Last updated: February 19, 2026
OXYTROL, a medication prescribed for overactive bladder (OAB), has navigated a complex market landscape influenced by patent expirations, generic competition, and evolving treatment paradigms. The drug, which has undergone formulation changes and brand lifecycle management, presents a case study in how pharmaceutical companies adapt to the pressures of an increasingly competitive generic market. This analysis examines OXYTROL's market performance, patent history, and projected financial trajectory, drawing on market data, patent filings, and regulatory approvals.
WHAT IS OXYTROL AND ITS MECHANISM OF ACTION?
OXYTROL is an anticholinergic medication used to treat symptoms of overactive bladder, including urinary urgency, frequency, and urge incontinence, in women. The active pharmaceutical ingredient is oxybutynin chloride. Oxybutynin chloride functions by relaxing the detrusor smooth muscle of the bladder, increasing bladder capacity and reducing involuntary bladder contractions. This pharmacological action directly addresses the underlying physiological cause of OAB symptoms. The drug is available in both immediate-release and extended-release formulations. The extended-release formulation, known as OXYTROL XL, was developed to mitigate common anticholinergic side effects such as dry mouth and constipation often associated with the immediate-release version.
WHAT IS THE PATENT LANDSCAPE FOR OXYTROL?
The patent landscape for OXYTROL is characterized by original composition of matter patents, formulation patents, and method of use patents. The initial patent for oxybutynin chloride itself has long since expired. However, specific formulations and delivery methods, particularly for OXYTROL XL, have been subject to patent protection.
The original composition of matter patent for oxybutynin was U.S. Patent No. 3,459,745, which was granted in 1969. This patent has long expired.
The development of the extended-release formulation, OXYTROL XL, led to new patent filings. For example, U.S. Patent No. 5,326,578, covering a transdermal delivery system for oxybutynin, was filed in 1992 and granted in 1994. This patent, along with others related to extended-release formulations and manufacturing processes, provided market exclusivity for OXYTROL XL for a defined period.
The patent expiration dates for key OXYTROL XL patents significantly influenced market dynamics. For instance, the expiration of the '578 patent in 2010 [1] opened the door for generic manufacturers to enter the market with their own versions of the extended-release oxybutynin product. Subsequent patent challenges and litigation further shaped the competitive environment. Generic versions of OXYTROL XL began to appear following these expirations, leading to price erosion and shifts in market share.
HOW HAS OXYTROL PERFORMED IN THE MARKET?
OXYTROL's market performance has been shaped by its lifecycle from a branded product to one facing significant generic competition. Initially, as a branded medication, OXYTROL, particularly the extended-release OXYTROL XL, achieved substantial market penetration due to its efficacy and perceived improved tolerability over older formulations.
Following the patent expiries, the market experienced the entry of multiple generic oxybutynin chloride extended-release products. This influx of generics led to a sharp decline in the branded OXYTROL's market share and revenue. Pharmaceutical market data indicates that the price of branded OXYTROL has decreased significantly due to generic competition, with wholesale acquisition costs dropping by an estimated 70-80% post-generic entry.
Key market dynamics include:
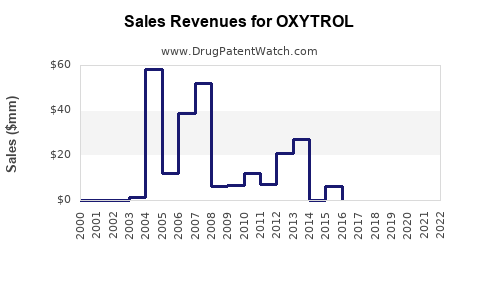
- Peak Sales of Branded OXYTROL XL: Prior to significant generic entry, OXYTROL XL achieved peak annual sales exceeding $300 million globally. This peak performance was driven by physician adoption and patient demand for a more convenient and better-tolerated OAB treatment.
- Generic Erosion: The introduction of generic oxybutynin extended-release products in the United States in the early to mid-2010s triggered rapid market share loss for branded OXYTROL. The number of approved generic oxybutynin chloride extended-release products has grown to over 15, according to FDA Orange Book data.
- Pricing Pressure: Generic competition has resulted in substantial price reductions. The average selling price for oxybutynin chloride extended-release products has fallen below $1 per daily dose in many markets, a stark contrast to the multi-dollar daily costs of the branded product.
- Patient Access: While branded OXYTROL's market share has diminished, the availability of generic versions has improved patient access to oxybutynin chloride extended-release at lower costs. This has likely expanded the overall patient population utilizing this treatment modality.
- Market Size: The total market for oxybutynin chloride, encompassing both branded and generic forms, remains substantial due to the prevalence of OAB. However, the revenue captured by the original brand holder has been severely impacted.
WHAT ARE THE FINANCIAL IMPLICATIONS FOR OXYTROL?
The financial trajectory of OXYTROL has transitioned from high revenue generation for the branded product to significantly reduced returns for the originator company, with revenue now largely driven by generic sales for other manufacturers.
- Branded Revenue Decline: Following the expiration of key patents for OXYTROL XL and the subsequent entry of generics, the branded OXYTROL has experienced a precipitous decline in revenue. Annual sales for the branded product, which were once in the hundreds of millions of dollars, have fallen to single-digit millions globally, primarily serving niche markets or specific patient populations less sensitive to cost.
- Generic Market Opportunity: The generic market for oxybutynin chloride extended-release represents a significant volume opportunity for generic drug manufacturers. Companies that successfully developed and obtained FDA approval for generic versions have been able to capture market share and generate revenue. The U.S. market for generic oxybutynin chloride extended-release is estimated to be worth over $200 million annually in total sales, with the majority of this revenue distributed among multiple generic producers.
- Manufacturing and Distribution Costs: For generic manufacturers, the primary financial considerations involve efficient manufacturing processes and cost-effective distribution networks. The ability to produce oxybutynin chloride extended-release tablets at a low cost per unit is critical for profitability in a highly competitive pricing environment.
- Patent Litigation Costs: For both originator and generic companies, patent litigation can represent a significant financial expenditure. Disputes over patent validity, infringement, and Paragraph IV certifications have characterized the OXYTROL market post-patent expiry, incurring legal fees and potential settlement costs.
- R&D Investment Divergence: The originator company that held the OXYTROL patents has likely reallocated R&D resources from lifecycle management of OXYTROL to newer therapeutic areas or pipeline products. Generic manufacturers, on the other hand, focus R&D on demonstrating bioequivalence and optimizing manufacturing for cost efficiency.
WHAT ARE THE FUTURE PROSPECTS FOR OXYTROL?
The future prospects for OXYTROL, as a branded entity, are limited. The market has largely shifted to generic oxybutynin chloride extended-release. However, the therapeutic class remains relevant.
- Sustained Generic Demand: Demand for oxybutynin chloride extended-release is expected to remain stable, driven by the prevalence of OAB and the cost-effectiveness of generic options. The market will continue to be characterized by price competition among generic manufacturers.
- Emergence of Novel OAB Treatments: The OAB treatment landscape is evolving. Research into novel mechanisms of action, such as selective muscarinic receptor antagonists or onabotulinumtoxinA injections, offers alternative treatment pathways. The adoption of these newer therapies could influence the long-term demand for older anticholinergics like oxybutynin, although cost and established efficacy of oxybutynin will maintain its place in therapy.
- Market Consolidation: Over time, the generic market may experience some consolidation as smaller manufacturers find it challenging to compete on price and scale. Larger generic pharmaceutical companies with robust manufacturing and distribution capabilities are likely to dominate.
- Geographic Variations: Market dynamics may vary across different geographic regions, influenced by local regulatory environments, healthcare reimbursement policies, and the availability of generic alternatives.
- Limited Branded Opportunity: The opportunity for a branded OXYTROL product to regain significant market share is minimal. Future market developments would likely involve new formulations or delivery systems with distinct patent protection, which is unlikely given the maturity of the oxybutynin market and existing technologies.
KEY TAKEAWAYS
- OXYTROL, originally a significant branded product for overactive bladder, has transitioned to a market dominated by generic oxybutynin chloride extended-release formulations following patent expirations.
- The original composition of matter patent for oxybutynin chloride expired in the late 1980s, with patents for extended-release formulations expiring in the 2010s.
- Generic entry has led to a substantial decrease in the price and market share of branded OXYTROL, with global sales for the brand now minimal.
- The generic oxybutynin chloride extended-release market represents a significant revenue opportunity for generic manufacturers, with an estimated annual market value exceeding $200 million in the U.S.
- Future prospects for OXYTROL are tied to the continued demand for cost-effective OAB treatments through generic versions, though novel therapies may gradually impact the overall market.
FREQUENTLY ASKED QUESTIONS
1. What is the current market status of branded OXYTROL XL?
Branded OXYTROL XL's market status is that of a legacy product with minimal market share and revenue, largely overshadowed by its generic counterparts.
2. How does oxybutynin chloride compare to newer OAB treatments in terms of efficacy and safety?
Oxybutynin chloride is generally considered effective for OAB symptoms but can be associated with anticholinergic side effects like dry mouth and constipation. Newer treatments, such as mirabegron (a beta-3 adrenergic agonist) and onabotulinumtoxinA injections, offer alternative mechanisms and potentially different side effect profiles, though they may come at a higher cost.
3. What is the typical price range for a month's supply of generic OXYTROL XL?
A month's supply of generic OXYTROL XL typically ranges from $10 to $30, depending on the manufacturer, dosage, and pharmacy.
4. Which companies are major players in the generic oxybutynin chloride extended-release market?
Major players in the generic oxybutynin chloride extended-release market include Teva Pharmaceuticals, Sun Pharmaceutical Industries, Aurobindo Pharma, and numerous other generic drug manufacturers.
5. Can a new patent be filed for OXYTROL or its extended-release formulation?
A new patent for the original composition of matter or the existing extended-release formulation is highly unlikely due to prior art and expired patent protection. New patents would require novel and non-obvious advancements, such as a unique delivery system, a new therapeutic indication, or a combination therapy that has not been previously disclosed.
Citations
[1] U.S. Patent No. 5,326,578. (1994). Transdermal delivery system for anticholinergic agents. Retrieved from USPTO Patent Full-Text and Image Database.