IBUPROFEN Drug Patent Profile
✉ Email this page to a colleague
When do Ibuprofen patents expire, and when can generic versions of Ibuprofen launch?
Ibuprofen is a drug marketed by Amneal Pharms, Ascent Pharms Inc, Aurobindo Pharma Ltd, Bionpharma, Contract Pharmacal, Humanwell Puracap, Marksans Pharma, Onesource Specialty, P And L Dev Llc, Patheon Softgels, Sofgen Pharms, Guardian Drug, L Perrigo Co, Strides Pharma, Sun Pharma Canada, Tris Pharma Inc, Actavis Mid Atlantic, Annora Pharma, Arise, P And L, Padagis Us, Pai Holdings Pharm, Rubicon Research, Perrigo, Abbott, Adaptis, Alkem Labs Ltd, Amneal Pharms Ny, Ani Pharms, Aurobindo Pharma, Avema Pharma, Dr Reddys, Dr Reddys Labs Inc, Granules, Granules India, Halsey, Ivax Sub Teva Pharms, J And J Consumer Inc, Lederle, Leiner, LNK, Mcneil, Merro Pharm, Northstar Hlthcare, OHM, Ohm Labs, Par Pharm, Perrigo R And D, Ph Health, Pharmobedient, Pliva, Purepac Pharm, Rising, Sandoz, Shandong Xinhua, Strides Pharma Intl, Sun Pharm Industries, Sunshine, Superpharm, Teva, Ultratab Labs Inc, Watson Labs, Yichang Humanwell, Dr Reddys Labs Ltd, Pld Acquisitions Llc, Teva Pharms Usa, Torrent, Zydus Lifesciences, and Xgen Pharms. and is included in one hundred and eighty-four NDAs.
The generic ingredient in IBUPROFEN is ibuprofen sodium. There are sixty-four drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the ibuprofen sodium profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for IBUPROFEN?
- What are the global sales for IBUPROFEN?
- What is Average Wholesale Price for IBUPROFEN?
Summary for IBUPROFEN
Recent Clinical Trials for IBUPROFEN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Brigham and Women's Hospital | PHASE1 |
| National Institutes of Health (NIH) | PHASE1 |
| Armed Forces Post Graduate Medical Institute (AFPGMI), Rawalpindi | NA |
Pharmacology for IBUPROFEN
| Drug Class | Nonsteroidal Anti-inflammatory Drug |
| Mechanism of Action | Cyclooxygenase Inhibitors |
Medical Subject Heading (MeSH) Categories for IBUPROFEN
Anatomical Therapeutic Chemical (ATC) Classes for IBUPROFEN
US Patents and Regulatory Information for IBUPROFEN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dr Reddys Labs Inc | IBUPROFEN | ibuprofen | TABLET;ORAL | 076112-001 | Oct 31, 2001 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Marksans Pharma | IBUPROFEN | ibuprofen | TABLET;ORAL | 091239-001 | Feb 1, 2011 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Alkem Labs Ltd | IBUPROFEN | ibuprofen | TABLET;ORAL | 214699-001 | Sep 13, 2021 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sun Pharma Canada | IBUPROFEN | ibuprofen | SUSPENSION;ORAL | 209204-001 | Jun 23, 2017 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sun Pharm Industries | IBUPROFEN | ibuprofen | TABLET;ORAL | 070493-001 | Dec 24, 1985 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strides Pharma | IBUPROFEN | ibuprofen | TABLET;ORAL | 206989-001 | Jun 29, 2018 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for IBUPROFEN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Recordati Rare Diseases | Pedea | ibuprofen | EMEA/H/C/000549Treatment of a haemodynamically significant patent ductus arteriosus in preterm newborn infants less than 34 weeks of gestational age. | Authorised | no | no | no | 2004-07-28 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
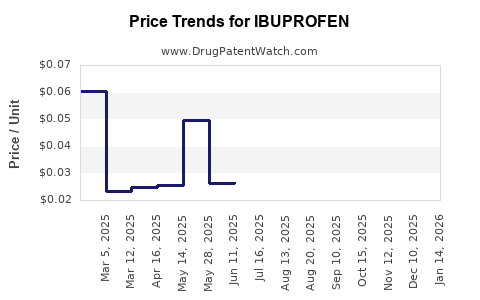
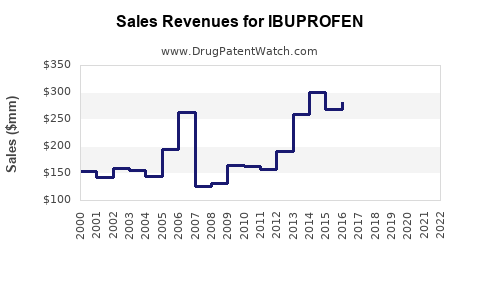
Market Dynamics and Financial Trajectory for Ibuprofen
More… ↓