ESOMEPRAZOLE Drug Patent Profile
✉ Email this page to a colleague
When do Esomeprazole patents expire, and what generic alternatives are available?
Esomeprazole is a drug marketed by Alkem Labs Ltd, Amneal Pharms Ny, Aurobindo Pharma Ltd, Cipla, Cisen, Cspc Ouyi, Dr Reddys, Ethypharm, Glenmark Speclt, Granules, Graviti Pharms, Guangzhou Novaken, Hec Pharm, Hetero Labs Ltd Iii, Indchemie Health, Lannett Co Inc, Macleods Pharms Ltd, Mylan, Prinston Inc, Sun Pharm, Torrent, Zhejiang Yongtai, Zydus Pharms, Aurobindo Pharma, Guardian Drug, Marksans Pharma, Perrigo R And D, Pharmobedient, P And L, Dexcel, Accord Hlthcare, Deva Holding As, Epic Pharma Llc, Eugia Pharma, Gland, Slate Run Pharma, and Belcher. and is included in fifty-nine NDAs. There are two patents protecting this drug.
The generic ingredient in ESOMEPRAZOLE is esomeprazole strontium. There are seventy-four drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the esomeprazole strontium profile page.
DrugPatentWatch® Generic Entry Outlook for Esomeprazole
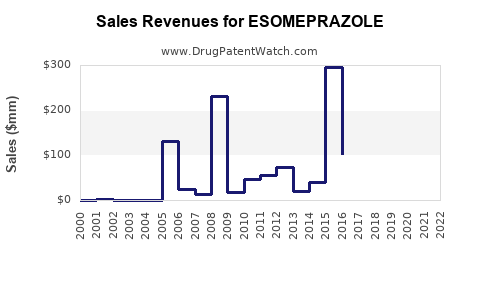
Annual sales in 2022 were $61mm indicating the motivation for generic entry (peak sales were $295mm in 2015).
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ESOMEPRAZOLE?
- What are the global sales for ESOMEPRAZOLE?
- What is Average Wholesale Price for ESOMEPRAZOLE?
Summary for ESOMEPRAZOLE
| US Patents: | 0 |
| Applicants: | 37 |
| NDAs: | 59 |
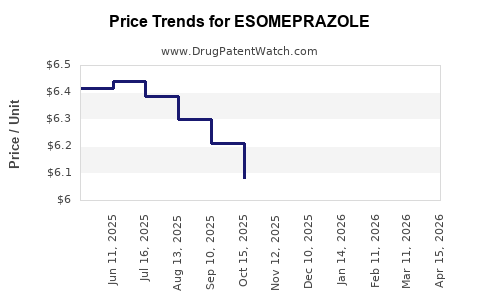
| Drug Prices: | Drug price information for ESOMEPRAZOLE |
| Drug Sales Revenues: | Drug sales revenues for ESOMEPRAZOLE |
| What excipients (inactive ingredients) are in ESOMEPRAZOLE? | ESOMEPRAZOLE excipients list |
| DailyMed Link: | ESOMEPRAZOLE at DailyMed |
US Patents and Regulatory Information for ESOMEPRAZOLE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Torrent | ESOMEPRAZOLE MAGNESIUM | esomeprazole magnesium | FOR SUSPENSION, DELAYED RELEASE;ORAL | 212651-002 | Mar 5, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sun Pharm | ESOMEPRAZOLE MAGNESIUM | esomeprazole magnesium | CAPSULE, DELAYED REL PELLETS;ORAL | 209735-001 | Apr 30, 2018 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Indchemie Health | ESOMEPRAZOLE MAGNESIUM | esomeprazole magnesium | CAPSULE, DELAYED REL PELLETS;ORAL | 210559-002 | Feb 26, 2021 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Epic Pharma Llc | ESOMEPRAZOLE SODIUM | esomeprazole sodium | INJECTABLE;INTRAVENOUS | 214046-001 | Jan 8, 2025 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Amneal Pharms Ny | ESOMEPRAZOLE MAGNESIUM | esomeprazole magnesium | CAPSULE, DELAYED RELEASE;ORAL | 209716-001 | Jun 5, 2019 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |