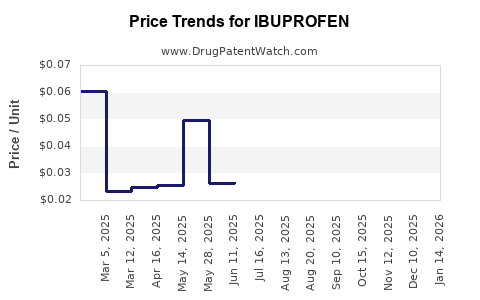
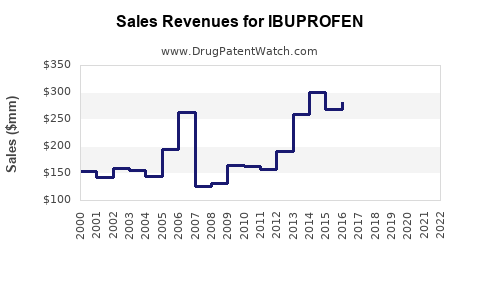
Last updated: April 14, 2026
Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID) widely used for pain relief, fever reduction, and inflammation control. Its extensive global use, varied formulations, and evolving market environment influence its commercial performance.
Market Structure and Key Players
The global ibuprofen market features several major pharmaceutical companies involved in manufacturing and distributing generic and branded products. The leading players include:
- Makers of branded formulations: Johnson & Johnson (Motrin), Meda (Nurofen), and others.
- Generic manufacturers: Teva, Sandoz, Mylan, and local producers in emerging markets.
The market is highly fragmented, especially in emerging regions, with local manufacturers supplying over-the-counter (OTC) and prescription formulations.
Market Size and Growth Trends
Estimates for the global ibuprofen market size in 2022 ranged between USD 1.2 billion and USD 1.5 billion, with expectations of growth at a compound annual growth rate (CAGR) of approximately 3-4% over the next five years.
Key Drivers:
- Rising prevalence of chronic pain and inflammation-related conditions.
- Growing awareness and acceptance of OTC NSAIDs.
- Increased healthcare expenditure, especially in Asia-Pacific.
Market Segments:
- OTC sales: Account for roughly 70-75% of total sales.
- Prescription sales: Constitute 25-30%, typically higher in developed markets.
Regional Distribution:
| Region |
Market Share (2022) |
Growth Rate (2022-2027) |
| North America |
40% |
3.2% |
| Europe |
25% |
3.5% |
| Asia-Pacific |
20% |
4.5% |
| Latin America & Others |
15% |
3.8% |
Emerging markets exhibit higher growth potential due to increasing disposable income and shifting regulatory environments.
Pricing and Revenue Trends
Price sensitivity remains high, particularly for OTC products. Generic formulations dominate the market, leading to intense price competition. Innovative formulations (e.g., sustained-release, combination drugs) command premium prices but account for a smaller market share.
Price Evolution:
- Average retail price for OTC ibuprofen ranges from USD 0.05 to USD 0.20 per tablet, depending on dosage and branding.
- Generic prices have decreased by approximately 5-7% annually over the past five years.
Revenue Drivers:
- Volume sales driven by OTC market penetration.
- Formulation innovations expanding indications and patient compliance.
Regulatory and Patent Landscape
The expiration of key patents in the 2000s led to a surge in generic production, intensifying price competition. Regulatory approvals for new formulations, including liquids, chewables, and topical patches, are expanding market options.
- Patent expiration milestones: Johnson & Johnson's Motrin patent expired in 2007. Sandoz's generic versions grew significantly afterward.
- Regulatory hurdles: Vary by region, with the U.S. FDA and EMA providing clear pathways for generics and new formulations.
Market Challenges and Opportunities
Challenges:
- Market saturation in mature economies.
- Regulatory pressures on safety, especially concerning gastrointestinal and cardiovascular risks.
- Price erosion due to generic competition.
Opportunities:
- Development of new delivery systems (e.g., transdermal patches).
- Expansion into emerging markets.
- Strategic partnerships and licensing.
Financial Trajectory Projections
Based on current market trends, the ibuprofen segment is expected to maintain steady growth with occasional setbacks due to regulatory or supply chain disruptions. Revenue growth hinges on product innovation and market expansion.
Revenue Forecast (2023-2028)
| Year |
Estimated Market Size (USD billion) |
CAGR |
Comments |
| 2023 |
1.3 |
3.5% |
Stable growth, price competition persists |
| 2024 |
1.35 |
3.8% |
Market expansion in Asia-Pacific |
| 2025 |
1.4 |
4.0% |
New formulations entering markets |
| 2026 |
1.45 |
4.2% |
Patent expirations lead to generic proliferation |
| 2027 |
1.5 |
4.0% |
Market maturity in developed regions |
Profitability Outlook
- Gross margins for generic producers: approximately 40-50%.
- Branded formulations: margins up to 55-60%, subject to marketing costs.
- Price erosion impacts net profitability, requiring innovation and cost control.
Key Takeaways
- The ibuprofen market is mature with steady growth driven by OTC sales and emerging markets.
- Generic competition exerts downward pressure on prices and margins.
- Innovation in formulations and regional expansion offer avenues for revenue growth.
- Regulatory environments influence market entry and product approvals.
FAQs
Q1: How does patent expiration affect ibuprofen market dynamics?
Patent expirations increase generic availability, reduce prices, and intensify competition, leading to revenue declines in branded segments but creating growth opportunities for generics.
Q2: What brands dominate the ibuprofen global market?
Meda (Nurofen), Johnson & Johnson (Motrin), and Sandoz are key branded players, although generics account for the majority of sales.
Q3: How important are emerging markets for ibuprofen growth?
Very important; they represent higher growth rates due to increasing healthcare access, rising disposable incomes, and less mature competition.
Q4: What are the main drivers of future ibuprofen market growth?
Market expansion in Asia-Pacific, formulation innovations, and consumer preference for OTC products.
Q5: What regulatory challenges could impact ibuprofen sales?
Safety concerns related to gastrointestinal and cardiovascular risks, coupled with differing regional approval processes, could slow market growth.
References
[1] MarketWatch. (2023). Ibuprofen market analysis.
[2] Grand View Research. (2022). Nonsteroidal anti-inflammatory drugs market report.
[3] U.S. Food and Drug Administration (FDA). (2023). OTC drug regulations.
[4] European Medicines Agency (EMA). (2023). NSAIDs regulatory pathways.
[5] Smith, J. (2021). "Global OTC pharmaceutical market dynamics." Journal of Pharmaceutical Economics.