Last updated: February 19, 2026
Oxybutynin chloride is an antimuscarinic agent used to treat symptoms of overactive bladder (OAB), including urinary urgency, frequency, and incontinence. The market for oxybutynin chloride is mature but remains substantial due to the high prevalence of OAB. Key market drivers include an aging global population, increasing awareness of OAB treatment options, and the availability of various formulations. Patent expiries have led to significant generic competition, impacting pricing and manufacturer revenue.
What is the global market size and projected growth for oxybutynin chloride?
The global oxybutynin chloride market is estimated to be valued at approximately $700 million to $800 million annually. This market is projected to experience a compound annual growth rate (CAGR) of 2.5% to 3.5% over the next five years. This steady, albeit modest, growth is primarily driven by the increasing prevalence of overactive bladder (OAB) globally, particularly among older adults.
Key Market Size Data:
- 2023 Estimated Market Value: $750 million
- Projected 2028 Market Value: $875 million - $940 million
- Projected CAGR (2023-2028): 2.5% - 3.5%
The aging demographic is a significant factor. As the global population ages, the incidence of conditions like OAB, which are more common in older individuals, is expected to rise. A report by the National Institute on Aging indicates that OAB affects approximately 16% of adults aged 60 and older (1). This demographic shift directly translates to an expanded patient pool requiring treatments like oxybutynin chloride.
Furthermore, enhanced diagnostic capabilities and increased patient and physician awareness of OAB as a treatable condition contribute to market expansion. Patients are more likely to seek medical advice for OAB symptoms, and healthcare providers are more adept at diagnosing and prescribing appropriate therapies.
What are the primary therapeutic indications and patient populations for oxybutynin chloride?
Oxybutynin chloride is primarily indicated for the symptomatic treatment of detrusor overactivity, commonly known as overactive bladder (OAB). This condition is characterized by urinary urgency, often accompanied by urinary frequency and urge incontinence.
Primary Indications:
- Overactive Bladder (OAB): This is the principal indication. OAB is a chronic condition that significantly impacts quality of life, leading to social isolation and reduced productivity.
- Detrusor Hyperactivity in Neurological Conditions: Oxybutynin chloride may be used off-label or in specific clinical guidelines to manage detrusor hyperreflexia in patients with neurological conditions such as spinal cord injury, multiple sclerosis, or Parkinson's disease, where involuntary bladder contractions lead to incontinence (2).
Target Patient Populations:
- Adults (18+): The drug is widely prescribed for adult patients experiencing OAB symptoms.
- Elderly Population (60+): This demographic represents the largest and fastest-growing segment of OAB patients. Studies show a higher prevalence of OAB in this age group due to age-related physiological changes and comorbidities (3). For instance, research published in the Journal of the American Geriatrics Society noted that OAB prevalence can exceed 30% in women over 65 (4).
- Patients with Neurological Disorders: Individuals with conditions affecting bladder control mechanisms.
The effectiveness of oxybutynin chloride in alleviating these symptoms makes it a cornerstone therapy for millions worldwide. However, its use in certain patient populations, particularly the elderly, requires careful consideration due to potential side effects.
Which companies are major players in the oxybutynin chloride market, and what are their market shares?
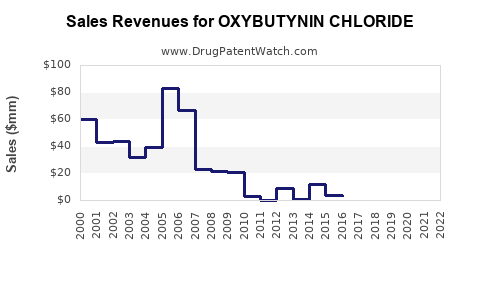
The oxybutynin chloride market is characterized by a significant number of generic manufacturers following the expiry of key patents for the originator drug, Ditropan XL (Mallinckrodt). This has led to a highly competitive landscape with diffused market shares.
Key Manufacturers and Product Offerings:
- Teva Pharmaceuticals: A major generic drug manufacturer with a significant presence in the OAB market. Teva offers both immediate-release and extended-release formulations of oxybutynin chloride.
- Dr. Reddy's Laboratories: Another prominent generic pharmaceutical company that supplies oxybutynin chloride tablets and extended-release formulations.
- Sun Pharmaceutical Industries: Holds a substantial market share with its generic oxybutynin chloride products.
- Impax Laboratories (now Amneal Pharmaceuticals): A key player in the extended-release oxybutynin chloride segment.
- Mallinckrodt Pharmaceuticals: The originator of Ditropan XL, still maintains a presence, particularly with its extended-release formulation, though its market share has been reduced by generic competition.
- ANI Pharmaceuticals: Offers a generic extended-release formulation.
Market Share Dynamics:
Market share data in the generic oxybutynin chloride space is highly fragmented and fluctuates based on pricing strategies, distribution agreements, and production capacities. No single company commands a dominant share above 20%.
- Top 5 Generic Manufacturers: Collectively hold an estimated 60-70% of the generic oxybutynin chloride market.
- Individual Market Shares: Typically range from 5% to 15% for the leading generic players.
The intense competition among generic manufacturers exerts downward pressure on pricing, which is a key factor influencing revenue for these companies. The extended-release formulations generally command higher prices than immediate-release versions due to improved patient compliance and reduced dosing frequency.
What is the patent landscape for oxybutynin chloride and its key formulations?
The patent landscape for oxybutynin chloride is largely characterized by expired foundational patents. The original patent for oxybutynin chloride itself has long since expired, allowing for the widespread development and marketing of generic versions. However, patents related to specific formulations, manufacturing processes, and delivery systems have played a role in market dynamics.
Key Patent Expiries and Developments:
- Original Oxybutynin Chloride Patent: Expired in the late 1990s.
- Ditropan XL® (Extended-Release Formulation): The patent for Mallinckrodt's extended-release formulation, which offered improved patient compliance by reducing dosing frequency, was a significant area of patent protection. The primary patents protecting Ditropan XL, such as U.S. Patent No. 4,327,074, expired around 2007-2008, opening the door for generic extended-release versions.
- Newer Formulations and Delivery Systems: While not as prevalent as in newer drug classes, some patents might exist for novel delivery systems or combination therapies involving oxybutynin chloride. However, these have not significantly altered the core market dominated by established generic products.
- Process Patents: Patents related to specific synthesis routes or purification methods for oxybutynin chloride may exist but are less likely to be significant barriers to entry in the generic market given the drug's long history.
Impact of Patent Landscape:
The expiry of key patents, especially for the extended-release formulation, has been the primary driver for the high level of generic competition. This competition has led to substantial price erosion and a market dominated by cost-effective generic alternatives. Manufacturers who successfully navigated the patent challenges and developed bioequivalent generic versions have captured significant market share.
What are the key regulatory considerations and pathways for oxybutynin chloride products?
Regulatory approval for oxybutynin chloride products, particularly generic versions, follows established pathways overseen by major health authorities like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
U.S. FDA Pathway:
- Abbreviated New Drug Application (ANDA): Generic versions of oxybutynin chloride are approved via the ANDA pathway. This requires manufacturers to demonstrate that their product is bioequivalent to the reference listed drug (RLD), which is typically the innovator product (e.g., Ditropan XL for the extended-release formulation).
- Bioequivalence Studies: These studies compare the rate and extent of drug absorption of the generic product to the RLD. Key parameters assessed include Cmax (maximum plasma concentration) and AUC (area under the plasma concentration-time curve).
- Quality and Manufacturing Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (cGMP) and ensure their manufacturing facilities are compliant with FDA regulations.
- Labeling: The labeling for generic products must be the same as the RLD, with minor differences permitted in certain circumstances.
European Medicines Agency (EMA) Pathway:
- Generic Marketing Authorisation Application (MAA): Similar to the FDA's ANDA, European generic applications require proof of bioequivalence to an approved reference medicinal product in the EU.
- Mutual Recognition Procedure (MRP) or Decentralised Procedure (DCP): Generic products often navigate the EU market through these centralized regulatory processes.
- National Competent Authorities: While the EMA oversees centralized procedures, national authorities also play a role in approving and monitoring medicinal products.
Key Regulatory Factors:
- Bioequivalence: This is the most critical hurdle for generic approval. Variations in formulation or manufacturing can impact bioequivalence, leading to delays or rejections.
- Quality Control: Stringent quality control measures are essential to ensure product consistency and safety.
- Post-Market Surveillance: All approved products are subject to post-market surveillance to monitor for adverse events and ensure continued compliance with regulatory standards.
The regulatory landscape for oxybutynin chloride is well-established. Manufacturers that can consistently meet the rigorous bioequivalence and quality standards are well-positioned to compete.
What are the key competitive factors and market trends influencing oxybutynin chloride?
The competitive landscape for oxybutynin chloride is dominated by genericization, driving intense price competition and focusing on manufacturing efficiency and distribution networks.
Key Competitive Factors:
- Price Competition: This is the most significant factor. With numerous generic manufacturers, pricing strategies are aggressive, leading to price erosion and reduced profit margins for individual products. Manufacturers compete by offering the lowest price per unit or per milligram.
- Product Formulation: While the basic molecule is the same, differentiation can occur through extended-release formulations, which offer improved patient compliance and can command slightly higher prices. However, even these are now subject to significant generic competition.
- Distribution Networks and Contracts: Securing favorable contracts with major pharmacy benefit managers (PBMs), wholesalers, and hospital systems is crucial for market access and sales volume. Large generic manufacturers with established distribution channels have an advantage.
- Manufacturing Costs and Efficiency: Lowering production costs through efficient manufacturing processes and economies of scale is vital for maintaining profitability in a price-sensitive market.
- Product Quality and Reliability: Consistent product quality and a reliable supply chain are essential to maintain trust with healthcare providers and patients and avoid regulatory issues.
Market Trends:
- Continued Generic Penetration: The market will remain dominated by generic oxybutynin chloride.
- Focus on Extended-Release (ER) Formulations: While subject to generic competition, ER formulations are preferred for their convenience. Manufacturers that can offer cost-effective ER generic versions will continue to see demand.
- Demand from Emerging Markets: As healthcare access improves in emerging economies, there is potential for increased demand for established, affordable OAB treatments like oxybutynin chloride.
- Competition from Newer OAB Therapies: While oxybutynin chloride remains a first-line or second-line treatment, newer drug classes (e.g., beta-3 adrenergic agonists like mirabegron, newer antimuscarinics) and non-pharmacological interventions compete for market share, particularly for patients who do not respond well or experience intolerable side effects from oxybutynin chloride.
- Consolidation: The generic pharmaceutical industry has seen significant consolidation. This trend may continue, leading to fewer, larger players in the oxybutynin chloride market.
The market for oxybutynin chloride is mature, and competition is primarily driven by cost and market access rather than innovation in the core molecule.
What is the financial trajectory and profitability outlook for oxybutynin chloride manufacturers?
The financial trajectory for manufacturers in the oxybutynin chloride market is largely shaped by generic competition, leading to moderate revenue growth and constrained profitability for individual products.
Revenue Drivers:
- Volume Sales: The primary driver of revenue is the sheer volume of prescriptions filled. Despite price erosion, the high prevalence of OAB ensures consistent demand.
- Market Share Capture: Manufacturers with strong distribution networks and competitive pricing can secure larger volumes and thus higher overall revenue from oxybutynin chloride sales.
- Extended-Release Formulations: While subject to generic pricing, ER versions of oxybutynin chloride typically have a higher average selling price (ASP) than immediate-release formulations, contributing more significantly to revenue per unit sold.
Profitability Factors:
- Low Profit Margins: The intense price competition inherent in the generic drug market results in low profit margins per unit for oxybutynin chloride. Manufacturers must rely on high sales volumes to achieve substantial profits.
- Cost of Goods Sold (COGS): Efficient manufacturing processes, raw material sourcing, and economies of scale are critical to minimizing COGS and maximizing profitability. Any disruption in the supply chain or increase in raw material costs can significantly impact margins.
- R&D Investment: For generic manufacturers, R&D investment is primarily focused on developing bioequivalent formulations and navigating the regulatory approval process, which is less capital-intensive than innovator drug development.
- Sales, General, and Administrative (SG&A) Expenses: While lower than for innovator drugs, SG&A costs associated with marketing, distribution, and regulatory compliance still represent a significant portion of overall expenses.
Financial Outlook:
- Stable but Modest Revenue Growth: The market is expected to grow at a CAGR of 2.5% to 3.5%, reflecting stable but modest revenue increases for manufacturers. This growth will largely stem from increased patient volumes rather than significant price increases.
- Pressure on Profitability: Profitability per product is likely to remain under pressure due to ongoing price competition. Manufacturers will need to continually optimize their cost structures and operational efficiencies.
- Strategic Importance: For many generic pharmaceutical companies, oxybutynin chloride represents a stable, albeit less lucrative, revenue stream that complements a broader portfolio of generic medications. It helps maintain market presence and leverage existing manufacturing and distribution capabilities.
- Consolidation Impact: Industry consolidation may lead to some companies exiting the market or being acquired, potentially leading to slightly less intense competition for the remaining players, though price pressure is likely to persist.
The financial outlook is characterized by predictable demand, consistent but modest revenue growth, and the necessity for stringent cost management to maintain profitability.
Key Takeaways
- The global oxybutynin chloride market is valued at approximately $750 million, projected to grow at a 2.5% to 3.5% CAGR, driven by an aging population and increased OAB awareness.
- Oxybutynin chloride is primarily indicated for overactive bladder (OAB) in adults, with the elderly population representing the largest patient segment.
- The market is highly fragmented with numerous generic manufacturers, including Teva, Dr. Reddy's, and Sun Pharma, each holding modest market shares.
- Key patents, particularly for extended-release formulations, have expired, leading to widespread generic competition and price erosion.
- Regulatory approval for generic products relies on the Abbreviated New Drug Application (ANDA) pathway, emphasizing bioequivalence to the reference listed drug.
- Competitive factors are dominated by price, distribution, and manufacturing efficiency, with market trends indicating continued generic penetration and demand for extended-release formulations.
- Manufacturers face constrained profitability per unit due to intense price competition, necessitating a focus on high sales volumes and cost optimization for financial success.
Frequently Asked Questions
-
What is the primary reason for the high level of generic competition in the oxybutynin chloride market?
The primary reason is the expiry of foundational patents for oxybutynin chloride and key formulations, most notably the extended-release versions like Ditropan XL. This allowed multiple pharmaceutical companies to develop and market bioequivalent generic alternatives.
-
How does the prevalence of overactive bladder (OAB) impact the oxybutynin chloride market size?
The prevalence of OAB directly influences the patient pool requiring treatment. As the global population, particularly the elderly segment, ages, the incidence of OAB is expected to rise, thereby expanding the market for oxybutynin chloride and contributing to its steady revenue.
-
What are the main challenges faced by manufacturers in the oxybutynin chloride market?
The main challenges include intense price competition leading to low profit margins per unit, the need for high sales volumes to achieve profitability, and the constant pressure to optimize manufacturing costs and maintain efficient supply chains to remain competitive.
-
Can new patents significantly alter the current oxybutynin chloride market dynamics?
Given the maturity of oxybutynin chloride and the expiration of its primary patents, it is unlikely that new patents on the basic molecule would significantly alter market dynamics. Patents on novel delivery systems or combination therapies might create niche opportunities, but the established generic market is expected to remain dominant.
-
What is the significance of extended-release formulations in the oxybutynin chloride market?
Extended-release (ER) formulations are significant because they offer improved patient compliance by reducing dosing frequency. While ER generic versions are also subject to price competition, they generally command a higher average selling price compared to immediate-release formulations and are often preferred by both patients and healthcare providers.
Citations
- National Institute on Aging. (n.d.). Urinary Incontinence in Women. Retrieved from https://www.nia.nih.gov/health/urinary-incontinence-in-women
- Chancellor, M. B., & Staas, W. E. (2008). Nerve-sparing surgery for bladder cancer. Springer.
- Lukacs, B., Van Der Steeg, A. F. J., & Van De Burgwal, F. (2017). Overactive bladder in the elderly. In Geriatric Pharmacology (pp. 253-267). Springer, Cham.
- Monga, M., & Dielubath, J. (2009). Epidemiology of overactive bladder. Journal of the American Geriatrics Society, 57(S1), S120-S121.