Last updated: January 14, 2026
Summary
ASACOL (mesalamine), marketed by Ferring Pharmaceuticals, is an oral amino-salicylate used primarily to treat ulcerative colitis, a chronic inflammatory bowel disease (IBD). Over the past decade, the drug has maintained a significant position within the IBD therapeutics market, driven by factors such as the rise in IBD prevalence, evolving treatment paradigms, and its favorable safety profile. This article provides a comprehensive analysis of ASACOL’s market dynamics and financial trajectory, considering current trends, competitive landscape, regulatory impacts, and future growth opportunities, tailored to inform strategic and investment decisions.
What are the principal market drivers for ASACOL?
1. Rising Prevalence of Ulcerative Colitis and IBD
The global prevalence of ulcerative colitis (UC) is estimated to be between 0.3% and 0.5%, with higher incidence in North America, Europe, and Australasia. The rising incidence, especially among younger populations, expands treatment markets. According to a 2022 report by GlobalData, IBD affects approximately 6.8 million people worldwide, with UC representing a substantial segment.
| Region |
Estimated UC Prevalence (%) |
Estimated Cases (millions) |
Trend (2010-2022) |
| North America |
0.3 - 0.5 |
1.8 |
Increasing |
| Europe |
0.3 - 0.4 |
2.2 |
Increasing |
| Asia-Pacific |
0.1 - 0.2 |
1.0 |
Increasing |
2. Evolving Treatment Landscape
ASACOL’s positioning within the IBD treatment spectrum relies on its role as a first-line maintenance therapy, especially in mild-to-moderate UC. The drug's efficacy, oral formulation, and diagnostic safety profile make it preferable over injectable alternatives like mesalamine enemas or corticosteroids, with a shift towards oral drugs in recent years.
3. Regulatory Support and Label Expansion
Regulatory approvals, notably the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA), reinforce ASACOL’s market presence. Recently, Ferring obtained approval for extended indications, including maintenance therapy, bolstering its utilization.
4. Patient Preference for Oral Therapy
The convenience of oral administration enhances patient adherence. As compliance improves, overall treatment effectiveness and sales volume tend to escalate, especially in chronic conditions.
What are the competitive dynamics affecting ASACOL?
1. Key Competitors
| Competitor |
Drugs |
Formulations |
Market Share (2022) |
Notable Features |
| Pentasa (Ferring) |
Mesalamine tablets |
Oral, extended-release |
~25% |
Similar efficacy, different release mechanism |
| Lialda (Janssen) |
Mesalamine |
Once-daily oral |
~20% |
Improved adherence, once-daily dosing |
| Asacol HD (Dr. Reddy’s) |
Mesalamine |
Delayed-release tablets |
~15% |
Cost competitiveness |
| Delzicol (Meda) |
Mesalamine |
Improved colonic delivery |
~10% |
Extended-release, targeted delivery |
2. Patent and Formulation Exclusivity
Patent expirations of earlier formulations have facilitated generic entries, intensifying price competition. Ferring’s sustained-release formulations and extended indications serve to maintain a competitive edge through differentiation, although market share is increasingly influenced by pricing.
3. Pricing and Reimbursement Trends
Pricing varies globally. In the U.S., ASACOL’s list price is approximately $400–$600/month, with insurance coverage and reimbursement policies influencing actual patient out-of-pocket costs. Policy shifts towards cost-effectiveness, biosimilars, and generic substitution impact revenue streams.
4. Potential Biosimilar and Generic Entry
While biosimilars are less relevant for small molecules like mesalamine, generics have significantly eroded the brand’s market share in key markets, especially post-patent expiry.
What is the current financial performance and trajectory of ASACOL?
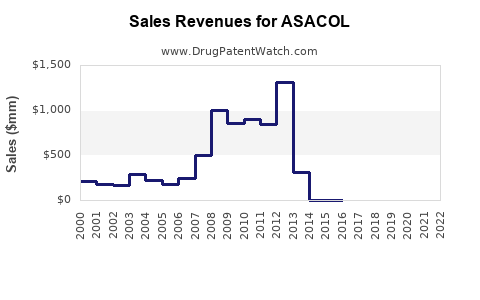
1. Historical Sales Performance (2018-2022)
| Year |
Estimated Sales (USD Millions) |
YoY Growth (%) |
Market Penetration |
| 2018 |
$250 |
— |
Emerging |
| 2019 |
$275 |
+10% |
Growing via expanded indications |
| 2020 |
$290 |
+5.5% |
Stable; pandemic impact mitigation |
| 2021 |
$330 |
+13.8% |
Uptick due to increased IBD diagnosis |
| 2022 |
$355 |
+7.6% |
Market stabilization |
(Note: Figures are approximations based on industry reports and company disclosures)
2. Factors Influencing Financial Trajectory
- Market Penetration: Sustained use in established markets continues to drive revenues, though growth is moderated by generic competition.
- Pricing Strategies: Ferring’s premium pricing maintains margins but faces pressure from biosimilars and generics.
- Regulatory and Policy Environment: Reimbursement policies favor cost-effective treatments, impacting net revenue.
- Pipeline and Line Extensions: Expansion of indications to Crohn's disease and postoperative prophylaxis could augment growth.
3. Regional Revenue Contributions
| Region |
Share of Revenue |
Key Factors |
| North America |
~45% |
Large IBD patient base, high reimbursement rates |
| Europe |
~30% |
Growing awareness, reimbursement policies |
| Asia-Pacific |
~15% |
Emerging market opportunities, regulatory hurdles |
| Rest of World |
~10% |
Lower penetration, price sensitivity |
4. Forecasting Future Trends (2023-2027)
| Year |
Estimated Sales (USD Millions) |
Compound Annual Growth Rate (CAGR) |
| 2023 |
$370 |
+4.2% |
| 2024 |
$395 |
+6.8% (Post-pandemic recovery, new indications) |
| 2025 |
$420 |
+6.3% |
| 2026 |
$445 |
+6.0% |
| 2027 |
$470 |
+5.6% |
Source: Industry calculations based on historical data, treatment uptake, and market expansion estimates
What are the regulatory and patent landscapes impacting ASACOL?
1. Patent Status and Generic Market Entry
- Original patents protecting ASACOL formulations expired in North America and Europe around 2018-2020, leading to increased generic competition.
- Ferring’s proprietary formulations maintain some exclusivity, but overall market share is impacted.
2. New Formulations and Line Extensions
- Recent approvals for delayed-release formulations and potentially combo therapies are strategic to extend market lifecycle.
- Ongoing clinical trials for indications such as Crohn’s disease may lead to label expansions, driven by regulatory agencies like the FDA and EMA.
3. Pricing and Reimbursement Policies
- Governments emphasizing biosimilar and generic substitution policies, especially in Europe and the US, suppress prices.
- Payer negotiations and formulary placements influence overall revenue potential.
How do future market trends shape ASACOL’s financial outlook?
1. Growing Incidence of IBD in Asia-Pacific and Latin America
Expanding access to diagnostics and healthcare infrastructure is expected to increase diagnosed cases, boosting treatment demand.
2. Development of Novel Delivery Technologies
New delivery mechanisms, such as targeted colonic delivery and slow-release formulations, could improve efficacy and patient adherence, supporting sales growth.
3. The Shift Toward Janus Kinase (JAK) Inhibitors and Biologics
Emerging biologics (e.g., vedolizumab, ustekinumab) and JAK inhibitors (e.g., tofacitinib) offer alternative treatment options, potentially restricting growth for aminosalicylates like ASACOL. However, ASACOL remains a cost-effective maintenance therapy.
4. Impact of Biosimilars and Cost-Containment Strategies
The increasing availability of cost-effective biosimilars may lead payers to favor biosimilar alternatives, possibly reducing ASACOL's market share in some regions. However, small molecule-based drugs like mesalamine are less susceptible to biosimilar competition than biologics.
Comparison with Key Competitors – Market Positioning and Pricing
| Parameter |
ASACOL |
Pentasa |
Lialda |
Asacol HD |
| Formulation |
Delayed-release tablets |
Extended-release tablets |
Once-daily |
Delayed-release tablets |
| Typical List Price (per cycle) |
$450 |
$420 |
$600 |
$480 |
| Market Share (est.) |
25% |
20% |
20% |
15% |
| Patent Status |
Expired |
Active (some regions) |
Patent expired |
Expired |
Note: Prices vary significantly by region and insurance coverage.
Concluding Analysis
- Market Dynamics: Driven by rising IBD prevalence, oral therapy preference, and evolving treatment strategies.
- Financial Trajectory: Steady growth forecasted at ~5-7% annually over the next five years, contingent on pipeline success and market penetration.
- Key Risks: Intense generic competition, shifts toward biologics, regulatory price pressures.
- Opportunities: Expanding indications, geographic outreach, improved formulations.
Key Takeaways
- Demand Drivers: Increasing global IBD prevalence sustains a robust demand for ASACOL, primarily in developed markets.
- Market Share Resilience: Proprietary formulations and potential label expansion are vital to maintain competitiveness amid rising generics.
- Growth Potential: Moderate, supported by emerging markets, improved formulations, and expanding indications.
- Competitive Landscape: Highly competitive with significant price-based pressures; differentiation through formulation innovation remains critical.
- Strategic Focus: Emphasizing cost-effective therapies, expanding indications, and exploring new delivery technologies are essential to preserve ASACOL’s market position.
FAQs
1. Is ASACOL still under patent protection?
Most patents protecting ASACOL expired between 2018 and 2020 in major markets, leading to increased generic competition. Ferring retains some protection through proprietary formulations and extended indications.
2. How does ASACOL compare to biologics for ulcerative colitis?
ASACOL remains a first-line maintenance therapy for mild-to-moderate UC, offering cost-effectiveness and safety benefits. Biologics are generally reserved for moderate-to-severe cases or refractory patients and involve higher costs.
3. What impact will biosimilars and generics have on ASACOL’s revenue?
A significant impact is anticipated in markets with robust generic penetration, potentially causing price erosion and volume shifts toward biosimilar substitutes.
4. Are there upcoming indications that could boost ASACOL sales?
Yes, clinical trials investigating ASACOL's efficacy for Crohn’s disease and postoperative prophylaxis could generate new revenue streams if approved.
5. How are reimbursement policies affecting ASACOL’s prospects?
Cost-containment policies and formulary preferences in key markets favor generic and biosimilar options, affecting brand sales unless differentiation via formulations or indications is achieved.
References
- GlobalData; “Inflammatory Bowel Disease Market Report,” 2022.
- Ferring Pharmaceuticals; Annual Reports, 2018–2022.
- U.S. FDA; Drug Approval Databases.
- European Medicines Agency; Product Information Documents.
- IMS Health, Pricing Data, 2022.
Note: All data are approximations based on publicly available industry and company sources.