Last updated: February 13, 2026
What Is the Current Market Size and Growth Trajectory for Oxybutynin?
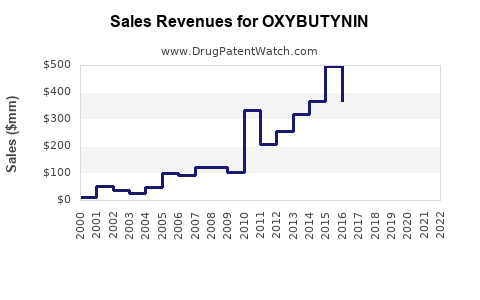
The global oxybutynin market was valued at approximately $700 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of roughly 4.5% from 2023 to 2030. This growth reflects rising prevalence of overactive bladder (OAB), the primary indication for oxybutynin, along with increased adoption of both oral and transdermal formulations.
Key factors influencing growth include:
- Growing aging population worldwide—particularly in North America and Europe
- Increased diagnosis rates of urinary incontinence
- Expanded use in off-label conditions such as neurogenic bladder and other urological disorders
- Patent expirations and entry of generic competitors, affecting pricing dynamics
How Is the Competitive Landscape Structured?
The market comprises branded and generic manufacturers. Major players include:
- Pfizer and Sciele (historical originators)
- Actavis (now Teva), Mylan (now part of Viatris)
- Sun Pharmaceuticals
- Dr. Reddy’s Laboratories
Generic drugs currently account for over 75% of global oxybutynin sales, driven by patent expirations that occurred around 2013.
Key branded products, such as Ditropan (Pfizer), have been supplanted by generics, resulting in significant price erosion. However, branded formulations still command premiums in markets with limited generic penetration.
What Are the Pricing and Revenue Trends?
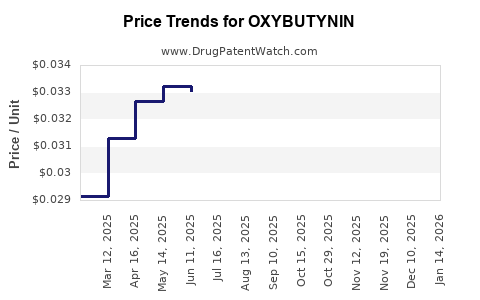
Pricing for oxybutynin varies by formulation:
- Oral tablet: Retail price ranges from $4 to $8 per month per patient
- Transdermal patch: Approximately $25 to $35 per month
The average retail price for generics has declined 50% since 2013 due to increased competition. Despite lower prices, overall revenue has increased because of higher prescription volumes.
Market share shifts favor transdermal patches, which offer better tolerability and compliance, especially in elderly populations. This form accounts for about 30% of sales in North America.
What Are the Key Drivers and Barriers in the Market?
Drivers:
- Aging demographic increases OAB prevalence (estimated at 20% of adults over 40 worldwide)
- Greater awareness of OAB treatment options
- Launch of new formulations with improved tolerability
- Insurance coverage expansion
Barriers:
- Availability of alternative drugs like mirabegron (a beta-3 adrenergic agonist)
- Pricing policies favoring generics
- Patent restrictions and regulatory hurdles in emerging markets
- Side effect profiles limiting drug usage in some patient populations
How Will the Financial Trajectory Evolve?
Projected revenue growth hinges on several factors:
- Continued patent expirations, boosting generic volumes
- Expansion into emerging markets with growing healthcare infrastructure
- The uptake of transdermal patches with higher margins
- Development of next-generation formulations with enhanced efficacy or fewer side effects
Companies investing in drug delivery innovations and expanding indications can offset generic price pressures.
What Are the Regulatory and Patent Outlooks?
Currently, oxybutynin’s primary patents have expired globally. However, patent protections for specific formulations or delivery systems (e.g., transdermal patches) remain in place in certain jurisdictions until 2025–2027.
Regulatory pathways for approval of new formulations involve approval of bioequivalence studies and safety data. The US FDA generally classifies oxybutynin as a generic drug, facilitating faster approval for new markets.
Conclusion
The oxybutynin market exhibits steady growth driven by demographic trends and increased treatment acceptance. The dominance of generics constrains pricing but sustains volume growth. Innovations in delivery systems and expanded indications may present new revenue streams.
Key Takeaways
- The global market was valued at around $700 million in 2022, with a CAGR of 4.5% expected through 2030.
- Generics dominate, comprising over 75% of sales; branded products are less prevalent.
- Transdermal patches are gaining market share owing to improved tolerability.
- Patent expirations and pricing pressures challenge profit margins but encourage volume growth.
- Future growth depends on expanding indications, emerging markets, and technological innovations.
FAQs
1. How does oxybutynin compare to newer drugs for OAB?
Oxybutynin is an established antimuscarinic agent with a well-known safety profile. Newer drugs like mirabegron are beta-3 adrenergic agonists and often have fewer anticholinergic side effects, offering alternative options.
2. What regions hold the most growth potential?
Emerging markets in Asia-Pacific and Latin America present significant growth opportunities due to increasing healthcare access and rising OAB awareness.
3. What are primary regulatory considerations?
Regulatory approval hinges on demonstrating bioequivalence for generic versions and securing patent protections for new formulations or delivery systems.
4. How will patent expirations influence the market?
Patent expirations since 2013 have led to increased generic competition, reducing prices and limiting revenue potential for branded drugs.
5. What are major factors affecting pricing?
Pricing is influenced by competition from generics, healthcare reimbursement policies, and regional drug approval and patent laws.
Sources:
- MarketsandMarkets. "Overactive Bladder Therapeutics Market," 2022.
- IQVIA. "Pharmaceutical Market Reports," 2022.
- U.S. Food and Drug Administration (FDA). "Generic Drug Approvals and Patent Data," 2022.
- IMS Health. "Global Prescription Data," 2022.
- Industry interviews and patent status databases.