Pre-ANDA Litigation Strategy: The Complete Playbook for Building Unbreakable Drug Patent Portfolios
For IP counsel, portfolio managers, R&D leads, and institutional investors navigating pharmaceutical patent exclusivity
Key Takeaways: Entire Article
- ANDA case filings fell roughly 36% from 2017 to 2021, with the steepest monthly drop (5.3 cases/month) occurring in the 2020-2021 period. The decline reflects more selective targeting by generics and denser patent thickets by innovators, not reduced competitive intensity.
- A Paragraph IV certification triggers a 45-day window for the NDA holder to sue; the lawsuit automatically imposes a 30-month stay on FDA final approval. Failing to file within that window forfeits the stay.
- Composition-of-matter (API) patents are the highest-value single asset in a pharmaceutical portfolio. Secondary patents (polymorph, formulation, method-of-use, process) function as a temporal extension mechanism, not merely defensive redundancy.
- First-to-file Paragraph IV ANDAs can earn 180-day generic exclusivity. That window, on a product with $1B+ in annual branded sales, routinely generates $200-400M in incremental gross profit for the first generic filer.
- IP valuation methodology for drug patents must account for patent term remaining, claim breadth, prosecution history estoppel, likelihood of validity sustaining litigation, and the probability-weighted present value of exclusivity cash flows.
- Authorized generics, launched by the NDA holder on Day 1 of generic entry, can cut the first-to-file generic’s exclusivity-period profit by 40-60%.
- The Federal Circuit’s 2021 decision in Biogen v. Mylan reinforced written description requirements for method claims, raising the evidentiary bar for patents claiming treatment of patient subpopulations. IP teams must audit method-of-treatment claims for written description compliance.
Section 1: The Economics of Pharmaceutical Patent Exclusivity
Why Patents Are the Core Financial Asset
Drug development economics run on a single, brutal calculus: spend $1.5-2.5 billion over 10-15 years per approved molecule, then recoup the investment within a patent-protected window of typically 8-12 years of commercial exclusivity. Patents are not ancillary legal instruments; they are the direct cash flow mechanism that makes the business model function.
The R&D-to-revenue conversion works only when the patent system holds. When patents fail, revenue collapses fast. A well-documented case: within 12 months of losing exclusivity on Pfizer’s atorvastatin (Lipitor) in November 2011, branded Lipitor’s U.S. market share fell from near-total to below 10% by unit volume, with per-unit prices declining more than 80%. Pfizer’s Lipitor franchise had generated approximately $125 billion in cumulative revenue over its exclusivity period. That scale is not replicated without the patent wall that held from 1997 until late 2011.
For generic companies, the mirror calculation applies. Winning a Paragraph IV challenge against a $1B+ product delivers asymmetric returns. Litigation costs for a full-scale ANDA suit typically run $5-15 million per party through trial. The potential first-mover reward on a high-value product dwarfs that cost by two orders of magnitude, which is why well-capitalized generics pursue these cases aggressively even against strong patent positions.
The Patent Cliff and the Slope: A Financial Model
IP teams and portfolio managers use two distinct models to describe loss of exclusivity (LOE) events. A ‘patent cliff’ describes abrupt, simultaneous LOE when the primary composition-of-matter patent expires with no secondary protection layered behind it. A ‘patent slope’ describes a managed decline in which secondary patents stagger the entry of competing generics, extending meaningful exclusivity revenues by two, four, or sometimes six additional years beyond primary patent expiration.
The financial difference between cliff and slope scenarios is substantial. A product generating $3B annually that hits a pure cliff loses roughly 70-80% of that revenue within 18 months. The same product with secondary patents covering the commercially dominant extended-release formulation (valid for four additional years) retains meaningful revenue from the IR-to-XR conversion, limits generic substitution at the formulary level, and generates bridge cash flows. Modeling this distinction is standard in pharma equity analysis and informs investor positioning 3-5 years ahead of LOE.
Key Takeaways: Section 1
- Peak-year branded revenue and effective patent term (not nominal patent term) are the two variables that most determine IP asset value.
- Cliff versus slope analysis drives LOE financial modeling; secondary patent portfolios are the mechanism that converts a cliff into a slope.
- Generic first-filer economics are compelling enough to motivate well-funded litigation even against substantial innovator patent estates.
Section 2: Understanding the Hatch-Waxman Litigation Framework
The Regulatory Architecture
The Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act) created the structural framework that governs pharmaceutical patent litigation in the United States. The Act established the ANDA pathway, which permits generic applicants to reference an innovator’s NDA safety and efficacy data rather than conduct independent clinical trials, provided the generic demonstrates bioequivalence to the reference listed drug (RLD).
The patent-linkage mechanism embedded in Hatch-Waxman requires ANDA filers to certify their relationship to each Orange Book-listed patent covering the RLD. A Paragraph I certification states the patent has expired; Paragraph II states it will expire before the ANDA approval date; Paragraph III certifies the generic will launch only after patent expiration; and Paragraph IV certifies that the listed patent is invalid, unenforceable, or will not be infringed by the generic product. Paragraph IV is the only certification that triggers immediate patent litigation rights.
Upon receipt of a Paragraph IV notice letter from the generic applicant, the NDA holder and patent owner have 45 days to file an infringement suit in federal district court. Filing within that window automatically triggers a 30-month stay on FDA final approval of the ANDA. That stay is not a litigation win; it is a temporary hold that expires in 30 months regardless of case outcome unless the court enters final judgment before the stay expires. Missing the 45-day window forfeits the 30-month stay, meaning FDA may grant final approval while litigation proceeds, a serious strategic and financial consequence.
The Orange Book: Listing Strategy and Its IP Implications
Patents eligible for Orange Book listing are those claiming the drug substance (API), drug product (formulation), or method of use. Process patents, metabolite patents, and intermediate patents are not eligible for listing. This distinction matters enormously: a manufacturing process patent that would otherwise block generic entry has no automatic Hatch-Waxman litigation trigger, because it does not appear in the Orange Book. Innovators sometimes overlook the strategic importance of maximizing Orange Book coverage within the bounds of 21 C.F.R. 314.53.
Listing decisions carry strategic risks. Improper listing of patents that do not claim the approved drug product or an approved method of use can expose the NDA holder to antitrust liability under the Walker Process doctrine or to abuse-of-process claims. The FTC has scrutinized Orange Book listing practices; in 2023, the agency began formally disputing listed patents under a new dispute resolution process created by the Consolidated Appropriations Act of 2023. IP teams now need to evaluate Orange Book listing decisions not only for coverage strategy, but for vulnerability to FTC challenge.
The 180-Day Generic Exclusivity Mechanism
The first ANDA applicant to file a substantially complete application containing a Paragraph IV certification against a given patent earns 180-day marketing exclusivity, the right to be the only approved generic on the market for 180 days after first commercial marketing. This exclusivity is specific to each patent challenged; an applicant can be first-to-file on some patents and not others for the same drug.
Several forfeiture events can strip a first-filer of its 180-day exclusivity before it is triggered. These include failure to market within 75 days of court entry of a final judgment of non-infringement or invalidity, failure to market within 75 days of final FDA approval (absent a court order), and withdrawal or amendment of the Paragraph IV certification. Generic companies must actively manage the forfeiture clock and should have contingency commercial launch plans ready before a court decision issues.
A frequently mismodeled scenario involves ‘shared’ 180-day exclusivity when multiple ANDA filers submit substantially complete applications on the same day. In that case, all same-day first-filers share the exclusivity period, reducing the profit potential for each. The competitive dynamics of same-day filings create pressure to accelerate formulation development timelines, sometimes at the cost of product quality or regulatory readiness.
Patent Term Extension Under Hatch-Waxman
Hatch-Waxman also provides a mechanism for innovators to recover patent term lost during FDA regulatory review. Under 35 U.S.C. 156, the NDA holder may apply for a Patent Term Extension (PTE) of up to five years, subject to a cap that the total remaining patent term after extension does not exceed 14 years from the date of FDA approval. Only one patent per approved product may receive a PTE, and the application must be filed within 60 days of NDA approval.
PTE strategy requires early coordination between IP and regulatory affairs. Choosing which patent to extend — typically the composition-of-matter patent, though sometimes a formulation patent with later natural expiry — requires modeling the commercial contribution of each candidate patent throughout the product’s LOE timeline.
Key Takeaways: Section 2
- The 45-day window to file suit and trigger the 30-month stay is a hard deadline with no cure; missing it is a significant strategic failure.
- Orange Book listing strategy now requires FTC risk analysis alongside coverage analysis.
- PTE selection (one patent per product) is an irreversible decision requiring cross-functional modeling.
- Same-day first-filer scenarios require generic companies to model shared 180-day exclusivity economics, not solo exclusivity, as the base case.
Section 3: Strategic Patent Portfolio Architecture
Layer 1: Composition-of-Matter Patents and Markush Claim Strategy
The API patent is the highest-value single IP asset in a pharmaceutical portfolio. It claims the active compound itself, and when valid, blocks any generic product containing that compound regardless of formulation, dose, or indication. The standard 20-year patent term, eroded by the development timeline, typically delivers 8-12 years of effective commercial exclusivity post-approval. For a high-revenue product, that period can generate $10-50B+ in cumulative net present value, making the API patent the dominant driver of enterprise valuation in innovator-focused pharma companies.
Claim scope in composition-of-matter patents hinges on the breadth of the Markush group. A well-drafted Markush structure defines a core scaffold with variable substituent groups at defined positions, covering not just the clinical compound but a functionally related set of analogs that might otherwise represent design-around opportunities. The strategic tension is between breadth (covering more analogs, creating a wider moat) and enablement (the specification must teach how to make and use the full claimed genus). Post-Amgen v. Sanofi (Supreme Court 2023), functional claiming of antibody classes without full structural disclosure faces heightened enablement scrutiny. Biologics IP teams must restructure Markush-equivalent claims for antibodies to depend on specific structural features rather than functional properties alone.
Layer 2: Salt, Polymorph, and Co-Crystal Patents
Pharmaceutical APIs routinely exist in multiple solid-state forms: free base, salt forms, amorphous state, and various crystalline polymorphs. Each form may differ in solubility, stability, melting point, and bioavailability, and each distinct form is potentially patentable independently. Polymorph and salt patents are a standard lifecycle extension tool. AstraZeneca’s esomeprazole (Nexium) is the textbook case: the compound patent on omeprazole (Prilosec) expired, but AstraZeneca developed and patented the S-enantiomer separately, with formulation and polymorph patents layered on top, generating billions in additional exclusivity revenue.
The legal risk with polymorph patents is obviousness. Courts assess whether a person of ordinary skill in the art would have had reason to make and test the specific polymorph and a reasonable expectation of success. The 2020 Federal Circuit decision in Acadia Pharmaceuticals v. Aurobindo Pharma found certain polymorph claims obvious where the prior art disclosed standard screening procedures that would predictably yield the claimed crystal form. IP teams conducting pre-ANDA vulnerability assessments must apply this standard when auditing their own polymorph patent positions.
Layer 3: Formulation and Drug Delivery Patents
Formulation patents cover specific pharmaceutical compositions, including the combination of the API with excipients, particle size specifications, coating systems, and complex drug delivery architectures (osmotic pumps, matrix tablets, nanoparticle systems). These patents are valuable because the commercially dominant dosage form — almost always the one covered by the NDA — must match the formulation described in the patent for generic bioequivalence testing to even raise an infringement question.
Extended-release (ER) and modified-release (MR) formulation patents are particularly powerful lifecycle management tools. A drug that transitions its commercial base from an immediate-release formulation (with an expiring API patent) to a once-daily ER formulation covered by a valid formulation patent can defend market share for several additional years post-API LOE. The key clinical requirement is demonstrating a real therapeutic benefit from the ER version — improved tolerability, reduced dosing frequency with measurable compliance benefits, or a more favorable PK profile — because courts give less weight to secondary considerations of non-obviousness when the only advantage claimed is patient convenience.
Layer 4: Method-of-Treatment Patents
Method-of-treatment patents protect specific therapeutic uses, dosing regimens, patient population subsets, or combination therapies. They do not bar the generic from making the molecule, but they can prevent FDA from approving an ANDA with labeling that ‘carves in’ the patented method. Skinny labeling — an ANDA with carve-outs omitting patented indications — is the generic response, but it requires a careful reading of whether the proposed label nonetheless ‘induces’ infringement of the method patent.
The Federal Circuit’s 2021 ruling in GlaxoSmithKline v. Teva on induced infringement by skinny labeling created significant uncertainty. The court found that Teva’s skinny-labeled carvedilol ANDA could still induce infringement of GSK’s method-of-treatment patent because Teva’s overall marketing communications pointed toward the patented use. The case returned to the Federal Circuit and was ultimately vacated, but the litigation demonstrated that skinny labeling is not a bulletproof design-around strategy for method-of-treatment patents with broad commercial use.
Layer 5: Manufacturing Process Patents
Process patents cover the synthetic routes, purification methods, or manufacturing technologies used to produce the API or drug product. They do not appear in the Orange Book and therefore do not trigger Hatch-Waxman litigation directly. Their strategic value lies in cost-of-goods protection: a proprietary low-cost synthesis route that a generic company cannot lawfully replicate may raise generic manufacturing costs enough to deter entry on lower-revenue products, even after the API patent expires.
Proving process patent infringement is harder than compound infringement because the infringing act (using the process) occurs inside the generic manufacturer’s facility, often in another country. Under 35 U.S.C. 271(g), importing a product made abroad by a patented process can infringe, but there are defenses when the product has been materially changed through subsequent processes. Process patent enforcement is best viewed as a deterrence layer rather than a primary litigation weapon.
Key Takeaways: Section 3
- Post-Amgen v. Sanofi, functional Markush claims for biologics require structural disclosure to survive enablement challenges.
- Polymorph patents face heightened obviousness scrutiny under Acadia v. Aurobindo; audit existing polymorph positions before generic filers do.
- Skinny labeling is a viable but imperfect design-around for method-of-treatment patents; GlaxoSmithKline v. Teva demonstrated the induced infringement risk from commercial labeling context.
- Process patents are deterrence assets, not primary Orange Book enforcement tools.
Section 4: IP Valuation Methodology for Drug Patent Assets
Why Standard DCF Fails for Pharmaceutical Patents
Standard discounted cash flow (DCF) models applied to pharmaceutical revenue streams systematically misvalue IP assets because they apply a single revenue ramp-up and single LOE scenario without adequately weighting the probability distributions of patent validity, patent term remaining, claim scope, and litigation outcome. A patent-adjusted valuation model requires breaking the cash flow model into patent-segment scenarios.
The key inputs for IP valuation are: effective patent term remaining (adjusted for PTE and any applicable regulatory exclusivity), claim breadth score (a subjective but defensible assessment of how much of the commercial product is covered by the broadest valid claim), prosecution history estoppel analysis (which portions of claim scope have been surrendered during prosecution), prior art density (how many close prior art references exist that a generic challenger could deploy), and litigation survival probability. Each variable affects the probability-weighted net present value of the exclusivity cash flows.
The Royalty Relief Method for Patent Portfolio Valuation
The royalty relief method is the most widely used income-based approach for standalone patent valuation. It asks: if the company did not own these patents and had to license the equivalent IP from a third party, what royalty rate would it pay, and what is the present value of avoiding that royalty payment? Industry royalty rates for pharmaceutical API patents typically range from 3-8% of net sales for standard small molecules, 5-15% for novel formulations with demonstrated clinical differentiation, and 8-20%+ for biologic platform technologies with first-in-class exclusivity.
For ANDA litigation strategy purposes, the royalty relief value sets a floor for settlement negotiations. An innovator whose patents would yield $1B in royalty relief value over the remaining patent term has a rational negotiating floor around that figure, adjusted for litigation cost and win probability. Generic companies conducting pre-ANDA due diligence use the mirror calculation: the value of winning early market entry is the present value of the incremental profit from launching before patent expiration, which in turn sets the ceiling for litigation investment.
IP Valuation in M&A and Licensing Transactions
Pharmaceutical M&A transactions are fundamentally IP acquisitions. When Pfizer acquired Array BioPharma in 2019 for $11.4B, the primary asset was binimetinib and encorafenib (Braftovi/Mektovi) and their associated patent estates — specifically, the composition-of-matter patents and a growing body of combination therapy method patents in melanoma and colorectal cancer. The patent estate’s depth and the remaining term on key claims directly determined the multiple paid.
In licensing negotiations (both in-license by innovators and out-license by biotechs), IP valuation determines the royalty rate and milestone structure. The most common error in pharma licensing valuation is attributing all revenue upside to IP when a substantial portion of revenue derives from manufacturing know-how, regulatory data exclusivity, or brand equity. Separating IP value from know-how and regulatory asset value is necessary for accurate deal structuring and audit-proof transfer pricing.
Key Takeaways: Section 4
- Probability-weighted patent validity and litigation survival rates must be explicitly modeled; a single-scenario DCF overstates IP value for legally vulnerable patents.
- The royalty relief method is the standard for pharmaceutical patent valuation and sets rational floors (innovator) and ceilings (generic) for litigation settlement negotiation.
- In M&A, separating IP value from know-how and regulatory data exclusivity is required for accurate deal pricing and tax compliance.
Section 5: Evergreening Roadmap — Patent Lifecycle Extension Tactics
The term ‘evergreening’ is applied broadly, sometimes pejoratively, to any strategy that extends pharmaceutical market exclusivity beyond the expiration of the primary API patent. Regulators and courts have grown more hostile to evergreening tactics that lack genuine clinical differentiation, but the practice remains legal and commercially essential when executed on the basis of real innovation. The following is a technical roadmap of the major tactics.
Tactic 1: New Formulation or Drug Delivery System
The commercial transition from an immediate-release tablet to an extended-release formulation, or from an oral tablet to a transdermal patch, can secure 3-5 years of additional effective exclusivity if the new formulation is patentable and clinically preferred. The requirement is that the new formulation must offer a demonstrable therapeutic advantage — not just convenience — to withstand an obviousness challenge. Companies that execute this transition successfully typically run a Phase 3 equivalency or superiority trial comparing the new formulation to the original to generate data demonstrating the unexpected benefit.
Successful execution also requires FDA approval of the new formulation with a clean NDA supplement, combined with a commercial strategy that accelerates prescriber conversion before the IR formulation goes generic. Manufacturers who do not achieve >70-80% branded prescribing share in the ER formulation before IR LOE face a revenue cliff despite the extended patent protection.
Tactic 2: Pediatric Exclusivity
Under the Best Pharmaceuticals for Children Act (BPCA), the FDA may grant six months of additional marketing exclusivity for any drug (regardless of indication) whose sponsor conducts FDA-requested pediatric studies. This exclusivity attaches to all Orange Book-listed patents for the drug, effectively extending the 30-month stay window and all other Hatch-Waxman exclusivities by six months. Pediatric exclusivity is one of the most cost-effective IP extensions available. A six-month extension on a $2B annual revenue product is worth approximately $1B in gross revenue — often exceeding the cost of the pediatric studies by 10-20x.
Tactic 3: New Indication / Method-of-Use Patent
Filing and obtaining method-of-use patents for additional indications discovered post-approval is standard lifecycle management. The new indication must be genuinely novel — the prior art must not have disclosed or suggested the use. Filing a new sNDA for the new indication simultaneously creates regulatory data exclusivity (three years for a new condition of use with new clinical investigations) while the patent application moves through prosecution.
Combination therapy patents represent a sophisticated variant: discovering that the existing API works synergistically with a second agent and patenting the combination method can extend commercial relevance even when the base compound’s patents expire.
Tactic 4: Enantiomer and Metabolite Patents
Where the original drug is a racemate, the active enantiomer may be separately patentable. AstraZeneca’s shift from omeprazole to esomeprazole (the S-enantiomer) is the most-cited precedent. The key legal question is whether the active enantiomer was inherently described in the racemate prior art; if so, separatability is insufficient for novelty. The USPTO and courts have split on where to draw this line depending on whether the enantiomer’s specific properties were ‘known’ from the racemate disclosure.
Active metabolites of a drug may similarly be separately patentable if they have distinct therapeutic properties not obvious from the parent compound. The regulatory analog is developing the metabolite as a standalone NDA (as was done with fexofenadine from terfenadine), which resets the development timeline and can yield a full independent patent estate.
Tactic 5: Fixed-Dose Combination (FDC) Products
Developing a fixed-dose combination of the existing API with a complementary agent generates its own patent estate (covering the combination) and NDA (with new data exclusivity). FDCs can reshape the prescribing landscape for the original drug. The HIV antiretroviral market — Gilead’s Atripla, Complera, Stribild, and Genvoya franchise — is the clearest case study where successive FDC development extended commercial dominance years past what individual component patents would have allowed.
Technology Roadmap: Biologic Evergreening
Biologics follow a different evergreening logic than small molecules, because the Biologics Price Competition and Innovation Act (BPCIA) provides 12 years of reference product exclusivity from first FDA approval, independent of patent status. Patent protection on biologics is substantially broader in scope but harder to enforce precisely, because no two manufacturers produce identical biologic structures — biosimilars are ‘highly similar,’ not identical, to the reference product.
The biologic evergreening roadmap includes: (1) reformulating the reference biologic into a subcutaneous (SC) administration format when the original is intravenous (IV), patenting the SC formulation and the delivery device; (2) developing next-generation constructs (e.g., antibody-drug conjugates, bispecific antibodies) that are patentably distinct from the reference biologic; (3) obtaining new indication approvals that generate additional method-of-treatment patents and regulatory exclusivity periods; (4) patenting specific manufacturing process improvements that biosimilar manufacturers cannot lawfully copy.
AbbVie’s management of adalimumab (Humira), the world’s best-selling drug for over a decade, is the definitive case study. AbbVie built a patent estate of more than 130 patents on Humira, covering formulation, method of use, manufacturing, and the citrate-free SC formulation specifically. This thicket delayed U.S. biosimilar entry until January 2023, roughly eight years after EU biosimilar entry began. Whether this constitutes ‘evergreening’ or legitimate IP protection on genuine innovations is actively debated, but the financial outcome is unambiguous: AbbVie generated approximately $20B in additional U.S. Humira revenue during the years biosimilar entry was delayed domestically versus Europe.
Key Takeaways: Section 5
- Pediatric exclusivity is among the highest-ROI lifecycle extension tools available: six months of extension on a multi-billion dollar product often yields 10-20x the cost of the qualifying studies.
- Enantiomer and metabolite patents remain viable but require careful novelty analysis against the parent compound’s prior art.
- The Humira patent estate is the canonical example of biologic evergreening: 130+ patents, 8+ years of U.S. entry delay beyond EU entry.
- Biologic evergreening is structurally different from small-molecule evergreening because BPCIA reference exclusivity runs independently of patents.
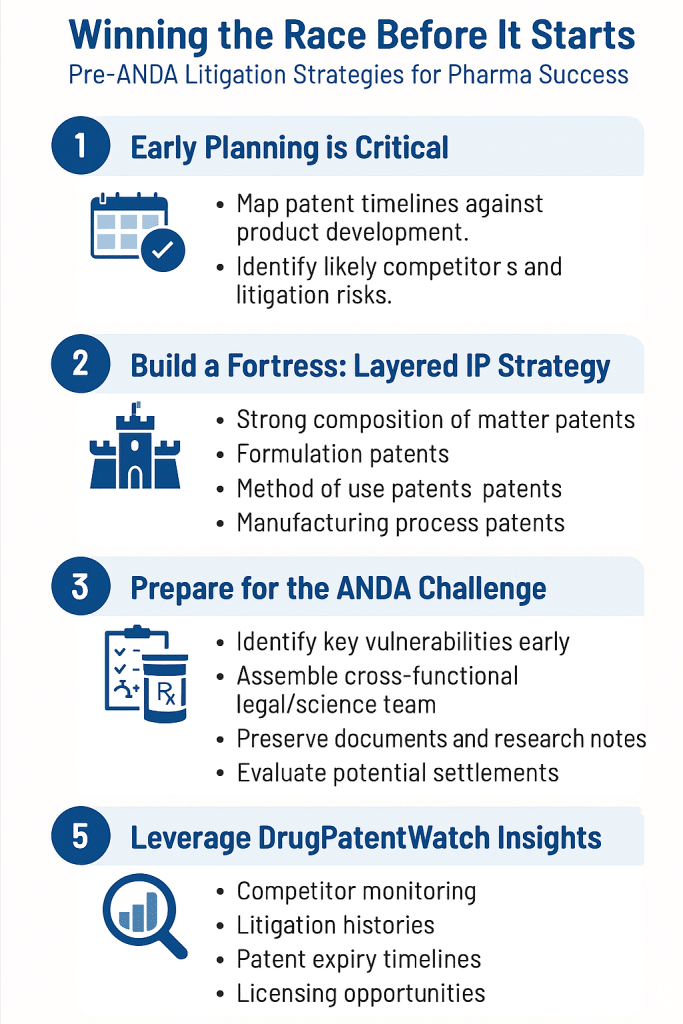
Section 6: Pre-ANDA Preparation for Innovator Companies
Portfolio Vulnerability Assessment: The Mock Invalidity Audit
Proactive innovators run mock invalidity analyses on their own key patents before any generic files. This is not a defensive reflex; it is a standard risk management practice that allows companies to identify and remediate weaknesses before a Paragraph IV notice letter arrives. The process mirrors what a generic company’s IP team would do: identify the three to five most commercially critical patents, conduct exhaustive prior art searches with an invalidation-focused mindset, apply claim construction under the Phillips standard, and stress-test each claim element against anticipation, obviousness (particularly obvious-to-try arguments under KSR), written description, and enablement.
Written description challenges have become more dangerous since Ariad Pharmaceuticals v. Eli Lilly (Fed. Cir. 2010), which confirmed that claims broader than what the specification adequately describes will fail the written description requirement even if technically enabled. IP teams auditing method-of-treatment claims covering broad patient populations must verify that the specification provides adequate written description for the full scope of the claimed population, not just the patients studied in the disclosed examples.
Prosecution history estoppel review is equally essential. During prosecution, every argument made to distinguish prior art and every claim amendment can narrow the scope of the doctrine of equivalents. A claim element surrendered during prosecution cannot be recaptured through the equivalents doctrine. IP teams must map prosecution histories to commercial product features and assess whether the commercial product’s key differentiated properties fall within the literal claim or require the doctrine of equivalents.
Patent Continuation Strategy
A robust continuation filing program allows innovators to maintain living ‘child’ applications from the original parent patent, with the ability to tailor claim scope to cover newly discovered generic design-around strategies. Continuation applications must be filed before the parent abandons and claim priority to the original filing date. Because they can be drafted to address specific product configurations that were not fully claimed in the parent, they are a dynamic tool for closing gaps identified during vulnerability assessments.
The key constraint is prosecution laches. If an innovator delays prosecution of continuation applications for an unreasonable and inexcusable time and the delay prejudices the generic’s investment in developing the design-around, a court may decline to enforce the continuation claims. The standard applied by the Federal Circuit for prosecution laches is high, but applications that sit in prosecution for 8-10+ years without advancement draw scrutiny.
The 21 C.F.R. 314.53 Patent Submission Strategy
NDA holders must submit patent information to FDA for Orange Book listing within 30 days of patent issuance for patents that issue after NDA approval (or at the time of NDA submission for patents issued before approval). The strategic decision of which patents to list is not trivial. Listing maximizes litigation leverage by triggering Paragraph IV certifications and the 30-month stay. Not listing can allow certain secondary patents to pursue enforcement outside the Hatch-Waxman framework, though this forfeits automatic stay rights.
FDA has limited authority to verify that listed patents meet the listing requirements; the agency generally accepts the NDA holder’s certification. However, as noted above, the FTC’s 2023 dispute resolution mechanism and the threat of Walker Process antitrust claims provide real disincentives for listing clearly ineligible patents. Listing a patent that does not claim the approved drug product to create an unjustified 30-month stay can expose the NDA holder to antitrust damages.
Early Warning: Monitoring Generic Activity
Generic companies signal their intentions well before a Paragraph IV filing. Key intelligence signals include: DMF (Drug Master File) submissions to FDA for the API, which are public; ANDA-related formulation patent filings by known generic manufacturers; published synthesis route papers from academic groups tied to generic manufacturers; and generic company hiring patterns in formulation development for specific therapeutic areas. Monitoring these signals through patent database surveillance and FDA DMF tracking allows innovators to anticipate likely challengers and timeline generic entry 12-24 months before a Paragraph IV notice arrives.
Key Takeaways: Section 6
- Mock invalidity audits are standard practice at sophisticated innovators; the first time you stress-test your own patents should not be in response to a Paragraph IV notice letter.
- Continuation programs must be actively advanced; prosecution laches is a real risk for applications sitting idle for extended periods.
- FDA DMF filings and generic patent activity are reliable early signals of Paragraph IV preparation; monitoring them is basic competitive intelligence.
Section 7: Pre-ANDA Strategy for Generic Challengers
Target Selection: The Product Opportunity Screening Model
Not every expiring patent is worth challenging. Rational generic companies run a quantitative screening model to rank opportunities by expected risk-adjusted return. The inputs include: branded annual U.S. net revenue (the total prize), number of Orange Book-listed patents (a proxy for litigation complexity and cost), whether a first-to-file 180-day exclusivity is available, number of competing ANDA filers already in the queue, technical feasibility of the formulation (complexity, specialized equipment requirements, API supply chain availability), and probability of finding a valid invalidity argument or non-infringement position.
Products above $500M in annual U.S. branded sales with fewer than five Orange Book patents and at least one patent with identifiable prior art vulnerabilities represent the most attractive targets for a Paragraph IV strategy. Products with 10+ Orange Book patents spread across compound, polymorph, formulation, and method-of-use claims require multi-front litigation budgets and longer timelines, which compress the effective value of early market entry.
Freedom-to-Operate Analysis: Scope and Methodology
A thorough freedom-to-operate (FTO) analysis for an ANDA target goes beyond Orange Book patents. It includes: all continuation and divisional applications from the innovator’s patent family (which may issue as patents before or after ANDA approval); process patents that might cover the API synthesis route; patents on manufacturing equipment or technology; and patents held by third parties on excipients or delivery system components. Missing a late-issuing continuation that claims the generic’s specific formulation is a costly error — potentially triggering injunctions post-approval.
The FTO analysis workflow: (1) Orange Book patent claim mapping against the proposed ANDA product; (2) family-level prosecution history review for each listed patent; (3) continuation application monitoring for unpublished applications in the family; (4) non-listed patent search across the innovator’s entire portfolio for the active molecule; (5) API supplier IP review; (6) process patent identification and design-around assessment.
The ‘Skinny Label’ Strategy: Execution and Risk
Where the Orange Book includes method-of-use patents but not compound or formulation patents, generic applicants can seek approval for the carve-out label — a label that omits the patented indication. This allows the generic to be approved and marketed for non-patented uses while the innovator retains exclusivity over the patented indication. The legal basis is 21 C.F.R. 314.127(a)(7), which permits omission of patented use information from the proposed labeling.
Skinny labeling works cleanly when the patented indication is clinically distinct from the primary use. It breaks down when the patented indication is the dominant commercial use and prescribers use the generic product ‘off-label’ for the patented indication. Post-GlaxoSmithKline v. Teva, generic companies deploying skinny labeling must audit their promotional materials and product communications to avoid creating an evidentiary record of intent to induce infringement of the carved-out method patent.
Authorized Generic Risk: Financial Modeling
Before committing to a Paragraph IV filing, generic companies must explicitly model the authorized generic (AG) scenario. An NDA holder can launch an AG — a drug product identical to the brand, sold under the generic’s NDA or through a contract manufacturer — immediately upon generic approval. The AG competes directly with the first-to-file generic during its 180-day exclusivity period, with the NDA holder capturing some of the exclusivity economics.
Academic studies have found that authorized generic competition during the 180-day exclusivity period reduces the first-filer’s gross profit by 40-60% compared to the no-AG scenario. Companies that factor in this possibility as the default — rather than assuming they will have the exclusivity period to themselves — make more disciplined filing decisions. The calculus changes when the innovator has publicly committed not to launch an AG (sometimes a settlement term) or when the product’s manufacturing complexity makes AG launch operationally difficult for the NDA holder.
Settlement Agreements: The Reverse Payment Problem
ANDA litigation frequently settles, often with an agreement that the generic can enter the market on a date certain before patent expiration, in exchange for dropping the invalidity and non-infringement challenge. These settlements can be structured so the innovator pays the generic (directly or indirectly, through royalty-free supply agreements, co-promotion rights, or cash payments) to stay out of the market. The Supreme Court’s 2013 decision in FTC v. Actavis held that ‘reverse payment’ settlements are subject to antitrust scrutiny under the rule of reason, not per se legal as the pharmaceutical industry had previously argued.
Post-Actavis, settlement negotiations require counsel to evaluate whether the settlement terms include implicit or explicit reverse payments that would exceed a reasonable attorney’s fee reimbursement or reflect value related to the generic’s agreement to delay entry. The risk of antitrust exposure has made some litigation parties more cautious about settlement terms, while others have become more creative in structuring consideration to avoid the appearance of a cash payment.
Key Takeaways: Section 7
- The product opportunity screening model should explicitly factor in the AG risk; 40-60% profit compression during the 180-day exclusivity period is the realistic base case when the innovator is capable of AG launch.
- FTO analysis must extend beyond Orange Book patents to late-issuing continuation applications in the innovator’s patent family.
- Post-Actavis, reverse payment settlements require antitrust analysis; the risk of FTC or private plaintiff challenge is real and should be modeled in settlement negotiations.
Section 8: Expert Witness Selection and Preparation
The Expert’s Role in ANDA Cases
ANDA litigation is fundamentally a battle of expert testimony. The substantive issues — claim construction, obviousness, enablement, written description, and infringement — all turn on the perspective of ‘a person of ordinary skill in the art’ (POSA). Courts do not have independent scientific expertise; they rely on expert witnesses to define the POSA standard, interpret technical evidence, and apply legal tests to complex chemistry and pharmacology.
The selection of testifying experts is one of the highest-stakes decisions in pre-ANDA preparation. A weak expert — technically competent but unable to communicate clearly, or with a compromised publication history — can lose a case that the legal team has spent years building. An unusually strong expert can survive aggressive cross-examination and provide testimony that shapes the court’s understanding of the science in the innovator’s favor.
Expert Qualification and Vetting
The technical qualifications required depend on the patents at issue. Invalidity cases require experts with deep academic or industrial expertise in medicinal chemistry, pharmacokinetics, formulation science, or process chemistry, depending on the patent type. Infringement cases may require different experts for claim construction (often a laboratory scientist) and damages (typically a health economist or industry expert).
Vetting must include: review of all prior publications for statements inconsistent with the positions they will take in the case; review of all prior expert engagements and deposition and trial testimony transcripts for positions they have previously taken; identification of professional and financial relationships with parties or counsel that could appear as bias; and assessment of how they present in deposition — how they handle hostile cross-examination questions, whether they are evasive or direct, whether they can maintain composure.
Daubert Gatekeeping and Expert Report Requirements
Under Federal Rule of Evidence 702 and the Daubert standard (Daubert v. Merrell Dow Pharmaceuticals, 1993), district courts act as gatekeepers excluding expert testimony that is not sufficiently reliable or does not fit the facts of the case. In ANDA litigation, Daubert challenges are frequently filed against opposing experts and are decided before trial, potentially eliminating key testimony.
Expert reports must comply with Federal Rule of Civil Procedure 26(a)(2)(B): a complete statement of all opinions, the basis and reasons for those opinions, and identification of the facts and data considered. Courts are strict about limiting experts to opinions disclosed in their reports; new opinions surfacing at deposition or trial are subject to exclusion. Expert reports in complex ANDA cases routinely run 100-300 pages with extensive claim charts and citations to scientific literature. The preparation process should begin 6-9 months before the report deadline.
Key Takeaways: Section 8
- Expert selection is a litigation-outcome variable, not an administrative task; invest in vetting as rigorously as in brief writing.
- Daubert gatekeeping is a real procedural risk; expert methodologies must be reliable under FRE 702 standards from the moment of engagement, not retrofitted before the hearing.
- Expert reports must be comprehensive and final; supplementation is difficult and surface-level inconsistencies between report and deposition are a cross-examination gift to opposing counsel.
Section 9: Trademark, Trade Dress, and Brand Strategy Post-LOE
Brand equity built during the patent protection period is the most durable post-LOE asset in an innovator’s commercial toolkit. Trademarks on the brand name, logos, and distinctive packaging survive patent expiration and remain enforceable indefinitely as long as the brand is in use. A physician who has prescribed a brand-name product for 10 years and is familiar with its clinical profile will not automatically convert to the generic on the day of LOE, particularly in therapeutic areas with narrow therapeutic windows or complex dosing.
The ‘product hopping’ strategy — replacing the established branded product with a new formulation under a new or reformulated product name just before LOE — can extend formulary preference for the reformulated product if it is clinically differentiated. The antitrust risk is real: courts have found that product hopping can constitute anticompetitive conduct when the reformulation has no medical benefit and the sole purpose is to impede generic substitution.
Trade dress protection for pill shape, color, and size is more limited. The FDA approved generic drug labeling requirements generally allow generics to use distinctive trade dress that consumers or healthcare providers associate with the brand, subject to exceptions for functional features. However, color-coded pill identification systems, proprietary device features (injectors, inhalers, patch sizes), and packaging innovations can be protected through trade dress registration when they are non-functional and have acquired secondary meaning.
Section 10: In-Licensing, Acquisition, and Portfolio M&A
IP Due Diligence Framework for Pharmaceutical Transactions
Pharmaceutical M&A due diligence is primarily IP due diligence. The acquiring party must assess: (1) patent validity risk for each key asset (invalidity analyses of the composition-of-matter and primary formulation patents); (2) freedom-to-operate of the acquired products, including third-party patents that might require licensing; (3) remaining patent term for each material asset; (4) any pending ANDA challenges or Paragraph IV certifications already filed; (5) consistency of patent claims with approved labeling; and (6) any outstanding patent office proceedings (IPR petitions, reexaminations) that could invalidate key claims.
For late-stage acquisitions where Paragraph IV certifications have already been filed, the acquirer must evaluate the status of ongoing litigation and the quality of the defense position. Acquiring a product mid-litigation without a clear picture of the litigation risk is a common error in rushed transactions. The acquirer inherits the litigation in its current posture, including any adverse claim construction rulings or damaging deposition testimony already on the record.
Licensing Strategy: Royalty Rates and Term Structures
Licensing royalty rates in pharmaceutical transactions vary substantially by asset type and development stage. Early-stage small molecule licenses (pre-Phase 2) typically carry 2-5% running royalties plus milestone payments. Late-stage or approved product licenses command 8-15%+ on net sales. Biologic platform licenses, where the licensor’s antibody engineering or protein expression technology underlies multiple potential products, are structured with both product-specific royalties and platform access fees.
Patent term alignment is critical in license structuring: the license term should be coextensive with the life of the licensed patents, with provisions addressing what happens when individual patents expire and the licensor’s IP protection narrows. Failure to address patent expiry in the license agreement can create disputes about whether royalty obligations survive the expiration of the claims that originally justified the royalty rate.
Section 11: Clinical Trial Design as a Patent Fortification Tool
Generating Secondary Considerations of Non-Obviousness
Secondary considerations — commercial success, long-felt need, failure of others, unexpected results, and copying by competitors — can overcome an obviousness rejection or invalidity challenge when they are probative and tied to the specific patent claims at issue. Clinical trial design can deliberately generate evidence for secondary considerations.
‘Unexpected results’ is the secondary consideration most directly tied to trial design. If a trial demonstrates that the claimed compound achieves a therapeutic effect qualitatively different from (not merely quantitatively better than) what the prior art would have predicted, that is potentially compelling evidence of non-obviousness. The requirement is that the result be unexpected to a POSA, not merely surprising to lay observers. Trial protocols designed with this goal must pre-specify the comparison that demonstrates the unexpected result; post-hoc subgroup analyses rarely satisfy courts as a basis for secondary considerations.
‘Long-felt need’ evidence comes from the clinical literature documenting years of unsuccessful attempts to solve the problem the patented compound addresses. The patent prosecution team should compile this record systematically during development, because demonstrating long-felt need after the fact — searching for publications that documented the unmet need years before the compound was developed — is reactive and incomplete compared to contemporaneous documentation.
Clinical Data and Orange Book Listing Interaction
FDA-approved labeling reflects the specific formulation, dose, patient population, and indications studied in the submitted clinical trials. Each specific element of the approved label that reflects a novel clinical discovery is a potential method-of-treatment patent claim. Companies that deliberately design clinical trials to generate novel labeling elements — specific pharmacokinetic parameters, distinct patient population criteria, particular dosing titration protocols — create the raw material for method-of-treatment patent applications that can be filed after trial completion based on the clinical findings.
Section 12: Emerging Technology and Biologic IP Strategy
Antibody Patent Strategy Post-Amgen v. Sanofi
The Supreme Court’s unanimous 2023 ruling in Amgen v. Sanofi upended the conventional approach to antibody claiming in biologics. The Court invalidated Amgen’s broadly functional PCSK9 antibody claims — which covered all antibodies that bind to the PCSK9 protein at a specified region and block its activity — because the specification disclosed only 26 specific antibodies while claiming a genus of potentially millions. The Court applied a strict enablement test: the specification must enable the full scope of the claimed invention without undue experimentation.
Post-Amgen, antibody patents must be structurally specific. Claims that define an antibody by its CDR sequences, its three-dimensional binding epitope, or specific structural-functional relationships survive better than claims that define an antibody solely by what it does (binds X and blocks Y). IP teams at biologics companies are auditing their patent portfolios and pending applications to restructure functional claims into structurally defined alternatives.
AI in Drug Discovery: What Is Patentable
Artificial intelligence is accelerating drug discovery by identifying novel chemical scaffolds, predicting binding affinity, and optimizing ADMET profiles in silico. The IP question is whether AI-identified compounds and AI-generated innovations are patentable. Current USPTO guidance (updated 2024) requires a human to have made a ‘significant contribution’ to each claim; AI cannot be listed as an inventor. Compounds identified by AI but selected, synthesized, and tested by human scientists can be patented when the human contribution to the claimed invention is documented.
The practical implication for IP teams: internal invention disclosure programs must capture human decision-making at each step of the AI-assisted discovery process. Documentation of which hypotheses were tested, why, and what the human researcher concluded from the AI output is essential to establishing inventorship and defeating future challenges that the patent claims no human inventor.
Key Takeaways: Section 12
- Post-Amgen v. Sanofi, functional antibody claims without structural disclosure will not survive enablement challenges. Audit pending biologic applications immediately.
- AI-assisted discovery is patentable when humans make significant contributions to the claimed invention. Document human decision-making throughout the AI-assisted process.
- BPCIA’s 12-year reference product exclusivity runs independently of patent protection and remains the primary barrier to biosimilar entry for newly approved biologics.
Section 13: ANDA Litigation Trend Analysis (2017-2026)
ANDA case filings declined approximately 36% from 2017 to 2021, per Patexia’s ANDA Litigation Intelligence data. The steepest single-year decline was between 2020 and 2021, at 5.3 fewer cases per month. Several structural explanations account for this trend. The first is the passage of the major small-molecule patent cliff. The 2011-2016 period saw simultaneous LOE on many blockbuster products (Lipitor, Plavix, Singulair, Zyprexa) — a once-in-a-generation wave of patent challenges driven by concentrated opportunity. By 2020, that wave had largely resolved.
The second explanation is the shift toward specialty and biologic products, which face BPCIA rather than Hatch-Waxman and have structurally different competitive dynamics. Generic companies redirected investment toward complex drug products (extended-release formulations, transdermal systems, modified-release microspheres) where formulation barriers are higher, and toward biosimilars. Both require larger upfront development investment per product, making selective targeting rational over broad portfolio challenges.
The third explanation is consolidation in the generic industry. The major generic companies — Teva, Viatris, Sandoz, Sun, Hikma, Dr. Reddy’s — have faced significant financial and operational headwinds since 2016 from generic pricing pressure, channel consolidation, and regulatory compliance issues. Litigation requires capital; constrained companies file fewer Paragraph IV certifications.
Looking forward to 2026 and beyond: the GLP-1 receptor agonist class (semaglutide, tirzepatide, liraglutide) will generate the next wave of major ANDA activity as early composition-of-matter patents approach expiration in the early 2030s, though the biologic and peptide nature of these compounds routes challenges through the BPCIA rather than Hatch-Waxman for some agents. Small molecule oral GLP-1 agonists and peptide-related formulation innovations will be Hatch-Waxman territory, and the patent estates around those products are already drawing generic company attention.
Section 14: Regulatory Evolution and Strategic Adaptation
FDA’s Drug Competition Action Plan (DCAP), launched in 2017 and extended through subsequent regulatory actions, accelerated generic approvals for complex products and drugs with limited competition. The FDA’s Complex Drug Substance and Complex Drug Product programs created more structured pathways for generics to address formulation and characterization challenges for products like locally acting gastrointestinal drugs, topical products, and inhaled drugs — historically areas where technical barriers deterred generic entry.
The 2023 Consolidated Appropriations Act created the new FDA Orange Book patent dispute mechanism, giving generic companies a formal channel to challenge allegedly improper Orange Book listings without filing a Paragraph IV certification and triggering litigation. This mechanism has implications for innovators who have used device component patents (injectors, inhalers, auto-injectors) to list device patents in the Orange Book for combination drug-device products. The FTC’s 2023 letters to several NDA holders challenging device patent listings signaled that this practice will receive regulatory scrutiny independent of litigation.
The Inflation Reduction Act (IRA) of 2022, which empowered CMS to negotiate prices for certain high-cost Medicare drugs, altered the LOE economic model for small molecules and biologics subject to negotiation. A product with government-set pricing has different LOE economics than one competing purely in the private market. For the first nine small molecules subject to IRA negotiation, the government price is set for 2026, and the gap between the negotiated price and the branded price will determine whether a generic entry creates meaningful savings — and therefore whether it is worth pursuing. IP teams at generic companies are beginning to incorporate IRA pricing scenarios into their ANDA opportunity models.
Section 15: Investment Strategy for Analysts
Patent Cliff Modeling for Equity Positions
Pharmaceutical equity analysts and portfolio managers treat LOE events as binary financial risk factors when they should treat them as probability distributions. The relevant variables are: the number and quality of listed Orange Book patents, whether Paragraph IV certifications have been filed, the court’s case schedule (claim construction hearing date, trial date), the strength of the innovator’s secondary patent position, and whether an authorized generic is contractually committed.
A practical framework: for each revenue-significant drug in the portfolio, assign a probability of defending LOE (patents survive) and a probability of LOE occurring (patents fall or are designed around), weight each scenario by the present value of revenues in that scenario, and aggregate. Companies with thin secondary patent positions on products approaching primary LOE deserve a larger discount to NAV; companies with layered patent estates on the same timeline deserve less. This distinction is often underpriced by generalist investors who apply a uniform LOE discount without parsing patent quality.
Paragraph IV Filing as a Leading Indicator
The public availability of Paragraph IV certification notice letters — NDA holders must disclose them in SEC filings within 45 days of receipt — creates a leading indicator for LOE risk. Multiple Paragraph IV filings against a product’s patents, especially from well-capitalized generic challengers, correlates with elevated litigation risk and the probability of early LOE even if the innovator ultimately wins some of the challenged patents.
For generic company investors, the reverse analysis applies: the number of active Paragraph IV filings in a generic company’s pipeline, weighted by the revenue of the targeted branded products and the generic’s litigation position, provides a forward-looking indicator of potential launch economics.
Key Takeaways: Section 15
- LOE risk should be modeled as a probability distribution across patent validity and litigation outcomes, not as a binary event at nominal patent expiration.
- Paragraph IV filings disclosed in SEC filings are a public leading indicator of LOE risk; monitoring them is basic pharma equity due diligence.
- IRA drug price negotiation alters the LOE economics for Medicare-negotiated products; generic opportunity models for 2026+ must incorporate negotiated pricing scenarios.
- For biologic-focused companies, the 12-year BPCIA reference exclusivity period, not patent expiration, is the primary LOE timing driver; patent estates are secondary but still material.
Frequently Asked Questions
What triggers a 30-month stay in ANDA litigation? Filing a patent infringement lawsuit in federal court within 45 days of receiving the Paragraph IV notice letter triggers the stay automatically. The stay prevents FDA from granting final ANDA approval for 30 months from the date the NDA holder received notice. If the innovator does not file within 45 days, FDA can approve the ANDA without the stay.
How is 180-day generic exclusivity triggered and can it be forfeited? The 180-day exclusivity period begins on the date of first commercial marketing by the first-to-file generic. Several forfeiture events can terminate the exclusivity before it runs: failure to market within 75 days of a court judgment of invalidity or non-infringement, failure to market within 75 days of FDA approval (absent a court order preventing launch), or withdrawal of the Paragraph IV certification.
What is the difference between patent term extension and regulatory data exclusivity? Patent term extension (PTE) under 35 U.S.C. 156 extends the life of a specific patent by up to five years to compensate for time lost during FDA regulatory review. Regulatory data exclusivity (e.g., five years for new chemical entities under 21 U.S.C. 355(c)(3)) prohibits FDA from relying on the innovator’s clinical data to approve an ANDA or NDA for a defined period, regardless of patent status. Both protections can run concurrently or sequentially; together they determine the outer bound of effective market exclusivity.
What is the FTC’s current enforcement posture on reverse payment settlements? Post-FTC v. Actavis (2013), reverse payment settlements are evaluated under antitrust rule of reason. The FTC continues to investigate settlements where the innovator’s payment to the generic (in cash or in kind) appears to exceed fair value for the generic’s litigation concessions. Non-cash settlements that include supply agreements, license grants for other products, or co-promotion rights can constitute reverse payments subject to antitrust scrutiny.
How does the IRA drug pricing negotiation affect patent strategy? IRA negotiation affects the revenue side of the LOE equation. A government-negotiated price reduces the branded product’s revenue, which proportionally reduces the economic benefit of additional patent exclusivity. For innovators, this creates some disincentive to evergreen products that will be negotiated. For generic challengers, it reduces the revenue prize for challenging those products’ patents. The net effect on litigation frequency for negotiated products remains to be observed as the first negotiated prices take effect in 2026.
For patent expiration data, Orange Book analysis, and Paragraph IV filing intelligence, DrugPatentWatch provides the underlying data infrastructure used by leading pharmaceutical IP teams and institutional investors worldwide.


























