Last updated: April 24, 2026
Doxycycline is a high-volume, long-established antibiotic with broad generics penetration. Market dynamics are driven by (1) persistent demand for respiratory and skin infections, (2) cycling of guideline preferences between tetracyclines, beta-lactams, and macrolides, (3) specialty and non-infectious uses that sustain prescription and channel pull, and (4) generic pricing pressure that caps growth to volume-led expansion and premium formulations.
What is doxycycline’s demand base and why does it keep demand resilient?
Core infection demand
Doxycycline is widely used for community-acquired bacterial infections, with recurring use-cases that support stable prescribing across geographies:
- Respiratory infections: bronchitis and other non-severe lower respiratory infections where tetracyclines remain guideline options.
- Dermatologic infections: acne (notably via sub-antimicrobial strategies in specific formulations), rosacea subtypes, and other bacterial skin conditions.
- Vector-borne and atypical pathogens: where doxycycline is a standard option for certain endemic risks and empiric therapy contexts.
Non-infectious and specialty-drug demand drivers
Doxycycline also sustains demand beyond routine acute infection treatment through:
- Acne and inflammatory dermatology: doxycycline is used for acne control and other dermatology indications in multiple product formats.
- Rosacea: doxycycline-based therapies are key in rosacea management pathways, supporting repeat demand even when acute antibiotic use softens.
Supply and channel structure
Doxycycline’s market has:
- High generic coverage in most major markets
- Multiple dosage forms (tablets, capsules, liquids, and delayed/modified release formats)
- Large-scale manufacturing that keeps baseline supply strong, supporting patient access and limiting supply-driven pricing spikes
How does the pricing and margin profile typically evolve under generic dominance?
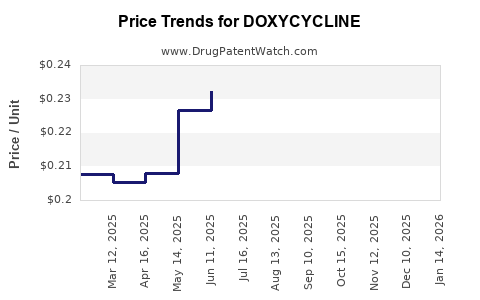
Generic-driven price compression
In a mature antibiotic with extensive patent expiration and broad generics competition, the commercial pattern typically looks like:
- Initial brand pricing followed by step-down after generics entry
- Continued price erosion as additional ANDA entrants and authorized generics increase buyer leverage
- Margin stabilization only when a product has (a) formulation differentiation, (b) dosing convenience, or (c) payer preference in specific indications
What still supports revenue beyond pure commodity pricing
Revenue can hold up through mix shift, not headline price increases:
- Premium and modified-release formulations: delayed/controlled-release products can sustain higher net pricing where payer formularies prefer them.
- Formulation substitution: liquid/weight-based pediatric dosing or specific gastro-tolerability profiles can keep channel pull.
- Dermatology chronic use: dermatology regimens can be more resistant to total category substitution than episodic acute antibiotics.
What market forces shape doxycycline’s financial trajectory over time?
1) Guideline and payer behavior
- Antibiotic stewardship programs can reduce unnecessary antibiotic prescribing, but they also keep doxycycline in recommended lists for eligible indications.
- Formulary decision-making favors low-cost therapeutics and consistent tolerability. This increases share for the lowest-cost clinically acceptable options and penalizes higher-cost branded products unless they maintain payer-preferred status.
2) Resistance patterns and clinical substitution
- Doxycycline resistance has varied by pathogen and geography. When resistance data deteriorates for specific organisms, prescribers shift to other classes, reducing volumes in those niches.
- When resistance remains manageable, doxycycline retains usage as a cost-effective option, which stabilizes volumes.
3) Safety and administration constraints
Tetracyclines are associated with known class-specific constraints (e.g., photosensitivity; gastrointestinal intolerance). Commercially, this creates demand for:
- Improved tolerability formats
- Dose regimens with better adherence
- Patient counseling support through product labels
4) Competitive class dynamics
Doxycycline competes against:
- Beta-lactams (penicillins/cephalosporins) in many empiric indications
- Macrolides in certain respiratory indications
- Other tetracyclines and newer agents in niche uses
Under stewardship and payer constraints, the market tends to reward the lowest total-treatment-cost option with sufficient clinical performance.
5) Specialty channel: dermatology and repeat dosing
Chronic dermatology use dampens volatility:
- Acute antibiotic markets swing with seasonal infection patterns and prescribing restrictions.
- Dermatology use patterns are more stable, which smooths revenue for products positioned for those indications.
Where does the growth potential actually come from in doxycycline?
For a generic-heavy molecule, growth typically comes from mix and lifecycle execution:
- New formulations that improve tolerability or adherence
- Indication expansion into dermatology pathways where doxycycline is used as a standard-of-care or preferred option
- Geographic expansion in markets with evolving access and formularies
- Channel optimization for institutional contracting (long-term care, ambulatory clinics, retail)
How does the financial trajectory usually present for a mature generic antibiotic?
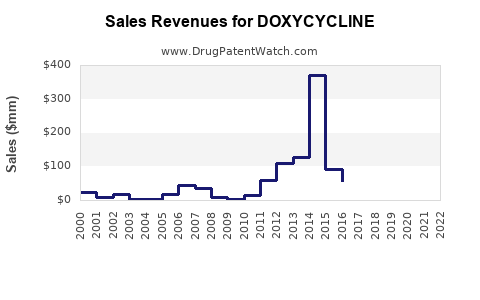
Typical trajectory shape
Doxycycline’s financial trajectory in most markets follows a maturity curve:
- Revenue stabilizes as branded demand transitions to generics.
- Nominal revenue declines if payer net prices fall faster than volume increases.
- Volume can partially offset price through mix shift to preferred generics and specific formulations.
- Later-stage declines flatten when category demand is large and stable (respiratory and dermatology baseline).
- Periodic disruptions occur from formulation supply constraints or short-term shifts in guideline preference.
Investor and business implication
- Upside is limited to share gains, premium mix, and formulation differentiation.
- Downside is predictable through net price compression from additional generic entrants and increased buyer negotiation leverage.
What does competition look like and how does it affect revenue stability?
Competitive landscape structure
Doxycycline’s market is characterized by:
- Multiple generic manufacturers across major dosage forms
- Frequent substitution at the pharmacy level when cost-sharing drives switch behavior
- Payer-driven formulary preference that locks in certain SKUs
Pricing power reality
Pure doxycycline generics have low pricing power. Revenue stability tends to depend on:
- Contracting strength (ability to supply at target net prices)
- Preferred formulary placement for certain SKUs
- Therapeutic equivalence acceptance (no meaningful clinical differentiation)
How does formulation strategy change the revenue equation?
Differentiation levers
Revenue resilience improves when a product:
- Provides gastrointestinal tolerance via formulation design
- Enables simplified dosing or better adherence
- Targets dermatology dosing patterns that align with chronic therapy schedules
Competitive effect
Even within generics, improved formulations can:
- Reduce patient discontinuations
- Keep higher utilization against lower-cost immediate-release alternatives
- Support payer preference for tolerability-centered switches
What are the key risks to the financial trajectory?
The principal risks are structural and recurring:
- Generic net price erosion through continued entry and contracting pressure
- Guideline changes that move empiric treatment away from tetracyclines in certain pathogen settings
- Resistance-driven shifts that reduce effective use in specific indications
- Stewardship tightening that reduces total antibiotic prescribing (volume headwind)
- Safety perception and adherence barriers that favor alternative drug classes
What is the likely outlook for doxycycline revenues and margins?
Revenue outlook
- Demand remains durable due to established infection and dermatology use cases.
- Total market growth is modest and largely volume-led.
- Revenue is sensitive to payer net price due to generic dominance.
Margin outlook
- Margins stay compressed relative to branded specialty drugs.
- Execution wins come from mix (preferred SKUs), supply reliability, and contracting rather than pricing.
Lifecycle outlook
- The molecule’s core economics are mature.
- Near-term outcomes are determined by competitive intensity in specific SKUs and the sustained uptake of dermatology positioning for doxycycline formulations.
Market checkpoints that matter commercially
Business and investment decisions for doxycycline should track the same set of variables:
- Generic entry intensity by SKU and dosage form (net price and share shifts)
- Payer formulary changes (preferred listing and prior authorization behavior)
- Dermatology growth and retention (repeat-use stability vs seasonal infection swings)
- Guideline updates by geography (treatment pathway changes that shift volume)
- Resistance reports by pathogen class (label-relevant clinical performance)
Key Takeaways
- Doxycycline is a high-volume, mature antibiotic with generic-dominated economics and low pricing power.
- Market stability is supported by persistent infection use and repeat-demand dermatology use, which can smooth volatility.
- Financial trajectory is driven more by mix and contracting than by price growth, with revenue constrained by ongoing generic price pressure.
- Upside is mainly tied to formulation differentiation (tolerability/adherence) and payer-preferred positioning in dermatology pathways.
- Downside is predictable: net price compression, stewardship volume reduction, and guideline or resistance shifts that alter prescribing share.
FAQs
-
Is doxycycline growth mainly price-led or volume-led?
It is primarily volume-led with mix effects, because pricing power is limited under generic dominance.
-
Which therapeutic areas stabilize doxycycline demand?
Respiratory and dermatology use cases, especially chronic dermatology regimens, stabilize utilization.
-
What determines net pricing for doxycycline in formularies?
Generic contracting strength, preferred SKU placement, and payer cost-sharing dynamics.
-
What is the biggest financial risk for doxycycline?
Continued net price erosion from increased generic competition and formulary substitution.
-
Do resistance trends materially change doxycycline’s commercial outlook?
Yes, pathogen-specific resistance shifts can reduce effective use in certain indications and move share to alternative classes.
References
[1] U.S. Food and Drug Administration (FDA). National Drug Code (NDC) Directory. https://www.fda.gov/drugs/drug-approvals-and-databases/national-drug-code-directory
[2] FDA. Drug Shortages. https://www.accessdata.fda.gov/scripts/drugshortages/default.cfm
[3] Centers for Disease Control and Prevention (CDC). Antibiotic Use and Stewardship Resources. https://www.cdc.gov/antibiotic-use/
[4] Infectious Diseases Society of America (IDSA). Clinical practice guideline and stewardship-related publications. https://www.idsociety.org/guidelines/
[5] World Health Organization (WHO). Antimicrobial resistance and antibiotics use resources. https://www.who.int/health-topics/antimicrobial-resistance