MESALAMINE Drug Patent Profile
✉ Email this page to a colleague
When do Mesalamine patents expire, and when can generic versions of Mesalamine launch?
Mesalamine is a drug marketed by Teva Pharms Usa, Alembic, Alkem Labs Ltd, Amta, Annora Pharma, Aurobindo Pharma Ltd, Difgen Pharms, Mylan, Novast Labs, Sun Pharm, Zydus Pharms, Encube, G And W Labs Inc, Klm Labs Pvt, Novitium Pharma, Padagis Israel, Actavis Mid Atlantic, Amneal, Amring Pharms, Quagen, Rising, Sandoz, Actavis Labs Fl, Sinotherapeutics Inc, and Teva Pharms Inc. and is included in thirty-five NDAs.
The generic ingredient in MESALAMINE is mesalamine. There are twenty-eight drug master file entries for this compound. Thirty-seven suppliers are listed for this compound. Additional details are available on the mesalamine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Mesalamine
A generic version of MESALAMINE was approved as mesalamine by PADAGIS ISRAEL on September 17th, 2004.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for MESALAMINE?
- What are the global sales for MESALAMINE?
- What is Average Wholesale Price for MESALAMINE?
Summary for MESALAMINE
| US Patents: | 0 |
| Applicants: | 25 |
| NDAs: | 35 |
| Finished Product Suppliers / Packagers: | 31 |
| Raw Ingredient (Bulk) Api Vendors: | 130 |
| Clinical Trials: | 106 |
| Patent Applications: | 3,973 |
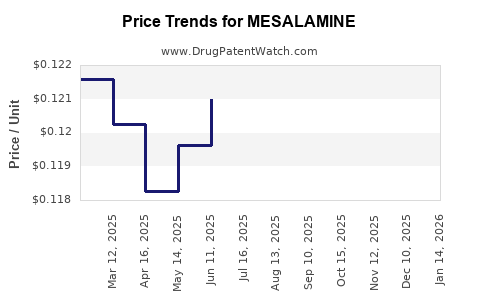
| Drug Prices: | Drug price information for MESALAMINE |
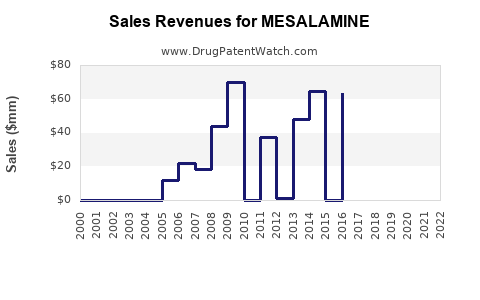
| Drug Sales Revenues: | Drug sales revenues for MESALAMINE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for MESALAMINE |
| What excipients (inactive ingredients) are in MESALAMINE? | MESALAMINE excipients list |
| DailyMed Link: | MESALAMINE at DailyMed |
Recent Clinical Trials for MESALAMINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Grupo Espanol de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa | PHASE4 |
| Ihab Elsayed Hassan | PHASE2 |
| Prof. Dr. Ahmed Ibrahim Mohammed El Mallah, faculty of pharmacy, Alexandria University. | PHASE2 |
Pharmacology for MESALAMINE
| Drug Class | Aminosalicylate |
Medical Subject Heading (MeSH) Categories for MESALAMINE
Paragraph IV (Patent) Challenges for MESALAMINE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| DELZICOL | Delayed-release Capsules | mesalamine | 400 mg | 204412 | 1 | 2014-06-17 |
| CANASA | Suppository | mesalamine | 1000 mg | 021252 | 1 | 2013-05-24 |
| APRISO | Extended-release Capsules | mesalamine | 0.375 g | 022301 | 1 | 2012-04-03 |
| ASACOL HD | Delayed-release Tablets | mesalamine | 800 mg | 021830 | 1 | 2011-07-13 |
| LIALDA | Delayed-release Tablets | mesalamine | 1.2 g | 022000 | 1 | 2009-12-16 |
| ASACOL | Delayed-release Tablets | mesalamine | 400 mg | 019651 | 1 | 2007-06-22 |
US Patents and Regulatory Information for MESALAMINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sinotherapeutics Inc | MESALAMINE | mesalamine | TABLET, DELAYED RELEASE;ORAL | 217337-001 | May 12, 2023 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Actavis Labs Fl | MESALAMINE | mesalamine | TABLET, DELAYED RELEASE;ORAL | 203817-001 | Mar 23, 2018 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sun Pharm | MESALAMINE | mesalamine | TABLET, DELAYED RELEASE;ORAL | 211858-001 | Jan 25, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Amneal | MESALAMINE | mesalamine | TABLET, DELAYED RELEASE;ORAL | 215067-001 | Feb 18, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Teva Pharms Usa | MESALAMINE | mesalamine | CAPSULE, DELAYED RELEASE;ORAL | 207873-001 | May 9, 2019 | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Padagis Israel | MESALAMINE | mesalamine | ENEMA;RECTAL | 076751-001 | Sep 17, 2004 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Rising | MESALAMINE | mesalamine | SUPPOSITORY;RECTAL | 207448-001 | Apr 19, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Mesalamine
More… ↓