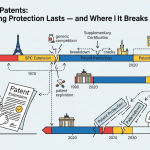
Who this is for: Pharma and biotech IP teams, portfolio managers, R&D leads, M&A counsel, and institutional investors with exposure to branded pharmaceutical revenue.
What this covers: Patent architecture, evergreening tactics, Hatch-Waxman mechanics, PTAB strategy, biologic IP, AI inventorship doctrine, global SPC and Bolar frameworks, and three blockbuster case studies in full technical detail.
Section 1: The Economics of Exclusivity: Why the Patent Portfolio Is the Business
A pharmaceutical company’s patent portfolio is not a legal department artifact. It is the primary mechanism by which a company converts R&D expenditure into revenue. Strip out the patents and you have a manufacturing operation without pricing power, a drug without a monopoly, and a business without a defensible moat.
The foundational model runs as follows. A company invests, on average, north of $2 billion to bring a single new molecular entity from discovery through FDA approval, a process that consumes 10 to 15 years. That investment generates no revenue during its course. The patent grants a statutory exclusivity window, nominally 20 years from the filing date, during which the company holds a monopoly on the drug. Within that window, often compressed to eight or ten years of effective commercial exclusivity after accounting for development time, the company must price the drug high enough to recoup its investment, fund its next pipeline, pay its cost of capital, and generate returns for shareholders. The moment exclusivity ends and generics enter, branded drug revenues typically fall 80 to 90 percent within 12 months.
This compression creates an existential tension. The patent system offers the temporary monopoly as an incentive to take the discovery risk. Society tolerates the monopoly in exchange for eventual generic access. Every strategic, legal, and regulatory maneuver in the pharmaceutical industry flows from this tension: how to extend the monopoly as long as legally permissible, how to collapse a competitor’s monopoly as quickly as possible, and how to do both simultaneously across dozens of products in dozens of jurisdictions.
Between 2025 and 2029, an estimated $350 billion in industry revenue faces patent expiration, concentrated in drugs like Merck’s Keytruda (pembrolizumab), Bristol Myers Squibb’s Eliquis (apixaban), and Johnson & Johnson’s Darzalex (daratumumab). That figure dwarfs the patent cliff of a decade ago. Every major pharma company is simultaneously managing an offensive LCM program for its revenue-generating assets and scouting acquisitions to fill the gaps those expirations will open.
Key Takeaways, Section 1: The patent portfolio is not a legal cost center. It is the primary profit engine of any innovator pharmaceutical or biotech company. Revenue quality, pipeline durability, and M&A optionality all reduce to a single question: what does the patent estate look like, and for how long does it hold?
Section 2: Architecting a Fortress: Patent Types, Claim Strategies, and Portfolio Construction
2.1 The Composition of Matter Patent: The Cornerstone
The composition of matter (CoM) patent, also called the API patent or compound patent, covers the chemical molecule or biological compound that produces the drug’s therapeutic effect. It is the strongest and broadest form of pharmaceutical IP because it attaches to the substance itself, regardless of how the substance is formulated, dosed, or administered. If the patented molecule is in a competitor’s product in any form, the claim applies.
For Orange Book listing purposes in the US, a patent’s claims must explicitly recite the active pharmaceutical ingredient of the approved drug. This requirement is not procedural housekeeping. It is the mechanism that activates the Hatch-Waxman litigation framework, specifically the 30-month stay that blocks FDA approval of a generic ANDA upon an infringement suit. A CoM patent that is correctly drafted and Orange Book-listed triggers the entire defensive architecture of US pharmaceutical IP law.
Claim drafting strategy for CoM patents centers on breadth versus defensibility. The Markush structure, named after inventor Eugene Markush, allows a single claim to define a core molecular scaffold with variable substituents, covering potentially millions of related compounds. A well-drawn Markush claim makes designing around the invention technically and commercially unattractive. The tradeoff is validity risk: broader claims are more likely to encounter prior art that renders them obvious or anticipated, particularly as patent examiners and PTAB judges scrutinize the scope of blocking claims.
A sophisticated filing strategy deploys multiple independent claims covering the core compound at different levels of structural specificity, with dependent claims narrowing toward the precise marketed molecule. This creates a layered claim set where invalidating the broadest claim still leaves narrower claims intact, preserving some degree of protection even after a successful patent challenge.
2.2 The Secondary Patent Arsenal: Formulation, Method, Polymorph, Device, and Combination
No blockbuster drug survives on a single patent. The effective commercial life of an API patent is routinely less than 10 years after accounting for clinical development and regulatory review time. Secondary patents are the instruments by which companies construct a defense-in-depth around a drug, creating multiple overlapping barriers that generic manufacturers must overcome simultaneously.
Formulation patents cover the specific pharmaceutical composition: the combination of the API with excipients, the release mechanism, the particle size distribution, the pH, the vehicle for delivery. An extended-release formulation, a transdermal patch, or a subcutaneous auto-injectable that replaces an intravenous infusion can each carry its own 20-year patent term while simultaneously improving the product’s clinical profile. Formulation patents are the bread and butter of lifecycle management because they offer genuine clinical differentiation in addition to IP protection.
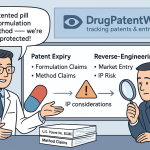
Method of use patents cover the application of the drug to treat a specific disease or condition. They do not protect the molecule itself, so a generic manufacturer can in theory produce and sell the compound for uses not covered by the method patent. But most diseases of commercial interest are patented, and “skinny labeling” strategies that carve out patented indications from generic labels have faced increasing legal scrutiny in US courts. The Federal Circuit’s 2021 decision in GlaxoSmithKline LLC v. Teva Pharmaceuticals USA, Inc. held that Teva could be liable for induced infringement of GSK’s use patent for carvedilol in heart failure even though Teva’s label carved out that indication, because Teva’s promotional conduct directed physicians toward the patented use. That ruling raised the bar for generics pursuing skinny labels and added durability to method of use patent strategies.
Process patents cover manufacturing methods. They are harder to enforce because proving a competitor uses a patented synthesis route requires access to proprietary manufacturing data that is typically unavailable before litigation. Some jurisdictions, including Germany and Japan, apply a “burden of proof reversal” where the alleged infringer must disprove use of the patented process rather than the patentee having to prove it. In practice, process patents are strongest as a negotiating tool, raising the legal cost and complexity of generic entry rather than functioning as absolute blocks.
Polymorph patents cover specific crystalline forms of the API. Different polymorphs can have different solubility, stability, bioavailability, and manufacturability profiles. AstraZeneca’s esomeprazole (Nexium) is the canonical chiral switch case, where the company patented the S-enantiomer of omeprazole (Prilosec) and built a commercial franchise around that single-isomer product, extending its proton pump inhibitor franchise well beyond Prilosec’s patent expiration. The India Supreme Court’s 2013 rejection of Novartis’s polymorph patent on imatinib mesylate (Gleevec) under Section 3(d) of India’s patent law illustrates that polymorph strategies face jurisdiction-specific legal risk, particularly in markets where regulators require demonstration of enhanced efficacy, not just improved physical properties.
Delivery device patents cover the hardware required to administer the drug. Auto-injectors, prefilled syringes, inhalers, and drug-eluting stents can each carry separate, long-lived patent protection. For biologics administered subcutaneously, the device patent may outlast every pharmaceutical patent on the molecule itself, preserving brand loyalty and pricing leverage long after the compound is off-patent.
Combination therapy patents protect the use of the drug alongside another active ingredient, either as a fixed-dose combination product or as a combination regimen. These patents have become increasingly valuable in oncology and HIV, where combination regimens are standard of care and the addition of a novel agent to an existing backbone can justify a new use patent with a fresh 20-year term.
2.3 Timing, Claim Scope Variation, and Global Coverage
Filing timing is a variable companies routinely mismanage. The 20-year term starts on the filing date. A compound patent filed at the time of discovery but not reaching FDA approval for 12 years has eight years of effective commercial exclusivity remaining. Filing a provisional application early establishes a priority date while deferring examination costs and allowing an additional year to gather data before the formal prosecution clock starts.
The tension between filing early to establish priority and filing late to maximize remaining exclusivity requires company-specific analysis of the likely development timeline. A drug in a fast-moving regulatory environment like oncology with Breakthrough Therapy Designation might reach approval in seven years rather than twelve, shifting the calculus toward an earlier filing date. A drug in a chronic disease indication facing a lengthy Phase 3 program might benefit from a later formal filing.
Claim scope variation within the same portfolio is a risk management strategy. Broad Markush claims provide maximum blocking power but face higher invalidation risk on prior art and obviousness grounds. Narrow species claims covering the exact marketed compound and its pharmaceutically relevant salts are harder to invalidate but easier for competitors to design around. A well-constructed portfolio contains both, such that successful invalidity of the broad genus claim still leaves species-level protection intact.
Global coverage requires territory-by-territory prioritization because patent rights are strictly national. The Patent Cooperation Treaty (PCT) application allows companies to file a single international application that reserves the right to enter over 150 national phase proceedings, providing a 30-month window after the priority date to make national filing decisions. That window is valuable because it coincides with early clinical data that informs how commercially important each market will be and how aggressively competitors are filing in the same space.
Beyond the US and EU, China is now the second-largest pharmaceutical market by volume and has significantly strengthened its IP enforcement posture since joining the World Trade Organization. Prosecution and enforcement strategy in China warrants dedicated attention. Similarly, Japan’s patent linkage system, while less rigid than the US Orange Book framework, creates opportunities for innovators to delay generic market authorization through administrative challenge proceedings.
2.4 Portfolio Architecture: Key Takeaways
A high-value drug patent portfolio has five structural characteristics: it anchors on a well-drafted CoM patent with Orange Book listing, it builds multiple secondary patent layers covering different product attributes with staggered expiration dates, it varies claim scope across individual patents to create redundant defenses, it achieves coverage in all jurisdictions generating material revenue, and it is subject to active management including scheduled reviews, pruning of maintenance-fee-intensive low-value assets, and gap-filling new filings aligned with commercial priorities.
Section 3: IP Valuation as a Core Asset: Methodologies and Deal-Level Mechanics
3.1 Why Patent Valuation Is Not an Accounting Exercise
Standard balance sheet accounting treats patents as intangible assets subject to amortization on a straight-line basis over their useful life. This treatment is almost entirely useless for strategic decision-making. The book value of a patent tells you its historical cost, which is a function of prosecution fees and attorney time, not its economic significance. AbbVie’s Humira patent estate had a book value in the hundreds of millions. Its economic value was tens of billions. The gap between those figures is the entire point.
Patent valuation for pharmaceutical IP purposes requires three distinct analytical lenses: the income approach, the market approach, and the cost approach. Each is useful in different contexts. None is definitive alone.
3.2 The Income Approach: Discounted Cash Flow and the RRNPV Model
The income approach calculates the net present value of projected future cash flows attributable to the patent, discounted at a rate reflecting the risk profile of those cash flows. For pharmaceutical patents, the standard implementation uses a risk-adjusted net present value (rNPV) model that explicitly accounts for clinical, regulatory, and commercial probability factors.
The model starts with a peak sales estimate for the drug in each major market, derived from epidemiological data, patient segmentation, pricing assumptions, and penetration curves. It then applies phase-specific probability of success (PoS) factors to adjust for clinical attrition risk. A Phase 1 asset might carry a 10 to 15 percent PoS to approval; a Phase 3 asset with clean interim data might carry 60 to 80 percent. The resulting expected revenue stream is discounted at a rate reflecting the company’s weighted average cost of capital plus an asset-specific risk premium.
The patent’s contribution to this cash flow is the period of market exclusivity it enables. When the patent expires, revenue typically declines 80 to 90 percent rapidly. The economic value of the patent is therefore the present value of the difference between monopoly-period cash flows and post-exclusivity cash flows, adjusted for the time remaining on the patent. Adding one year of effective exclusivity to a drug generating $5 billion annually at a 30 percent operating margin adds roughly $1.5 billion in gross margin, discounted appropriately.
For M&A purposes, this calculation drives deal premiums. Bristol Myers Squibb paid $74 billion for Celgene in 2019, a transaction justified substantially by the residual exclusivity and pipeline optionality of Revlimid (lenalidomide), Pomalyst (pomalidomide), and Opdivo (nivolumab) combinations. The effective price per year of exclusivity purchased can be calculated, and that ratio serves as a benchmark for evaluating subsequent deals.
3.3 The Market Approach: Comparable License Transactions
The market approach values a patent by reference to arm’s-length transactions involving comparable IP. In pharmaceutical licensing, royalty rates for API patents in major therapeutic categories have been studied extensively. Median royalty rates for drug patents range from 3 to 10 percent of net sales, with biologics commanding the upper end of that range and mature small-molecule franchises at the lower end.
The challenge is comparability. A patent on a first-in-class mechanism in a large indication is not comparable to a patent on a reformulation of an established drug in a niche indication. Deal structure further complicates comparison: upfront payments, milestones, royalties, sublicensing rights, and co-promotion arrangements all affect the economic content of reported transactions. Databases like Cortellis, Evaluate Pharma, and GlobalData Pharma Deals aggregate licensing transaction data, but raw comparables require extensive normalization before they are analytically useful.
The market approach is most reliable when applied to out-licensing of a specific compound at a defined clinical stage, where comparable deals involving similar stage assets in similar indications exist and are disclosed in sufficient detail. For whole-portfolio acquisitions or unique platform technology, the income approach generally produces more defensible estimates.
3.4 The Cost Approach: Floor Value and Replacement Cost
The cost approach values the patent at the cost required to recreate the underlying invention: the R&D investment, clinical costs, prosecution fees, and regulatory costs necessary to bring a comparable asset to the same stage. It functions as a valuation floor: no rational acquirer would pay more than replacement cost for a patent if the alternative is recreating the invention.
In practice, the cost approach is most useful for early-stage assets where clinical proof-of-concept data does not yet exist to support an income approach estimate, and where comparable market transactions are sparse. For mature, revenue-generating assets, replacement cost dramatically understates economic value.
3.5 M&A Due Diligence: What Acquirers Get Wrong
Patent due diligence in pharmaceutical M&A is a systematic risk assessment, not a box-checking exercise. A properly structured due diligence covers chain of title (confirming the target actually owns what it claims to own), prosecution history estoppel (identifying statements made during patent prosecution that limit the scope of the granted claims and cannot be reclaimed in litigation), prior art landscaping (assessing whether any third-party publications or patents could successfully invalidate key claims), and Orange Book listing accuracy (verifying that listed patents meet FDA requirements and are correctly described).
Common errors include underweighting prosecution history estoppel risk. When a patent applicant amends claims or makes arguments to overcome a prior art rejection during prosecution, the doctrine of prosecution history estoppel prevents the patentee from later arguing that the patent covers subject matter that was surrendered during prosecution. Acquirers who analyze only the granted claim language without reading the full prosecution file history routinely overpay for patents whose effective scope is substantially narrower than the granted claims suggest.
A second common error is failure to identify potential PTAB vulnerability. The inter partes review (IPR) petition rate at the PTAB for pharmaceutically relevant Orange Book patents has accelerated since 2012. An acquirer who models exclusivity through a patent’s nominal expiration date without stress-testing the patent’s ability to survive an IPR challenge is building a financial model on a potentially fragile foundation.
Investment Strategy Note for Analysts:
When building a pharma equity model, treat the rNPV of the patent estate as the primary valuation input, not trailing EBITDA multiples. EBITDA multiples do not capture the step-change revenue decline that occurs upon generic entry. A company trading at 10x EBITDA might look cheap relative to peers, but if its lead product loses exclusivity in 18 months and biosimilar entry follows in 36 months, that multiple reflects a business that is about to shrink by 40 percent. The patent expiration schedule is the earnings model.
Section 4: The Offensive Playbook: Lifecycle Management, Evergreening, and Patent Thickets
4.1 The Patent Cliff: Scale of the Problem
The “patent cliff” is the shorthand for the revenue implosion that follows patent expiration for a major drug. When Lipitor lost exclusivity in 2011, Pfizer’s revenue fell by $10 billion over the subsequent two years. When Humira’s US biosimilar exclusivity ended in 2023, AbbVie faced the prospect of a drug that had generated over $20 billion annually facing competition for the first time. The 2025-2029 wave represents a concentration of cliff exposure that exceeds the prior decade’s losses, with Keytruda alone generating over $25 billion annually at risk.
Revenue does not gradually decline when generics enter. Pharmacy benefit managers and insurers immediately shift formulary preference to the generic, which is priced at 10 to 20 percent of the branded price. Retail pharmacy substitution laws in most US states allow, and in practice require, pharmacists to dispense a generically equivalent drug unless the prescribing physician specifically writes “dispense as written.” The combination of formulary pressure and automatic substitution collapses branded unit volume within months of generic entry.
4.2 Evergreening: The Mechanics and the Controversy
Evergreening refers to the practice of obtaining secondary patents on incremental modifications to an existing drug in order to extend effective market exclusivity beyond the original API patent’s term. The term is used pejoratively by critics and defensively by industry, but the mechanics are commercially precise.
The typical evergreening technology roadmap for a small-molecule drug proceeds through several stages. During Phase 2 and 3 clinical development, the formulation team identifies modifications that improve the drug’s performance or patient adherence, such as a once-daily extended-release formulation versus a three-times-daily immediate release, or a fixed-dose combination that simplifies a multi-pill regimen. Each improvement generates new patent applications. The company then conducts clinical studies comparing the improved formulation to the original, generating data to support an updated label and sometimes a new indication. Shortly before the original API patent expiration, the company transitions prescribers to the new formulation through marketing, often simultaneously withdrawing or de-emphasizing the original.
This “product hopping” strategy works because state pharmacy substitution laws typically require bioequivalence between the dispensed generic and the specific product described in the prescription. A prescription written for the once-daily extended-release formulation cannot be filled with a generic of the three-times-daily immediate-release formulation, even if both contain the same API. If the company has successfully transitioned the market to the new formulation before the original patent expires, the first-generation generics entering the market against the old formulation find a much smaller commercial target.
AstraZeneca’s transition from omeprazole (Prilosec) to esomeprazole (Nexium) is the textbook illustration. Prilosec was a $6 billion drug facing generic entry in 2001. AstraZeneca launched Nexium, the S-enantiomer, in 2001 and invested heavily in transitioning prescriptions to the new brand, which carried patent protection through 2014. Critics argued the single-enantiomer formulation offered no meaningful clinical advantage for most patients. AstraZeneca countered that esomeprazole achieved higher plasma concentrations and more consistent acid suppression. Both arguments contain some truth, which is precisely why the chiral switch strategy occupies contested legal and ethical territory.
4.3 Patent Thickets: Volume as Strategy
The patent thicket takes evergreening to its logical extreme. Rather than filing a handful of carefully chosen secondary patents, the thicket strategy involves accumulating dozens or hundreds of patents covering every conceivable aspect of the drug and its use, with overlapping claims and staggered expiration dates. The goal is not necessarily to win every patent dispute. The goal is to make the total cost of litigation so high that no generic or biosimilar manufacturer will attempt to enter the market before the thicket eventually clears.
The economics of this deterrence are stark. Filing and prosecuting each patent in the thicket costs $25,000 to $100,000. Challenging a single patent through an IPR proceeding at the PTAB costs a generic challenger an average of $774,000. Full federal district court litigation of a single patent runs $3 to $10 million per side through trial. When a generic must simultaneously challenge 20 or 30 patents, the financial exposure to the challenger exceeds what many companies can sustain, particularly smaller generic manufacturers without deep balance sheets.
The thicket strategy’s legitimacy was affirmed by a US District Court ruling in 2021 in the antitrust challenge to AbbVie’s Humira thicket. The court held that accumulating a large number of patents, even weak ones, does not itself constitute an antitrust violation. The focus of antitrust analysis remains specific anticompetitive conduct in enforcing those patents, such as sham litigation or objectively baseless infringement claims. Pure accumulation is legal.
Section 5: Case Study: AbbVie’s Humira, the Archetypal Patent Thicket
5.1 The Asset: Humira’s Commercial Scale
Adalimumab, marketed as Humira, is a fully human monoclonal antibody targeting tumor necrosis factor alpha (TNF-alpha). It received FDA approval in 2002 for rheumatoid arthritis and subsequently gained approval across 15 additional indications including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, and plaque psoriasis. By 2022, Humira was generating over $21 billion in global annual revenue, making it the best-selling drug in history by cumulative revenue. It accounted for over 35 percent of AbbVie’s total revenue.
The IP estate protecting Humira’s US market position is the most studied patent thicket in the pharmaceutical industry.
5.2 The Thicket Architecture: 247 Applications, 73 Granted Patents, 14 Distinct Inventions
Humira’s primary composition of matter patent was filed in the 1990s and expired in December 2016. AbbVie filed a total of 247 US patent applications related to adalimumab. The critical statistical fact is that 89 percent of those applications were filed after the drug received FDA approval in 2002, meaning they are explicitly secondary patents, not foundational discovery patents.
From those 247 applications, AbbVie obtained 73 granted US patents. An independent analysis by academic researchers and I-MAK found that 80 percent of those 73 patents were “non-patentably distinct” from each other, meaning they did not represent separate inventions. The 73 patents effectively covered only 14 genuinely distinct inventions, duplicated across the thicket through a mechanism called terminal disclaimers. A terminal disclaimer links the expiration date of a later-filed patent to an earlier one, allowing a company to overcome the USPTO’s “obviousness-type double patenting” rejection by agreeing that both patents will expire together. The practical effect is that multiple granted patents cover the same underlying invention while each presents a separate litigation obstacle.
The patents in Humira’s thicket cover adalimumab formulations (the citrate-free, high-concentration formulation launched in 2016 as a pre-filled syringe), methods of treatment for specific indications, dosing regimens, manufacturing processes, and the antibody’s mechanism of interaction with TNF-alpha. Each category carries different expiration dates, with the last patents not expiring until 2034.
5.3 The IP Valuation of the Thicket as a Strategic Asset
The thicket’s economic value is best understood not as the sum of individual patent values but as the option value of US market exclusivity it preserved. European biosimilars launched in October 2018 following the expiration of AbbVie’s primary European patents. US biosimilar entry did not occur until January 2023, after AbbVie negotiated settlement agreements with all major biosimilar developers, granting each a US license effective January 1, 2023, in exchange for agreement not to challenge the thicket patents.
That five-year differential between European and US biosimilar entry translates to roughly $100 billion in cumulative additional US Humira revenue that AbbVie generated during the period when biosimilars were on the market in Europe but not in the US. Even discounting for litigation costs, settlement payments, and the per-unit royalties embedded in the license agreements, the thicket preserved extraordinary economic value.
5.4 The Settlement Strategy: Converting Litigation Risk into Licensed Revenue
AbbVie’s endgame was not to litigate the thicket to the last patent. Its strategy was to use the thicket as leverage to negotiate delayed entry agreements with biosimilar developers, each accepting a 2023 US launch date in exchange for a license rather than spending years in litigation with uncertain outcomes. The companies that settled include Amgen (Amjevita), Samsung Bioepis (Hadlima/Imraldi), Sandoz (Hyrimoz), Pfizer (Abrilada), Mylan (Hulio), and others.
These settlements are legally distinct from the “reverse payment” or “pay-for-delay” settlements that courts have scrutinized under antitrust law in the small-molecule generic context, because they did not involve direct cash payments from AbbVie to the biosimilar developers to delay entry. AbbVie’s leverage came entirely from the cost and uncertainty of patent litigation, not from explicit compensation for delay.
5.5 Key Takeaways and Investment Implications, Humira
The Humira case demonstrates that a patent thicket’s value is asymmetric: its construction cost is trivial relative to the exclusivity value it preserves. The strategic requirement is identifying the window between primary patent expiration and biosimilar readiness, then building a thicket dense enough that rational economic actors choose settlement over litigation. For AbbVie investors, the thicket delivered five years of US monopoly economics on a $20+ billion revenue product, contributing to AbbVie’s ability to fund the Allergan acquisition ($63 billion, 2020) and build Skyrizi (risankizumab) and Rinvoq (upadacitinib) as post-Humira revenue pillars. The template has been studied and partially replicated by other biologic manufacturers facing biosimilar competition.
Section 6: The Defensive Doctrine: Freedom to Operate, Hatch-Waxman, and PTAB
6.1 Freedom to Operate Analysis
Freedom to operate (FTO) analysis is the systematic assessment of whether a company’s proposed product or process infringes a valid, in-force patent held by a third party. It is a prerequisite for any rational R&D investment decision, partnership negotiation, or product launch.
A properly structured FTO analysis begins with a comprehensive search of patent databases in relevant jurisdictions, using both keyword and structure-based search methodologies. For small molecules, structure-based searches using tools like STN, SciFinder, or Reaxys identify patents covering the compound itself and structurally related analogs. For biologics, sequence-based searches identify patents covering related proteins, antibody sequences, and binding epitopes. The search output feeds into a legal opinion assessing whether identified patents are valid, in-force, and whether the company’s specific product or process falls within the scope of the claims.
FTO opinions are not binary. They produce a risk-stratified assessment: red (high infringement risk, blocking patent likely valid and infringed), yellow (material uncertainty, claim construction or validity is contestable), and green (low risk, no blocking patent identified or blocking patents are clearly invalid or not infringed). The appropriate response to each outcome differs. A red-light opinion may require the company to seek a license, redesign the product to fall outside the claim scope, or initiate an IPR challenge to invalidate the blocking patent before launch.
FTO analysis is not a one-time event at project initiation. The patent landscape in any active therapeutic area changes continuously. Competitor filings, patent grants, PTAB decisions, and court rulings all alter the risk profile of a given development program. Best practice is FTO updates at key development milestones: IND filing, end-of-Phase 2, NDA submission, and within 12 months of projected commercial launch.
6.2 The Hatch-Waxman Act: Structural Analysis
The Drug Price Competition and Patent Term Restoration Act of 1984, known as Hatch-Waxman, created the modern framework governing generic drug competition in the United States. Its provisions divide the generic pharmaceutical litigation landscape into a structured sequence that both innovators and generic manufacturers must navigate with precision.
The Act established the Abbreviated New Drug Application (ANDA) pathway, which allows a generic manufacturer to seek FDA approval based on bioequivalence to an already-approved reference listed drug (RLD), without conducting independent clinical trials. The ANDA applicant must certify its position with respect to each patent listed in the Orange Book for the RLD. The four certification options are Paragraph I (patent is not listed), Paragraph II (patent is expired), Paragraph III (generic will not launch until patent expires), and Paragraph IV (patent is invalid, unenforceable, or will not be infringed by the generic).
A Paragraph IV (P-IV) certification is a declaration of challenge. Filing a P-IV ANDA constitutes an “artificial act of infringement” that gives the innovator the immediate right to sue the generic applicant before a single pill is sold. If the innovator files suit within 45 days of receiving notification of the P-IV filing, the FDA is automatically stayed from granting final ANDA approval for 30 months, or until the litigation concludes if sooner. That 30-month window is the operational core of the Hatch-Waxman litigation game: it allows innovators to litigate their most important patents without the existential pressure of competing in the market during the dispute.
The first generic applicant to file a substantially complete ANDA with a P-IV certification receives 180 days of marketing exclusivity upon launch, during which the FDA cannot approve any other generic application for the same drug. This 180-day exclusivity is the economic prize that motivates generic companies to invest in P-IV challenges. It is exclusively valuable: the first generic can price at approximately 80 percent of the brand’s price during the exclusivity period, generating a substantial margin before the market opens to the 10 to 30 additional generic entrants that typically follow.
Patent Term Extension (PTE) under Hatch-Waxman partially compensates innovators for time lost during FDA regulatory review. The PTE can restore up to five years of patent term, capped such that total remaining exclusivity (including the PTE) does not exceed 14 years from the date of NDA approval. The PTE calculation is complex and applies only to one patent per approved drug. Companies must file PTE applications within 60 days of NDA approval, a deadline that is frequently missed by companies unfamiliar with the procedural requirement.
6.3 The PTAB: From “Patent Death Squad” to Nuanced Reality
The Patent Trial and Appeal Board was created by the America Invents Act (AIA) of 2011 to provide a faster, less expensive alternative to district court litigation for challenging issued patents. The two primary proceedings relevant to pharmaceutical patents are Inter Partes Review (IPR), which challenges validity based on prior art patents and publications, and Post-Grant Review (PGR), which allows broader invalidity grounds but must be filed within nine months of patent grant.
PTAB gained its “patent death squad” characterization from early invalidation rates that were dramatically higher than district court rates. Institutional Patent Challengers, hedge funds, and generic manufacturers filed IPR petitions against blockbuster drug patents as both commercial tools and, in some cases, as part of short-selling strategies. The Allergan/Saint Regis Mohawk Tribe patent sovereignty transfer attempt in 2017 (where Allergan transferred dry eye drug Restasis patents to a Native American tribe to exploit tribal sovereign immunity from IPR proceedings) illustrated the creativity, and legal fragility, of strategies designed to circumvent PTAB jurisdiction.
The actual performance data at PTAB for pharmaceutical patents is more nuanced than the “death squad” characterization suggests. Analysis of Orange Book-listed patent challenges at PTAB shows that the overall rate of all challenged claims being invalidated at PTAB (approximately 23 percent) is statistically similar to the rate in district court (approximately 24 percent). The critical variable is patent type. API and composition of matter patents are exceptionally resistant to IPR invalidity: they are almost never fully invalidated in either forum. Method of treatment patents are the most vulnerable category at PTAB, with invalidation rates substantially above average. Formulation patents occupy the middle ground.
This means PTAB’s practical utility for generic challengers is greatest when attacking the secondary patents that form the outer layers of a patent thicket. Stripping the thicket of its formulation and method patents through IPR proceedings narrows the litigation target to the core compound patent, which is easier to evaluate and settle around. This explains why many PTAB proceedings in pharmaceutical IP ultimately accelerate settlement negotiations rather than producing final decisions.
6.4 Orange Book Listing Litigation: A Growing Battleground
A separate line of defensive litigation concerns the accuracy of Orange Book patent listings. Section 505(b)(1) of the FD&C Act requires that listed patents have claims that cover the approved drug. The FDA has historically taken a non-adjudicative approach to Orange Book listings, accepting what innovators submit without independent review of whether listed patents actually meet the listing requirements.
Generic manufacturers have increasingly challenged improper Orange Book listings through “delisting petitions” to the FDA and through private antitrust litigation asserting that improper listings create an anticompetitive 30-month stay. A 2023 Federal Trade Commission policy statement asserted that improperly listed patents are presumptively anticompetitive. This enforcement posture has prompted several delisting controversies, including challenges to AstraZeneca’s Symbicort inhaler device patents and to several biologic combination product device patents listed for drugs including Novo Nordisk’s Ozempic.
For innovators, the Orange Book listing question requires careful pre-launch analysis. The FDA’s 2021 and 2023 guidance documents on Orange Book listing have tightened requirements, particularly for device patents on combination products. A patent on the injection device component of a combination drug product must have claims covering the approved combination product, not just the device in isolation, to qualify for listing.
Section 7: Case Study: Pfizer’s Lipitor, Defensive Lifecycle Management at Scale
7.1 The Asset: $12 Billion and the Approaching Cliff
Atorvastatin, marketed as Lipitor, was the best-selling drug in the world for most of the decade before its patent expiration. At peak, it generated over $12 billion annually for Pfizer, approximately 27 percent of the company’s total revenue. The loss of exclusivity scheduled for November 2011 was, at the time, the largest revenue exposure any single company had ever faced from a patent expiration.
Pfizer knew the cliff was coming for years. Its preparation illustrates what comprehensive defensive lifecycle management looks like when executed with sufficient resources and institutional commitment.
7.2 Pre-Expiry Preparation: The Full Toolkit
Pfizer’s pre-expiry strategy for Lipitor involved simultaneous execution across clinical, legal, commercial, and regulatory dimensions.
On the clinical front, Pfizer conducted over 500 clinical studies involving more than 20,000 patients to broaden Lipitor’s evidence base and expand the drug’s labeled indications and risk reduction claims. This is not just academic science. A richer label with more specific indication carve-outs makes skinny labeling harder for generics and keeps prescribers engaged with brand-specific data.
The pediatric exclusivity strategy was the single most impactful regulatory maneuver. Under the Best Pharmaceuticals for Children Act, a company that conducts FDA-requested pediatric studies for an approved drug receives an additional six months of market exclusivity for the entire product, including all adult indications. Pfizer conducted the required pediatric study in familial hypercholesterolemia and earned a six-month pediatric exclusivity extension for Lipitor. Those six months represented approximately $6 billion in additional revenue at Lipitor’s US run rate.
On the formulation front, Pfizer developed and launched Caduet, a fixed-dose combination of atorvastatin and amlodipine besylate (a calcium channel blocker). Caduet carried its own patent protection and offered prescribers a simplification for patients taking both drugs separately. While Caduet never became a major commercial product in its own right, it served as a vehicle for transitioning atorvastatin prescriptions to a product with a different patent profile.
The legal strategy involved settlement negotiations with Ranbaxy, the first P-IV ANDA filer for atorvastatin, which had been in litigation with Pfizer since 2003. The settlement, ultimately reached in 2008, granted Ranbaxy a US atorvastatin license effective November 30, 2011, the day Lipitor’s patent expired, and separately resolved antitrust exposure from an FDA hold on Ranbaxy’s Indian manufacturing facilities. The settlement delayed the risk of a court ruling that might have accelerated generic entry and gave Pfizer a known, fixed timeline to plan against.
7.3 The 180-Day War: Post-Expiry Market Defense
Pfizer’s post-expiry strategy during the 180-day exclusivity period represents the most sophisticated generic defense campaign in pharmaceutical marketing history.
Pfizer launched an authorized generic version of atorvastatin through Watson Pharmaceuticals on the same day generic atorvastatin entered the market. The authorized generic participates in the 180-day exclusivity alongside Ranbaxy’s generic, ensuring that Pfizer captured a share of the generic revenue stream rather than ceding it entirely. The authorized generic strategy is now standard practice but was relatively novel in Lipitor’s case.
The direct consumer component of the defense was the Lipitor for You program, which offered branded Lipitor to patients for a $4 per month copay. The program bypassed standard insurance reimbursement structures by having Pfizer directly subsidize the copay differential between branded and generic pricing. For insured patients, this made branded Lipitor cost less out-of-pocket than the generic version, which carried a standard generic copay of $10 to $20. The program enrolled over 600,000 patients.
Simultaneously, Pfizer negotiated with major pharmacy benefit managers (PBMs) to maintain formulary coverage for branded Lipitor at parity with generic pricing for a defined period. These negotiations required Pfizer to accept deep rebates, but the market share preservation justified the economics. Pfizer also continued investing in direct-to-consumer advertising for Lipitor through the expiry period, contra the industry standard of winding down DTCA spending as a patent nears expiration.
7.4 Outcome and Key Takeaways
Pfizer retained approximately 30 percent branded market share at the end of the 180-day exclusivity period. For a drug that had just lost patent exclusivity to a commodity generic, this outcome dramatically exceeded analyst expectations. The financial result cushioned Pfizer’s revenue trajectory and demonstrated that aggressive post-expiry commercial defense, rather than passive acceptance of generic displacement, could preserve material value.
The Lipitor defense also illustrates a key portfolio management principle. Defensive lifecycle management requires resourcing commitment proportional to the drug’s commercial significance. Pfizer invested hundreds of millions of dollars in post-expiry Lipitor defense. The return on that investment was measured in billions. Companies that default to standard wind-down protocols for their patent cliff assets leave value on the table.
Investment Strategy Note: When modeling a pharma company’s revenue trajectory through a patent cliff, build a scenario analysis that captures the difference between passive generic entry response and active defense. The spread between these scenarios can be material, particularly for drugs with strong brand recognition, an established patient community, and a formulary position that can be defended through rebate negotiation.
Section 8: The Global Chessboard: US vs. EU Frameworks, SPCs, and Bolar Divergence
8.1 The Principle of Territoriality and Its Strategic Consequences
A US patent generates no rights in Europe, Japan, China, or any other territory. A company that builds a strong US patent estate but neglects Europe exits the world’s second-largest pharmaceutical market six months earlier than necessary for every drug it sells. Patent rights are national, which means global pharmaceutical patent strategy requires territory-by-territory execution and territory-by-territory monitoring.
The strategic complexity is that patentability standards, claim scope interpretation, enforcement speed, and the relationship between patents and regulatory approval differ significantly across jurisdictions. A patent on a specific polymorph that would be granted in the US may be rejected in India under Section 3(d). A method of treatment patent that is valid and enforceable in the US may not be listable in a patent linkage registry in a European country that does not maintain one. An evergreening strategy that delays generic entry in the US for three years may fail entirely in Australia, where the Therapeutic Goods Administration does not maintain an equivalent to the Orange Book.
8.2 Supplementary Protection Certificates in Europe: The SPC as a Sui Generis Right
The European equivalent of the US Patent Term Extension is the Supplementary Protection Certificate (SPC). An SPC is not an extension of the patent itself but a separate intellectual property right that activates the day after the underlying patent expires. Its purpose is identical to the US PTE: compensating the patentee for effective patent term consumed by the regulatory review and clinical trial process required to obtain a marketing authorization.
An SPC extends protection by up to five years. A six-month pediatric extension is available for drugs that have completed a Pediatric Investigation Plan (PIP) approved by the European Medicines Agency. The total maximum protection from an SPC, including the pediatric extension, is five and a half years beyond the underlying patent’s expiration.
SPCs are national rights administered by each EU member state’s national patent office. The legal requirements for grant are governed by EU Regulation 469/2009, but national interpretations of that regulation have diverged significantly, producing a fragmented landscape where the same product can obtain an SPC in one country and be refused in another. The Court of Justice of the European Union (CJEU) has issued multiple preliminary rulings to harmonize SPC interpretation, including Neurim Pharmaceuticals (C-130/11), Teva UK (C-121/17), and Royalty Pharma (C-650/17), each resolving specific disputes about what products and product combinations qualify for SPC protection. These rulings affect the SPC eligibility analysis for every new SPC application and warrant careful monitoring by European IP teams.
The European Commission’s 2023 Pharmaceutical Reform Package includes a proposed revision to the SPC regulation that would introduce a centralized SPC procedure under the Unitary Patent system and potentially modify SPC eligibility criteria. The reform is still in legislative process as of the date of this publication. IP teams managing European assets should track its progress, as the reform could materially alter SPC filing strategy for products currently in development.
8.3 The Bolar Exemption in Europe: Lack of Harmonization as Both Risk and Opportunity
The Bolar exemption, codified in Article 10(6) of Directive 2001/83/EC, permits generic and biosimilar manufacturers to conduct studies and trials needed for regulatory approval without those activities constituting patent infringement, even while the innovator’s patent or SPC remains in force. The explicit policy objective is to allow generic manufacturers to be ready for immediate market launch the day after exclusivity expires.
The critical problem for innovators in Europe is the absence of harmonized national implementation. EU member states have implemented the Bolar exemption with substantially different scope. Italy has adopted one of the broadest implementations, permitting Bolar activities in support of regulatory filings in countries outside the EU and extending the exemption to innovative drug development, not just generics. Germany, the Netherlands, and France have implemented more moderate versions. Some member states maintain strict readings that limit the exemption to activities directly supporting EU marketing authorization applications for generic products.
This divergence creates both risk and opportunity. Generic manufacturers can conduct Bolar-exempt development activities in Italy to support global regulatory filings, including US ANDA submissions, potentially accelerating their global launch readiness. Innovators should understand which member states offer the broadest exemptions and factor that into their competitive intelligence on generic manufacturers’ likely development timelines.
The European Commission’s 2023 Pharmaceutical Reform Package includes a proposal to harmonize Bolar exemption scope across the EU, specifically to confirm that Bolar activities in support of non-EU regulatory submissions are covered. If adopted, this harmonization would formalize Italy’s broader approach across the EU, affecting innovators’ ability to use manufacturing injunctions in other member states to slow generic development programs targeting markets outside Europe.
8.4 India’s Section 3(d): A Different Standard for Patentability
The Novartis v. Union of India decision by the Indian Supreme Court in 2013 is the most cited case illustrating the divergence between Indian and Western patent standards for pharmaceutical products. Novartis sought a patent in India for the beta-crystalline form of imatinib mesylate, the active ingredient in its leukemia drug Gleevec (Glivec in some markets). The Court rejected the patent application under Section 3(d) of India’s Patents Act, which excludes from patentability a new form of a known substance unless it demonstrates significantly enhanced efficacy compared to the known substance.
The Court held that improved bioavailability of the crystalline form over the free base did not constitute the “enhanced efficacy” required by Section 3(d), which the Court read to mean enhanced therapeutic efficacy, not merely better physical properties. This interpretation effectively bars most standard evergreening strategies, including polymorph patents, from obtaining Indian patent protection unless the applicant can demonstrate superior clinical outcomes.
India is not an isolated case. Brazil’s National Institute of Industrial Property (INPI) has applied similar standards under Article 10 of Brazil’s Industrial Property Law. Argentina, Thailand, and several other developing-country jurisdictions have enacted or enforced analogous provisions. For companies building global patent strategies, these markets require a separate analysis of which secondary patents are likely to be granted and enforced, rather than assuming that a US or European patent strategy will translate globally.
Section 9: Pricing Power, Public Backlash, and the Limits of Patent Rights
Patents grant the legal power to exclude competition and set price without direct competitive constraint. They do not insulate companies from payer negotiating power, regulatory intervention, public opinion, or congressional action. The history of pharmaceutical pricing controversies since 2014 illustrates that the social contract underlying the patent system can fracture when companies are perceived to have exercised pricing power beyond what the innovation justifies.
The Inflation Reduction Act (IRA) of 2022 represents the most direct structural challenge to the pharmaceutical patent system’s pricing assumptions in the US since Hatch-Waxman. The IRA grants Medicare the authority to directly negotiate prices for a defined set of high-expenditure drugs that lack generic or biosimilar competition. The first ten drugs subject to negotiation were announced in August 2023, with negotiated prices taking effect in 2026. Drugs facing IRA price negotiation include Eliquis, Jardiance (empagliflozin), Xarelto (rivaroxaban), and Januvia (sitagliptin).
The IRA’s impact on patent strategy is still developing. A drug that loses exclusivity and faces generic entry is removed from negotiation eligibility, which could in theory reduce companies’ incentive to pursue aggressive lifecycle extension through secondary patents that delay generic entry. If generic entry occurs sooner, the drug exits the IRA’s scope sooner, avoiding years of administratively set pricing. That analysis is product-specific and requires modeling the comparative economics of LCM revenue versus IRA pricing for each affected drug.
Separately, the political and reputational risks of aggressive patent strategies have increased. Congressional oversight of pharmaceutical patent thickets, triggered by the Humira case, produced multiple proposed bills targeting terminally disclaimed patents, pay-for-delay settlements, and improper Orange Book listings. None has yet been enacted into law, but the legislative attention has changed the risk calculus for companies designing LCM strategies that could generate adverse headlines.
Section 10: Case Study: Gilead’s Sovaldi, When Value-Based Pricing Meets Mass-Market Disease
10.1 The Asset: A Curative Therapy at Scale
Sofosbuvir, approved by the FDA as Sovaldi in December 2013, was a nucleotide prodrug inhibitor of the NS5B polymerase of the hepatitis C virus (HCV). It achieved sustained virologic response (SVR), which is the clinical definition of cure, in over 90 percent of HCV-infected patients when combined with ribavirin or other direct-acting antivirals, in a 12-week all-oral treatment course. The prior standard of care, pegylated interferon plus ribavirin, required 24 to 48 weeks of treatment, generated substantial adverse effects, and achieved SVR rates well below 60 percent in difficult-to-treat genotypes.
Gilead Sciences acquired sofosbuvir through its $11.2 billion acquisition of Pharmasset in 2011. The acquisition was the highest-priced pharmaceutical M&A deal in history at the time, priced at a 89 percent premium to Pharmasset’s pre-announcement trading price, and was derided by some analysts as a dramatically overpriced bet on unproven science. It turned out to be one of the most financially successful pharmaceutical acquisitions ever executed.
10.2 The Pricing Decision and Its Consequences
Gilead launched Sovaldi at $84,000 for a 12-week treatment course, or $1,000 per pill. The pricing was explicitly value-based: Gilead’s analysis demonstrated that curing HCV prevented the long-term progression to cirrhosis and hepatocellular carcinoma, conditions that generate lifetime healthcare costs, including the $300,000 to $500,000 cost of liver transplantation, that dwarfed Sovaldi’s price. The economic argument was analytically sound.
The political and commercial reality was different. Approximately 3.2 million Americans were chronically infected with HCV. State Medicaid programs, faced with obligations to treat beneficiaries who had the disease, calculated aggregate drug costs in the hundreds of millions to billions of dollars annually, an impossible budget line for programs already operating under fiscal stress. Several state Medicaid programs responded by implementing treatment restrictions limiting Sovaldi access to patients with advanced fibrosis or cirrhosis (F3-F4 on the Metavir scale), effectively rationing a curative therapy based on disease severity rather than medical indication.
The controversy triggered a Senate Finance Committee investigation, which in 2015 published a report concluding that Gilead’s pricing was based primarily on revenue maximization rather than value analysis or cost recovery. The report’s disclosure of Gilead’s internal pricing deliberations, which explicitly acknowledged the company was pricing at the maximum the market would bear, damaged Gilead’s credibility in subsequent policy discussions.
10.3 The Patent Estate and Its Vulnerabilities
Sovaldi’s IP protection rested primarily on composition of matter and use patents covering sofosbuvir and its prodrug structure. I-MAK filed IPR petitions at the PTAB in 2017 challenging the core sofosbuvir patents on grounds of obviousness, arguing that sofosbuvir was a predictable modification of prior nucleoside analog antiviral chemistry. The PTAB petitions were ultimately withdrawn or procedurally resolved without final written decisions, leaving the patents intact.
The more consequential patent dispute was Gilead v. Merck. Merck’s subsidiary Idenix Pharmaceuticals held patents on nucleoside analogs for HCV treatment. A federal jury in Delaware awarded Merck $2.54 billion in 2016, one of the largest patent verdicts in US history, finding that Gilead’s manufacturing of sofosbuvir infringed Idenix’s patents. The verdict was subsequently complicated by the district court’s finding of inequitable conduct by Merck’s attorneys during patent prosecution, and the case’s ultimate resolution involved years of additional litigation.
10.4 Tiered Pricing and Global Access Licensing
Gilead implemented a tiered pricing strategy for Sovaldi in lower-income markets. The drug was licensed to generic manufacturers in India under non-exclusive licenses for distribution in approximately 100 low- and middle-income countries at prices as low as $900 per treatment course. While this approach expanded access dramatically in developing markets, it also established a global price differential that became a reference point in negotiations with high-income country payers.
In the UK, the National Institute for Health and Care Excellence (NICE) approved Sovaldi’s successor fixed-dose combination, Harvoni (ledipasvir/sofosbuvir), at a confidential discounted price from the list price of $94,500 per course. The NHS’s confidential rebate mechanism allowed NICE to approve the drug for reimbursement while maintaining a list price level that preserved global reference pricing structures.
10.5 Key Takeaways and the IRA Implication
Sovaldi established that a patent monopoly on a curative therapy for a mass-market infectious disease at a population health scale creates a different political and social environment than pricing of a niche oncology drug. The healthcare system absorbs high prices for drugs treating rare diseases because the absolute aggregate expenditure is contained. When a curative drug addresses a condition affecting millions of patients, the aggregate budget impact overwhelms the per-patient value justification.
The IRA’s negotiation framework targets precisely the drugs in Sovaldi’s profile: high expenditure, no generic or biosimilar competition, and significant aggregate Medicare spending. Companies developing curative or highly effective therapies for common chronic diseases must now build IRA negotiation risk into their commercial models from the outset of clinical development, not as an afterthought at launch.
Section 11: Biologics, the BPCIA, and the Purple Book Transparency Gap
11.1 What Makes Biologic IP Different
Biologics are large, complex molecular entities, typically proteins, peptides, monoclonal antibodies, or nucleic acid therapeutics, manufactured in living cells. They differ from small-molecule drugs in every dimension relevant to patent strategy: their molecular complexity, the manufacturing process dependence of their structure, the analytical challenge of demonstrating biosimilarity, and the regulatory framework governing their approval and competition.
A small-molecule generic must be chemically identical to its reference listed drug. A biosimilar must be “highly similar” to its reference biologic with no clinically meaningful differences in safety, purity, and potency. The regulatory standard for biosimilarity is inherently probabilistic and involves comparative analytical, clinical pharmacology, and clinical trial data requirements that add years and hundreds of millions of dollars to development relative to a generic ANDA.
These development economics reshape the competitive dynamics around biologic patent expiration. Generic companies with generic drug development infrastructure are not well-positioned for biosimilar development. The biosimilar competitive set is smaller, more concentrated among large pharmaceutical and biotechnology companies with biologic manufacturing capabilities, and characterized by higher entry costs that limit the number of competitors any given biologic will ultimately face.
11.2 The BPCIA: 12-Year Data Exclusivity and the Patent Dance
The Biologics Price Competition and Innovation Act (BPCIA) of 2009 established the abbreviated approval pathway for biosimilars in the US. Its key features differ materially from the Hatch-Waxman small-molecule framework.
Data exclusivity under the BPCIA lasts 12 years from the date of biologic FDA approval, compared to five years for small-molecule drugs. The longer period reflects the higher cost and complexity of biologic development. An additional four-year period of regulatory data exclusivity (separate from the 12-year period) prevents FDA from accepting a biosimilar application for the first four years after biologic approval. The 12-year exclusivity period is independent of patent status: a biologic without any remaining patent protection still cannot face biosimilar competition until 12 years after its original approval.
The “patent dance” is the BPCIA’s mechanism for identifying and resolving patent disputes. Unlike the Orange Book framework where innovators list patents and generics certify against them in a public and standardized way, the BPCIA’s patent dance involves a confidential exchange of information between the reference product sponsor and the biosimilar applicant. The biosimilar applicant provides its application and manufacturing process information to the innovator. The innovator then identifies patents it believes would be infringed. The parties negotiate which patents to litigate, with unresolved disputes potentially proceeding to court.
The patent dance has been criticized for its opacity and complexity. The Supreme Court’s 2017 ruling in Amgen v. Sandoz clarified that the patent dance is optional for biosimilar applicants, but that innovators retain the right to sue for infringement upon biosimilar launch regardless of whether the dance occurred. The practical consequence is continued uncertainty about the sequence and timing of biologic patent disputes.
11.3 The Purple Book Transparency Gap
The FDA’s “Purple Book,” which lists FDA-approved biological products, has historically provided far less patent information than the Orange Book. An analysis found that only approximately 2 percent of Purple Book entries included patent information, compared to comprehensive Orange Book listings for small molecules. This transparency gap forces biosimilar developers to make investment and manufacturing decisions with substantial uncertainty about which patents they may infringe upon launch.
The BPCIA’s drug product listing requirements for biologics have been strengthened by FDA guidance issued in 2021 and 2022, which requires innovators to list certain patents in the Purple Book that cover approved biological products. The listing requirements are narrower than Orange Book requirements and do not create an automatic stay equivalent to the Orange Book’s 30-month stay mechanism. Biosimilar developers still must conduct comprehensive landscape searches across patent databases and published prosecution histories to identify potential blocking patents.
For innovators, the absence of a mandatory, comprehensive Purple Book patent listing system has strategic implications: patents that are not listed cannot trigger a patent dance or formal pre-launch litigation framework, but they remain enforceable upon biosimilar launch. Companies that rely on unlisted patents as a surprise enforcement tool risk FTC scrutiny under evolving anticompetitive conduct standards.
Section 12: AI-Assisted Drug Discovery and the Inventorship Crisis
12.1 What AI Changes About Drug Discovery IP
Artificial intelligence in drug discovery is not a theoretical future application. It is active, commercially deployed, and generating novel drug candidates that are entering clinical trials. Insilico Medicine developed a fibrosis drug candidate (INS018_055) using AI in 18 months at a fraction of conventional discovery cost. Exscientia has multiple AI-designed compounds in clinical development. Recursion, Schrödinger, and AbSci use computational approaches to design and optimize molecules at a pace and scale that human medicinal chemistry teams cannot match.
The IP implications of AI-assisted discovery center on a fundamental legal requirement: US patent law mandates that inventors be human beings. This requirement was reaffirmed in Thaler v. Vidal (Fed. Cir. 2022), where the Federal Circuit ruled that DABUS, an AI system developed by Stephen Thaler, could not be listed as a patent inventor because the Patent Act’s use of the word “whoever” in 35 U.S.C. § 101 refers exclusively to natural persons.
12.2 USPTO Guidance and the “Significant Contribution” Standard
In February 2024, the USPTO issued guidance on AI-assisted inventions. The guidance confirmed that AI-assisted inventions are patentable, but only if a human being made a “significant contribution” to the conception of each claimed invention. Conception, in patent law, is the complete mental formation of the definite and permanent idea of the complete and operative invention. AI systems that generate molecular structures through algorithmic search are doing something analogous to conception, but they are not thinking in the legal sense.
The USPTO’s guidance attempts to operationalize the significant contribution standard. Human contributions that likely qualify include designing the AI model and training it for a specific inventive purpose, constructing a specific and insightful prompt that directs the AI toward solving a particular technical problem, and critically selecting and refining the AI’s output based on technical judgment. Human activities that likely do not qualify include merely operating standard AI software, using AI as a literature search tool, or following AI output without exercising independent technical judgment.
This standard creates a documentation imperative. Companies using AI in drug discovery need contemporaneous records that capture human inventors’ specific technical contributions at each stage of the AI-assisted process: the selection of the target, the design of the screening protocol, the criteria used to evaluate AI-generated candidates, the modifications made to AI-suggested structures, and the technical reasoning for each human decision. Without this documentation, patent applications may face rejection or post-grant invalidity challenges on inventorship grounds.
12.3 Enablement and Written Description Challenges for AI-Generated Chemical Space
Beyond inventorship, AI-assisted drug discovery creates challenges under the enablement and written description requirements of 35 U.S.C. § 112. Broad Markush claims covering vast chemical spaces are standard in pharmaceutical prosecution, but they must be enabled: the specification must teach a person of ordinary skill in the art how to make and use the full scope of the claimed invention without undue experimentation.
AI discovery systems can efficiently search enormous chemical spaces and identify promising candidates across broad structural categories. But a patent claiming the entire category defined by an AI search may not be enabled if the specification cannot provide sufficient experimental data or predictive guidance across the claim scope. Examiners at the USPTO and EPO are increasingly scrutinizing broad Markush claims in AI-assisted applications under the post-Amgen v. Sanofi (US Supreme Court, 2023) enablement framework, which held that Sanofi and Regeneron’s broadly claimed antibody patents covering an entire functional class of PCSK9 antibodies were not enabled because the specification described only a small fraction of the claimed class.
12.4 Key Takeaways, AI and Drug Discovery IP
Pharmaceutical companies using AI in drug discovery need four things from their IP teams: a documentation protocol that captures human contributions at each stage of AI-assisted work, claim drafting strategies that balance the desire for broad scope against enablement risk under the post-Amgen standard, an inventorship attribution process that can defend against post-grant challenges, and ongoing monitoring of USPTO and EPO examination trends for AI-assisted applications to adapt prosecution strategy as standards develop.
Section 13: Personalized Medicine and Diagnostic Patent Eligibility After Mayo and Myriad
13.1 The Section 101 Problem
The promise of personalized medicine rests on companion diagnostics: tests that identify which patients will respond to a specific therapy, based on genetic, proteomic, or biomarker characteristics. The commercial case for companion diagnostics is straightforward. A drug that works in 60 percent of unselected patients works in 90 percent of biomarker-positive patients. Identifying those patients before treatment improves outcomes, reduces adverse events in non-responders, and justifies premium pricing for the drug-diagnostic combination.
The legal case for patenting companion diagnostics is considerably more complicated following two Supreme Court decisions.
In Mayo Collaborative Services v. Prometheus Laboratories (2012), the Court invalidated patents claiming a method of optimizing treatment with thiopurine drugs by measuring metabolite levels in a patient’s blood and correlating those levels to drug efficacy and toxicity. The Court held that the claims were directed to a law of nature (the relationship between metabolite levels and clinical outcomes) and added only conventional steps (measuring, deciding) that did not transform the natural law into patent-eligible subject matter.
In Association for Molecular Pathology v. Myriad Genetics (2013), the Court held that naturally occurring DNA sequences are products of nature and therefore not patent-eligible, invalidating Myriad’s claims covering isolated BRCA1 and BRCA2 gene sequences. The Court distinguished cDNA (complementary DNA synthetically created from messenger RNA) as patent-eligible because it does not exist naturally.
Together, these decisions have made it very difficult to obtain US patents on diagnostic methods that correlate naturally occurring biomarkers or genetic sequences with disease states or therapeutic outcomes. The Federal Circuit has applied Mayo broadly, invalidating numerous diagnostic patents on Section 101 grounds. The result is a systematic disincentive to invest in diagnostic development: companies that develop novel companion diagnostics may be unable to obtain meaningful US patent protection for the core innovation.
13.2 Prosecution Strategies Under the Post-Mayo Framework
Patent prosecution for diagnostic methods requires drafting claims that go beyond the natural correlation itself and add specific, non-routine steps that the Mayo framework would recognize as inventive. Strategies include: claiming a specific method of performing the diagnostic test using novel reagents or detection technologies (where the novelty lies in the technical means, not the correlation); claiming a treatment method that includes the diagnostic step as an integrated component of a specific therapeutic intervention; and claiming the diagnostic kit or device rather than the diagnostic method.
The last approach has some success. A novel array or next-generation sequencing protocol that implements the diagnostic in a non-obvious way can be claimed as a method of performing the assay, which the USPTO has been more willing to allow than pure correlational claims. The limitation is that these claim types protect the specific technical implementation rather than the underlying diagnostic concept, which competitors can design around with alternative assay technologies.
The American Invents Act’s transitional program for covered business method patents (CBM review at PTAB) has been used to challenge diagnostic patents as well as financial method patents. While the CBM program expired in September 2020 for newly filed petitions, the PTAB’s Section 101 analysis in remaining cases continues to influence how the USPTO examines diagnostic method claims.
13.3 The Strategic Gap for IVD Companies and Pharma Partners
The diagnostic patent eligibility problem creates a misaligned incentive in the development of precision medicine. Pharmaceutical companies need companion diagnostics to demonstrate that their targeted therapies work in biomarker-selected populations, which is increasingly required by FDA for oncology drug approvals. But the in vitro diagnostic (IVD) companies that develop those companion diagnostics cannot reliably patent the diagnostic methods, creating an uncertain return on their investment.
Several commercial structures have emerged to address this gap. Pharmaceutical companies have entered into co-development agreements with IVD companies where the pharma partner funds the diagnostic development in exchange for co-exclusive rights to the diagnostic for use with the specific drug. In this model, the diagnostic is protected not by the IVD company’s own patents but by contractual exclusivity and the pharmaceutical partner’s drug patents, which are far easier to obtain and enforce. The FDA’s requirement for a companion diagnostic as a condition of drug approval creates a de facto exclusivity for the approved companion diagnostic within the context of the approved drug, because physicians prescribing the drug will use the FDA-cleared companion diagnostic test rather than a non-cleared alternative.
Section 14: Investment Strategy: Reading a Portfolio Like a Sell-Side Analyst
14.1 The Patent Estate as a Financial Model Input
Pharmaceutical equity analysis requires treating the patent estate as the primary determinant of revenue duration and quality, not a footnote to the DCF model. The following analytical framework covers the variables that determine whether a patent position is durable or fragile.
Start with the Orange Book. Every approved drug’s Orange Book listing is publicly available at FDA.gov. The listing shows each patent number, expiration date (before any PTE), whether a PTE has been granted, and whether any P-IV certifications have been filed. A drug with five patents listed in the Orange Book and no pending P-IV certifications has a very different risk profile than a drug with two patents listed and three active P-IV litigations.
The PTAB’s patent trial management system (PTAB.usptO.gov) is publicly searchable by patent number. Checking whether Orange Book-listed patents have been the subject of IPR petitions, and if so, whether petitions were instituted and what the outcome was, is basic due diligence that reveals how aggressively the patent position is being challenged and how well it has held up under scrutiny.
FDA’s Paragraph IV certification notification records, published in the Federal Register, provide public disclosure of active ANDA challenges. These records identify which generic companies have filed against a specific branded drug and when the 30-month stay expires (45 days after notification, if the innovator sues within the window). The stay expiration date is the operative market entry date for modeling purposes if the litigation does not resolve in the innovator’s favor.
14.2 Valuing Lifecycle Extension Patents vs. Core Compound Patents
Market consensus often undervalues lifecycle extension patents that are in active litigation. When AstraZeneca’s second-generation cancer drug Tagrisso (osimertinib) faced PTAB challenges to its formulation patents, analysts who modeled only nominal patent expiry dates and ignored the IPR proceedings were building models that did not capture the actual downside scenario. Conversely, analysts who assumed IPR challenges would succeed without examining the specific prior art and the technical strength of the patent claims were also producing inaccurate models.
The forensic work required is: read the IPR petition and identify the prior art arguments, read the patent owner’s response, assess whether the claim construction being argued is the broadest reasonable interpretation or a narrower construction that might survive, and compare the challenged claims to the actual commercial embodiment of the drug (many formulation patent claims cover specific parameters that the commercial product clearly meets, making design-around very difficult).
14.3 Biosimilar Entry Timing: The Key Revenue Inflection Point
For biologic-heavy companies, the biosimilar entry timing model is more complex than the small-molecule generic entry model. Key variables include: the BPCIA 12-year data exclusivity expiration date (the hard floor on biosimilar entry), the patent estate beyond data exclusivity (which could extend effective exclusivity further), the FDA’s biosimilar interchangeability designation status (interchangeable biosimilars can be substituted by pharmacists without prescriber authorization, materially accelerating market penetration), and the number of biosimilar developers that have completed development and received approval (competitive intensity directly correlates with branded price erosion).
Merck’s Keytruda (pembrolizumab) loses US data exclusivity in 2028. The first biosimilar approvals for pembrolizumab are expected in the 2028 to 2030 window. The depth and speed of revenue erosion will depend on how many biosimilar manufacturers enter, whether any achieve interchangeability status, and how aggressively Merck pursues lifecycle management through additional combinations and indications that may carry independent patent protection.
14.4 M&A Premium Attribution to IP Assets
When a pharmaceutical M&A transaction is announced, the premium paid over the target’s unaffected market capitalization reflects the acquirer’s assessment of strategic value. For transactions driven primarily by IP acquisition, decomposing the premium into its components (clinical-stage pipeline assets, marketed drug patent estate, manufacturing capabilities, and commercial infrastructure) reveals the implied value assigned to specific patent positions.
AstraZeneca’s $39 billion acquisition of Alexion Pharmaceuticals in 2021 was driven substantially by Alexion’s complement pathway inhibitor franchise, anchored by Soliris (eculizumab) and its successor Ultomiris (ravulizumab). At the time of acquisition, Soliris was generating approximately $4 billion annually but facing biosimilar competition risk. Ultomiris, with a longer patent runway and a more convenient dosing schedule, represented the lifecycle management pivot. The implied value assigned to Ultomiris’s patent estate and the complement pathway platform determined whether AstraZeneca’s price was rational, and subsequent performance of the franchise has validated the acquisition’s strategic logic.
Section 15: Key Takeaways and Strategic Recommendations
15.1 For IP Teams
Build secondary patent programs into Phase 2 clinical design, not as an afterthought at NDA submission. Every formulation study, every dose-ranging trial, every biomarker analysis generates potential patentable innovations. IP teams embedded in clinical operations capture these innovations contemporaneously. IP teams working from completed clinical study reports often find that the statutory bar has closed, the prosecution window has passed, or the most commercially valuable claims are no longer available.
Orange Book listing accuracy is a legal compliance requirement and a competitive tool. Each patent listed in the Orange Book covers a different commercial embodiment and triggers different defensive rights. A patent that should be listed but is not forfeits the 30-month stay protection for the claims it covers. A patent that is incorrectly listed exposes the company to antitrust risk under the FTC’s evolving enforcement posture.
PTAB vulnerability analysis should be part of every product launch review. Before committing to a product’s exclusivity-dependent revenue forecast, the patent position supporting that forecast should undergo a simulated IPR analysis: what is the best prior art argument a generic company could make, and how well does the patent survive it? Patents that would not survive a rigorous IPR analysis are not reliable revenue protection assets.
15.2 For Portfolio Managers
The patent expiration schedule is a more reliable financial model input than management guidance on pipeline timelines. Orange Book data, PTAB proceedings, and P-IV notification records are public. Build your models on those facts, then stress-test against scenarios where secondary patents fail in PTAB or litigation. The spread between optimistic (all patents hold) and base (secondary patents fail, effective exclusivity ends at first P-IV challenge resolution) revenue scenarios defines the uncertainty band around your pharma equity positions.
Biologic companies with entrenched positions in autoimmune and oncology franchises face a different patent cliff profile than small-molecule companies. The 12-year data exclusivity floor, the manufacturing complexity that limits biosimilar entrant count, and the slower market penetration of biosimilars relative to small-molecule generics all support longer revenue durability assumptions for biologic franchises. Price them accordingly.
15.3 For R&D Leads
Drug discovery programs that use AI tools need patent documentation protocols from day one. The USPTO’s 2024 guidance on AI-assisted inventions places the burden on patent applicants to demonstrate human significant contribution. That burden is easiest to meet with contemporaneous records: lab notebooks, computational run logs, internal communications capturing the technical reasoning behind candidate selection decisions.
Diagnostic development programs for companion diagnostics should engage IP counsel early to identify whether the planned diagnostic method is likely to survive Section 101 scrutiny. If the method is directed to a natural correlation, the prosecution strategy should be built around novel technical implementation claims rather than pure correlational claims. Co-development agreements with drug partners provide an alternative protection mechanism through contractual exclusivity and FDA approval linkage.
15.4 Final Assessment
The pharmaceutical patent portfolio is the center of gravity for everything that matters in the industry: revenue, pipeline, M&A, pricing, and competitive position. Managing it as a static legal artifact is the most expensive mistake a company can make. The companies that consistently outperform their patent cliff exposure, that extract the most value from their portfolios through licensing and partnerships, and that acquire pipeline most efficiently through IP-driven M&A are the ones that treat patent strategy as a real-time operational priority integrated into every major business decision.
The legal frameworks governing pharmaceutical patents, from Hatch-Waxman to the BPCIA to Section 101, are complex, jurisdiction-specific, and continuously evolving. The analytical tools for monitoring and exploiting those frameworks, from PTAB filings to Orange Book data to Purple Book listings, are increasingly accessible and quantitatively rich. Companies that build the institutional capability to extract insight from those tools and translate that insight into IP strategy will operate with a structural advantage over those that rely on external counsel for periodic static analysis.
The patent system offers a time-limited monopoly in exchange for public disclosure of the invention. The companies that win are not those that simply obtain the monopoly. They are those that maximize every day of it.
This analysis is intended for informational purposes for pharmaceutical industry professionals, institutional investors, and IP practitioners. It does not constitute legal advice. Readers should consult qualified legal counsel for guidance on specific patent strategy decisions.