Generic drug companies that win in multiple markets simultaneously share one trait: they treat the regulatory submission itself as a competitive asset, not an administrative function. Filing an ANDA with the FDA while simultaneously progressing a Marketing Authorization Application with the EMA and a Type II variation in Japan demands a level of strategic architecture that most regulatory affairs teams still approach as three separate projects. The ones who do it as one coordinated machine capture first-mover pricing, lock in exclusive windows, and drain competitor resources in litigation before a rival’s dossier clears its first review cycle.
This guide covers the full operational and strategic stack: how to build a global regulatory plan that actually functions across jurisdictions, which harmonization frameworks carry real weight versus bureaucratic theater, where automation creates durable competitive advantage, and how the IP landscape around key molecules directly shapes submission timing and market-entry sequencing decisions.
Part I: Global Regulatory Plan Architecture
Why Most Global Regulatory Plans Fail Before the First Submission
A global regulatory plan (GRP) is not a project timeline with agency logos attached. A real GRP is a living document that integrates IP expiry dates, clinical data ownership, product-specific guidance (PSG) updates from FDA, bioequivalence (BE) study design tolerances, and country-specific post-approval variation categories into a single decision framework. Most regulatory teams produce Gantt charts. The ones who build actual GRPs produce submission sequences that front-load data where it creates IP leverage and defer resource-intensive markets to the back half of the schedule.
The FDA’s Center for Drug Evaluation and Research (CDER) publishes PSGs for roughly 1,700 drug products, and each PSG is a direct map of what the agency considers scientifically adequate for an ANDA approval. Ignoring a PSG and substituting your own BE study design is how companies accumulate Complete Response Letters (CRLs) at a rate that destroys project economics. As of early 2026, approximately 60% of ANDA CRLs cite BE deficiencies that a current PSG would have prevented.
The EMA operates differently. Rather than a product-specific database of guidance documents, the EMA’s scientific guideline system routes generic applicants through therapeutic area-specific guidelines, with biowaiver provisions for lower-risk BCS Class I and III compounds codified under Guideline EMA/CHMP/QWP/49313/2010. These two frameworks, FDA PSG and EMA BCS biowaiver doctrine, are not the same standard applied to the same compounds, and treating them interchangeably is how a multi-market GRP accumulates preventable divergences between dossiers.
Building the Submission Sequence Around IP, Not Convenience
The sequence in which you file across markets should be dictated by patent expiry dates, Paragraph IV certification windows, and country-specific data exclusivity periods, not by which regulatory package is cheapest to prepare first.
Take the example of linagliptin (Boehringer Ingelheim’s Tradjenta). The compound’s FDA Orange Book patent listings included a use patent (US8883835) covering specific dosing methods that remained in force years after the compound patent expired. Generic filers who sequenced their ANDA Paragraph IV certifications against the compound patent without simultaneously building a design-around strategy for the method-of-use patent faced 30-month statutory stays that neutralized their first-to-file advantage. The IP analysis has to precede the GRP schedule, not run parallel to it.
For ANDA filers, the 180-day exclusivity window awarded to the first Paragraph IV filer against each Orange Book patent is the single most valuable piece of temporary IP in the small-molecule generic space. FDA data from 2023 shows that a generic product capturing 180-day exclusivity in a high-volume therapeutic category generates peak-year revenues between $150M and $400M, compared to $30-$80M for the same product entering a fully open generic market. Structuring the GRP to optimize Paragraph IV timing is not a legal afterthought; it is the primary economic variable the plan should solve for.
In the EU, data exclusivity under Article 10 of Directive 2001/83/EC runs for eight years of data protection from original marketing authorization, followed by two years of market exclusivity, with a possible one-year extension for a new therapeutic indication. For reference compounds first authorized in a major EU member state, this 8+2+1 framework determines when a generic applicant can file a marketing authorization application (MAA) under the abbreviated procedure. Mapping these windows for 10-15 target molecules simultaneously requires purpose-built IP analytics, not a spreadsheet.
Cross-Functional GRP Governance
The GRP requires joint ownership between regulatory affairs, IP/legal, clinical development, commercial, and supply chain at the governance level. That is not a standard operating procedure; it is an organizational design choice with cost implications. Companies that run regulatory as a downstream recipient of commercial and IP decisions consistently produce GRPs that need reworking when a Paragraph IV certification triggers unexpected litigation or when a PSG update invalidates a planned BE study design.
The FDA pre-ANDA meeting program (available under GDUFA II) gives complex generic applicants a direct channel to clarify BE study methodology, complex product characterization requirements, and CMC expectations before a dossier is drafted. Companies that use pre-ANDA meetings systematically, across their entire development portfolio rather than for individual products in crisis, reduce first-cycle approval rates well below the industry average of approximately 60% for complex generics. The FDA’s MAPP 5200.21 formalized a structured pre-ANDA meeting agenda, and working fluently within that structure means arriving with specific questions, not vague requests for feedback.
Key Takeaways: Global Regulatory Plan Architecture
The GRP must be patent-sequenced, not convenience-sequenced. ANDA Paragraph IV timing against individual Orange Book entries determines whether a first-to-file advantage translates into 180-day exclusivity revenue or a 30-month stay that resets the economics entirely. Pre-ANDA meetings are a systematic planning tool, not a crisis intervention. And the FDA PSG database and EMA BCS biowaiver doctrine are distinct frameworks that produce different dossier requirements for the same compound.
Part II: Harmonization Frameworks That Actually Matter
ICH Guidelines: Where the Real Leverage Is
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) produces guidelines that most regulatory teams know by name and fewer actually exploit by design. ICH Q1 (stability testing), Q6A (specifications for new drug substances and drug products), M4 (Common Technical Document structure), and E6 (Good Clinical Practice) are not interchangeable across agencies in their implementation, but they create enough common architecture that a strategically assembled dossier module can satisfy FDA, EMA, Health Canada, TGA (Australia), and Swissmedic with jurisdiction-specific adaptations rather than complete rewrites.
ICH M2 defines the eCTD (Electronic Common Technical Document) format. eCTD compliance is mandatory for FDA NDAs, ANDAs, and BLAs as of May 2018, mandatory for EMA procedures, and is the accepted format for Health Canada, TGA, and most high-income markets. Filing in eCTD from the outset, with a well-structured backbone that allows regional Module 1 (administrative) sections to be swapped without disturbing the scientific content in Modules 2-5, compresses multi-market submission timelines by eliminating reformatting cycles late in the process.
What the harmonization literature underplays is the divergence between ICH guidance and agency-specific implementation. FDA’s clinical pharmacology requirements for complex oral dosage forms routinely exceed ICH M12 biopharmaceutics guidance. EMA’s guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CPMP/EWP/280/96 Rev1) imposes food-effect study requirements and dose-dumping assessment standards that FDA’s equivalent guidance does not mirror exactly. Companies filing a modified-release generic simultaneously in both markets without running parallel BE study arms to satisfy both agencies’ specific standards commonly find that the FDA-qualified study design fails to generate the in vivo data profile EMA requires, necessitating a separate study and adding 9-18 months to the EU timeline.
Mutual Recognition Agreements and Their Practical Limits
Mutual Recognition Agreements (MRAs) for GMP inspections cover the FDA-EU MRA (operational since November 2019 for EMA member states), the FDA-Canada MRA, and several bilateral arrangements with Australia, Japan, Singapore, and Switzerland under the PIC/S framework. What MRAs do in practice is allow the FDA to accept EMA member state GMP inspection reports for qualifying facilities in lieu of conducting its own inspection, and vice versa. This matters because FDA foreign inspections carry a 18-24 month scheduling backlog in some therapeutic categories, and a manufacturing site relying on FDA pre-approval inspection clearance before a first-cycle ANDA approval is looking at timeline slippage that MRA coverage can eliminate.
The limits are specific. MRAs do not cover all facility types. Biologics manufacturing sites, sterile fill-finish facilities for complex injectables, and active pharmaceutical ingredient (API) manufacturers in countries outside the agreement (notably China and India, which supply the majority of small-molecule generic APIs) are not covered. An ANDA that relies on an API sourced from a Chinese manufacturer not previously inspected by FDA may wait 18+ months for a foreign drug manufacturing inspection if the facility has not been inspected within the preceding compliance cycle. Building a GRP without explicit foreign inspection risk-mapping for every API and finished dosage form manufacturer in the supply chain is how approvals slip from 18 months to 36.
WHO Prequalification as a Market-Access Multiplier
WHO Prequalification (WHO-PQ) is the regulatory approval pathway that unlocks procurement by UNICEF, PEPFAR, the Global Fund, and dozens of national health authorities in low- and middle-income countries (LMICs). For companies with a therapeutic portfolio that includes essential medicines (antiretrovirals, antimalarials, antibiotics, insulin analogs), WHO-PQ approval is worth including in the GRP as a distinct workstream rather than an afterthought.
The WHO-PQ submission standard has converged significantly with FDA and EMA requirements over the past decade. A dossier assembled for FDA ANDA and adapted for EMA via the centralized procedure can be further adapted for WHO-PQ with approximately 20-30% incremental documentation effort, primarily around labeling, stability data to ICH zone IVa/IVb conditions, and site master file formatting. Companies like Cipla, Aurobindo Pharma, and Mylan (now Viatris) have built WHO-PQ into their standard GRP template for essential medicines because the incremental revenue from LMIC tender procurement is highly predictable and lightly competed.
Key Takeaways: Harmonization Frameworks
ICH eCTD compliance structured at the outset, with modular Module 1 compartmentalization, is the single-highest-return documentation investment in a multi-market GRP. MRAs provide real GMP inspection relief in US-EU routes but do not cover the China and India API supply chains where inspection backlog risk is highest. WHO-PQ deserves a dedicated GRP workstream for essential medicines portfolios because the marginal documentation cost is low and the LMIC procurement revenue is durable.
Part III: The IP Valuation Architecture Underneath Every Generic Filing
How Patent Expiry Schedules Drive Submission Economics
The market value of a generic drug’s regulatory approval is not a function of the dossier’s technical quality. It is a function of timing relative to the reference listed drug’s (RLD’s) patent expiry schedule and the number of Paragraph IV certifications filed against the same Orange Book entries. A first-to-file Paragraph IV ANDA against a product with a single, unchallenged compound patent and $2B in annual US sales is worth fundamentally different economics than the same filing against a product ringed by 12 Orange Book patents, four of which cover formulation methods that survived a prior inter partes review (IPR) challenge.
The FDA Orange Book lists patents by category: drug substance (DS), drug product (DP), and method of use (MoU). DS patents are the primary expiry clock. DP patents covering formulation technology, crystalline polymorphs, or particle size distributions routinely extend effective exclusivity 3-7 years past the DS patent expiry, which is where the evergreening phenomenon generates real litigation economics. MoU patents are the most complex for generic filers because a Section viii carve-out of the patented indication from the ANDA label is a valid alternative to a Paragraph IV certification, but carve-outs narrow the commercial opportunity and can create off-label prescribing dynamics that are difficult to predict.
Vyvanse (lisdexamfetamine dimesylate, Takeda/Shire) illustrates how layered Orange Book listings translate into submission economics. The compound patent covering lisdexamfetamine expired in 2023, but Shire held DP patents covering the abuse-deterrent prodrug mechanism that multiple Paragraph IV filers challenged over a 5-year litigation cycle. Teva Pharmaceutical, Amneal Pharmaceuticals, and Impax Laboratories (now part of Ampio/Amneal) each navigated different patent claim constructions in separate District Court proceedings before generic market entry was fully cleared. The Hatch-Waxman 30-month stay triggered by Shire’s infringement suit against first-to-file challengers ran through mid-2023 in several cases, and the litigation settlement terms in those cases, which have become standard Hatch-Waxman settlement practice, included authorized generic supply agreements that diluted the 180-day exclusivity economic value for challengers.
IP Valuation Methodology for Generic Portfolio Planning
Quantifying the IP asset value of a generic development candidate requires four inputs: the probability-weighted time to market entry (accounting for both regulatory review timelines and litigation duration), peak-year revenue in an open generic market, the expected number of generic entrants at day one of open competition, and the probability of capturing 180-day exclusivity.
For high-value reference products, the standard discounted cash flow (DCF) approach applies a risk-adjusted net present value (rNPV) to the generic entry. A product with $1.5B in US branded sales, a single DS patent expiring in 30 months, and no DP or MoU Orange Book listings presents a relatively clean rNPV profile. A product with the same branded sales but six Orange Book listings, two of which cover recently issued continuation patents claiming specific crystalline forms, requires a Scenario Tree that models litigation outcomes, IPR petition success probabilities (using PTAB grant and invalidation base rates by technology class), and the probability of a carve-out strategy succeeding commercially before first-cycle rNPV is credible.
PTAB grant rates for petitions against pharmaceutical patents have averaged around 60-65% for institution decisions over the trailing five years, with invalidity findings in Final Written Decisions running roughly 40-55% of all instituted reviews depending on patent vintage and claim breadth. For DP patents covering formulation technology filed post-2010, the invalidity rate at PTAB is somewhat lower because claim drafters have become more precise. Generic filers and their IP counsel who build litigation probability distributions from PTAB outcome data, rather than using flat assumptions, produce materially more accurate rNPV models for Paragraph IV programs.
Evergreening Tactics and the Defender’s Technology Roadmap
Brand companies extend effective exclusivity through a sequence of IP maneuvers that generic regulatory teams need to model explicitly. The standard evergreening toolkit runs from new polymorph patents (covering crystalline forms of the active pharmaceutical ingredient discovered during formulation development), to new composition-of-matter patents on prodrug formulations (as in Vyvanse), to method-of-treatment patents covering specific patient populations or dosing titration schedules, to pediatric exclusivity awards under PREA or BPCA that tack an additional six months onto any remaining patent or exclusivity period.
Humira (adalimumab, AbbVie) is the canonical evergreening case for biologics. AbbVie built a patent thicket exceeding 250 granted US patents around adalimumab, covering formulation, manufacturing processes, dosing regimens, prefilled syringe delivery devices, and concentration-specific claims. The result was that biosimilar entry in the US was delayed until 2023 despite European biosimilar competition beginning in October 2018, a five-year gap attributable in large part to settlement agreements with biosimilar developers that delayed US launch in exchange for European entry rights. The commercial cost of that delay to the US healthcare system runs into the tens of billions in foregone savings.
For small-molecule generics, the evergreening roadmap is somewhat more constrained because the FDA’s Orange Book listing criteria for method-of-use patents have been contested. The FTC’s 2023 report on pharmaceutical patent settlements and its broader Orange Book delisting challenge program, in which the FTC formally contested more than 100 Orange Book patent listings between 2023 and 2025 as potentially improper, has introduced a new layer of tactical uncertainty for brand companies building defensive patent portfolios and a new layer of opportunity for generic developers who can cross-reference FTC challenge targets against their own Paragraph IV pipelines.
Investment Strategy: IP-Driven Generic Portfolio Construction
Portfolio managers evaluating generic drug companies need three metrics that most sell-side research underweights: the ratio of Paragraph IV certifications to total ANDA filings (a higher ratio indicates a company systematically targeting first-mover positions rather than me-too commodity generics), the litigation win rate in Hatch-Waxman proceedings over the prior five years (a proxy for IP counsel quality and early-stage patent analysis rigor), and the concentration of ANDA approvals by therapeutic category relative to market size (category concentration in a contracting generic pricing environment, such as oral solid dosage forms in established primary care categories, carries margin compression risk that category diversification into injectables, ophthalmics, or transdermal platforms partially offsets).
Companies like Hikma Pharmaceuticals, which has systematically built its US injectables ANDA portfolio through both organic filings and acquisitions, and Amneal Pharmaceuticals, which has used Paragraph IV certifications in CNS and respiratory categories to drive 180-day exclusivity capture, illustrate what a disciplined IP-sequenced portfolio strategy looks like at the organizational level.
Part IV: The ANDA Submission Technical Stack
Building a Deficiency-Resistant ANDA
The FDA’s ANDA review process under GDUFA II operates on a two-tiered review goal: 10-month review for standard applications and 8-month review for priority applications, measured from the receipt date. The gap between goal and actual median review time has historically been 6-12 months wider than goal dates due to information request (IR) cycles, and the single largest driver of IR issuance is CMC deficiencies, specifically dissolution method inadequacy, container closure system incomplete characterization, and API reference standard documentation gaps.
FDA CDER’s Office of Pharmaceutical Quality (OPQ) publishes Quality Assessment metrics annually. In fiscal year 2024, approximately 48% of ANDAs received at least one IR during the review cycle, with CMC-related IRs comprising the majority. The FDA’s Good ANDA Assessment Practices (GAAP) program, which released an updated guidance in 2022, identifies the specific CMC elements most frequently cited in IRs: stability data incompleteness (particularly at accelerated conditions for ANDAs filed without all required timepoints), comparative dissolution profile documentation for modified-release products, and elemental impurity testing under ICH Q3D.
A deficiency-resistant ANDA requires a pre-submission quality gate that maps every required data element against the current PSG and the most recent GAAP guidance before the application is assembled. This is not standard practice at most generic companies; it requires a dedicated pre-submission review function that operates independently of the authoring team. Companies that have implemented structured pre-submission quality gates report first-cycle approval rates of 70-80%, compared to an industry average closer to 55-60%.
Bioequivalence Study Design Precision
BE study design is where most regulatory timeline slippage originates. An inadequate BE design identified post-study requires protocol amendment, IRB resubmission, subject recruitment restart, and study execution before a CRL can be resolved, adding 12-18 months minimum to an ANDA timeline. Getting BE design right before IND-equivalent pre-clinical work begins is the highest-leverage point in the entire ANDA development cycle.
The FDA’s BE study design requirements for complex oral dosage forms, inhaled products, topical dermatologics, and ophthalmic solutions differ substantially from the standard two-period, two-sequence crossover design adequate for conventional oral immediate-release products. For orally inhaled drug products (OIDPs), FDA’s draft guidance on BE testing for dry powder inhalers (DPIs) and metered dose inhalers (MDIs) requires pharmacokinetic (PK) studies and in vitro aerosol performance characterization that together constitute a multi-study, multi-site program often running 24-36 months. For topical dermatologics, FDA’s BE recommendations increasingly rely on pharmacodynamic (PD) methods, tape stripping methods, or comparative clinical endpoint studies, none of which are interchangeable methodologically and each of which carries different statistical power requirements.
Spiriva Respimat (tiotropium, Boehringer Ingelheim) illustrates OIDP complexity in practice. The Respimat inhaler device is subject to device-specific claims that are separately Orange Book-listed from the drug compound claims. Generic filers have had to navigate both the BE study complexity for tiotropium and the IP question of whether a generic delivered via a different device platform constitutes a carve-out from the device-specific Orange Book listings or a Paragraph IV challenge. The first generic approval for tiotropium HandiHaler (an earlier delivery platform) came via Mylan in 2021, with the Respimat formulation following under a distinct regulatory and IP pathway.
CMC Documentation: What FDA Actually Reviews First
CDER’s OPQ filing review checklist, which is publicly available in the MAPP 5200.14 series, reveals where reviewers spend the most time at the filing acceptance stage: drug substance specification completeness (acceptance criteria for identity, assay, impurities, and residual solvents under ICH Q3C), drug product specification alignment with the dosage form guidance, manufacturing process description adequacy, and container closure system description with extractables/leachables data where applicable.
For ANDAs relying on a Type II API Drug Master File (DMF), the DMF must be in current eCTD format and must have been assessed as acceptable for reference by FDA before the ANDA can advance past filing review. DMF deficiency letters issued by the DMF staff are not automatically transmitted to the ANDA applicant; the ANDA holder must proactively monitor DMF assessment status via FDA’s DMF status portal and coordinate with the API supplier to resolve deficiencies on a timeline that does not create rate-limiting delays for the ANDA review.
This coordination requirement has real business implications. Generic companies that source APIs from suppliers with multiple open DMF deficiency letters carry latent approval risk that does not appear in a standard milestone schedule. Due diligence processes for API supplier qualification need to include DMF status review as a standard input, particularly for suppliers serving multiple ANDA programs simultaneously.
Key Takeaways: ANDA Technical Stack
First-cycle approval rates track directly with pre-submission quality gate discipline, particularly around CMC completeness against PSG and GAAP frameworks. BE study design for complex dosage forms requires expert-level guidance during protocol development, not post-study correction. DMF status monitoring for API suppliers is an active approval risk management function, not a passive vendor management task.
Part V: EMA Marketing Authorization Application Strategy
Procedure Selection and Its Consequences
The EMA offers four procedures for generic marketing authorization: the centralized procedure (CP), the decentralized procedure (DCP), the mutual recognition procedure (MRP), and the national procedure. For a generic applicant seeking approval in multiple EU member states simultaneously, the DCP is the standard route. It designates one Reference Member State (RMS) to conduct the primary assessment and allows Concerned Member States (CMSs) to accept or object to the RMS opinion within a 90-day recognition phase.
Procedure selection has dossier design consequences that are not reversible mid-process. The DCP runs on a strict Day 70/Day 120/Day 180 Day 210 timetable, with clock-stops for applicant responses to List of Outstanding Issues (LOIs). An applicant that misses the Day 70 response window or provides an inadequate response to a major objection faces a clock restart that extends the DCP timeline by months, not weeks. The EMA’s target for DCP completion (including recognition phase) is approximately 180 days from Day 1, but applications with major objections routinely run 270-360 days from start to opinion.
RMS selection is a strategic choice. Some EU member states have pharmaceutical assessment agencies with specific therapeutic area expertise and more predictable LOI practices than others. Netherlands Medicines Evaluation Board (MEB), the Medicines and Healthcare products Regulatory Agency (MHRA, now operating outside EU procedures post-Brexit), Denmark’s DKMA, and Germany’s BfArM are well-established RMS choices with qualified assessor pools. Selecting an RMS without assessing its assessor backlog, its LOI issuance rate for the relevant dosage form category, and its historical clock-stop frequency is where DCP timelines become unpredictable.
EU Bioequivalence Requirements Versus FDA: The Key Divergences
The EMA Guideline on the Investigation of Bioequivalence (EMA/CPMP/EWP/QWP/1401/98 Rev. 1) is the operative standard for most generic MAAs in the EU. The 90% confidence interval criterion for Cmax and AUC bioequivalence is the same as FDA’s (80.00-125.00% for standard products), but the implementation details diverge in ways that affect study design and sample size.
For highly variable drug substances (HVDS), EMA permits reference-scaled average bioequivalence (RSABE) with a widened acceptance criterion, scaled to the reference product’s within-subject variability, provided specific conditions are met including a minimum intra-subject CV of 30% for the reference. FDA uses a similar RSABE approach for Cmax (not AUC) for narrow therapeutic index drugs under separate criteria. These are not the same framework, and a single BE study designed to FDA RSABE specifications for Cmax will not satisfy EMA’s RSABE criteria unless the protocol prospectively addresses both agencies’ requirements including replicate-design subject numbers.
For BCS-based biowaivers, EMA grants biowaivers for immediate-release solid oral dosage forms containing BCS Class I and III substances under specific solubility, permeability, and dissolution criteria. FDA’s BCS biowaiver guidance (issued 2017) covers Class I and Class III substances but applies additional conditions around excipient identity, particularly for BCS Class III biowaivers where membrane permeability is lower and excipient effects on intestinal transporters are more relevant. The practical effect is that a compound that qualifies for both FDA and EMA biowaivers can often share dissolution data but rarely can use a single biowaiver justification document without jurisdiction-specific tailoring.
Key Takeaways: EMA MAA Strategy
DCP RMS selection should be based on assessor backlog, therapeutic area expertise, and historical clock-stop frequency, not proximity or relationship familiarity. HVDS BE study protocols need to be designed from the outset to satisfy both FDA RSABE and EMA RSABE criteria if both markets are targets, because retrofitting FDA-optimized protocols for EMA requirements typically requires a new study. BCS biowaiver justifications are agency-specific documents that share data but not argumentation structure.
Part VI: Submission Technology and Operational Infrastructure
eCTD Publishing Infrastructure
The eCTD format requires a validated publishing system capable of generating conformant eCTD sequences, managing lifecycle actions (new, replace, append, delete) across submission sequences for the same application, and producing validation-clean submissions for FDA’s Electronic Submissions Gateway (ESG) and EMA’s Common Repository. FDA requires eCTD validation against the FDA Validation Criteria document, which is updated with each technical specification revision; submissions with eCTD validation errors at the ESG are refused without formal receipt, which means a validation failure on a Paragraph IV ANDA means losing the filing date and the first-to-file position.
The technical tools most commonly used in the industry include Lorenz Life Sciences (formerly Liquent InSight), Ennov Clinical (formerly Amplexor Life Sciences), DocuBridge, and Extedo eCTD Manager. The software category has consolidated significantly since 2020 through M&A. The competitive differentiators that matter for high-volume generic filers are multi-regional publishing capability (generating FDA, EMA, Health Canada, and TGA conformant outputs from a single authoring environment), integrated validation against all target agencies’ technical criteria, and automated lifecycle management for post-approval change submissions.
Regulatory information management (RIM) systems, such as Veeva Vault RegulatoryOne or OpenText Documentum for Life Sciences, sit one layer above the eCTD publisher and manage the content management, workflow routing, submission planning, and post-approval lifecycle tracking functions. For a generic company managing 200+ ANDA applications simultaneously across multiple markets, a RIM system is not optional; without structured tracking of submission histories, regulatory correspondence, and outstanding commitment deadlines, the compliance risk associated with missed post-approval reporting deadlines is material.
Automation in Document Authoring and Validation
Robotic process automation (RPA) in regulatory submissions is most mature for metadata tagging, document format validation, submission unit leaf mapping, and automated XML backbone generation. Pharmaceutical regulatory teams at Teva, Mylan/Viatris, and Sun Pharma have reported 40-60% reduction in document preparation cycle time for standard ANDA submissions using RPA tools integrated with RIM systems, primarily by automating the validation checks that previously required manual reviewer time.
Generative AI tools are beginning to enter the document authoring workflow for structured content generation, particularly for Module 2 quality summaries and Module 3 narrative sections. The FDA has not issued formal guidance on the use of AI-generated content in regulatory submissions as of early 2026, but the agency’s 2023 AI/ML action plan for drug development noted that content authenticity and traceability are areas the agency expects applicants to address. Companies using generative AI for regulatory document drafting should establish version-controlled, human-reviewed audit trails that demonstrate the submitted content reflects human scientific judgment, not unreviewed AI output.
Real-Time Submission Tracking and Portfolio Visibility
For regulatory affairs teams managing multi-market portfolios, real-time visibility into submission status across agencies is the operational foundation for timeline management. FDA’s ANDA portal provides applicant-accessible status data including receipt date, review division assignment, outstanding IR status, and tentative/final approval dates. The EMA’s CESP (Common European Submission Portal) provides similar tracking for EU procedures. What neither system provides adequately is cross-portfolio analytics: which submissions are at risk of IR cycles based on historical review patterns, which DMFs are in open deficiency status for referenced applications, and which post-approval commitment deadlines are approaching across the portfolio simultaneously.
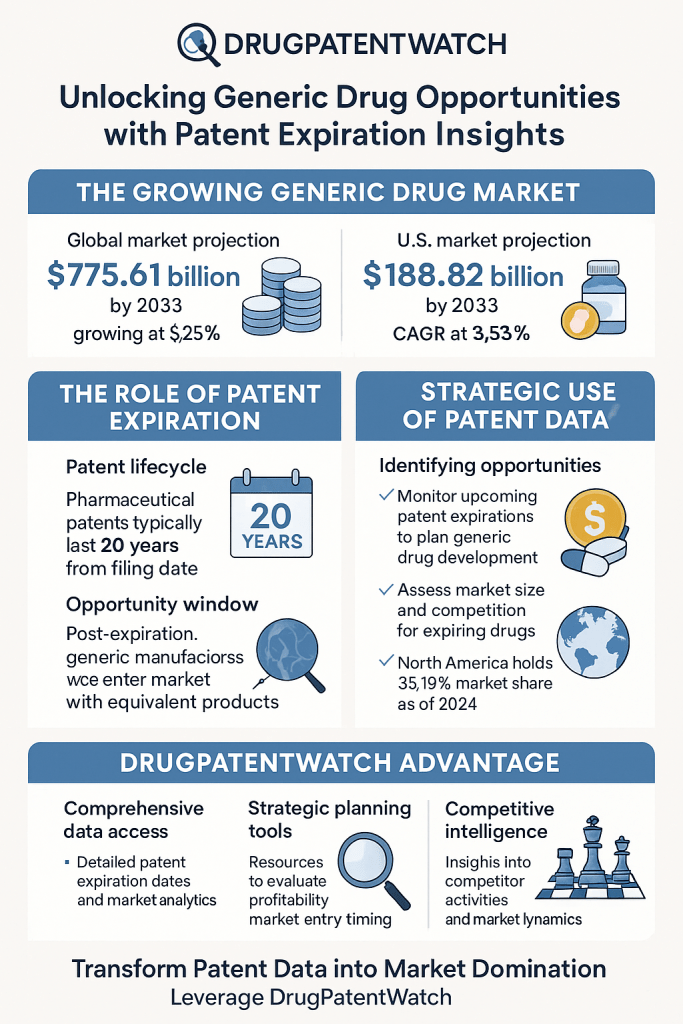
Companies building cross-portfolio regulatory analytics functions typically integrate FDA public ANDA database data (available through FDA’s ANDA status dataset), proprietary submission status feeds, and patent/litigation data from sources such as DrugPatentWatch, Derwent Innovation, or Docket Alarm into centralized analytics environments. The output is a portfolio-level regulatory risk dashboard that functions as an operational management tool rather than a project status report.
Key Takeaways: Submission Technology
eCTD validation failure at the ESG is a filing-date loss event with direct first-to-file commercial consequences; validation infrastructure quality is not a back-office IT question. RIM system implementation is a prerequisite for managing 100+ ANDA portfolios without material compliance risk from missed post-approval commitments. AI-generated regulatory content requires human review audit trails to satisfy FDA’s emerging content authenticity expectations.
Part VII: Post-Approval Lifecycle Management
Post-Approval Change Management: FDA CBE-0, CBE-30, and Prior Approval
Post-approval changes to ANDAs are governed by 21 CFR 314.70 and the FDA guidance on Scale-Up and Post-Approval Changes (SUPAC), which covers manufacturing equipment changes, scale changes, site transfers, and formulation changes with change-level-specific reporting categories. Changes are categorized as Prior Approval Supplements (PAS), requiring FDA approval before implementation; Changes Being Effected in 30 Days (CBE-30), allowing implementation 30 days after FDA receipt unless the agency objects; and Changes Being Effected (CBE-0), allowing immediate implementation with concurrent notification.
Site transfers for finished dosage form manufacturing from one FDA-registered facility to another require at minimum a CBE-30 or PAS depending on dosage form complexity, and the transferring facility must be included in the approved ANDA before the commercial transfer can occur. For generic products where manufacturing is contracted to a CDMO, post-approval site transfers add a regulatory coordination layer between the ANDA holder and the CDMO that must be managed with explicit timelines built into the CDMO contract. Generic companies that negotiate CDMO contracts without explicit post-approval change notification timelines and cost allocations routinely discover that a site optimization decision by the CDMO triggers a PAS requirement they had not budgeted.
Synchronized Multi-Market Variation Management
Post-approval changes in the EU are governed by Commission Regulation (EC) No 1234/2008 on the examination of variations to the terms of marketing authorizations. Variations are classified as Type IA (minor, immediate notification), Type IB (minor, 30-day notification), and Type II (major, requiring approval), with a separate Urgent Safety Restriction category for immediate risk-mitigation changes.
A manufacturing site transfer approved via a CBE-30 in the US and a Type II variation in the EU does not happen on the same schedule: the FDA CBE-30 can be implemented 30 days after submission, while an EU Type II variation takes 60 days at minimum and typically 90-180 days for complex manufacturing changes. If the supply chain decision requires synchronized implementation across both markets, the minimum common timeline is constrained by the slower EU variation approval, meaning the planning horizon for supply chain changes in a multi-market portfolio must account for EU variation timelines as the rate-limiting factor, not FDA CBE-30 timelines.
Indian generic manufacturers supplying both the US and EU markets, including Dr. Reddy’s Laboratories, Cipla, and Aurobindo, have developed internal variation management systems that track outstanding variation approvals across markets and flag supply decisions that require synchronized multi-market change management. The operational complexity scales with the number of markets and the frequency of manufacturing changes, which is a direct argument for manufacturing platform stability in established generic products rather than continuous optimization that generates ongoing variation submission burdens.
GDUFA Commitment Tracking and Compliance
GDUFA user fees fund FDA’s generic drug review program. Under GDUFA II (FY2018-FY2022) and GDUFA III (FY2023-FY2027), ANDA applicants pay annual program fees for approved ANDAs and one-time filing fees per ANDA submission. The fee structure for GDUFA III introduces portfolio-level fee calculations for companies with large approved ANDA portfolios, with reduced per-product fees above threshold portfolio sizes. For the largest generic drug companies by approved ANDA count (Teva with approximately 600+ approved ANDAs, Mylan/Viatris with a similarly sized portfolio, and Sun Pharma, Amneal, and Sandoz in the 200-400 range), GDUFA program fees represent a material operating cost line that requires active portfolio rationalization to manage.
Post-approval commitments (PACs) required as conditions of ANDA approval, typically covering stability studies at extended timepoints, pediatric labeling updates where required, and risk evaluation and mitigation strategy (REMS) implementation where applicable, carry compliance tracking requirements with specific FDA reporting dates. Missing a PAC deadline without prior FDA communication is a cGMP compliance event that can trigger warning letter risk for repeat failures. For a large generic portfolio, PAC deadline management is an enterprise compliance function, not a product-level regulatory affairs task.
Key Takeaways: Post-Approval Lifecycle Management
Multi-market post-approval change management is constrained by the slowest jurisdiction’s variation timeline, which is typically EU Type II at 90-180 days, not FDA CBE-30 at 30 days. CDMO contracts must specify post-approval change notification timelines and cost allocations before contract execution, not during a site transfer event. GDUFA III portfolio-level fee structures reward active ANDA portfolio rationalization for large generic companies.
Part VIII: Competitive Generic Therapy Designation and the 180-Day Exclusivity Playbook
What CGT Designation Actually Does
The FDA’s Competitive Generic Therapy (CGT) designation, created under the FDA Reauthorization Act of 2017 (FDARA), provides an expedited review pathway for generic drugs with inadequate generic competition, defined as no approved ANDA or a single approved ANDA for the reference listed drug at the time of filing. The designation awards 180-day first-generic exclusivity to the first CGT-designated applicant to receive final approval, separate from and non-cumulative with the Paragraph IV Hatch-Waxman 180-day exclusivity.
The commercial logic is straightforward. Products with no generic competition have branded pricing that generic entry can undercut by 70-90%, generating the largest single-entry generic revenue events in the industry. CGT-designated products, where FDA commits to 8-month review goals (same as standard ANDA priority review timelines under GDUFA III), generate first-generic exclusivity revenue in the range of the original 180-day Hatch-Waxman exclusivity for high-volume products without requiring Paragraph IV litigation. The litigation risk is absent because the reference product often has simple or expired patent coverage, which is why no prior generic had filed.
Between 2018 and 2024, FDA received over 1,000 CGT designation requests and granted approximately 60-65% of them. Not all CGT-designated ANDAs convert to approval within the target review period, because CGT designation does not waive CMC, BE, or labeling requirements. Companies that treat CGT designation as an approval guarantee rather than a review priority end up submitting technically deficient ANDAs that miss their window and watch a second filer receive CGT final approval instead.
Investment Strategy: 180-Day Exclusivity as a Portfolio Construction Tool
For portfolio managers evaluating generic drug companies, the 180-day exclusivity pipeline is the most direct proxy for near-term revenue visibility. A generic company with five products in active 180-day exclusivity periods simultaneously, across therapeutic categories with different branded price points, has a revenue profile that is analytically separable from the commodity generic business and warrants separate valuation treatment.
The valuation methodology for 180-day exclusivity revenue uses a product-specific revenue model: branded annual US sales multiplied by a generic penetration rate assumption (typically 70-85% market share at 30% of branded price for the exclusivity holder alone), then risk-adjusted for the probability of litigation settlement, authorized generic competition from the brand (which does not break the exclusivity period but does reduce the exclusivity holder’s market share within the period), and patent challenge failure risk.
Amneal Pharmaceuticals’ 2022-2023 period, when the company was in or entering 180-day exclusivity for several CNS generics including generic versions of branded extended-release products, illustrates the revenue concentration effect. A single 180-day exclusivity period for a $500M branded product can contribute $80-150M in incremental revenue to a generic company with $2B in total annual revenues, a contribution magnitude that temporarily distorts operating margin and cash generation metrics that should be analyzed on an ex-exclusivity basis for normalized valuation.
Key Takeaways: CGT and Exclusivity Strategy
CGT designation provides 180-day first-generic exclusivity without litigation risk but requires technically complete ANDAs to convert designation into on-schedule approval. The 180-day exclusivity pipeline is the most direct measure of near-term generic revenue visibility and should be valued separately from commodity generic operations in portfolio company analysis. Authorized generic competition during the exclusivity period is the primary commercial risk that most first-filer revenue models inadequately account for.
Part IX: Japan, China, and Emerging Market Regulatory Strategy
Japan’s PMDA and the Generic Drug Market Reforms
Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) oversees generic drug approval under a bioequivalence-centered framework that has undergone substantial reform since 2020. The Japanese government’s target of achieving an 80% generic substitution rate (by volume) in the national health insurance market has driven policy changes that accelerated PMDA review timelines and simplified bioequivalence requirements for standard oral solid dosage forms.
PMDA’s BE requirements align broadly with ICH E6 and ICH E8 for study conduct, but the specific dissolution testing conditions required for BCS-based biowaivers in Japan differ from FDA and EMA standards: PMDA requires dissolution testing at pH 1.2, 6.8, and 7.5 (rather than pH 4.5 or 6.8 used in FDA and EMA BCS biowaiver frameworks), which means a three-condition dataset that satisfies PMDA’s dissolution requirements must be prospectively collected even when the FDA and EMA datasets are otherwise reusable. Building this Japan-specific dissolution condition into the standard reference product dissolution profiling conducted early in development costs two additional analytical batches and approximately 6-8 weeks of timeline; not building it in costs a 12-18 month delay when Japan enters the GRP as a target market mid-development.
China’s NMPA and the Generic Drug Consistency Evaluation Program
China’s National Medical Products Administration (NMPA) launched the Generic Drug Consistency Evaluation program in 2016, requiring generic drugs on the national essential medicines list to demonstrate bioequivalence to reference listed drugs that meet current international manufacturing and quality standards. The program has transformed China’s domestic generic market, eliminating low-quality generics that lacked adequate BE data and creating market consolidation dynamics that parallel the US generics industry evolution of the 1980s-1990s.
For international generic companies seeking NMPA approval for China market entry, the Consistency Evaluation framework requires a full BE study conducted in Chinese subjects (NMPA does not currently accept non-Chinese BE studies as primary evidence for most drug classes), GMP certification of the manufacturing site under NMPA standards, and dossier submission in NMPA’s eCTD-equivalent format. The requirement for China-specific BE studies represents a material additional investment for any international company targeting the China market, typically $3-8M per product including study execution, regulatory translation, and site qualification costs.
The China market opportunity for off-patent small molecules is substantial: the National Healthcare Security Administration (NHSA) volume-based procurement (VBP) program has negotiated dramatic price reductions (typically 50-90% off pre-VBP prices) for generics that win VBP tenders, but the volume commitments attached to VBP contracts provide production scale and revenue predictability that partially offsets margin compression. Sun Pharma, Dr. Reddy’s, and Zydus Lifesciences have all invested in China Consistency Evaluation programs for priority molecules with sufficient VBP volume potential to justify the regulatory and manufacturing investment.
Emerging Market Regulatory Convergence
Brazil (ANVISA), South Korea (MFDS), Saudi Arabia (SFDA), South Africa (SAHPRA), and Indonesia (BPOM) collectively represent emerging market generic drug revenues exceeding $30B annually. Each has moved toward varying degrees of regulatory convergence with ICH, FDA, and EMA standards since 2015, but each retains jurisdiction-specific requirements that prevent straightforward dossier reuse.
ANVISA’s RDC 204/2017 for generic drug registration aligns closely with FDA ANDA requirements for BE and CMC standards, and ANVISA accepts FDA or EMA clinical BE data as supplementary evidence in some categories. The South Korean MFDS is an ICH full regulatory member as of 2016 and has progressively aligned its BE guideline with ICH M12. Saudi SFDA accepts submissions in formats aligned with ICH M4 (CTD format) and frequently relies on FDA or EMA regulatory assessments as supporting documentation.
For companies with portfolios that include molecules with high emerging market clinical demand (antiretrovirals, fixed-dose combination antimalarials, second-line tuberculosis drugs), building a WHO-PQ approval into the GRP creates a documentation asset that simplifies ANVISA, SAHPRA, and BPOM submissions through those agencies’ reliance pathways on WHO-PQ decisions. The convergence of WHO-PQ, ANVISA, and SAHPRA regulatory requirements is not complete, but a WHO-PQ-approved dossier typically satisfies 70-80% of ANVISA’s technical requirements, reducing the marginal documentation cost for Brazil entry from full dossier to targeted gap analysis.
Key Takeaways: Japan, China, and Emerging Markets
PMDA’s Japan-specific dissolution condition (pH 7.5) must be built into the standard reference product profiling done early in development, not retrofitted. China’s NMPA Consistency Evaluation requires China-specific BE studies that represent a $3-8M per-product incremental investment with volume commitments from NHSA VBP contracts as the primary revenue justification. WHO-PQ approval is a documentation leverage asset for emerging market entries into ANVISA, SAHPRA, and BPOM reliance pathways.
Part X: Regulatory Intelligence and Competitive Monitoring
Tracking Competitor Paragraph IV Pipelines
The FDA ANDA database, updated monthly, lists all pending and approved ANDAs by applicant, reference drug, and certification type. Paragraph IV certifications trigger public notification requirements under Hatch-Waxman, and while the specific patent claims being challenged are not publicly disclosed in the application, the combination of Orange Book listing data and ANDA applicant history makes it possible to model competitor IP strategy with reasonable precision.
DrugPatentWatch, Citeline (formerly Informa Pharma Intelligence), and Evaluate Pharma aggregate FDA ANDA data, Orange Book patent listings, litigation records, and market authorization data into commercial intelligence platforms used by regulatory affairs teams, IP counsel, and institutional investors for competitive tracking. The specific analytical outputs that matter for GRP development are: the number of Paragraph IV filers per Orange Book patent, the filing date distribution among filers (to model shared versus solo 180-day exclusivity), and the historical litigation outcomes for the patent holder in question (to calibrate the probability that the brand will sue and prevail).
For EMA procedures, the European public assessment reports (EPARs) for reference medicines and their generic counterparts, combined with the EMA’s annual report on generic applications received and approved, provide the competitive landscape data necessary to model EU market entry timing relative to competitor MAA filings. The EU does not have an equivalent of the US 180-day exclusivity mechanism, but the EU’s ‘first generic’ market dynamics still reward early market entry through volume capture before price erosion accelerates with each additional generic entrant.
Patent Landscape Analysis as a GRP Input
A patent landscape for a generic development target requires analysis across multiple patent databases (USPTO full-text, EPO’s Espacenet, WIPO’s PatentScope, and the JPO J-PlatPat for Japan), correlation of patent families with FDA Orange Book listings and EMA reference product status, and freedom-to-operate (FTO) analysis for the proposed generic’s dosage form, synthesis route, and formulation components.
The FTO analysis for a generic product is distinct from the Paragraph IV certification strategy: FTO identifies patents that could be infringed by the generic product’s commercialization regardless of whether those patents are Orange Book-listed. A process patent covering the API synthesis route, held by a third party other than the brand company, is not Orange Book-listed but is fully enforceable against a generic manufacturer. High-profile examples include litigation over synthetic routes for API intermediates in generic oncology products, where third-party process patents not associated with the brand have delayed or restructured manufacturing plans for generic entrants.
Regulatory Decision Monitoring and Label Carve-Out Tracking
FDA’s DailyMed and Orange Book, combined with FDA’s Approved Drug Products database (the electronic Orange Book), are the primary sources for tracking labeling changes to reference listed drugs that could affect generic label carve-out strategies. When a brand company receives a new indication added to the product label, the Orange Book patent listings may be updated to include a new method-of-use patent for that indication. If a generic applicant has already filed an ANDA with a carve-out of the original indication but not the new indication, the label scope of the approved generic may be narrower than initially planned.
Tracking Orange Book updates, new patent listings, and reference product label amendments is a continuous function for any ANDA program in active development. Patent listing challenges under 21 CFR 314.53(f)(1), which allow ANDA applicants to challenge improper Orange Book listings directly with FDA without separate patent litigation, have become more common since the FTC’s public delisting challenge campaign drew attention to the mechanism. As of 2025, FDA has responded to over 300 listing challenges filed by generic applicants and third parties, delisting several patents that did not meet the statutory criteria for inclusion.
Investment Strategy: Regulatory Intelligence as a Moat
Regulatory intelligence capability, meaning the systematic capacity to track competitor ANDA pipelines, monitor Orange Book listing changes, and model patent expiry scenarios across a 5-10 year horizon, is a durable competitive moat for generic drug companies that invest in it as an organizational function rather than purchasing point-in-time external reports.
Companies that build proprietary regulatory intelligence infrastructure consistently identify CGT designation opportunities, first-to-file Paragraph IV windows, and RLD labeling change risks before competitors do, which compounds into portfolio construction advantages that persist for years. For institutional investors, the organizational sophistication of a generic company’s regulatory intelligence function is a proxy for execution quality that is more predictive of long-term performance than pipeline size alone.
Conclusion: The Submission Optimization Stack as a Competitive System
Multi-market generic drug approval optimization is not a collection of individual best practices. It is a system where patent expiry sequencing, GRP architecture, BE study design, eCTD technical infrastructure, and post-approval variation management are interdependent variables that must be solved simultaneously. Companies that manage these as separate workstreams, coordinated informally, consistently underperform on first-cycle approval rates, miss first-to-file windows, and absorb variation management costs that could have been designed out of the system at the GRP stage.
The economic stakes are not abstract. A 6-month compression in ANDA approval timeline for a product with $800M in branded annual US sales, where 180-day exclusivity is at stake, is worth $40-80M in incremental revenue net of the additional regulatory investment required to achieve it. That arithmetic applies across every molecule in a 200-ANDA pipeline, which is why the most capable generic drug organizations treat regulatory strategy as a core profit driver and staff and capitalize it accordingly.
The companies that consistently outperform on multi-market generic approvals, including Hikma in injectables, Amneal in CNS and respiratory, Cipla in respiratory and antiretrovirals, and Viatris across its legacy Mylan and Upjohn portfolios, share a structural characteristic: regulatory affairs functions that operate with IP intelligence, commercial context, and supply chain visibility as standing inputs, not as information requested after a submission strategy is already committed. Building that organizational architecture is the actual optimization problem. The tactics in this guide are the tools; the organizational design is the system.