A single ClinicalTrials.gov entry can redirect millions in legal fees. When a specialty pharma company quietly posts a Phase II 505(b)(2) study, the legal business development clock starts ticking. Firms that read clinical registries as competitive intelligence capture engagements that span patent prosecution, NDA strategy, Orange Book listing, Hatch-Waxman litigation, and eventual licensing deals. Firms that wait for press releases do not.
This guide covers the full operational playbook: how the 505(b)(2) regulatory pathway generates IP complexity, how attorneys convert pipeline data into fee-bearing work, how to structure triage and outreach, and what the real-world engagement economics look like across the drug’s commercial lifecycle.

What the 505(b)(2) Pathway Actually Is, and Why It Generates So Much Legal Work
The 505(b)(2) New Drug Application is a statutory creation of the Drug Price Competition and Patent Term Restoration Act of 1984, commonly called Hatch-Waxman. Section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act allows an applicant to submit an NDA that relies, at least in part, on data or published literature not developed by the applicant. That reliance triggers a specific cascade of IP and regulatory obligations that creates work for attorneys at every stage.
In practical terms, 505(b)(2) covers reformulations of approved drugs (new dosage forms, new delivery systems, new salt forms), fixed-dose combinations of approved active ingredients, new indications for approved molecules, and certain new chemical entities that can rely on published pharmacology data. It is not a generic pathway — the applicant must demonstrate clinical relevance of the modification, not just bioequivalence.
The IP Architecture of a 505(b)(2) Product
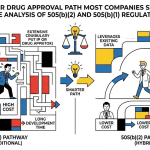
A 505(b)(2) applicant enters a landscape already cluttered with the reference drug’s Orange Book-listed patents. Those patents may cover the active ingredient, the formulation, the method of use, or the dosing regimen. The 505(b)(2) sponsor must certify to each listed patent in one of four ways: a Paragraph I certification (patent has expired), Paragraph II (patent will expire before the applicant’s commercial marketing date), Paragraph III (applicant will not market until the patent expires), or Paragraph IV (the listed patent is invalid or will not be infringed).
The Paragraph IV route is contentious by design. Filing a Paragraph IV certification immediately exposes the 505(b)(2) applicant to patent infringement litigation under 35 U.S.C. Section 271(e)(2). The brand company has 45 days to sue, triggering a 30-month stay on FDA approval if they do. For attorneys on the brand side, monitoring for incoming 505(b)(2) Paragraph IV filers is itself a practice area. For attorneys representing the 505(b)(2) applicant, building a litigation-ready freedom-to-operate position before the Paragraph IV certification drops is standard practice.
Beyond Orange Book patents, the 505(b)(2) sponsor must also think about what it can protect. A new extended-release formulation earns three years of marketing exclusivity under 21 U.S.C. Section 355(c)(3)(E)(iii) if it required new clinical investigations essential to approval. An orphan drug indication earns seven years under the Orphan Drug Act. A qualified infectious disease product can earn an additional five years. Each exclusivity type has distinct conditions and clock-start dates, and stacking them is a recognized lifecycle management tactic. The attorney who helps a sponsor identify and stack exclusivities early in development often generates more enterprise value than the attorney who simply files the NDA.
IP Valuation: Why the Patent Estate Is the Asset
For pharma IP teams and portfolio managers, understanding the monetary value of a 505(b)(2) patent estate is not optional. When a product like Bendeka (Eagle Pharmaceuticals’ reformulated bendamustine, approved 2015 via 505(b)(2)) hit the market, its IP value derived not from the molecule itself — bendamustine had been generic — but from the new formulation patents covering the propylene glycol-based ready-to-dilute formulation. Eagle’s Orange Book-listed patents for Bendeka became the target of Paragraph IV certifications from multiple ANDA filers. The resulting litigation and settlements generated licensing revenue that substantially exceeded Eagle’s development costs for the reformulation.
The core lesson for IP valuation is that a 505(b)(2) product’s patent estate should be modeled across several dimensions: the composition-of-matter coverage (often absent since the molecule is known), the formulation patents (the primary defensive layer), method-of-use patents (especially for new indications), and process patents (occasionally Orange Book-listable). An analyst or IP team that values a 505(b)(2) asset only by its regulatory exclusivity window, without accounting for the litigation-deterrent value of a robust formulation patent estate, systematically undervalues these products. A single Orange Book-listed formulation patent with claims broad enough to require a Paragraph IV certification can add 18 to 30 months of effective market exclusivity through litigation delay, even if the patent is ultimately found invalid.
Key Takeaways: The 505(b)(2) Pathway
The pathway sits between a full NDA and an ANDA, and that structural position means it carries elements of both brand IP strategy and generic challenge risk simultaneously. The formulation or indication improvement generates novel IP, but the underlying molecule’s patent history constrains freedom-to-operate. Law firms entering this space need fluency in both brand-side lifecycle management and ANDA/Paragraph IV litigation. That dual competency is what distinguishes practices that win 505(b)(2) mandates from those that merely understand the regulation.
Why Clinical Trial Registries Are the Earliest Legal Signal
Before an NDA is filed, before a product announcement, before an investor presentation, there is almost always a clinical trial registration. FDA regulations require registration of applicable clinical trials under 42 U.S.C. Section 282(j), and ClinicalTrials.gov has been the primary registry since 2000. A 505(b)(2) sponsor running a bridging pharmacokinetic study, a relative bioavailability study, or a Phase II efficacy trial for a new indication must register the study, and that registration is publicly visible, usually within 21 days of enrollment of the first participant.
That 21-day window, before any press release, before any SEC disclosure, before any FDA submission, is where the legal business development opportunity lives.
Reading a Trial Registration as an IP Intelligence Document
A ClinicalTrials.gov record contains more commercially relevant information than most attorneys extract from it. The official title and brief summary often describe the product category precisely enough to infer the 505(b)(2) strategy. A trial titled ‘Pharmacokinetics of an Extended-Release Formulation of Drug X in Healthy Adults’ tells a pharmaceutical IP attorney several things immediately: the sponsor is pursuing a new dosage form 505(b)(2), the primary regulatory hurdle is a PK bridging study, the Orange Book listing for Drug X needs to be reviewed for formulation patents approaching expiration, and a three-year marketing exclusivity period will be the primary market protection upon approval.
The intervention description identifies the active ingredient and often the dose, delivery mechanism, and target indication. The sponsor field names the company and sometimes the principal investigator, whose academic affiliations can indicate whether a university technology transfer office is involved (suggesting IP licensing complexity). The collaborator field may reveal a CDMO or CRO with proprietary formulation technology, which raises questions about who owns the manufacturing IP. The eligibility criteria often confirm the indication. The primary outcome measures indicate what FDA efficacy standard the sponsor is trying to meet.
When read together, these fields constitute an informal preliminary IP opinion on the incoming product. Attorneys who train their teams to perform this read systematically, rather than treating trial registrations as administrative notices, convert public data into proprietary lead intelligence.
Volume and Timing: The Statistical Case for Monitoring
In 2020, FDA’s Center for Drug Evaluation and Research approved 68 505(b)(2) NDAs, representing roughly 60 percent of all NDA approvals that year. Over one-third of orphan drug NDA approvals in the same period used the 505(b)(2) pathway. These are not aberrations. The proportion of NDAs using 505(b)(2) has grown consistently since the 2000s as branded companies pursue lifecycle management and specialty pharma companies built business models around value-added reformulations.
Each approved 505(b)(2) product was preceded by at least one registered clinical trial, and typically by several. The median time from first trial registration to NDA submission for a 505(b)(2) product is roughly 18 to 36 months for reformulations and three to five years for new indications. That window represents the period during which law firm engagement is both most valuable to the client and most competitively accessible. After NDA submission, the sponsor’s regulatory counsel is usually locked in. Before trial registration, the sponsor may not yet be thinking about patent strategy.
Key Takeaways: Using Trial Data as a Lead Source
The commercial intelligence value of a ClinicalTrials.gov record is highest at registration, not at approval. Firms that build monitoring infrastructure around early-phase 505(b)(2) trial registrations and run systematic IP reads on the records they flag operate with a structural lead-time advantage of 18 to 36 months over firms waiting for NDA-stage business development opportunities. That advantage directly translates to engagements won.
Investment Strategy Note
For portfolio managers and institutional investors, the same trial intelligence that alerts law firms to incoming products can serve as an early signal for secondary market positions or private financing opportunities. A Phase II 505(b)(2) trial registration in a high-value therapeutic area, filed by a company with a credible regulatory history and no current Orange Book presence for the target drug, often indicates a near-term inflection point. The combination of an early-stage trial and a clean IP competitive landscape — meaning the reference drug’s formulation patents are expiring or weak — creates a favorable risk-adjusted development scenario worth tracking.
The Operational Playbook: From Trial Alert to Client Engagement
Identifying a promising 505(b)(2) trial registration is step one. Converting that identification into a retained mandate requires a disciplined process that moves faster than competitors and engages clients with genuine technical credibility rather than cold solicitation.
Building the Monitoring Infrastructure
Effective monitoring requires more than a ClinicalTrials.gov keyword alert. The most operationally mature law firm practices layer multiple data sources to triangulate signal quality before committing resources to outreach.
ClinicalTrials.gov provides the primary alert. The registry’s API allows programmatic queries by intervention type, sponsor name, condition, or keyword, and it returns machine-readable JSON that can be piped into a law firm’s CRM. Firms that have not yet built this integration often use commercial pipeline databases — Clarivate’s Cortellis, Citeline’s Pharmaprojects, or DrugPatentWatch — which classify pipeline assets by regulatory pathway, including 505(b)(2), and provide alert functionality at the pathway level.
The Orange Book is the second layer. When a new trial appears for a reformulation of Drug X, the firm should immediately pull Drug X’s Orange Book patent listings and exclusivity database entry. This reveals which formulation patents are listed, their expiration dates, and any current ANDA or 505(b)(2) paragraph certifications already on file. A drug whose last listed formulation patent expires within three years, whose reference listed drug is the target of ongoing ANDA litigation, and whose indication has no current 505(b)(2) challenger represents a high-priority lead. One whose Orange Book listing is densely populated with active formulation patents expiring in a decade represents a more complex competitive landscape.
The USPTO Patent Center is the third layer. Searching for patent applications assigned to the 505(b)(2) sponsor, or to the formulation technology company identified in the trial registration, reveals whether the sponsor is already building a patent estate around the new product. A sponsor with active patent applications on the reformulation has thought about IP. A sponsor with no relevant patent applications may need the most help, or may be planning to rely on regulatory exclusivity alone, which is a strategy with meaningful commercial limitations that a good IP attorney can articulate precisely.
SEC filings, if the sponsor is publicly traded, provide a fourth layer. Form 10-K risk factor disclosures, 10-Q pipeline updates, and 8-K announcements of FDA interactions can corroborate what the trial registration suggests and add financial context about the sponsor’s capacity to retain outside counsel.
Lead Triage: Ranking the Opportunity
Not every 505(b)(2) trial registration warrants outreach. Firms that contact every sponsor they identify quickly develop a reputation for spam and dilute their credibility with the target universe. Effective triage uses a small number of high-signal variables.
Therapeutic area value matters significantly. A 505(b)(2) for a reformulated oncology drug in a market with multi-billion dollar peak sales potential generates proportionately larger legal economics — bigger patent prosecution budgets, higher litigation stakes, more complex licensing negotiations — than a reformulation in a crowded generic market. Similarly, rare disease indications pursued via 505(b)(2) carry the orphan designation overlay, which adds a seven-year exclusivity claim and often a pediatric extension, creating more IP architecture to build and defend.
Company stage matters. A 50-person specialty pharma company that has taken one product through FDA approval has demonstrated regulatory competence but likely lacks deep in-house IP expertise. This is the profile most likely to be in the market for outside patent counsel, regulatory attorneys, and licensing advisors simultaneously. A company filing its first 505(b)(2) NDA is in an even stronger position as a target — they know they need help and do not yet have entrenched counsel relationships in all the relevant practice areas.
Clinical phase matters. A Phase I pharmacokinetic bridging study indicates the sponsor is in early development; the engagement window is long but the sponsor’s urgency is lower. A Phase III 505(b)(2) study means NDA submission is likely 12 to 24 months out, the IP architecture needs to be in place now, and the sponsor is under real time pressure. Late-phase trials create the strongest urgency and the most compelling pitch context.
Patent cliff proximity matters. If the reference listed drug’s base patents expire within the trial’s development horizon, the window for building and filing new formulation and method-of-use patents is tight. Attorneys who frame their outreach around this specific timing pressure — ‘your formulation patents need to be on file before your PK data is public’ — provide an immediate, concrete reason to engage.
The Outreach Approach
Cold outreach from a law firm to a biotech company based on a clinical trial registration is legal and common. The trial registration is public information. No confidentiality obligation is triggered by reading it. The ethical issues arise only if the firm already represents a party with adverse interests to the sponsor, which standard conflict checks will surface.
Effective outreach does not lead with ‘we saw your trial registration.’ It leads with substantive expertise. The senior attorney reaching out should open with a specific observation about the regulatory or IP landscape relevant to the sponsor’s product — the expiration status of the reference drug’s formulation patents, the current state of litigation in the relevant technology class, or a recent FDA guidance that affects the sponsor’s development plan. This positions the firm as a knowledgeable advisor rather than a business development operation that scraped a database.
The most productive initial conversations focus on a specific, bounded question the sponsor likely has not fully resolved: whether a Section viii carveout is available to avoid a Paragraph IV certification for a particular listed patent, how to structure an orphan drug designation request to maximize exclusivity scope, or what data package FDA will require for the bridging study. These are questions an in-house team at a small specialty pharma company may genuinely be wrestling with, and a 30-minute call that provides real answers generates goodwill and credibility that a generic pitch about ‘full-service pharmaceutical law’ does not.
Key Takeaways: The Operational Playbook
The firms winning 505(b)(2) mandates at scale are not doing anything structurally exotic. They have built monitoring infrastructure that covers ClinicalTrials.gov, the Orange Book, and USPTO applications simultaneously. They have a triage rubric that ranks leads by therapeutic area value, company stage, clinical phase, and patent cliff proximity. And they lead outreach with substantive technical content rather than capability statements. That combination, executed consistently, produces a pipeline of engaged prospects that converts at rates meaningfully above cold solicitation averages.
The Full Legal Services Roadmap Across a 505(b)(2) Product’s Life
When a law firm engages early on a 505(b)(2) product, the relationship can span a decade or more. Each phase of the product’s regulatory and commercial life generates distinct legal work, and the firm that built the initial relationship is positioned to capture each successive engagement.
Phase One: Pre-Filing IP Architecture
The earliest and often most consequential work happens before the NDA is submitted. The core deliverable is a patent estate that will support both market exclusivity and Orange Book listing.
Formulation patent drafting is the primary task. For a new extended-release dosage form, claims should cover the specific polymer matrix or coating system, the drug release profile, and the manufacturing process. Broader claims covering the concept of extended-release of the active ingredient will face prior art challenges if extended-release formulations of the molecule have previously been described in the literature or in expired patents, so claim architecture must be tailored to what is genuinely novel in the specific formulation.
Method-of-use patent drafting is the secondary task. If the 505(b)(2) is pursuing a new indication, claims covering the method of treating the specific condition with the approved formulation can be Orange Book-listed and cited in the NDA. These patents are particularly valuable because they are difficult for a Paragraph IV filer to design around without also designing around the approved indication.
Freedom-to-operate analysis runs in parallel. The FTO opinion maps the reference drug’s existing patent landscape and identifies whether any claims of unexpired patents could be read to cover the new formulation or indication. Where potential issues are identified, the 505(b)(2) development team can adjust the formulation or the proposed labeling before the design is locked in, at a fraction of the cost of post-submission redesign or litigation-stage resolution.
Orange Book listing strategy requires early attention because FDA’s rules on what qualifies for listing are specific. Under 21 C.F.R. Section 314.53, only patents that claim the approved drug or an approved method of using the drug may be listed. Process patents do not qualify. A firm that advises the sponsor to file and prosecute only claims that will pass FDA’s listing requirements ensures the Orange Book estate is maximally protective.
Phase Two: NDA Submission and Exclusivity Procurement
The 505(b)(2) NDA submission triggers a set of legal and regulatory actions that run concurrently.
The Paragraph IV certification decision, if the sponsor cannot fully design around all Orange Book-listed patents of the reference drug, must be made before submission. Filing a Paragraph IV certification requires a detailed notice letter to each patent owner and NDA holder, explaining the factual and legal basis for the certification. Drafting that notice is skilled legal work that directly shapes the scope and duration of any resulting litigation.
Alternatively, a Section viii statement certifies that the applicant is not seeking approval for the uses claimed in a particular method-of-use patent and commits the applicant to carving that use out of the labeling. Section viii statements avoid the 30-month stay but require careful label carveout analysis to ensure the carvedout use will not be infringed by the expected prescribing patterns for the approved indication.
Orphan drug designation applications, if applicable, should be submitted well before the NDA. The Orphan Drug Office has its own review process and timeline, and designation is more readily granted before market approval than after. Attorneys with orphan drug experience know that the designation request must define the rare disease precisely, cite prevalence data supporting the under-200,000-patients threshold, and make the plausibility case for clinical benefit. Designation granted before NDA submission allows the sponsor to market the orphan exclusivity in licensing and partnership discussions as a confirmed asset rather than a pending application.
Pediatric exclusivity, which adds six months to all existing Orange Book-listed patent terms, is available to 505(b)(2) sponsors who receive a Written Request from FDA to conduct pediatric studies. Proactively requesting a Written Request, rather than waiting for FDA to issue one, can accelerate the pediatric exclusivity clock. Attorneys familiar with this process help sponsors identify whether their product is eligible and how to structure the request.
Phase Three: Post-Approval Defense and Monetization
Approval triggers the challenge period. Within 20 days of FDA’s publication of approval, generic companies monitoring the Orange Book for new 505(b)(2) approvals will assess whether to file ANDAs with Paragraph IV certifications targeting the newly-listed patents.
The first Paragraph IV ANDA filed against a 505(b)(2) product’s listed patents initiates a 45-day window during which the 505(b)(2) holder can sue for patent infringement and trigger the 30-month stay. The quality of the patent estate — the breath of the formulation claims, the strength of the method-of-use claims — determines how defensible the stay period is. A patent estate built with Orange Book listing strategy in mind, with claims drafted to be both infringed by any generic equivalent and novel enough to withstand invalidity challenges, can hold off generic competition through the full stay period and into settlement discussions.
Litigation settlements in the 505(b)(2) context frequently take the form of authorized generic agreements, where the 505(b)(2) holder licenses the ANDA filer to launch a co-branded generic at a future date in exchange for dropping the Paragraph IV challenge. These deals require skilled transactional and antitrust counsel, as they sit squarely in the antitrust enforcement crosshairs established by the Supreme Court’s FTC v. Actavis decision in 2013. Attorneys who advised the sponsor throughout development can model the settlement economics far more efficiently than outside counsel brought in cold at the litigation stage.
Out-licensing is a parallel monetization track. A 505(b)(2) sponsor with a strong patent estate and limited commercial infrastructure may find that licensing rights to a larger distributor or co-promotion partner generates more enterprise value than a fully internal commercial launch. Attorneys who understand both the IP and the regulatory exclusivity period can structure licenses that capture royalty streams through the exclusivity window, sub-license manufacturing rights while retaining method-of-use patent enforcement, and include milestone payments tied to Paragraph IV challenge outcomes.
Key Takeaways: The Full Services Roadmap
A law firm that engages at Phase I trial registration and maintains the relationship through post-approval generates work across at least six distinct practice areas: patent prosecution, FDA regulatory counsel, Hatch-Waxman litigation, antitrust review of settlements, licensing transactions, and potentially M&A advisory if the sponsor becomes an acquisition target after approval. The net present value of that full-lifecycle engagement, computed over the exclusivity period of a commercially successful 505(b)(2) product, can range from low seven figures for a niche specialty product to eight figures for an orphan drug with sustained pricing power.
Investment Strategy Note
For investors assessing a specialty pharma company with a 505(b)(2) asset, the patent estate quality is a more reliable predictor of post-approval cash flow duration than the regulatory exclusivity period alone. A company entering approval with only a three-year marketing exclusivity claim and no Orange Book-listed formulation patents faces generic entry on day one of year four. A company entering approval with five Orange Book-listed patents, two of which expire in year 12 and have been successfully defended in district court, has a fundamentally different earnings profile. IP audit quality — the depth of Orange Book coverage, the validity risk profile of listed patents, the breadth of pending claims — belongs in the financial model alongside peak sales assumptions.
Real-World 505(b)(2) IP Case Studies: The Legal and Commercial Record
Eagle Pharmaceuticals and Bendeka: Formulation IP as a Revenue Engine
Bendeka (bendamustine hydrochloride injection, 25 mg/mL) received FDA approval in December 2015 via the 505(b)(2) pathway. Eagle Pharmaceuticals developed Bendeka as a reformulation of Teva’s Treanda (bendamustine at 5 mg/mL, or in a lyophilized powder form), relying on Treanda’s clinical data to support Bendeka’s safety and efficacy while conducting new studies on the ready-to-dilute formulation. The reformulation reduced infusion time from 60 minutes to 10 minutes and required less saline diluent.
Eagle listed multiple patents covering the Bendeka formulation in the Orange Book. Paragraph IV certifications followed quickly from generic filers including Mylan, Fresenius Kabi, Accord Healthcare, and others. Eagle sued within the 45-day window in each case, triggering the 30-month stays. The resulting litigation went on for years and generated multiple settlement agreements. Eagle also entered a co-promotion arrangement with Teva for Bendeka, which produced significant revenue even before the generic challenges were resolved.
The Bendeka case is instructive for several reasons. The underlying molecule, bendamustine, was not patentable. The formulation innovation — the propylene glycol-based ready-to-dilute system and its specific concentration — was the entire IP asset. Eagle’s ability to generate substantial revenue from a product built on a decades-old chemotherapy drug depended entirely on the strength of the formulation patent estate. For attorneys, the Bendeka pattern is a template: identify a widely-used drug with a clinically meaningful reformulation opportunity, build a dense Orange Book-listed patent estate around the new formulation, and use the 30-month stay to generate settlement revenue and establish commercial market share.
For IP valuation purposes, Bendeka demonstrates that formulation patents on a 505(b)(2) product can support enterprise values many times the cost of development. Eagle’s market capitalization grew substantially following Bendeka’s approval, driven almost entirely by the patent-protected revenue stream from a drug that cost a fraction of a novel NDA program to develop.
Zuplenz (Ondansetron Oral Soluble Film): New Dosage Form, New IP
Zuplenz, the oral soluble film formulation of ondansetron developed by NovaBay Pharmaceuticals and later acquired by Strativa Pharmaceuticals, received 505(b)(2) approval in 2010. Ondansetron itself, the antiemetic best known as Zofran, had been generic since 2006. The Zuplenz formulation used a polymer film matrix to deliver the drug sublingually, which offered dosing convenience for patients experiencing nausea severe enough to make swallowing a tablet difficult.
The 505(b)(2) strategy relied on the new dosage form exclusivity (three years) and Orange Book-listed film formulation patents. The IP architecture supporting Zuplenz demonstrates the value of dosage form specificity in claim drafting: patents covering an oral soluble film of a 5-HT3 antagonist, with specific polymer compositions and dissolution characteristics, are considerably more defensible than generic claims to ‘a mucoadhesive formulation.’ Generic companies ultimately challenged the Zuplenz patents, but the patent estate provided meaningful commercial runway before generic entry.
For portfolio managers, Zuplenz shows that 505(b)(2) value-add strategies do not require therapeutic area novelty. The commercial case for Zuplenz was entirely a formulation and delivery story applied to a well-understood, widely-prescribed molecule. The IP strategy was correspondingly focused on formulation claims rather than mechanism-of-action claims.
Horizon Pharma and Vimovo: Fixed-Dose Combination IP
Vimovo (naproxen/esomeprazole magnesium, 375 mg/20 mg and 500 mg/20 mg) received 505(b)(2) approval in 2010. The product combined an approved NSAID (naproxen) with a proton pump inhibitor (esomeprazole) in a single bilayer tablet designed to reduce the gastric side effects of NSAID therapy. Both naproxen and esomeprazole were individually generic by the time of approval. The entire commercial value of Vimovo resided in the combination patent estate and the three-year marketing exclusivity for the new combination requiring new clinical investigations.
AstraZeneca (which licensed rights to Pozen, the originator, and later sold rights to Horizon Pharma) listed multiple patents in the Orange Book covering the specific bilayer combination, the release profile of the two components, and the method of treating pain while reducing NSAID-induced upper gastrointestinal injury. Numerous generic companies filed Paragraph IV ANDAs. Litigation was extensive. Horizon Pharma and its predecessors ultimately prevailed on some patent claims and settled others.
The Vimovo litigation history is required reading for any attorney building a fixed-dose combination 505(b)(2) practice. The validity challenges focused on obviousness — combining two approved drugs in a single tablet was argued to be an obvious design step. The claim drafting decisions made at prosecution stage, specifically how narrowly or broadly the combination and release profile claims were written, proved determinative in district court and appellate rulings. Attorneys advising on fixed-dose combination 505(b)(2) patent strategy need to anticipate obviousness challenges during prosecution and build file history that supports non-obviousness arguments from the outset.
Data Infrastructure: What Actually Works for Pipeline Intelligence
The monitoring tools that produce actionable 505(b)(2) leads require configuration, not just subscription. A law firm that pays for access to Clarivate’s Cortellis or DrugPatentWatch and runs no systematic workflow around the output will generate the same results as a firm using only ClinicalTrials.gov: occasional valuable sightings amid substantial noise.
ClinicalTrials.gov API Configuration
The ClinicalTrials.gov API (https://clinicaltrials.gov/api/v2/studies) supports structured queries with Boolean operators. Searches combining intervention name, study type, and phase with a date filter for recent registrations produce a manageable daily feed. A query filtering for studies with intervention type ‘Drug,’ phase ‘Phase 2 OR Phase 3,’ and first-posted date within the past 30 days, combined with keyword terms like ‘extended-release,’ ‘modified-release,’ ‘fixed-dose combination,’ or the name of a specific molecule approaching patent expiration, can be set up as an automated daily pull into a spreadsheet or CRM with minimal technical investment.
The most technically sophisticated practices use the API output to cross-reference sponsor names against a proprietary database of known pharma and specialty pharma companies by size and IP maturity, flagging registrations from companies in the target client profile — small to mid-size specialty pharma with a track record of one to three FDA approvals and a history of outside IP counsel engagement.
Orange Book Monitoring Protocols
FDA updates the Orange Book database continuously. The Electronic Orange Book is downloadable in flat file format and can be compared against a prior-period snapshot to identify new patent listings, new exclusivity entries, and new 505(b)(2) certifications. A monthly diff of the Orange Book flat files reveals which products received new patent listings (indicating a brand company is fortifying an existing product’s IP) and which products received new Paragraph IV certifications (indicating an incoming generic or 505(b)(2) challenge).
For law firms representing brand-side clients, the Paragraph IV certification diff is particularly valuable: it provides advance notice of incoming challenges that may require litigation counsel to be engaged. For firms without an existing brand relationship, a Paragraph IV certification against a product is an indicator that the brand company may need additional patent counsel to support the defense.
Patent Application Monitoring
USPTO’s Patent Center allows full-text searching of published patent applications. Monitoring published applications by sponsor company name, or by the technology class relevant to a known 505(b)(2) development program, reveals whether the applicant has filed new claims. If a sponsor registered a Phase II trial 18 months ago but has filed no patent applications since, that is a signal of an IP gap. If a sponsor has filed multiple continuation applications building on an original composition patent, that is a signal of a sophisticated IP operation that may not need outside prosecution counsel but may need litigation counsel once Paragraph IV certifications start arriving.
Key Takeaways: Data Infrastructure
The competitive advantage in clinical trial intelligence comes from systematic, multi-source monitoring configured around a clear client profile, not from subscribing to more databases. Firms that define their target client precisely — therapeutic area, company stage, IP maturity, trial phase — and configure their monitoring infrastructure around that profile generate higher-quality leads at lower noise ratios than firms attempting broad-spectrum monitoring without a triage framework.
Legal Ethics and Operational Risk
The clinical trial monitoring practice raises several compliance considerations that any firm building this capability needs to address explicitly.
Solicitation Rules
The Model Rules of Professional Conduct Rule 7.3 prohibits in-person, live telephone, or real-time electronic contact soliciting professional employment from a person when a significant motive is pecuniary gain, if the person has not already requested the firm’s representation or is not a lawyer. Most 505(b)(2) sponsors are sophisticated organizations represented by in-house counsel, and direct business-to-business outreach to in-house counsel or business development executives, offering legal services, is generally permissible. Firms should consult their state bar’s specific rules on attorney advertising and solicitation before building an outreach program, as rules vary by jurisdiction.
Conflict Screening
A firm monitoring ClinicalTrials.gov for 505(b)(2) opportunities will periodically identify trials sponsored by companies the firm already represents, or by companies with adverse interests to existing clients. Conflict screening should occur before any outreach and before any substantive review of the trial record that might constitute privileged analysis. The business development team and the client representation teams should not share unscreened trial intelligence.
Information Firewall Considerations
Some large firms create information barriers between the pipeline monitoring function and specific client representations. If a firm represents Company A in Orange Book patent litigation regarding Drug X, and the monitoring function identifies a 505(b)(2) trial for a reformulation of Drug X by Company B, the firm faces a potential conflict that requires analysis under Rule 1.7 before any outreach to Company B occurs.
Signal Quality and False Positives
Not every registered 505(b)(2) trial produces an approved product. FDA approval rates for Phase II trials overall are approximately 50 to 60 percent for small molecules, with lower rates in certain therapeutic areas. Firms that commit significant business development resources to every Phase II 505(b)(2) registration will find a meaningful portion of their pipeline disappears to trial failure or sponsor abandonment. Building a triage system that weights Phase III registrations more heavily, and that checks for Phase II data disclosure or interim results before committing to outreach, reduces false-positive rates.
Positioning the Firm: Authority Before Outreach
The firm that contacts a 505(b)(2) sponsor cold with a generic capabilities pitch faces an immediate credibility deficit. The firm that has published substantive, technically accurate analysis of 505(b)(2) regulatory strategy, Paragraph IV litigation trends, or Orange Book listing requirements enters the same conversation with pre-established credibility. The clinical trial monitoring practice functions most effectively when it operates downstream of an active thought leadership program.
Publishing analysis of specific 505(b)(2) approval trends, writing about the IP implications of recent FDA guidance on bioequivalence for complex drug products, or presenting at DIA, AAPS, or Bio conferences on 505(b)(2) patent strategy builds name recognition in the precise community the firm is trying to reach. When a specialty pharma company’s regulatory affairs director sees a firm’s attorney present at DIA on the Section viii carveout analysis framework, and that attorney’s name later appears in an outreach email referencing a just-registered trial, the email reads as expert outreach rather than cold solicitation.
Firms that have successfully used this model include Hyman, Phelps & McNamara, which built a large 505(b)(2) regulatory practice around published FDA regulatory intelligence; Alston & Bird, whose FDA and life sciences group has combined patent prosecution and regulatory work for 505(b)(2) clients; and Windels Marx, which has focused specifically on 505(b)(2) and Hatch-Waxman matters for specialty pharma companies. Each firm’s market position reflects a prior investment in specialty credibility that makes clinical trial outreach productive rather than merely intrusive.
The Economics of a Full-Lifecycle 505(b)(2) Engagement
To close the business case for this practice model, it helps to frame the fee economics concretely. A 505(b)(2) engagement that begins at Phase II trial registration and runs through post-approval generic defense generates work in multiple billing categories across a multi-year period.
Pre-filing patent prosecution, including freedom-to-operate analysis, formulation patent drafting and prosecution, method-of-use patent drafting, and Orange Book listing strategy, typically runs between $300,000 and $800,000 for a mid-complexity 505(b)(2) product. NDA regulatory support, including preparation of the patent certification analysis, support for Section viii carveout decisions, and orphan drug designation filings if applicable, runs an additional $150,000 to $400,000. Paragraph IV litigation, if it materializes, is the largest single engagement: major 505(b)(2) patent litigations routinely exceed $5 million in legal fees per party before settlement. Licensing negotiations for out-licensing arrangements add transactional fees ranging from flat-fee retainers to percentage-of-deal structures.
A firm that wins the full engagement on a successful 505(b)(2) product in a high-value therapeutic area can realistically collect $8 million to $20 million in fees across the product’s commercial life. A firm that enters at the NDA submission stage collects a fraction of that, primarily from Paragraph IV defense. The difference between early and late entry into a 505(b)(2) client relationship is the difference between capturing the full value chain and competing for a single, already-contested engagement.
That economics argument, made precisely and with reference to the client’s specific development timeline, is the most compelling case a law firm can bring to a 505(b)(2) sponsor in the Phase II stage.
What Changes When Biologics Enter the Picture: The 351(k) Parallel
The 505(b)(2) monitoring playbook has a structural analog in the biologic space that attorneys and IP teams should be building in parallel.
Section 351(k) of the Public Health Service Act governs biosimilar applications, which reference an innovator biologic’s approval in a manner analogous to how 505(b)(2) references the listed drug’s data. A 351(k) applicant must navigate the Biologics Price Competition and Innovation Act’s (BPCIA) patent resolution process — the ‘patent dance’ — which includes formal information exchange between the biosimilar applicant and the reference product sponsor regarding patents that could be asserted against the biosimilar.
Clinical trial registrations for biosimilar programs are publicly visible in the same way 505(b)(2) trials are. A Phase III pharmacokinetic comparability or clinical equivalence study for a proposed biosimilar of a named reference biologic signals incoming BPCIA patent dance engagement for the innovator. Law firms representing biologic innovators should monitor for biosimilar trial registrations against their clients’ reference products. Firms seeking biosimilar applicant clients should monitor for biosimilar Phase III registrations by sponsors who have not yet engaged established BPCIA patent dance counsel.
The biologic IP complexity exceeds 505(b)(2) substantially. Reference product exclusivity under BPCIA runs 12 years, during which no biosimilar can be approved regardless of patent status. The patent estate for a major biologic — adalimumab, for example, which is estimated to be covered by over 130 active patents — presents a scale of Orange Book analog (the Purple Book) management that dwarfs a typical small molecule 505(b)(2) product. But the same structural logic applies: clinical trial registrations are the earliest public signal, and the firm that reads them first and engages earliest controls the relationship.
Final Operational Summary
The 505(b)(2) clinical trial monitoring practice is not a speculative business development strategy. It is a systematic application of publicly available regulatory data to identify a defined client population at the moment of maximum legal need and minimum competitive coverage. The operational requirements — monitoring infrastructure, triage criteria, substantive outreach content, and thought leadership positioning — are well within the capacity of any life sciences practice group at a firm of 20 or more attorneys.
The firms that execute this consistently over a two-to-three year period build pipeline databases of hundreds of 505(b)(2) programs in various development stages, convert a meaningful fraction of those into retained engagements, and generate full-lifecycle fee revenues that dwarf what the same attorneys would produce through reactive client development. The ClinicalTrials.gov data is free. The Orange Book is free. The USPTO database is free. The competitive advantage comes entirely from building the workflow around them and executing it with technical credibility.
For pharma IP teams using this analysis from the sponsor side: the same intelligence that law firms use to find you early is available to you to use in managing your own IP strategy. Monitoring 505(b)(2) trial registrations in your therapeutic area reveals incoming competitive products before they are publicly announced, which informs your Orange Book patent listing strategy, your Paragraph IV litigation readiness, and your lifecycle management planning. The registry is not just a regulatory compliance requirement. It is a competitive intelligence document, and the companies that read it as such operate with a structural information advantage.
Data on 505(b)(2) approval volumes reflects CDER annual reports. Patent prosecution and litigation fee ranges are informed by industry survey data from AIPLA and published case records. Bendeka, Zuplenz, and Vimovo patent histories are based on public Orange Book records, district court dockets, and published appellate decisions.