Triamcinolone - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for triamcinolone and what is the scope of patent protection?
Triamcinolone
is the generic ingredient in thirty-one branded drugs marketed by Astellas, Delcor Asset Corp, Barr, Impax Labs, Ivax Sub Teva Pharms, Mylan, Purepac Pharm, Roxane, Sandoz, Teva, Watson Labs, Abbvie, Chattem Sanofi, Ivax Pharms, Solvay, Actavis Mid Atlantic, Alkem Labs Ltd, Alpharma Us Pharms, Ambix, Chartwell Rx, Cosette, Encube, Fougera Pharms, Glenmark Pharms Ltd, Macleods Pharms Ltd, Micro Labs, Morton Grove, Norvium Bioscience, Padagis Us, Pharmaderm, Pharmafair, Strides Pharma, Sun Pharma Canada, Taro, Topiderm, Crown Labs, Savage Labs, Pacira Pharms Inc, Apothecon, Alembic, Amneal, Caplin, Dr Reddys, Eugia Pharma, Hikma, Long Grove Pharms, Mylan Labs Ltd, Parnell, Teva Pharms Usa, Allergan, Harrow Eye, Beach Prods, Epic Pharma Llc, Pai Holdings Pharm, Pharmobedient, Quagen, Aurobindo Pharma Ltd, Extrovis, Glenmark Speclt, Padagis Israel, Senores Pharms, Cmp Pharma Inc, Rising, Sciegen Pharms, Lupin Atlantis, Sanofi Aventis Us, Apotex, Perrigo Pharma Intl, Sun Pharm Inds Inc, Bausch And Lomb Inc, Fosun Pharma, and Ethypharm, and is included in one hundred and seventy-one NDAs. There are eight patents protecting this compound. Additional information is available in the individual branded drug profile pages.There are fifty-one drug master file entries for triamcinolone.
Summary for triamcinolone
| US Patents: | 8 |
| Tradenames: | 31 |
| Applicants: | 72 |
| NDAs: | 171 |
| Drug Master File Entries: | 51 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 472 |
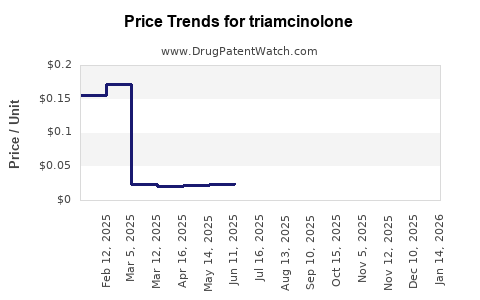
| Drug Prices: | Drug price trends for triamcinolone |
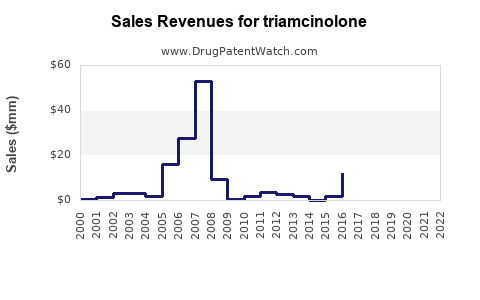
| Drug Sales Revenues: | Drug sales revenues for triamcinolone |
| DailyMed Link: | triamcinolone at DailyMed |
Recent Clinical Trials for triamcinolone
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Karachi Medical and Dental College | NA |
| University of California, San Francisco | PHASE4 |
| Research Insight LLC | PHASE4 |
Medical Subject Heading (MeSH) Categories for triamcinolone
Anatomical Therapeutic Chemical (ATC) Classes for triamcinolone
US Patents and Regulatory Information for triamcinolone
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caplin | TRIAMCINOLONE ACETONIDE | triamcinolone acetonide | INJECTABLE;INJECTION | 090166-001 | May 27, 2009 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Fougera Pharms | TRIAMCINOLONE ACETONIDE | triamcinolone acetonide | OINTMENT;TOPICAL | 085691-001 | Approved Prior to Jan 1, 1982 | AT | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Teva | TRIACET | triamcinolone acetonide | CREAM;TOPICAL | 084908-003 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Triamcinolone Market Analysis and Financial Projection
More… ↓