven process monitoring that flags deviations in real time, allowing operators to intervene before a batch failure. Lonza’s Ibex Insights platform incorporates ML-based process analytics for its biologics clients.
Formulation development acceleration: AI platforms — including Chemify, Intelligencia AI, and Kebotix — accelerate small molecule formulation development by predicting API solubility, permeability, crystal form stability, and excipient compatibility from molecular structure, reducing the number of physical experiments required to reach a developable formulation. CDMOs that have integrated these tools into their pre-formulation workflows can offer meaningfully shorter formulation development timelines, typically reducing the pre-IND formulation phase from 9-12 months to 4-6 months for standard BCS Class II compounds.
Regulatory intelligence and change management: AI-driven regulatory intelligence platforms track ICH guideline updates, FDA guidance document revisions, and EMA reflection papers in real time, alerting quality teams to upcoming compliance requirements before they take effect. This is operationally valuable for CDMOs manufacturing products for multiple regulatory jurisdictions, where a change in EU GMP Annex 1 (revised November 2023) or an FDA guidance update on particulate matter limits might require process changes across dozens of client programs simultaneously.
Agentic AI for manufacturing execution: The most advanced application currently in commercial CDMO deployment is agentic AI systems that autonomously monitor production workflows, detect parameter deviations, and initiate corrective actions within predefined control strategy bounds without human intervention. This is still early-stage in regulated pharmaceutical manufacturing, where any automated control action must be validated per 21 CFR Part 11 and GAMP 5, but CDMOs that have invested in the underlying manufacturing execution system (MES) infrastructure that supports validated automation are better positioned to integrate agentic AI as the regulatory framework matures.
Key Takeaways: Part V
Continuous flow chemistry is a commercially validated manufacturing technology with a clear FDA regulatory pathway; CDMOs lacking CM capability will become disadvantaged for programs with hazardous intermediates. Biologics manufacturing is evolving toward perfusion and continuous downstream processing — CDMOs investing in these platforms will deliver better economics and more consistent quality for high-volume programs. CGT manufacturing remains capacity-constrained; viral vector programs should begin CDMO engagement 18-24 months before clinical material is needed. ADC conjugation IP is complex; site-specific conjugation technologies held by Synaffix (now Lonza) and others require explicit licensing terms before program start. Evergreening through process patents requires deliberate IP capture in the CDMO MSA — this is not a legal formality but a commercial value question.
Investment Strategy: Part V
CDMOs with validated continuous manufacturing platforms are in a stronger competitive position for the next generation of small molecule launches, where FDA’s preference for CM-based processes will increasingly influence approval timelines. For biotech investors: any gene therapy company that cannot identify a qualified CGT CDMO with available capacity within the next 12 months faces a clinical timeline risk that should be reflected in valuation. The ADC CDMO market is a 2026-2028 capacity constraint story — Lonza, Abzena, and Piramal are the primary beneficiaries, and M&A in this space is likely as smaller biotech companies seek to secure supply.
Part VI: Capacity, Scalability, and Supply Chain Resilience
Capacity Planning Methodology
Capacity assessment for a CDMO engagement requires modeling three scenarios: current clinical demand, projected Phase III demand (typically 3-10x Phase I quantities for small molecules, 10-50x for biologics due to dose and patient count scaling), and commercial launch demand. The commercial demand model must account for patient prevalence (not incidence), treatment duration, dose per treatment cycle, and process yield assumptions that are specific to the manufacturing scale.
A CDMO that can comfortably produce 10kg of API for a Phase I trial may not have the equipment, facility footprint, or regulatory approvals to produce 500kg for commercial supply. Conversely, a very large commercial-scale CDMO may not have the flexibility to handle small Phase I batch sizes without significant cost overhead. The optimal CDMO for each stage of a program may differ, requiring a deliberate technology transfer plan built into the program strategy from the start.
For biologics, bioreactor scale is the primary capacity metric. A 500L single-use bioreactor can typically produce 0.5-3 kg of mAb per batch in a 14-day fed-batch run, depending on titer and process yield. A 2,000L bioreactor produces 2-12 kg per batch. A 15,000L stainless steel bioreactor at a large commercial facility (Lonza Portsmouth, Samsung Biologics) can produce 50-100 kg per batch for high-titer processes. Clinical trial material for a typical Phase I mAb program requires 1-5 kg; a high-volume commercial program may require 500-2,000 kg per year. The scale difference demands different facilities, different regulatory validation packages, and typically a different CDMO.
Supply Chain Dual-Sourcing and Reshoring
COVID-19 exposed the pharmaceutical industry’s single-source dependency on Asian manufacturers for key starting materials (KSMs), advanced intermediates, and finished APIs. The FDA’s Drug Shortage Staff database recorded over 120 active drug shortages as of Q1 2024, many of which traced to single-source API suppliers in China or India. CDMOs that have built dual-sourcing strategies — qualifying two geographically distinct suppliers for each critical raw material — are better positioned to maintain supply continuity during disruptions.
The BIOSECURE Act’s passage has accelerated reshoring of pharmaceutical manufacturing to the United States and Europe. The CHIPS and Science Act (2022) precedent and the Drug Security and Accountability Act (proposed) both signal continued U.S. policy support for domestic pharmaceutical manufacturing investment. CDMOs that are actively expanding U.S.-based capacity — Thermo Fisher Scientific’s Biologics facility in Lengnau, Switzerland and Greenville, NC; AGC Biologics’ Columbus, Ohio facility; and Emergent BioSolutions’ domestic fill-finish network — are positioned to capture reshoring demand.
For sponsors, dual-sourcing at the CDMO level — qualifying two CDMOs for clinical or commercial manufacturing — is a supply chain resilience strategy that most companies cannot afford at the API level but should consider for high-value, supply-critical commercial products. The cost of qualifying a second CDMO (technology transfer, process comparability studies, regulatory filings for additional manufacturing site) is typically USD 2-8 million depending on the complexity of the molecule, but the cost of a supply disruption from a single-source CDMO on a commercial product generating USD 500 million in annual revenue is an order of magnitude larger.
Viral Vector Capacity: The 6-24 Month Wait Problem
Wait times for viral vector manufacturing capacity are the most acute capacity problem in the current CDMO market, as noted above. The specific dynamics differ by vector type:
For AAV, the constraint is primarily downstream capacity — ultracentrifuges, analytical AUC instruments, and AVB Sepharose resin supply. Upstream HEK293 production at 200L scale is more widely available. CDMOs with the complete upstream-to-downstream AAV platform at Phase I scale include: Andelyn Biosciences (Columbus, OH), VGXI (The Woodlands, TX), Genezen (Indianapolis, IN), and Novatek International (now part of Catalent’s gene therapy business, post-Novo Holdings). Wait times at these facilities for new program slots have been 9-18 months as of early 2026.
For lentiviral vectors (used in ex vivo CAR-T and HSC gene therapies), the constraint is S2 biosafety-classified cell culture capacity and the regulatory complexity of manufacturing allogeneic versus autologous products. CDMOs producing lentiviral vectors for commercial CAR-T programs include Lonza (for Bristol-Myers Squibb’s Breyanzi/liso-cel and Novartis’s Kymriah/tisagenlecleucel historically), Waisman Biomanufacturing, and Pacific Biosciences.
Sponsors with CGT programs should initiate CDMO conversations no later than 24 months before their target IND submission date and should secure a manufacturing slot with a binding commitment before beginning preclinical studies that will generate the material specification requirements for the IND.
Key Takeaways: Part VI
Capacity planning requires three-scenario modeling: clinical, Phase III, and commercial. The right CDMO for Phase I is often not the right CDMO for commercial launch — plan for technology transfers as a program milestone, not an afterthought. Dual-sourcing at the CDMO level is the highest-value supply chain risk mitigation for commercial-stage programs. CGT sponsors must engage CDMOs 18-24 months before IND submission to secure capacity.
Part VII: Contracts, IP Protection, and Paragraph IV Considerations
Contract Architecture: MSA and Work Orders
The contractual framework governing a CDMO relationship consists of a Master Services Agreement (MSA) that establishes the general terms of the relationship and individual Statements of Work (SoWs) or Work Orders that govern each specific project. The MSA should be negotiated with the input of IP counsel, regulatory counsel, and commercial counsel — not just procurement.
Critical MSA provisions for pharma IP teams:
IP ownership: The MSA must unambiguously specify who owns IP generated during the CDMO engagement. The standard industry framework distinguishes between “background IP” (IP that each party owned before the engagement began, or developed independently), and “foreground IP” (IP generated specifically in performance of the work). The sponsor should negotiate to own all foreground IP related to the product, including process improvements, new analytical methods, and new formulations. The CDMO should retain background IP in its general platform technologies. Any joint inventions must have a defined ownership and licensing framework.
Technology transfer rights: The MSA should include an unconditional right for the sponsor to receive, within a defined timeframe (typically 30-60 days of written request), all batch records, analytical methods, cell banks, reference standards, regulatory filing data, and process descriptions necessary to transfer manufacturing to an alternative CDMO. This right should not be conditional on payment of any amounts beyond agreed technology transfer fees set in the MSA.
Data ownership and access: All batch records, quality records, stability data, and regulatory submission data belong to the sponsor. The CDMO should not have the right to use sponsor-specific manufacturing data for any purpose other than performance of the agreed services.
Regulatory filing rights: The MSA should specify that the sponsor has the right to reference the CDMO’s Drug Master File (DMF) or Biological Master File (BioMF) in regulatory filings, and that the CDMO will maintain those filings in good standing and provide letters of authorization for their use.
Supply continuity provisions: Include force majeure carve-outs for supply disruptions, step-in rights allowing the sponsor to take over manufacturing in the event of CDMO insolvency or regulatory shutdown, and safety stock provisions requiring the CDMO to maintain a defined number of months’ inventory of clinical or commercial supply.
Background IP vs. Foreground IP: The Hidden Risk
The distinction between background and foreground IP is where most CDMO IP disputes originate. A CDMO that has developed a proprietary perfusion cell culture platform — its background IP — will resist any contract language that assigns improvements to that platform to the sponsor. But if the sponsor’s process is developed on that platform, and the specific parameters and process conditions that achieve the required product quality emerge from that development work, the line between platform-level improvements (CDMO background IP) and product-specific process IP (sponsor foreground IP) is genuinely blurry.
The cleanest resolution is a contract provision specifying that any IP generated during the engagement that is specific to the sponsor’s molecule, product, or manufacturing process is assigned to the sponsor, while general platform improvements that could be applied to any product manufactured on the platform are retained by the CDMO. This formulation requires active monitoring during the development program to identify inventive contributions and assign them correctly as they arise — a function that the sponsor’s IP team must perform proactively rather than waiting until the program is complete.
CDMO-Generated Process Patents and Paragraph IV Exposure
When a CDMO generates process IP that is subsequently assigned to the brand sponsor and listed in the FDA Orange Book, that process patent becomes part of the sponsor’s exclusivity fence and must be cleared by any generic filer through a Paragraph IV certification (asserting the patent is invalid or not infringed) or a Paragraph II or III certification (accepting that the generic will not enter the market while the patent is valid). CDMOs that have been integral to building the sponsor’s Orange Book patent estate are therefore direct contributors to the commercial exclusivity period.
However, the reverse situation creates a different risk. If a CDMO retained process IP that it subsequently licensed to a generic manufacturer — or if a CDMO’s background IP on a manufacturing method is accessible to generic CDMOs who use that same method — then the CDMO’s process approach may inadvertently facilitate generic entry at scale. For sponsors doing Paragraph IV litigation defense, the manufacturing process of the generic filer must be examined against the full patent estate, including any process patents filed by the brand CDMO. If the generic filer uses a different CDMO with a different process, the process patents may not read on the generic product.
For R&D leads and IP teams managing a patent cliff: use the CDMO relationship proactively to identify all potentially patentable process innovations at the development stage, file applications during the manufacturing development phase, and ensure those patents are listed in the Orange Book as manufacturing method patents where applicable. The typical timeline from process development to granted U.S. patent is 2-4 years for pharmaceutical manufacturing method claims, meaning this work must begin no later than Phase II of clinical development to generate enforceable patents before the composition-of-matter patent expires.
Patent Intelligence Tools for CDMO Diligence
Patent intelligence platforms provide competitive information relevant to CDMO selection across multiple dimensions. DrugPatentWatch covers pharmaceutical drug patents, Paragraph IV filings, regulatory exclusivity periods, Orange Book listings, and biosimilar filings. For a sponsor using it in CDMO diligence, the tool can reveal: which CDMOs are named in patent litigation (as manufacturers of record for products involved in Paragraph IV disputes), which generic filers have used specific CDMOs as their manufacturers (a proxy for that CDMO’s exposure to the generics market and potential IP conflicts), and which approved products have manufacturing process patents with long remaining terms (informing evergreening opportunities).
For CDMOs using patent intelligence to develop business strategy: DrugPatentWatch’s pipeline monitoring capability allows identification of drugs approaching patent expiration, which signals upcoming need for generic or biosimilar manufacturing capacity; newly filed composition-of-matter patents, which signal early-stage programs likely to need CDMO services within 3-5 years; and Paragraph IV filing activity, which identifies programs in active Hatch-Waxman litigation where manufacturing scale-up for either brand or generic manufacture may become needed rapidly.
Key Takeaways: Part VII
The MSA is the most consequential document in a CDMO relationship from an IP perspective. Foreground IP ownership defaults to the CDMO in many standard contracts if not explicitly negotiated. CDMO-generated process patents can be valuable evergreening tools if correctly captured — or worthless liabilities if not captured at all. Paragraph IV defense strategy must account for the manufacturing process chosen by the CDMO and whether that process is distinguishable from the process available to generic filers.
Investment Strategy: Part VII
Patent cliffs present a systematic opportunity for CDMO-dependent generic and biosimilar manufacturers. Any NDA or BLA with an expiring composition-of-matter patent whose manufacturing process patent expires more than 2 years later represents a litigation-ready exclusivity extension — a target for Paragraph IV challenge. Generic CDMO operators (Teva’s API business, Amneal, Hikma’s injectables business) systematically identify such assets using patent intelligence databases. Biosimilar developers (Sandoz, Samsung Bioepis, Formycon) apply the same logic to biologics Orange Book listings and 351(k) biosimilar filing strategy.
Part VIII: Financial Due Diligence on CDMOs
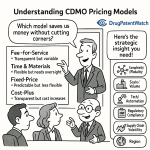
Pricing Model Analysis
CDMO pricing in 2026 spans three primary structures, each with a different risk allocation between sponsor and CDMO:
Fee-for-service (FFS): The CDMO is paid for specific deliverables — a batch of clinical material, a stability study, a process development report. Costs escalate if the scope changes. FFS is appropriate for early-phase programs where the scope is well-defined and the sponsor wants maximum cost visibility at the task level. The risk of scope creep is substantial: pre-formulation studies that reveal unexpected polymorphism, or upstream development that requires multiple CHO cell line screening campaigns, can multiply the original budget by 2-3x under FFS.
Time and materials (T&M): The CDMO is paid for labor hours and material consumption at agreed rates. T&M is appropriate for exploratory development programs where the scope is genuinely uncertain. The sponsor carries more cost risk than under FFS, but the CDMO has less incentive to artificially limit scope to protect its margin. T&M requires the sponsor to actively monitor labor hours against budget milestones, which demands dedicated internal project management capacity.
Fixed-price: The CDMO accepts full cost risk for defined deliverables at a fixed price. Fixed-price contracts are appropriate for well-characterized, reproducible manufacturing operations — e.g., production of clinical batches of a small molecule API where the process is validated and yield is predictable. CDMOs typically build a 15-25% risk premium into fixed-price bids for complex programs; for programs with high technical risk (novel molecule, unvalidated process), a fixed-price structure is often unavailable or priced high enough to make it economically inferior to T&M.
When evaluating quotes, the most common failure mode is comparing total headline costs across CDMOs without accounting for differences in what is included. One CDMO’s USD 2 million Phase I campaign cost may include regulatory starting material sourcing, analytical method development, and ICH Q1-compliant stability study initiation. Another CDMO’s USD 1.8 million quote may exclude all three, with those activities priced separately in addenda that only appear after contract signature. Require a line-item scope breakdown for every quote and reconcile it against a standard scope template developed by your project team.
CDMO Financial Health Red Flags
Private CDMOs — which constitute the majority of the global market — do not publish financial statements. The following proxies are available for assessing financial health:
Ownership structure and backing: CDMOs owned by private equity with leverage ratios above 5-6x EBITDA are at elevated risk of financial distress if market conditions tighten. The CDMO industry saw several PE-backed insolvencies in 2022-2024 as COVID-era demand for vaccine and therapeutics manufacturing collapsed and overleveraged platforms lost revenue. CDMOs with long-term corporate ownership (e.g., Boehringer Ingelheim’s contract manufacturing business, Thermo Fisher Scientific’s Patheon division) or sovereign-backed ownership (Samsung Biologics, within the Samsung Group) have structurally lower financial distress risk.
Revenue concentration: A CDMO where one or two clients represent more than 40-50% of revenue is exposed to single-client departure risk. That risk is not just the CDMO’s problem — it is the sponsor’s problem if the CDMO’s financial stability deteriorates when a major client’s program ends.
Facility investment trends: A CDMO that has not made significant capital investment in its manufacturing infrastructure in 3-5 years is either cash-constrained or strategically stagnant. Both signal risk. Conversely, a CDMO that is aggressively expanding capacity into a speculative market segment — as several CGT CDMOs did during the 2020-2021 gene therapy hype cycle — may be overextended if the market for that modality does not develop as projected.
Unrealistically low bids: A bid that is 30-40% below competitive quotes for equivalent scope is almost certainly missing scope, relying on optimistic yield assumptions, or pricing at or below cost to win the business. In pharmaceutical manufacturing, a CDMO that executes below the true cost of a program will either request contract amendments to cover overruns or make quality shortcuts to recover margin.
Key Takeaways: Part VIII
Pricing model selection should match technical risk profile — FFS for defined scopes, T&M for exploratory programs. All-in quote comparison requires a standardized scope template. Private CDMO financial health can be assessed through ownership structure, revenue concentration, and capital investment patterns. Below-market bids are nearly always a red flag, not an opportunity.
Part IX: Case Studies — What Works and What Fails
The Novo Holdings / Catalent Transaction and Its Supply Chain Implications
Novo Holdings acquired Catalent for approximately USD 16.5 billion, with the transaction closing in March 2024. The stated rationale was securing fill-finish capacity for Novo Nordisk’s semaglutide-based GLP-1 products (Ozempic, Wegovy, Rybelsus), which faced severe supply constraints driven by demand that significantly exceeded internal manufacturing capacity. Catalent’s Bloomington, Indiana and Anagni, Italy fill-finish facilities were identified as the primary strategic assets.
The transaction created a conflict of interest structure that has no clear precedent in the CDMO industry: a CDMO owned by the investment arm of a pharmaceutical company that is simultaneously one of the largest commercial players in the GLP-1 market. Catalent’s other clients — which include many large pharma and biotech companies — now manufacture products at facilities owned by a competitor’s corporate parent. The IP isolation implications are significant, and Catalent has publicly committed to maintaining confidentiality firewalls between Novo Nordisk-related operations and other client programs. Whether those firewalls are legally and operationally sufficient is a question that every existing and prospective Catalent client should address in their contractual agreements.
For portfolio managers: the transaction is a useful case study in how CDMO ownership changes can create immediate supply chain and IP risk for dependent pharma companies. The lesson for sponsors is to include CDMO change-of-control provisions in MSAs that give the sponsor the right to terminate, or at minimum to renegotiate, upon a change of ownership that creates a conflict of interest.
mRNA CDMO Partnerships: Moderna, Pfizer, and the Scale-Up Playbook
The COVID-19 vaccine manufacturing scale-up between 2020 and 2022 demonstrated both the potential and the limitations of CDMO partnerships in an extreme demand scenario. Moderna partnered with Catalent (for fill-finish) and Lonza (for mRNA drug substance at facilities in Geleen, Netherlands, and Portsmouth, NH). Pfizer-BioNTech partnered with Siegfried (fill-finish), Baxter BioPharma Solutions, and Rentschler Biopharma, while scaling its own Kalamazoo, MI and McPherson, KS facilities internally.
The technical challenges encountered in mRNA vaccine scale-up were: LNP formulation consistency at commercial scale (particle size distribution and encapsulation efficiency were sensitive to mixing parameters that changed non-linearly with scale), cold chain management (mRNA LNP formulations required storage at -70°C for Moderna’s vaccine and -20°C for Pfizer’s, imposing unusual logistics demands on CDMOs), and analytical method validation at speed (regulatory agencies granted Emergency Use Authorization with abbreviated analytical validation packages, creating post-authorization commitments that CDMOs had to fulfill).
The IP implications were significant. Moderna and Pfizer each claimed broad patent coverage on their mRNA and LNP technologies, with Moderna subsequently suing Pfizer-BioNTech for infringement of its LNP and mRNA modification patents (CureVac, as a third party, filed its own claims). The CDMO partners’ role in producing the infringing or allegedly infringing material was specifically addressed in the litigation: under the MSAs, the CDMOs were manufacturing under direction of the sponsor and were contractually indemnified from IP claims arising from following the sponsor’s process specifications. This is the standard indemnification structure and the one sponsors should require in their MSAs.
Common CDMO Failure Modes: A Pattern Analysis
Analysis of disclosed CDMO partnership failures across publicly available case studies, FDA enforcement records, and court filings reveals four recurring failure patterns:
Capability overstatement at the proposal stage: CDMOs that win contracts by claiming capabilities they do not yet have — or that are under development — expose sponsors to delays of 6-18 months when the gap becomes apparent during execution. This most commonly occurs in emerging modalities (CGT, mRNA, ADCs) where CDMOs are entering new technology areas to capture growing market segments. The mitigation is a qualification audit that tests specific technical capabilities before contract signature, not after.
Deviation management failure: Batch failures, sterility test failures, and out-of-specification (OOS) results are inevitable in pharmaceutical manufacturing. The failure mode is not the deviation itself but the CDMO’s response — inadequate root cause analysis, incomplete corrective action, or failure to notify the sponsor promptly. Under 21 CFR Part 314 and cGMP regulations, sponsors are responsible for the quality of their products regardless of whether manufacturing is outsourced. A CDMO that manages deviations poorly exposes the sponsor to regulatory liability, not just schedule risk.
Financial distress during program execution: Several small and mid-sized CDMOs entered financial distress during 2022-2024, following the COVID-era demand spike and subsequent normalization. Sponsors in mid-program with those CDMOs faced immediate supply disruptions, technology transfer emergencies, and in some cases loss of batch records and analytical method documentation when facilities closed without adequate data handover procedures. The mitigation is financial due diligence before program start and step-in right provisions in the MSA.
IP capture failure at program end: Sponsors who complete a manufacturing development program with a CDMO and then attempt to switch to an alternative manufacturer frequently discover that the process is not fully documented, that the CDMO retained ownership of analytical methods it considers proprietary, or that the cell bank was not contractually transferred along with the process description. These failures are entirely contractual — they result from MSAs that did not address technology transfer rights with adequate specificity.
Part X: The Future CDMO Landscape
Market Consolidation and M&A Trajectories
The CDMO industry consolidation trend, active since the early 2010s, is accelerating. The top five players hold only approximately 15% of the global market, leaving the remainder across hundreds of smaller platforms. That fragmentation is not sustainable — the investment required to build and maintain GMP-compliant facilities with advanced technology capabilities is beyond the reach of most small CDMO operators without external capital support.
The M&A trajectory favors: large-scale biologics CDMOs acquiring specialized modality players (Lonza’s acquisition of Synaffix for ADC bioconjugation IP; Thermo Fisher Scientific’s acquisition of Patheon and PPA for integrated pharma services); private equity rollup of regional small-molecule CDMOs to create scale; and pharma sponsor acquisitions of CDMO capacity as strategic supply assurance (Novo Holdings/Catalent being the most visible example, but Eli Lilly’s acquisition of CDMO capacity in the GLP-1 space follows the same logic).
The consolidation creates a bifurcated market: very large, fully integrated CDMOs capable of supporting multiple modalities across the full development-to-commercial lifecycle, and highly specialized boutique CDMOs with deep expertise in specific therapeutic areas or manufacturing technologies. The mid-market — CDMOs with single-modality capabilities at clinical scale but limited commercial capacity — will be the primary acquisition target.
The Evolving Regulatory Trajectory
Regulatory agencies are moving toward continuous oversight models that de-emphasize periodic inspection events and emphasize real-time data submission and ongoing manufacturing surveillance. FDA’s Pharmaceutical Quality/Manufacturing Science (PQ/MS) initiative is developing a framework for real-time reporting of manufacturing performance data, which would require CDMOs to maintain continuous electronic manufacturing records accessible to the agency. The EMA’s ongoing work on advanced therapy medicinal product (ATMP) manufacturing regulations is creating increasingly specific requirements for CGT CDMO GMP compliance.
ICH Q13 (continuous manufacturing, finalized 2023) and ICH Q14 (analytical procedure development, in implementation) are creating new regulatory expectations that CDMOs must embed into their quality systems. Sponsors should specifically ask CDMOs about their ICH Q13 and Q14 implementation roadmaps and timelines for any program that will be submitted after 2026.
Specialization vs. Integration: The Competitive Positioning Question
The CDMO industry faces a genuine strategic tension between integration (offering all services under one roof to eliminate technology transfer risk and streamline project management) and specialization (building superior capability in a specific modality or therapeutic area). Both models are commercially viable — the question is which is right for a specific program.
For sponsors with a single program in an advanced modality, a specialized CDMO with deep experience in that specific technology is usually superior to a large integrated platform with broad but shallow capability. For sponsors with multi-asset, multi-modality pipelines across development stages, an integrated CDMO partner — or a small number of preferred CDMO relationships — reduces the management overhead of coordinating multiple specialized vendors.
The “Goldilocks” positioning articulated by some CDMO executives — large enough to scale commercial programs, small enough to move fast on early-phase work — is aspirational but achievable for platforms in the USD 500 million to USD 2 billion annual revenue range that have made deliberate investments in both commercial-scale infrastructure and early-phase flexibility. Lonza’s Ibex Design-Build-Operate model, which allows sponsors to commission dedicated manufacturing suites within Lonza’s Swiss facilities, is one commercial implementation of this concept.
Master Key Takeaways
The right CDMO is not the largest, the cheapest, or the one with the best marketing materials. It is the one whose technical capabilities, IP posture, quality systems, financial stability, and cultural approach match the specific requirements of your program at its current development stage.
CDMO selection is an IP event, not a procurement event. A CDMO’s patent portfolio reveals its genuine technical depth. The MSA must protect foreground IP ownership, technology transfer rights, and data access unconditionally. Process patents developed during the CDMO relationship are potential Orange Book assets — they must be captured proactively.
Regulatory track record is predictive. FDA 483 observations and Warning Letters are diagnostic tools. Patterns of data integrity observations or inadequate investigations indicate systemic quality culture failures that rarely resolve quickly. A CDMO with an outstanding Warning Letter near your projected NDA submission window creates regulatory risk that will affect your timeline, not just theirs.
Technology choices have strategic consequences. Continuous manufacturing, perfusion biologics, site-specific ADC conjugation, and agentic AI process monitoring are not incremental improvements — they are technology platform choices that affect regulatory strategy, cost structure, and switching costs. Choose a CDMO whose technology roadmap aligns with your program’s commercial horizon.
The BioSecure Act has materially changed the global CDMO map. U.S.-based sponsors with WuXi Biologics or WuXi AppTec dependencies face technology transfer timelines of 12-24 months with real IP and regulatory risk. That risk should be reflected in program valuations.
Supply chain resilience requires active management. Dual-sourcing at the CDMO level, dual-sourcing of critical raw materials, and contractual step-in rights are the primary instruments. Viral vector capacity constraints require 18-24 month advance engagement for CGT programs.
Financial due diligence on CDMOs is not optional. PE-backed CDMOs with high leverage, revenue concentration in one client, and stagnant capital investment represent real financial distress risk. Below-market bids are scope gaps, not discounts.
Analyst FAQ
Q: How do I evaluate a CDMO’s IP portfolio for strength relative to its competitors in the same modality?
Pull the CDMO’s granted patents from Espacenet or the USPTO full-text database, filtered by IPC class for the relevant manufacturing technology (C12P for microbial/cell culture, A61K for formulation, C12N for cell biology). Compare the volume, citation index, and claim breadth of the CDMO’s patents against its primary competitors. Patents with high forward citation counts (cited frequently by subsequent filers) in the specific technical area are proxies for foundational IP. Also review whether the CDMO has licensed its platform IP to third parties — out-licensing revenue signals IP strength, while a portfolio with no licensing activity may be defensive rather than commercially productive.
Q: What contract terms most commonly lead to IP disputes when a sponsor attempts to switch CDMOs?
Three provisions generate the majority of IP disputes: overly broad definitions of “improvements to background IP” that allow the CDMO to claim ownership of product-specific process innovations; absence of unconditional technology transfer rights (some CDMOs include “right to withhold” provisions pending payment disputes); and cell bank ownership clauses that leave master cell bank ownership ambiguous when the cell line was developed collaboratively. Negotiate all three explicitly before program start.
Q: How should a portfolio manager assess the financial risk of a CDMO dependency in a biotech company’s pipeline valuation?
The key variables are: how close the dependent program is to an NDA/BLA submission (programs within 24 months face the highest regulatory risk from CDMO quality failures or BioSecure Act compliance issues), whether the MSA includes technology transfer rights that would allow alternative manufacturing within 12 months, and whether the CDMO has had any FDA enforcement actions in the past 3 years. A program without a qualified alternative CDMO and without technology transfer rights should carry a 15-25% schedule risk discount on its projected approval timeline.
Q: When should a sponsor consider building in-house manufacturing capacity rather than working with a CDMO?
In-house manufacturing is strategically justified when: the CDMO market lacks sufficient capacity or technical capability for the required modality at commercial scale (an increasingly relevant factor for CGT programs); the manufacturing process is so proprietary that CDMO exposure creates unacceptable IP risk; or the commercial program has a revenue profile and duration that makes the capital investment economically superior to ongoing CDMO costs over the product lifecycle (typically requiring USD 300 million-plus in annual revenue and a >10-year commercial horizon). Modular manufacturing solutions — which can be built and qualified in 18-24 months for USD 30-80 million at clinical scale — are an intermediate option that some CGT companies are pursuing as the market matures.
Q: What is the most important single provision to negotiate in a CDMO MSA from an IP perspective?
The unconditional right to receive all manufacturing data, analytical methods, and regulatory documentation within 30 days of written request, regardless of any outstanding commercial disputes. This provision — often called the “data access right” or “technology escrow” provision — ensures that the sponsor can effect a technology transfer to an alternative CDMO even if the relationship with the original CDMO deteriorates. Without it, a CDMO facing a contract dispute can effectively hold the program hostage by withholding documentation needed for the transfer.
Specific financial figures, regulatory actions, and CDMO capacity data should be independently verified before use in investment or strategic decisions.
Data sources include Fortune Business Insights, BioSpace, Healthcare Web Wire, IQVIA, FDA enforcement databases, Espacenet, USPTO full-text patent database, SEC filings, and company public disclosures.